3% Citanest Dental with Octapressin
Brand Information
| Brand name | 3% Citanest Dental with Octapressin |
| Active ingredient | Prilocaine hydrochloride + Felypressin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the 3% Citanest Dental with Octapressin.
Summary CMI
3% Citanest® DENTAL with Octapressin®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Citanest DENTAL
The medicine, 3% Citanest DENTAL with Octapressin, is hereafter referred to as Citanest DENTAL in this leaflet.
Citanest DENTAL contains the active ingredients prilocaine hydrochloride and felypressin. It is used to prevent or relieve pain during dental procedures.
For more information, see Section 1. Why am I using Citanest DENTAL? in the full CMI.
2. What should I know before I use Citanest DENTAL?
Do not use if you have ever had an allergic reaction to prilocaine, felypressin or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Citanest DENTAL? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Citanest DENTAL and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How Citanest DENTAL is given?
Citanest DENTAL is injected by your dentist into the gum near nerves. Depending on the amount used, Citanest DENTAL will either totally stop pain or will cause a partial loss of feeling. Dosage depends on your size, age, and procedure.
More instructions can be found in Section 4. How Citanest DENTAL is given? in the full CMI.
5. What should I know while being given Citanest DENTAL?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given Citanest DENTAL? in the full CMI.
6. Are there any side effects?
Tell your doctor or nurse as soon as possible if you do not feel well while you are being given Citanest DENTAL.
Some of these mild side effects include: nervousness, dizziness, blurred vision, drowsiness, ringing in ears, numbness, nausea and vomiting.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
3% Citanest® DENTAL with Octapressin®
Active ingredients: prilocaine hydrochloride and felypressin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Citanest DENTAL with Octapressin hereafter referred to as Citanest DENTAL in this CMI.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Citanest DENTAL.
Where to find information in this leaflet:
1. Why am I using Citanest DENTAL?
2. What should I know before I use Citanest DENTAL?
3. What if I am taking other medicines?
4. How Citanest DENTAL is given?
5. What should I know while being given Citanest DENTAL?
6. Are there any side effects?
7. Product details
1. Why am I using Citanest DENTAL?
Citanest DENTAL contains the active ingredients prilocaine hydrochloride and felypressin. Citanest DENTAL belongs to a group of medicines called local anaesthetics.
This medicine is used to prevent or relieve pain during dental procedures, but it will not put you to sleep.
It is injected into the mouth, where it makes the nerves unable to pass messages to the brain. Depending on the amount used, Citanest DENTAL will either totally stop pain or will cause a partial loss of feeling.
The felypressin makes it last longer. Felypressin makes the blood vessels at the site of injection narrower, which means you bleed less and it is easier for your dentist to see what is happening.
Your dentist will have explained why you are being treated with Citanest DENTAL. Follow all directions given to you by your dentist carefully. They may differ from the information contained in this leaflet. Ask your dentist if you want more information.
Citanest DENTAL is not addictive.
2. What should I know before I use Citanest DENTAL?
Warnings
Do not use Citanest DENTAL if:
- you are allergic to prilocaine hydrochloride, felypressin, or any of the ingredients listed at the end of this leaflet.
- the expiry date printed on the pack has passed.
- the solution is not clear, or the packaging is torn or shows signs of tampering.
If you have an allergic reaction, you may experience:
- a shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Check with your doctor if you:
- have any other medical conditions
- problems with your heart beat rhythm or a very slow heart beat
- anaemia (a decreased number of red blood cells)
- diabetes
- nerve problems
- heart problems
- liver problems
- kidney problems
- malignant hyperthermia
It may not be safe for you to be given Citanest DENTAL if you have any of these conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Talk to your doctor if you are pregnant or breastfeeding, or planning to. Your dental practitioner can discuss with you the risks and benefits involved.
Citanest DENTAL has been widely used during pregnancy and there have been no reports of any ill effects on the baby.
This medicine may enter breast milk, but in such small amounts that there is generally no risk of affecting the baby.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Citanest DENTAL and affect how it works. These include:
- medicines used to treat irregular heart activity (antiarrhythmics)
- a type of antibiotic medicine, known as sulphonamides
- medicines used to treat malaria, such as quinine
- medicines called nitric compounds such as nitroglycerin which is used to treat angina.
