Abrysvo
Brand Information
| Brand name | Abrysvo |
| Active ingredient | Respiratory syncytial virus pre-fusion F protein vaccine, recombinant |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Abrysvo.
Summary CMI
ABRYSVO®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This vaccine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I being given ABRYSVO?
ABRYSVO is a vaccine given to prevent disease of the respiratory tract (airways and lungs) caused by a virus called respiratory syncytial virus (RSV). ABRYSVO is given to pregnant individuals to protect their infants from birth through 6 months of age, or individuals 60 years of age or older.
ABRYSVO contains the active ingredient recombinant respiratory syncytial virus pre-fusion F protein.
For more information, see Section 1. Why am I being given ABRYSVO? in the full CMI.
2. What should I know before I am given ABRYSVO?
You should not be given ABRYSVO if you have ever had an allergic reaction to ABRYSVO or any of the ingredients listed at the end of the CMI. Tell your healthcare professional if you have or have had an allergic reaction after being given any other vaccine in the past, bleeding problems, have an infection with a high fever, anxiety or fainting following any needle injections or a weakened immune system.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given ABRYSVO? in the full CMI.
3. What if I am taking other medicines?
Tell your healthcare professional if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your healthcare professional if you have recently been given or plan to be given any other vaccine.
For more information, see Section 3. What if I am taking other medicines? in the full CMI.
4. How is ABRYSVO given?
- ABRYSVO will be given as a single dose injection into the muscle of your upper arm by a doctor, nurse or other trained healthcare professional.
More instructions can be found in Section 4. How is ABRYSVO given? in the full CMI.
5. What should I know after being given ABRYSVO?
| Things you should do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know after being given ABRYSVO? in the full CMI.
6. Are there any side effects?
- Side effects of ABRYSVO include: pain, swelling or redness in the area where the vaccine was given, muscle pain and headaches. Rare serious side effects include allergic reactions and Guillain-Barré syndrome, an autoimmune syndrome resulting in tingling or weakness, usually starting in the legs then spreading up your body.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I being given ABRYSVO?
ABRYSVO is a vaccine, which is a type of medicine that helps to protect (immunise) people from certain infectious diseases. It does this by preparing the body's defences to fight the infection before you catch the bacteria or virus.
ABRYSVO is used to prevent disease of the respiratory tract (lungs) caused by a virus called respiratory syncytial virus (RSV).
ABRYSVO contains a protein (antigen) from each of the two subgroups (A and B) of RSV.
RSV is a common virus which, in most cases, causes mild, cold-like symptoms such as a sore throat, cough or a blocked nose. However, some individuals are at higher risk of respiratory disease caused by RSV. This includes young infants, older adults and those with chronic medical conditions such as heart or lung disease. In infants RSV can cause serious lung problems such as bronchiolitis and pneumonia. In older adults it can make existing conditions such as chronic obstructive pulmonary disease (COPD) and congestive heart failure (CHF) worse. It can lead to hospitalisation in severe cases, and in some cases it can be fatal.
ABRYSVO is given to:
- pregnant individuals to protect their infants from birth through 6 months of age, or
- individuals 60 years of age or older.
As with any vaccine, ABRYSVO may not fully protect all those who receive it.
2. What should I know before I am given ABRYSVO?
Warnings
Do not use ABRYSVO if:
- you are allergic to ABRYSVO or any of the ingredients listed at the end of this leaflet
Check with your doctor if you:
- have any other medical conditions
- take any medicines for any other condition
- have an infection with a high fever
If this is the case, then vaccination will be postponed. There is no need to delay vaccination for a minor infection, such as a cold, but talk to your doctor first. - have any bleeding problems or disorders, or bruise easily
- are feeling nervous about the vaccination process or have ever fainted following any needle injection
- have a weakened immune system which may prevent you from getting the full benefit from ABRYSVO
- are less than 24 weeks pregnant
- are less than 18 years of age
During vaccination, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before you receive this vaccine.
Pregnant individuals can be given this vaccine in the late second or third trimester (weeks 24 to 36).
It is unknown whether ABRYSVO is excreted into breast milk. No side effects have been shown in breastfed newborns of vaccinated mothers.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your healthcare professional if you have recently been given or plan to be given any other vaccine.
4. How is ABRYSVO given?
How many injections will be given
For the vaccination of pregnant individuals to protect their infants
- One single dose is given in the late second or third trimester of pregnancy (24 - 36 weeks).
For the vaccination of adults aged 60 years or over
- One single dose is given.
Multiple vaccinations
If ABRYSVO is given at the same time as another vaccine, a different injection site will be used for each injection.
If you are given too much ABRYSVO
Overdose is unlikely as ABRYSVO is available in a single dose presentation and will be given by a trained healthcare professional.
5. What should I know after being given ABRYSVO?
Things you should do
Check with your healthcare professional before you receive any other vaccines or take other medicines.
Driving or using machines
ABRYSVO is unlikely to affect your ability to drive or use machines.
Looking after your medicine
- ABRYSVO will normally be stored at the doctor's surgery or in the pharmacy.
- If you need to store ABRYSVO, store the carton in a refrigerator (2°C to 8°C) in the original package.
- Do not freeze. Discard if the carton has been frozen.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
Your healthcare professional will dispose of any unused vaccine.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your healthcare professional if you have any further questions about side effects.
Side effects
| Side effects | What to do |
Injection site reactions:
| Speak to your doctor if you have any of these side effects and they worry you. |
Allergic reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your healthcare professional if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/safety/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What ABRYSVO contains
| Active ingredients (main ingredients) | Powder: RSV subgroup A stabilised pre-fusion F protein RSV subgroup B stabilised pre-fusion F protein |
| Other ingredients (inactive ingredients) | Powder: trometamol trometamol hydrochloride sucrose mannitol polysorbate 80 sodium chloride Diluent: water for injections |
| Potential allergens | n/a |
Do not use this medicine if you are allergic to any of these ingredients.
ABRYSVO does not contain lactose, gluten, tartrazine or any other azo dyes.
What ABRYSVO looks like
ABRYSVO is supplied as:
- a white powder in a glass vial, plus
- a diluent in a pre-filled syringe to dissolve the powder or a diluent in a vial
After dissolving the powder in the diluent, the solution is clear and colourless.
ABRYSVO is supplied in cartons containing:
- 1 vial of powder for injection, 1 pre-filled syringe of diluent and 1 vial adapter (AUST R 406624)
- 10 vials of powder for injection and 10 vials of diluent (AUST R 517945)
Who distributes ABRYSVO
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in February 2026.
Brand Information
| Brand name | Abrysvo |
| Active ingredient | Respiratory syncytial virus pre-fusion F protein vaccine, recombinant |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Recombinant respiratory syncytial virus pre-fusion F protein.
Water for injections diluent.
2 Qualitative and Quantitative Composition
After reconstitution, 1 dose (0.5 mL) contains:
Respiratory syncytial virus (RSV) subgroup A stabilised prefusion F protein1 60 micrograms.
Respiratory syncytial virus (RSV) subgroup B stabilised prefusion F protein1 60 micrograms.
