Acetylcysteine-Link
Brand Information
| Brand name | Acetylcysteine-Link |
| Active ingredient | Acetylcysteine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Acetylcysteine-Link.
Summary CMI
ACETYLCYSTEINE-LINK
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ACETYLCYSTEINE-LINK?
ACETYLCYSTEINE-LINK contains the active ingredient acetylcysteine. ACETYLCYSTEINE-LINK is used to treat paracetamol poisoning. For more information, see Section 1. Why am I using ACETYLCYSTEINE-LINK? in the full CMI.
2. What should I know before I use ACETYLCYSTEINE-LINK?
Do not use if you have ever had an allergic reaction to acetylcysteine or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ACETYLCYSTEINE-LINK? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ACETYLCYSTEINE-LINK and affect how it works.
For more information, see Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ACETYLCYSTEINE-LINK?
- ACETYLCYSTEINE-LINK must only be given by a doctor or a nurse as a slow infusion into the vein.
- Your doctor will decide what dose you will receive.
More information can be found in Section 4. How do I use ACETYLCYSTEINE-LINK? in the full CMI.
5. What should I know while using ACETYLCYSTEINE-LINK?
| Things you should do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ACETYLCYSTEINE-LINK? in the full CMI.
6. Are there any side effects?
Side effects may include nausea, vomiting, generally feeling unwell, fever or sweating, chills or shivering, anxiety, dizziness or lightheadedness, pain in your joints or bones, injection site pain and swelling, chest discomfort, bleeding or bruising more easily than normal, rash, or sweating. Severe side effects may include allergic reactions, difficulty breathing, chest pain, fast or irregular heartbeat, seizure, bluish-purple colour of skin, blurred vision or eye pain, pain in the face, swelling around the eyes.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ACETYLCYSTEINE-LINK
Active ingredient: acetylcysteine
Consumer Medicine Information (CMI)
This leaflet provides important information about using ACETYLCYSTEINE-LINK. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ACETYLCYSTEINE-LINK.
Where to find information in this leaflet:
1. Why am I using ACETYLCYSTEINE-LINK?
2. What should I know before I use ACETYLCYSTEINE-LINK?
3. What if I am taking other medicines?
4. How do I use ACETYLCYSTEINE-LINK?
5. What should I know while using ACETYLCYSTEINE-LINK?
6. Are there any side effects?
7. Product details
1. Why am I using ACETYLCYSTEINE-LINK?
ACETYLCYSTEINE-LINK contains the active ingredient acetylcysteine.
ACETYLCYSTEINE-LINK is used to treat paracetamol poisoning.
2. What should I know before I use ACETYLCYSTEINE-LINK?
Warnings
Do not use ACETYLCYSTEINE-LINK if:
- You are allergic to acetylcysteine, or any of the ingredients listed at the end of this leaflet.
Check with your doctor if you:
- have any other medical conditions, such as asthma or difficulty breathing, stomach ulcers, kidney problems or liver problems.
- weigh less than 40 kilograms.
- are on fluid restriction.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Discuss with your doctor if you are pregnant or intend to become pregnant.
Discuss with your doctor if you are breastfeeding or intend to breastfeed. It is not known if ACETYLCYSTEINE-LINK passes into breast milk. Breastfeeding is not recommended during treatment with ACETYLCYSTEINE-LINK.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
ACETYLCYSTEINE-LINK may interfere with the results of some urinary dipstick tests.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ACETYLCYSTEINE-LINK.
4. How do I use ACETYLCYSTEINE-LINK?
How much to inject
- Your doctor will decide what volume of solution is required to achieve the required dose.
How to inject ACETYLCYSTEINE-LINK
- ACETYLCYSTEINE-LINK must only be given by a doctor or nurse as a slow infusion into the vein.
- The duration of the treatment is approximately 20 hours.
If you use too much ACETYLCYSTEINE-LINK
As ACETYLCYSTEINE-LINK is given to you under the supervision of your doctor, it is very unlikely that you will receive too much.
If you think that you or anyone else may have been given too much ACETYLCYSTEINE-LINK, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ACETYLCYSTEINE-LINK?
Things you should do
Tell any doctor, dentist or pharmacist who treats you that you have been given ACETYLCYSTEINE-LINK.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ACETYLCYSTEINE-LINK affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Regular alcohol consumption may change the required dose of ACETYLCYSTEINE-LINK.
Looking after your medicine
ACETYLCYSTEINE-LINK will be stored in the pharmacy or on the ward. It is kept in a cool dry place, protected from light, where the temperature stays below 25°C.
When to discard your medicine
Once opened, the contents of a vial should be used immediately, and any unused portion of any vial should be discarded.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Psychiatric related
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs of allergic reaction such as:
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ACETYLCYSTEINE-LINK contains
| Active ingredient (main ingredient) | acetylcysteine |
| Other ingredients (inactive ingredients) | Sodium hydroxide Disodium edetate Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What ACETYLCYSTEINE-LINK looks like
ACETYLCYSTEINE-LINK is supplied as a clear, colourless solution. It is supplied in 10mL glass ampoules, in packs of 10. Each 10 mL ampoule contains 2g acetylcysteine.