You may need different amounts of your medicines, or you may need to take different medicines.
Your dental practitioner and pharmacist have more information on medicines to be careful with or avoid while being given this medicine.
4. How Citanest DENTAL is given?
Follow all directions given to you by your dental practitioner carefully. They may differ from the information contained in this leaflet.
How much is given
The dosage you will be given will depend on your body size, age and the type of dental work you will be having one.
Your dentist will have had a lot of experience injecting Citanest DENTAL or other local anaesthetics and will choose the best dose for you. They will be willing to discuss this decision with you.
How it is given/administered
Xylocaine DENTAL will be injected by your dentist into the gum inside your mouth near a single nerve, or into an area that contains a large number of nerves.
This will result in an area of numbness at the site or near the site of injection.
Xylocaine DENTAL should not be injected directly into the blood.
If you are given too much Citanest DENTAL
The dentist giving you Citanest DENTAL will be experienced in the use of local anaesthetics, so it is unlikely that you will be given an overdose.
However, if you are particularly sensitive to Citanest DENTAL, or the dose is accidentally injected directly into your blood, you may develop problems for a short time. You may get a numb feeling in or around the mouth, feel dizzy or stiff, or have twitchy muscles.
Whenever you are given Citanest DENTAL, equipment will be available to care for you if an overdose happens.
If an overdose occurs, you should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia; 0800 764 766 in New Zealand), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
5. What should I know while being given Citanest DENTAL?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you have been recently given Citanest DENTAL.
Things you should not do
Do not eat or drink anything until the feeling has returned to your mouth. You may burn or bite yourself.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Citanest DENTAL affects you.
Citanest DENTAL may cause dizziness in some people. You may be drowsy and your reflexes may be slow.
Looking after your medicine
Citanest DENTAL will be stored by your dentist under the recommended conditions.
It should be kept in a cool dry place where the temperature stays below 25°C. Protect from light.
When to discard your medicine
Any Citanest DENTAL from a single dose which is not used, will be disposed of in a safe manner by your dentist.
Do not use this medicine after the expiry date.
6. Are there any side effects?
Tell your doctor or nurse as soon as possible if you do not feel well while you are being given Citanest DENTAL. All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. Citanest DENTAL will help relieve pain in most people, but it may have unwanted side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these mild side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
If Citanest DENTAL is given wrongly, or you are very sensitive to it, it sometimes causes
| Call your doctor straight away, if you notice any of these serious side effects. You may need urgent medical attention or hospitalisation. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Citanest DENTAL contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Citanest DENTAL looks like
Citanest DENTAL is a clear, colourless, sterile solution for injection and is available as 2.2 mL dental cartridges in carton packs of 100 cartridges.
It contains the active ingredients: prilocaine hydrochloride 30 mg/mL and felypressin 0.54 microgram/mL.
Australian Registration Number: AUST R 220320
New Zealand Registration Number: TT50-1043
Who sponsors/distributes Citanest DENTAL
Australia
Dentsply Sirona Pty Ltd
11 – 21 Gilby Road
Mount Waverley, VIC 3149
Australia
Tel: 1300 552 929
New Zealand
Dentsply Sirona (NZ) Limited
c/o- Lowndes Jordan
Level 15, PWC Tower
188 Quay Street
Auckland 1010
Tel: 0800 336 877
® Trade Mark herein is the property of the AstraZeneca group
This leaflet was revised in December 2025 (Version 4).
Brand Information
| Brand name | 3% Citanest Dental with Octapressin |
| Active ingredient | Prilocaine hydrochloride + Felypressin |
| Schedule | S4 |
MIMS Revision Date: 01 February 2019
1 Name of Medicine
Prilocaine hydrochloride.
Felypressin.
2 Qualitative and Quantitative Composition
3% Citanest Dental with Octapressin is a clear, colourless sterile aqueous solution contains 30 mg/mL prilocaine hydrochloride and 0.54 micrograms/mL felypressin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
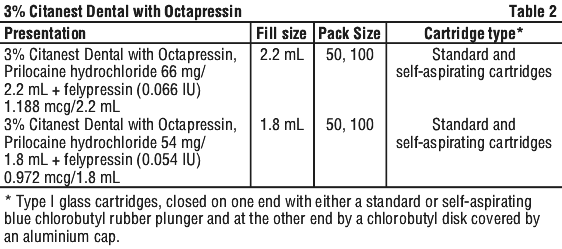
3% Citanest Dental with Octapressin injection solution in a standard and self-aspirating glass cartridge.