1 Produced in Chinese Hamster Ovary cells by recombinant DNA technology.
Excipient(s) with known effect. After reconstitution, 1 dose (0.5 mL) contains 11.3 milligrams of sucrose, 22.5 milligrams of mannitol and 0.43 milligrams of elemental sodium (as chloride).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Powder and diluent for solution for injection.
The powder or cake is white.
The diluent is a clear, colourless liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
Abrysvo is indicated for:
Active immunisation of pregnant women between 24-36 weeks of gestation for prevention of lower respiratory tract disease caused by respiratory syncytial virus (RSV) in infants from birth through 6 months of age.
Active immunisation of individuals 60 years of age and above for prevention of lower respiratory tract disease caused by respiratory syncytial virus (RSV).
Abrysvo should be used in accordance with official recommendations.
4.2 Dose and Method of Administration
Dosage. Pregnant women. Abrysvo is administered as a single dose (0.5 mL) in late second or third trimester of pregnancy (24-36 weeks of gestation).
Revaccination in subsequent pregnancies has not been studied.
Individuals 60 years of age and older. Abrysvo is administered as a single dose (0.5 mL).
The need for revaccination has not been established.
Paediatric population. The safety and efficacy of Abrysvo in children (from birth to less than 18 years of age) via active immunisation have not yet been established.
Method of administration. Abrysvo is for intramuscular injection only, preferably in the deltoid region of the upper arm.
Abrysvo is not to be administered intravascularly, intradermally or subcutaneously.
Do not mix Abrysvo with other vaccines/medicinal products in the same syringe.
Abrysvo is for single use in one patient only. Discard any residue.
Preparation for administration. For use of the powder for injection vial, diluent pre-filled syringe and vial adapter. The lyophilised vaccine (powder) must be reconstituted only with the diluent provided using the vial adapter to form Abrysvo.
Step 1. Attach vial adapter. Peel off the top cover from the vial adapter packaging and remove the flip off cap from the vial.
While keeping the vial adapter in its packaging, centre over the vial's stopper and connect with a straight downward push. Do not push the vial adapter in at an angle as it may result in leaking. Remove the packaging.
Step 2. Reconstitute lyophilised vaccine component to form Abrysvo. For all syringe assembly steps, hold the syringe only by the Luer lock adapter. This will prevent the Luer lock adapter from detaching during use.
Twist to remove the syringe cap, then twist to connect the syringe to the vial adapter. Stop turning when you feel resistance.
Inject the entire contents of the syringe into the vial. Hold the plunger rod down and gently swirl the vial until the powder is completely dissolved. Do not shake.
Step 3. Withdraw reconstituted vaccine. Invert the vial completely and slowly withdraw the entire contents into the syringe to ensure a 0.5 mL dose of Abrysvo.
Twist to disconnect the syringe from the vial adapter.
Attach a sterile needle suitable for intramuscular injection.
The prepared vaccine is a clear and colourless solution. Visually inspect the vaccine for large particulate matter and discolouration prior to administration. Do not use if large particulate matter or discolouration is found.
For use of the powder for injection vial and diluent vial. The lyophilised vaccine (powder) must be reconstituted only with the vial of diluent provided to form Abrysvo.
1. Using a sterile needle and sterile syringe, withdraw the entire contents of the vial containing the diluent.
2. Inject the entire contents of the syringe into the vial containing the powder. Gently swirl the vial in a circular motion until the powder is completely dissolved. Do not shake. Withdraw 0.5 mL from the vial containing the reconstituted vaccine.
The prepared vaccine is a clear and colourless solution. Visually inspect the vaccine for large particulate matter and discolouration prior to administration. Do not use if large particulate matter or discolouration is found.
4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients listed in Section 6.1.
4.4 Special Warnings and Precautions for Use
Guillain-Barré syndrome. Guillain-Barré syndrome has been reported rarely following vaccination with Abrysvo in individuals ≥ 60 years of age.
Healthcare professionals should be alert to signs and symptoms of Guillain-Barré syndrome to ensure correct diagnosis, in order to initiate adequate supportive care and treatment, and to rule out other causes.
Hypersensitivity and anaphylaxis. Medical treatment and supervision should always be readily available in case of a rare anaphylactic event following the administration of the vaccine.
Thrombocytopenia and coagulation disorders. Abrysvo should be given with caution to individuals with thrombocytopenia or any coagulation disorder since bleeding may occur following an intramuscular administration to these individuals.
Concurrent illness. Vaccination with Abrysvo should be postponed in individuals suffering from an acute febrile illness. However, the presence of a minor infection, such as a cold, should not result in the deferral of vaccination.
Anxiety-related reactions. Anxiety-related reactions, including vasovagal reactions (syncope), hyperventilation or stress-related reactions may occur in association with vaccination as a psychogenic response to the needle injection. It is important that procedures are in place to avoid injury from fainting.
Immunocompromised individuals. There are no data on the use of Abrysvo in immunocompromised individuals. Immunocompromised individuals, including individuals receiving immunosuppressant therapy, may have a diminished immune response to Abrysvo.
Individuals less than 24 weeks of gestation. Abrysvo has not been studied in pregnant individuals less than 24 weeks of gestation.
Limitations of vaccine effectiveness. As with any vaccine, vaccination with Abrysvo may not protect all vaccine recipients.
Excipients. This medicinal product contains less than 1 mmol sodium (23 mg) per dose, that is to say essentially 'sodium-free'.
Use in the elderly. See Section 4.8 Adverse Effects (Undesirable Effects).
Paediatric use. Abrysvo is not for active immunisation in children.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Use with other vaccines. Different injectable vaccines should always be given at different vaccination sites.
Do not mix Abrysvo with other vaccines/medicinal products in the same syringe. See Section 6.2 Incompatibilities.
Data on concomitant administration of Abrysvo and vaccines other than those listed below are not available.
For further guidance on vaccine interactions, please consult the most recent edition of the Australian Immunisation Handbook.
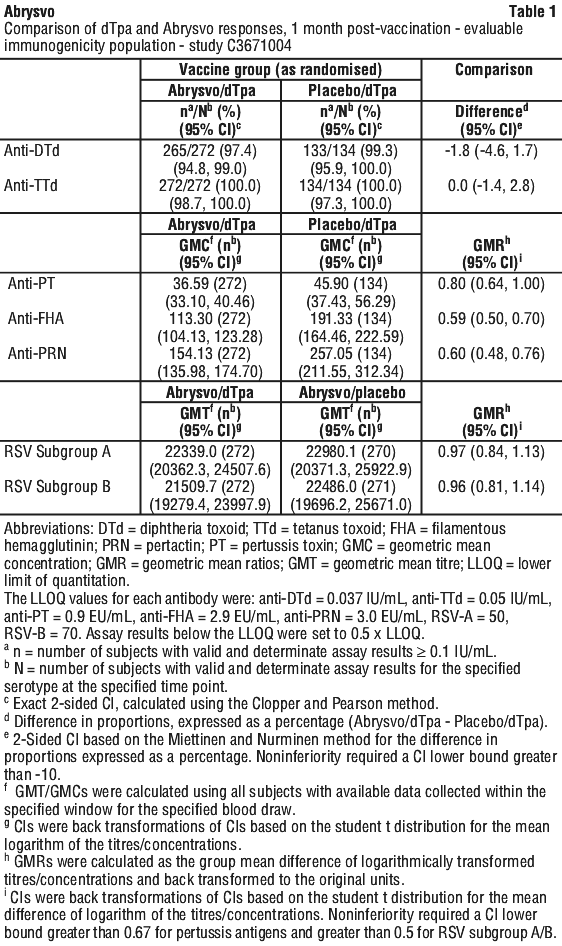
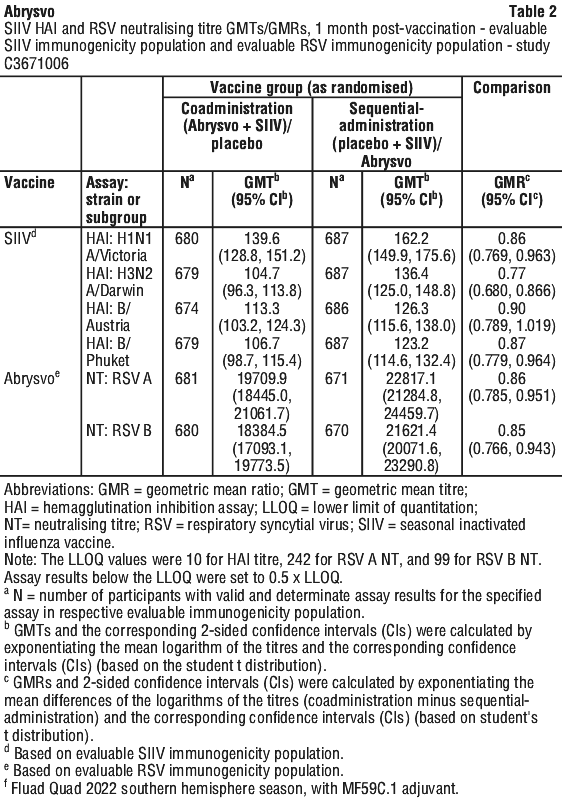
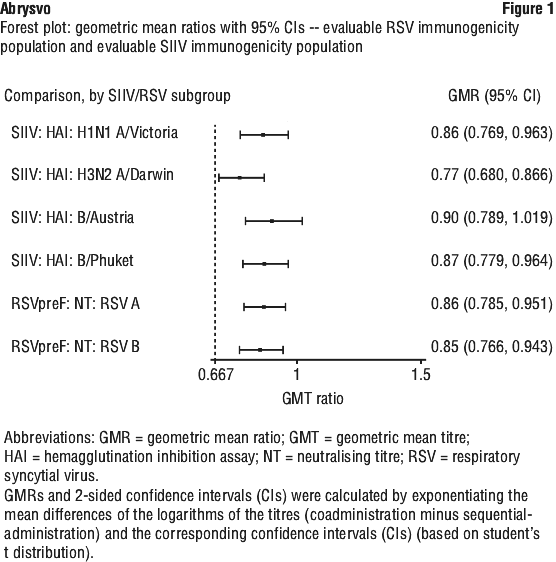
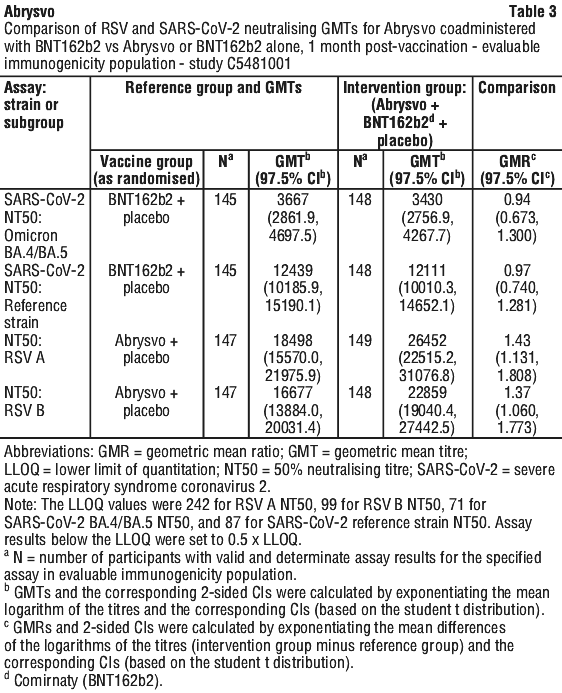
Use with tetanus, diphtheria and acellular pertussis vaccine (dTpa). Immunogenicity data in healthy non-pregnant women who received concomitant administration of Abrysvo and dTpa indicated the immune response induced by Abrysvo when administered concomitantly with dTpa was non-inferior to the immune response induced by Abrysvo alone. In addition, immunogenicity data indicated non-inferiority in immune response to the diphtheria and tetanus components. Immune response to the pertussis component of dTpa was lower when Abrysvo and dTpa were administered concomitantly as compared to dTpa administered alone. The clinical relevance of this observation is unknown. No safety concerns were identified in this concomitant administration study. See Table 1.
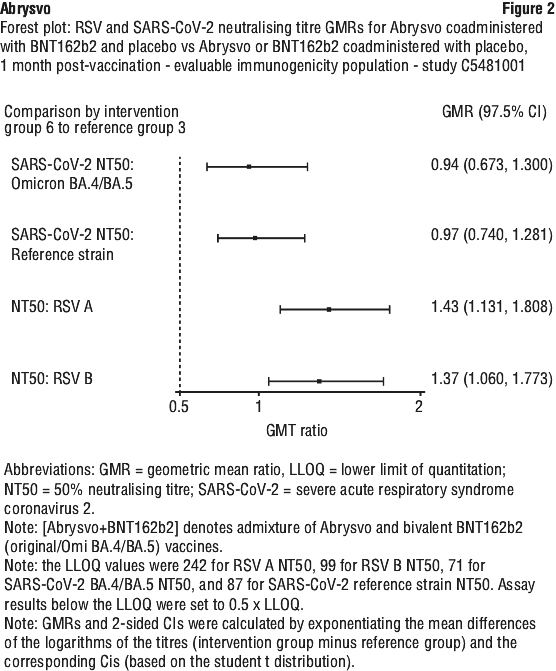

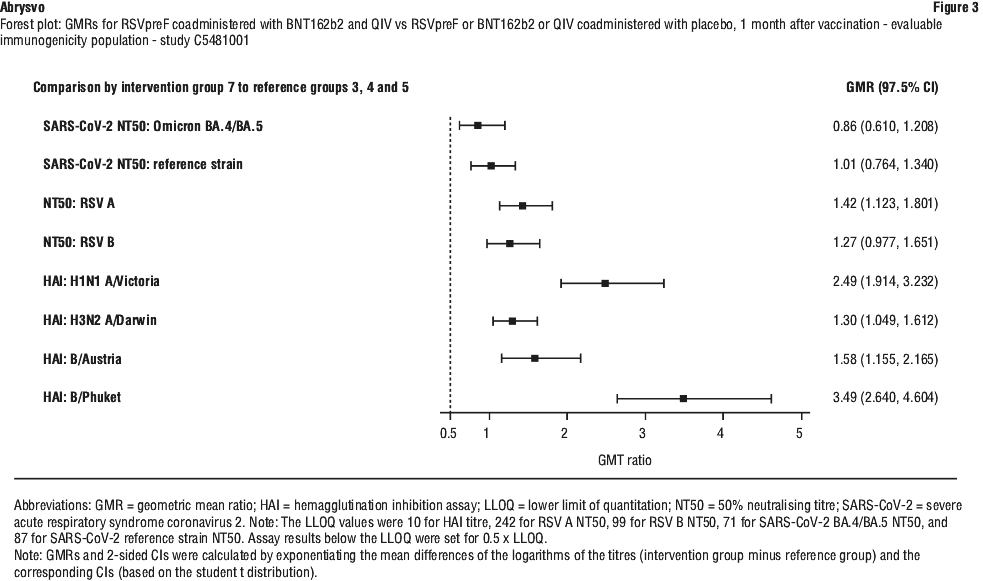
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No human data on the effect of Abrysvo on fertility are available.