The finished product is a clear and colourless solution packed in a carton containing either 50 or 100 units of 1.8 mL and 2.2 mL Type I glass cartridges, closed on one end with a self-aspirating bromobutyl rubber plunger and at the other end by a bromobutyl disk covered by an aluminium cap.
4 Clinical Particulars
4.1 Therapeutic Indications
3% Citanest Dental with Octapressin is indicated for the production of local anaesthesia in routine dental procedures and oral surgery by means of infiltration and nerve block techniques.
4.2 Dose and Method of Administration
The lowest dosage that results in effective anaesthesia should be used. The dosage will also depend on the area of the oral cavity to be anaesthetised, the vascularity of the oral tissues and the technique of anaesthesia. See Table 1.

2. Safe dose. The safe dose for people with acute or chronic disease, especially those on medications, may be substantially less.
4.3 Contraindications
1) Allergy or hypersensitivity to amide type local anaesthetics or other components of the injection solution which may be present (see Section 6.5 Nature and Contents of Container, Note). Detection of suspected sensitivity by skin testing is of limited value.
2) Congenital or idiopathic methaemoglobinaemia.
3) Local anaesthetic techniques must not be used when there is inflammation and/or sepsis in the region of the proposed injection.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
4.4 Special Warnings and Precautions for Use
1) When any local anaesthetic agent is used, resuscitative equipment and medicines, including oxygen, should be immediately available in order to manage possible adverse effects involving the cardiovascular, respiratory or central nervous systems.
2) Injection should always be made slowly with frequent aspirations to avoid inadvertent intravascular injection which can produce toxic effects. Multiple injections should be administered at spaced intervals.
3) The safety and effectiveness of 3% Citanest Dental with Octapressin depends on proper dosage, correct technique, adequate precautions and readiness for emergencies. Standard textbooks should be consulted for specific techniques and precautions regarding various anaesthetic procedures.
4) Prilocaine should be given with great caution to patients with severe bradycardia, cardiac conduction disturbances or severe digitalis intoxication.
5) Prilocaine and/or its metabolites may accumulate during prolonged or repeated administration in patients with hepatic, renal or cardiac disease, the elderly and patients in poor general condition. However, this is unlikely to occur at the doses normally used in dentistry.
6) Prilocaine should be used with caution in patients with known medicine sensitivities. Patients allergic to ester derivatives of para-aminobenzoic acid (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to agents of the amide type.
7) Prilocaine should be used with caution in patients with genetic predisposition to malignant hyperthermia as the safety of amide local anaesthetic agents in these patients has not been fully established.
8) In patients with hypoxaemia there is the potential for further hypoxic embarrassment as large doses of prilocaine may produce methaemoglobinaemia (see Section 4.8 Adverse Effects (Undesirable Effects)).
9) Although largely free of side effects as an additive to prilocaine, felypressin may cause a rise in blood pressure or coronary constriction if an overdose is given.
10) The patient should be advised to exert caution to avoid inadvertent trauma to the lips, tongue, cheek mucosa or soft palate when these structures are anaesthetised. The ingestion of food should therefore be postponed until normal function returns.
11) In the head and neck area the intravascular injection of even small doses of local anaesthetics may cause systemic adverse effects similar to those seen after the inadvertent intravascular injection of larger doses in other areas.
12) Local anaesthetics should be administered with caution to patients with severe or untreated hypertension, severe heart disease, advanced diabetes, severe anaemia or circulatory failure from whatever cause or any other pathological condition. Local anaesthetics should be avoided when there is inflammation in the region of the proposed injection.
Use in the elderly. Even if the dose of 3% Citanest Dental with Octapressin used in dental practice is generally small, the elderly may require special attention to reduce the risk of dangerous side effect.
Paediatric use. For children, the dose may have to be reduced commensurate with bodyweight. The dosage should be calculated for each patient individually and modified in accordance with the dentist's experience and knowledge of the patient.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
1. Antiarrhythmic medicines. Local anaesthetics of the amide type should be used with caution in patients receiving anti arrhythmic medicines, e.g. mexiletine, since the toxic effects may be additive.