A fertility and embryofetal and postnatal development study was conducted in female New Zealand white rabbits. The rabbits were administered IM 4 doses (twice prior to mating and twice during gestation) of a bivalent vaccine formulation containing F glycoproteins of RSV A and RSV B strains (120 micrograms of each preF protein). There were no effects on female fertility or embryofetal or postnatal development. Effects on male fertility have not been evaluated.
Use in pregnancy. (Category A)
A large amount of data on pregnant women (more than 4,000 exposed outcomes) indicate no malformative nor feto/neonatal toxicity.
No effects on embryofetal development were observed in rabbits (see Effects on fertility section above for details).
See Section 5.1 Pharmacodynamic Properties, Clinical trials.
Use in lactation. It is unknown whether Abrysvo is excreted in human milk. No adverse effects of Abrysvo have been shown in breastfed newborns of vaccinated mothers.
4.7 Effects on Ability to Drive and Use Machines
Abrysvo has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. The safety of Abrysvo was evaluated in 4,160 pregnant individuals ≤ 49 years of age in two clinical trials (one Phase 3 and one Phase 2b). It was also evaluated in 18,574 participants 60 years of age and older in a Phase 3 clinical trial.
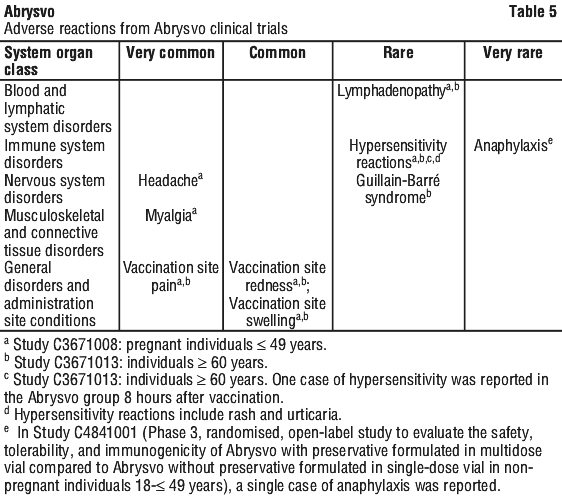
Tabulated list of adverse drug reactions. The following adverse reactions have been observed during clinical studies.
Adverse reactions are listed by system organ class, in decreasing order of seriousness.
The frequency is defined as follows: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000); not known (cannot be estimated from available data). See Table 5.
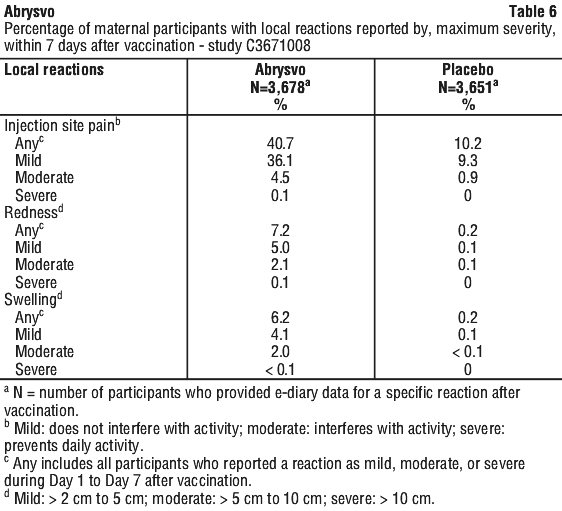
A Phase 2b study (C3671003) in pregnant women and their infants investigated the safety, tolerability and immunogenicity of two dose levels (120 micrograms and 240 micrograms), formulated with or without Al(OH)3) of Abrysvo. In this study, Abrysvo (120 micrograms, without Al(OH)3) was administered to 115 maternal participants and 114 infants were born to the maternal participants.
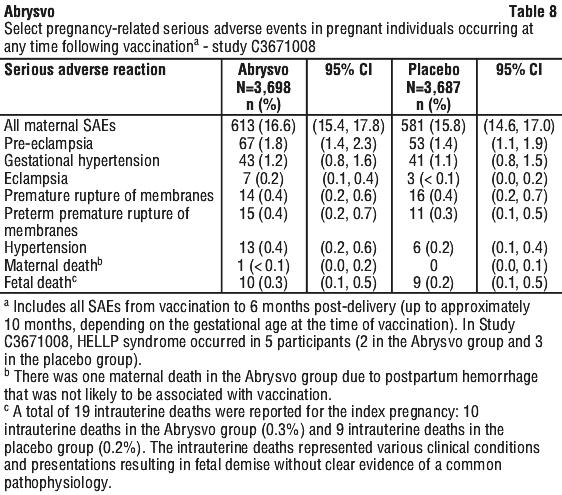
For all maternal participants, solicited local reactions and systemic events were collected for 7 days after vaccination (see Table 6 and Table 7), adverse events for 1 month and obstetric complications, serious adverse events (see Table 8) and adverse events of special interest for the duration of the study. For infant participants, the collection period for nonserious adverse events was from birth to 1 month. Serious adverse events were monitored for at least 1 year for all infant participants and for up to 2 years for half of the infants in Study C3671008.
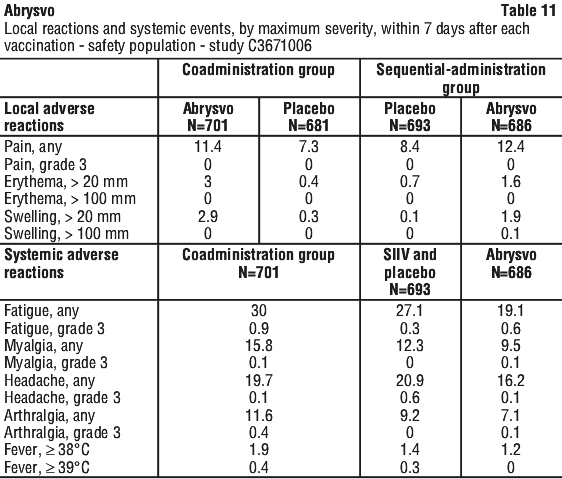
The most frequently reported adverse reactions in Study C3671008 were vaccination site pain, headache and myalgia.
The majority of solicited local and systemic reactions in maternal participants were mild to moderate in severity and resolved within 2-3 days of onset.
Solicited local and systemic reaction rates were similar among participants who received Abrysvo in Study C3671003.
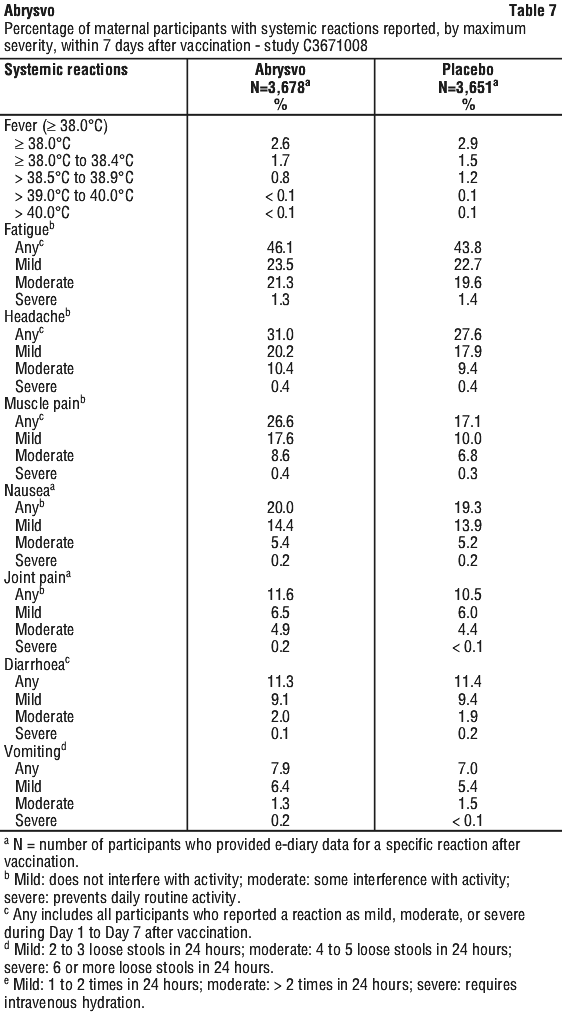
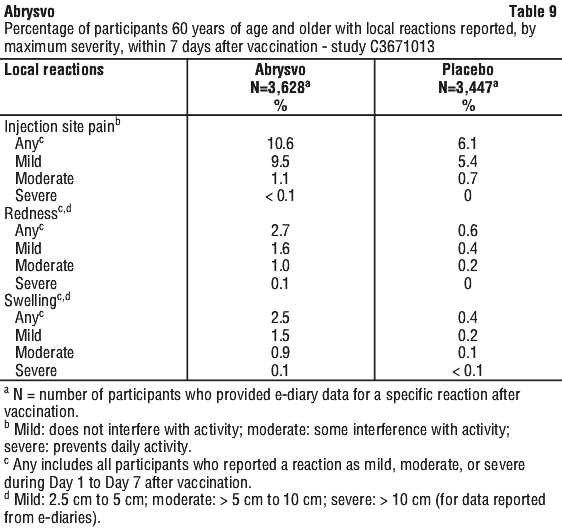
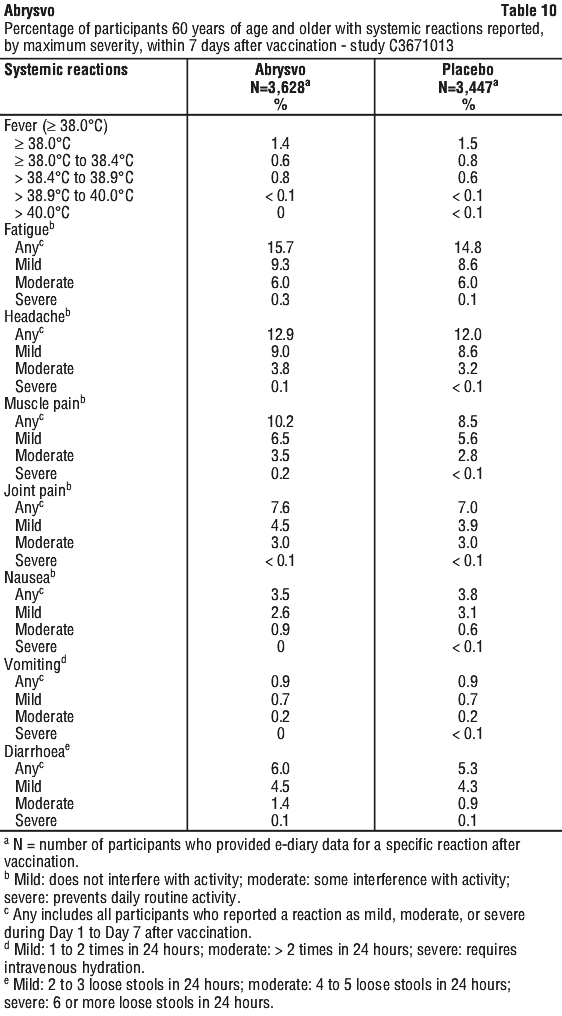
The most frequently reported adverse reaction in Study C3671013 was vaccination site pain. The majority of solicited local and systemic reactions were mild to moderate in severity and resolved within 1-2 days of onset.
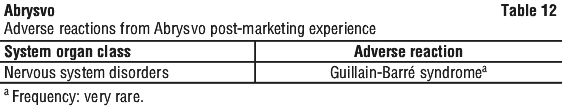
4.9 Overdose
Overdose with Abrysvo is unlikely due to its single dose presentation.
There is no specific treatment for an overdose with Abrysvo. In the event of an overdose, the individual should be monitored and provided with symptomatic treatment as appropriate.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Abrysvo is a bivalent formulation containing two recombinant stabilised RSV prefusion F antigens, each representing the two major virus subgroups and based on the genotype of major circulating strains, RSV A (Ontario) and RSV B (Buenos Aires). RSV F can exist in two antigenically distinct forms - prefusion and postfusion. Unlike postfusion F, prefusion F is the active form of the protein and is capable of mediating fusion of virus and host cell membranes during cell entry. Therefore, prefusion F is the primary target of the most potent neutralising antibodies that block RSV infection. Higher serum neutralising antibodies are associated with reduced risk of disease. Following intramuscular administration, the prefusion F antigens elicit an immune response, which protects against RSV-associated lower respiratory tract disease.
In pregnant individuals, the action of neutralising antibodies conferring protection is mediated through passive transfer of these antibodies from mother to infant. Adults 60 years of age and older are protected by active immunisation.
Clinical trials. Infants from birth through 6 months of age by active immunisation of pregnant individuals. Study C3671008 was a Phase 3, multicentre, randomised, double-blind, placebo-controlled study to assess the efficacy, safety and immunogenicity of Abrysvo in the prevention of RSV-associated medically attended lower respiratory tract disease in infants born to healthy individuals vaccinated during pregnancy, and safety and immunogenicity in pregnant individuals. This was a global study, including study sites in both the northern and southern hemispheres, and spanned multiple RSV seasons. Maternal participants received a single dose of Abrysvo or placebo (1:1 ratio) in the late second or third trimester of pregnancy. The dose of RSV prefusion F antigen in Abrysvo was 120 micrograms (60 micrograms A and 60 micrograms B). Revaccination for subsequent pregnancies has not been studied.