2. Methaemoglobinaemia. Medicines which may predispose to methaemoglobin formation, e.g. sulfonamides, antimalarials and certain nitric compounds, could potentiate this adverse effect of prilocaine.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Category A: Medicines which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed.
Although the safe use of prilocaine during pregnancy has not been established with respect to possible adverse effects upon foetal development, prilocaine has been used extensively for dental procedures during pregnancy with no reports of ill effects to mother or foetus. In large doses prilocaine may cause maternal and foetal methaemoglobinaemia which could lead to foetal hypoxia.
Use in lactation. Prilocaine may enter the mother's milk, but in such small amounts that there is generally no risk of this affecting the neonate. It is not known whether felypressin is excreted in breast milk.
4.7 Effects on Ability to Drive and Use Machines
Depending on the dose, local anaesthetics may have a very mild effect on mental function and may temporarily impair locomotion and coordination.
4.8 Adverse Effects (Undesirable Effects)
Reactions to prilocaine are rare in the doses used in dental procedures. If adverse effects occur, they are similar in character to those observed with other local anaesthetics of the amide type. Psychogenic reactions in anticipation of or during the dental procedures are, however, common and may mimic the symptoms of generalised systemic reaction to local anaesthetics.
Adverse effects may be due to high plasma levels as a result of excessive dosage, rapid absorption, delayed elimination or metabolism, or inadvertent intravascular injection. Such reactions are systemic in nature and involve the central nervous system and/or the cardiovascular system (see Section 4.9 Overdose). Pronounced acidosis or hypoxia may increase the risk and severity of toxic reactions.
The following adverse effects are rare when associated with dental use of prilocaine.
Nervousness, dizziness, blurred vision, tremor, drowsiness, tinnitus, numbness, disorientation, nausea, vomiting, headache, palpitations and tachycardia.
More serious reactions that reflect an overdosage of prilocaine are convulsions, unconsciousness, respiratory depression or arrest, hypotension, cardiovascular collapse and bradycardia which may lead to cardiac arrest.
Regarding methaemoglobinaemia, see below.
Allergy. Allergy to amide type local anaesthetics is very rare but may present as allergic dermatitis, bronchospasm or anaphylaxis.
Methaemoglobinaemia. Methaemoglobinaemia and cyanosis may occur following the administration of prilocaine solutions, particularly following a high dose. This is caused by the metabolite o-toluidine. Cyanosis of the nails and lips are clinical signs of methaemoglobinaemia.
In the dental dosage of prilocaine (1-5 mL 3% Citanest Dental with Octapressin, i.e. 30-150 mg prilocaine hydrochloride 3% with felypressin 0.03 IU/mL (0.54-2.7 microgram/mL) the occurrence of methaemoglobinaemia in dental practice appears remote. However, gross overdosage in dental practice has been reported to cause methaemoglobinaemia.
A dose response relationship appears to exist between the amount of prilocaine administered and the degree of methaemoglobin formation. In studies conducted in man, the incidence of methaemoglobinaemia at a dose of 400 mg prilocaine is not statistically significant. Cases of cyanosis at doses between 400 mg and 600 mg prilocaine are extremely rare.
At a dose of 600 mg prilocaine, methaemoglobin forms at levels less than 15% of the total haemoglobin content. This degree of methaemoglobinaemia is not associated with any adverse respiratory, cardiovascular or CNS symptoms. However, cyanosis has been reported at this dosage level.
If methaemoglobinaemia does occur, it may be treated by a single intravenous injection of a 1% methylene blue solution, given at a dose of 1 mg/kg bodyweight over a 5 minute period. This dose normally reverses methaemoglobinaemia within 15 minutes and should not be repeated as methylene blue in high concentrations acts as a haemoglobin oxidant.
In most patients receiving doses of 3% Citanest Dental with Octapressin within the recommended range, the appearance of clinical signs and symptoms of hypoxia may be due to cardiac or respiratory insufficiency and should be treated with oxygen and/or other appropriate measures. If cyanosis persists, methaemoglobinaemia should be suspected and methylene blue treatment initiated.
The reduction of the oxygen carrying capacity in normal patients is marginal; hence the cyanosis is usually symptomless. However, in severely anaemic patients it may cause significant hypoxaemia. It is important to rule out other more serious causes of cyanosis such as acute hypoxaemia and/or heart failure.