RSV-associated lower respiratory tract disease was defined as a medically attended visit with a reverse transcription-polymerase chain reaction (RT-PCR) confirmed RSV disease with one or more of the following respiratory symptoms: fast breathing, low oxygen saturation (SpO2 < 95%) and chest wall indrawing. RSV-associated severe lower respiratory tract disease was a subset defined as meeting the lower respiratory tract disease - RSV criteria plus at least one of the following: very fast breathing, low oxygen saturation (SpO2 < 93%), high-flow nasal cannula or mechanical ventilation, ICU admission for > 4 hours and/or failure to respond/unconscious.
The study enrolled healthy individuals ≤ 49 years of age who were between 24 and 36 weeks of gestation, with uncomplicated, singleton pregnancies. In the study, 3,711 maternal participants were randomised to the Abrysvo group and 3,709 to the placebo group. Maternal participants with certain high risk pregnancies were excluded from the study (BMI > 40 kg/m2 prior to pregnancy, pregnancies resulting after in vitro fertilisation, preeclampsia, eclampsia, or uncontrolled gestational hypertension, placental abnormalities, polyhydramnios or oligohydramnios, significant bleeding or blood clotting disorder, unstable endocrine disorders, including untreated hyperthyroidism, untreated hypothyroidism or untreated disorders of glucose intolerance).
The study objective was assessment of vaccine efficacy (VE), defined as the relative risk reduction of the endpoint in the Abrysvo group compared to the placebo group, for infant participants born to maternal participants receiving 1 dose of Abrysvo. At the primary analysis, there were two primary efficacy endpoints, assessed in parallel, severe RSV-positive medically attended lower respiratory tract disease and RSV-positive medically attended lower respiratory tract disease, occurring within 90/120/150/180 days after birth. Other efficacy endpoints included medically attended lower respiratory tract disease due to RSV in infants up to 360 days after birth, hospitalisation due to RSV up to 360 days after birth and medically attended respiratory tract disease due to RSV in infants occurring within 90/120/150/180 days after birth.
Demographic characteristics in Study C3671008 were generally similar with regard to age, race and ethnicity among participants who received Abrysvo and those who received placebo. Of the participants who received Abrysvo, 65% were White, 20% were Black or African American and 29% were Hispanic/Latino. The median age of participants was 29 years (range 16-45 years). There were 15 pregnant adolescents enrolled in the study (8 in the Abrysvo group and 7 in the placebo group). The median gestational age at vaccination was 31 weeks and 2 days. The median infant gestational age at birth was 39 weeks and 1 day (range 27 weeks and 3 days to 43 weeks and 6 days). Among the infants born to maternal participants 51% were male and 49% were female.
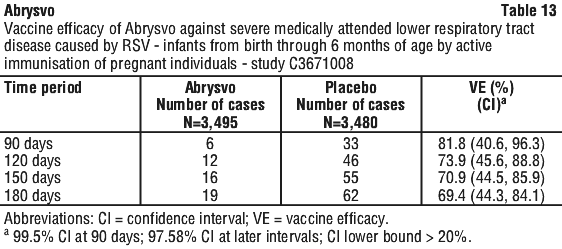
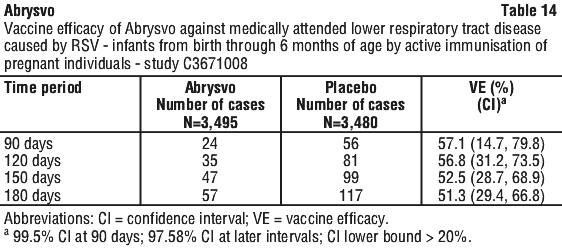
The VE results met the statistical criterion for success (a CI lower bound > 20%) for reducing severe medically attended lower respiratory tract disease due to RSV, at all timepoints through 180 days. The VE results did not meet the statistical criterion for success (a CI lower bound > 20%) for reducing medically attended lower respiratory tract disease due to RSV at 90 days; however, clinically meaningful efficacy was observed from 90 days through 180 days after birth.
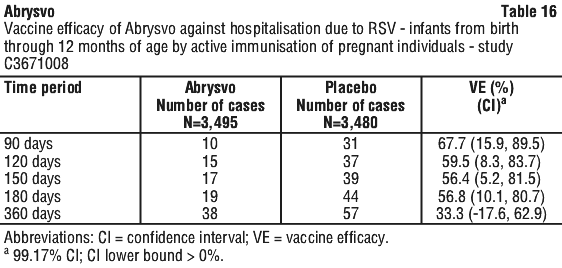
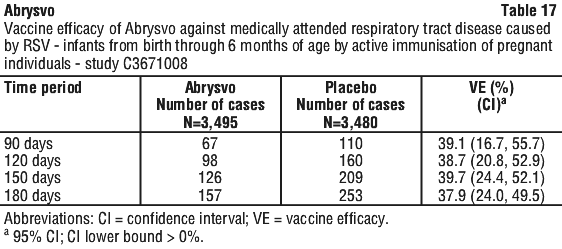
The VE results met the statistical criterion for success (a CI lower bound > 0%) for RSV-positive medically attended lower respiratory tract disease in infants, at all timepoints within 210 to 360 days after birth and for hospitalisation due to RSV in infants at all timepoints through 180 days; the statistical criterion for success was not met for hospitalisation at 360 days. VE was observed for RSV-positive medically attended respiratory tract disease, at all timepoints through 180 days after birth.
Vaccine efficacy information is presented in Table 13, Table 14, Table 15, Table 16 and Table 17.
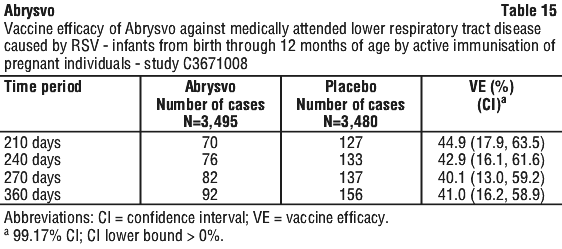
Maternal vaccination with Abrysvo yielded high RSV A- and B-neutralising titres in infants. Combined RSV A/B neutralising GMTs at birth and through 6 months of life were higher in infants whose mothers had received Abrysvo compared to infants whose mothers had received placebo.
In the pivotal maternal vaccine efficacy study (C3671008) a pertussis containing vaccine was allowed up to 14 days before and from 14 days after the administration of Abrysvo.
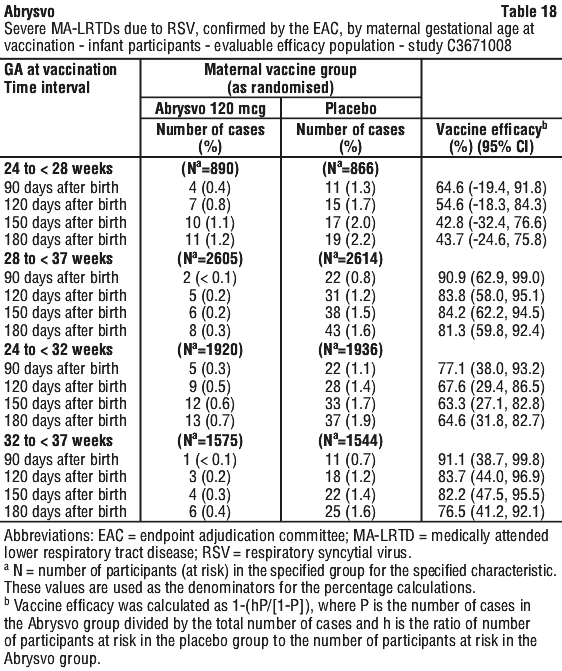
Vaccine efficacy by timing of vaccination according to weeks of gestation. A post-hoc analysis of VE by maternal gestational age was conducted. See Table 18.
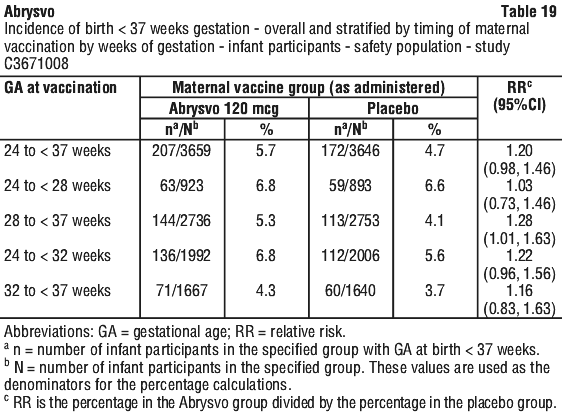
Other birth outcomes in the pivotal maternal study. The infant safety population included 3,659 and 3,646 infants born to individuals in the Abrysvo or placebo group, respectively. There were 10 (0.3%) fetal deaths in the Abrysvo group and 9 (0.2%) in the placebo group. Among the infants born to individuals in the Abrysvo group and in the placebo group, 207 (5.7%) and 172 (4.7%), respectively, were born preterm (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1, Clinical trials). Low birth weight was observed in 5.1% of participants in the Abrysvo group versus 4.3% in the placebo group, and neonatal jaundice was observed in 7.3% in the Abrysvo group versus 6.9% in the placebo group (see Section 4.8 Adverse Effects (Undesirable Effects)). For mortality in the neonatal period among infants born to pregnant individuals in Study C3671008, there were 3 deaths in the Abrysvo group and 5 in the placebo group, and for overall mortality including after the neonatal period there were 8 deaths in the Abrysvo group and 14 in the placebo group. Congenital abnormalities were reported in 5.6% in the Abrysvo group and 6.7% in the placebo group. In infants born preterm, 84 infants in the Abrysvo group and 81 infants in the placebo group remained hospitalised or were readmitted to the hospital in the neonatal period (up to 30 days after birth). Available data are insufficient to establish a causal relationship between preterm birth and Abrysvo.
Individuals 60 years of age and older by active immunisation. Study C3671013 was a Phase 3, multicentre, randomised, double-blind, placebo-controlled study to assess the efficacy, immunogenicity and safety of Abrysvo in the prevention of RSV-associated lower respiratory tract disease in individuals 60 years of age and older during the first RSV season and the long-term efficacy and immunogenicity of Abrysvo across two RSV seasons. The need for revaccination with a subsequent dose of Abrysvo has not been established.
RSV-associated acute respiratory tract disease was defined as RT-PCR confirmed RSV disease with one or more of the following respiratory symptoms within 7 days of symptom onset and lasting more than 1 day during the same disease - new or increased sore throat, nasal congestion, nasal discharge, cough, wheezing, sputum production or shortness of breath.
RSV-associated lower respiratory tract disease was defined as RT-PCR confirmed RSV disease with two or more, or three or more, of the following respiratory symptoms within 7 days of symptom onset and lasting more than 1 day during the same illness - new or increased cough, wheezing, sputum production, shortness of breath or tachypnea (≥ 25 breaths/min or 15% increase from resting baseline). RSV-associated severe lower respiratory tract disease was defined as meeting the lower respiratory tract disease-RSV criteria plus at least one of the following - hospitalisation due to RSV-associated lower respiratory tract disease, new or increased oxygen supplementation or mechanical ventilation including Continuous Positive Airway Pressure (CPAP).
The dose level of RSV prefusion F antigen in Abrysvo for this study was 120 micrograms (60 micrograms A and 60 micrograms B). Participants were randomised (1:1) to receive Abrysvo (n=18,487) or placebo (n=18,479). Enrolment was stratified by age, 60-69 years (63%), 70-79 years (32%) and ≥ 80 years (6%). Healthy adults and adults with stable chronic diseases were included. Participants (16%) were enrolled with stable chronic cardiopulmonary conditions such as chronic obstructive pulmonary disease (COPD), asthma or congestive heart failure (CHF).
The primary objective was assessment of vaccine efficacy (VE), defined as the relative risk reduction of first episode of RSV-associated lower respiratory tract disease in the Abrysvo group compared to the placebo group in the first RSV season. Secondary objectives were assessment of VE, defined as the relative risk reduction of first episode of RSV-associated severe lower respiratory tract disease and acute respiratory disease in the Abrysvo group compared to the placebo group in the first RSV season. Other efficacy endpoints include the efficacy of Abrysvo in preventing RSV-associated lower respiratory tract disease and acute respiratory disease across two RSV seasons following vaccination.
Demographic characteristics in Study C3671013 were generally similar with regard to age, gender, race and ethnicity among participants who received Abrysvo and those who received placebo. Of the participants who received Abrysvo, 51% were male and 80% were White, 12% were Black or African American and 42% were Hispanic/Latino. The median age of participants was 67 years (range 59-95 years). No overall differences in the safety or effectiveness of Abrysvo were observed between age groups.
The study met the primary objectives of reduction of lower respiratory tract disease RSV cases with ≥ 2 symptoms and lower respiratory tract disease RSV cases with ≥ 3 symptoms.
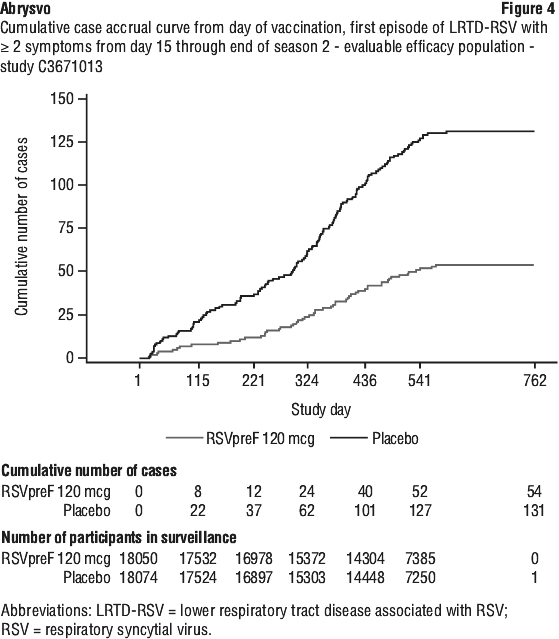
Vaccine efficacy information at the end of the first and second RSV seasons, and combined across the two RSV seasons is presented in Table 20, Table 21, Figure 4 and Figure 5. The average surveillance duration was 16.36 months. Vaccine efficacy is maintained through two RSV seasons.