Even low concentrations of methaemoglobin may interfere with pulse oximetry readings, indicating a false low oxygen saturation.
Neurological reactions. The incidence of adverse neurological reactions directly caused by the use of local anaesthetics is very low.
Neurological reactions may be related to the total dose of the local anaesthetic administered and are also dependent upon the particular medicine used, the route of administration and the physical status of the patient. Many of these effects may be related to local anaesthetic techniques, with or without contribution from the medicine.
Neurological reactions following regional nerve blocks have included persistent numbness, paraesthesia and sensory disturbances.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia) and 0800 764 766 (New Zealand).
Systemic toxicity is initially manifested as CNS excitation and may be characterised by nervousness, dizziness, blurred vision and tremors followed by drowsiness, convulsions, unconsciousness and possibly respiratory arrest.
Toxic cardiovascular reactions to local anaesthetics are usually depressant in nature, may occur rapidly and with little warning and can lead to peripheral vasodilation, hypotension, myocardial depression, bradycardia and possibly cardiac arrest.
Treatment of overdosage. Treatment of a patient with toxic symptoms consists of ensuring a patent airway and supporting ventilation with oxygen and assisted or controlled respiration as required. This usually will be sufficient in the management of most reactions.
Further treatment depends on diagnosis. Medical assistance should be summoned.
If ventricular fibrillation or cardiac arrest occurs, effective cardiovascular resuscitation treatment must be instituted and maintained for a prolonged period if necessary.
For treatment of reactions caused by adrenaline, consult standard textbooks.
For treatment of methaemoglobinaemia, see Section 4.8 Adverse Effects (Undesirable Effects).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Prilocaine is a membrane stabilising agent and a local anaesthetic of the amide type. Prilocaine stabilises the neuronal membrane and reversibly prevents the initiation and conduction of nerve impulses thereby producing local anaesthesia.
Prilocaine has a similar time of onset and potency to lignocaine. Prilocaine has a lower toxicity than lignocaine.
The onset and duration of anaesthesia depend on the route of administration, status of the patient and the dosage (volume and concentration) employed. The addition of felypressin reduces the rate of absorption of prilocaine from the site of injection thereby increasing its duration of action.
Felypressin is a synthetic hormone with similar properties to vasopressin. In contrast to adrenaline, felypressin does not produce ischaemia distal to or at the injection site. 3% Citanest Dental with Octapressin is therefore indicated for routine use. It is particularly suitable for use in patients for whom the use of solutions containing sympathomimetic agents is contraindicated.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
3% Citanest Dental with Octapressin contains the excipients sodium chloride and water for injections and may also contain sodium hydroxide and/or hydrochloric acid for pH adjustment to 3.5-5.2.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
3% Citanest Dental with Octapressin should be stored below 25°C. Protect from light.
Local anaesthetics react with certain metals and cause the release of their respective ions which, if injected, may cause severe local irritation. Adequate precautions should be taken to avoid prolonged contact between 3% Citanest Dental with Octapressin solutions and metal surfaces, e.g. cartridges should not be preloaded and connected to needles until just prior to use.
Cartridges showing discolouration or cracks should be discarded. 3% Citanest Dental with Octapressin solutions should not be autoclaved. Surface sterilisation using pure, undiluted isopropyl alcohol (91%) or 70% ethyl alcohol (USP) may be carried out if desired.
6.5 Nature and Contents of Container
See Table 2.
Note. 1. All Dentsply Sirona dental cartridges are paraben free and for single use in a single patient only. Remaining unused contents should be discarded.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
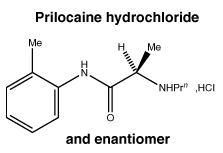
Prilocaine hydrochloride. The chemical name of prilocaine hydrochloride is 2-(propylamino)-o-propionotoluidide hydrochloride; and it is also known as propitocaine hydrochloride. It has a molecular weight of 256.8.
Chemical structure.
Felypressin. The chemical name of the vasoconstrictor octapressin is 2-(phenylalanine)-8-lysine vasopressin; also known as felypressin. It has a molecular weight of 1040.2.
Chemical structure.
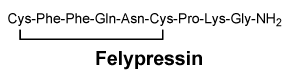
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
28 February 2008
Date of Revision
15 November 2018
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.