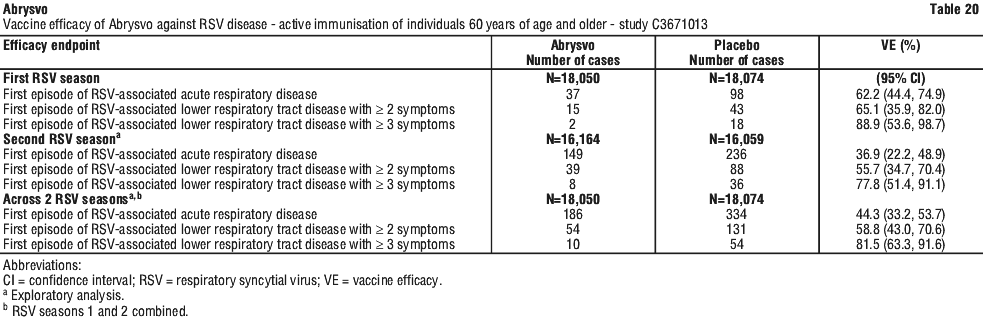
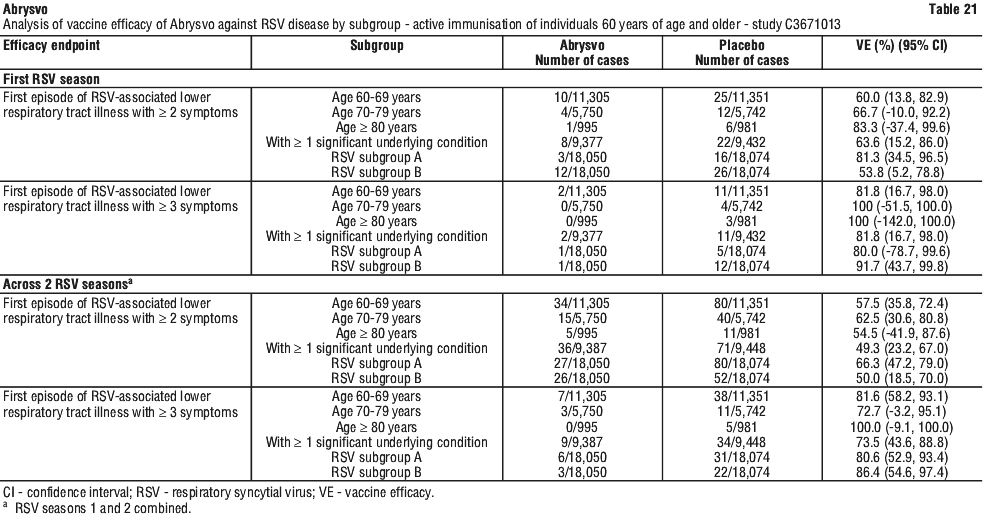
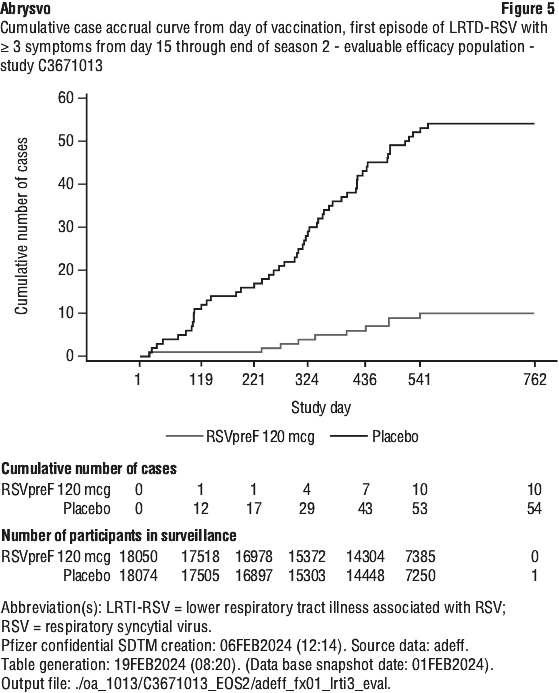
Immunogenicity in individuals 60 years of age and older - study C3671013. First RSV season. Immunogenicity data demonstrated that a single dose of Abrysvo elicited strong RSV A and RSV B neutralising responses at 1 month after vaccination. In the Abrysvo group (n=534), geometric mean fold rises (GMFRs) of neutralising titres (NTs) for RSV A, RSV B and combined RSV A/B were 11.6, 12.7 and 12.1, respectively.
Subgroup analyses by age and prespecified high risk groups in Abrysvo recipients were consistent with the main analyses and support consistent VE observed across different age and risk groups. GMFRs of NTs for RSV A, RSV B and combined RSV A/B were 11.4, 12.6 and 12.0, respectively, in the 60-69 years age group, 12.1, 12.8 and 12.4 in the 70-79 years age group and 11.2, 15.2 and 13.0 in the ≥ 80 years age group. GMFRs of NTs for RSV A, RSV B and combined RSV A/B were 12.3, 13.8 and 13.0, respectively, in recipients with > 1 significant underlying condition.
5.2 Pharmacokinetic Properties
Not applicable.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Powder for injection. Trometamol, trometamol hydrochloride, sucrose, mannitol, polysorbate 80, sodium chloride, hydrochloric acid (for pH adjustment).
Diluent. Water for injections.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
The unopened vial is stable for 5 days when stored at temperatures from 8°C to 30°C. At the end of this period Abrysvo should be used or discarded. This information is used to guide healthcare professionals in case of temporary temperature excursions only.
After reconstitution. Abrysvo should be administered immediately (within 4 hours) after reconstitution.
Do not store above 30°C. Do not freeze.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C - 8°C).
Do not freeze. Discard if the carton has been frozen.
Store in the original package.
For storage conditions after reconstitution of the medicinal product, see Section 6.3.
6.5 Nature and Contents of Container
Powder for injection: for 1 dose in a 2 mL vial (type 1 glass or equivalent) with a stopper (synthetic bromobutyl rubber or synthetic chlorobutyl rubber) plus either:
Diluent: for 1 dose in a 1 mL pre-filled syringe (type 1 glass) with a stopper (synthetic chlorobutyl rubber) and a tip cap (synthetic isoprene/bromobutyl blend rubber) or,
Diluent: for 1 dose in a vial (type 1 glass or equivalent) with a stopper (bromobutyl rubber).
Pack size. Carton containing 1 vial of powder for injection, 1 pre-filled syringe of diluent, 1 vial adapter.
Carton containing 5 vials of powder for injection, 5 pre-filled syringes of diluent, 5 vial adapters.
Carton containing 10 vials of powder for injection, 10 pre-filled syringes of diluent, 10 vial adapters.
Carton containing 10 vials of powder for injection and 10 vials of diluent.
No needles are included in the packs.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
No data available.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
20 March 2024
Date of Revision
24 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.