AUST R 170803
Who distributes ACETYLCYSTEINE-LINK
Link Medical Products Pty Ltd
5 Apollo Street
Warriewood, NSW, 2102
Australia
1800 181 060
linkhealthcare.com.au
This leaflet was prepared in October 2025.
Brand Information
| Brand name | Acetylcysteine-Link |
| Active ingredient | Acetylcysteine |
| Schedule | S4 |
MIMS Revision Date: 01 November 2020
1 Name of Medicine
Acetylcysteine.
2 Qualitative and Quantitative Composition
Acetylcysteine 2 g in 10 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Concentrated injection.
Acetylcysteine-Link is a clear, colourless, sterile, pyrogen free, aqueous solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Antidote for paracetamol poisoning.
Acetylcysteine is indicated in the treatment of paracetamol overdose to protect against hepatotoxity.
4.2 Dose and Method of Administration
To be most effective in protecting against liver damage, therapy with acetylcysteine should be started within ten hours of paracetamol ingestion. Although the role of acetylcysteine therapy in patients presenting later than 15 hours after paracetamol ingestion is not established, it may be considered as a clinical option in high risk patients.
Management of paracetamol overdosage. It should be noted that, after an ingestion of a potentially fatal dose of paracetamol, the patient may appear relatively well initially and may even continue normal activities for a day or two before the onset of hepatic failure. Hepatic damage is more likely to occur with a lower dosage of paracetamol in patients who have a history of chronic alcohol or enzyme inducing drug ingestion (e.g. isoniazid, rifampicin, anticonvulsants including carbamazepine, phenytoin, phenobarbitone, primidone, sodium valproate).
Patients are notoriously unreliable as to the amount ingested and the time of ingestion. Hepatic necrosis is preventable if treatment can be instituted within 10 to 12 hours of ingestion.
Note. Liver damage may not be biochemically apparent for 24 to 48 hours after ingestion.
Hepatic necrosis has been seen with 6 g of paracetamol, and death with 15 g.
Patients presenting within 15 hours of ingestion. Give activated charcoal (1 to 2 g/kg) if it is within one hour of paracetamol ingestion, and the patient's conscious state is not impaired.
Plasma paracetamol levels should be obtained no earlier than four hours after ingestion of the paracetamol overdose. Concentrations determined prior to this time are not reliable for assessing potential hepatotoxicity. If the time of ingestion is unknown, paracetamol levels should be measured immediately.
Measurements of plasma liver enzymes and bilirubin levels, and coagulation studies, should be performed as soon as possible after admission. Blood urea, electrolytes, glucose and blood gases should be obtained. The laboratory measurements are used to monitor hepatic and renal function and electrolyte balance. An ECG should also be performed.
Plasma paracetamol levels can be used to determine the likelihood of hepatic damage when compared with the nomogram (Figure 1).
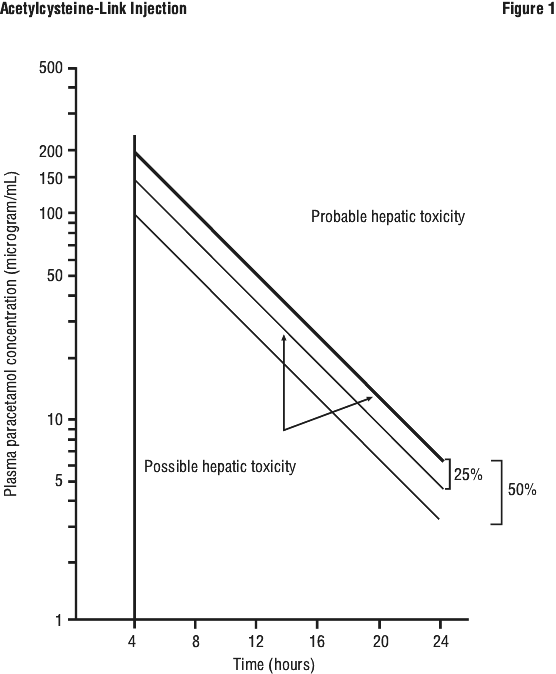
Note. To convert paracetamol concentrations from micromol/L to micrograms/mL, divide by 6.61.
Do not delay acetylcysteine therapy while awaiting the results of plasma assays. Once the results become available, treatment may be discontinued if the initial concentration is below the nomogram reference line.
Do not discontinue acetylcysteine therapy if the initial level is above the reference line and subsequent levels fall below the reference line.
The nomogram is designed to be used for single acute ingestions. It is not helpful in determining the need for acetylcysteine in multiple or chronic ingestions.
The nomogram may not be suitable for use when patients have taken sustained release preparations of paracetamol. Patients whose plasma paracetamol level is above the solid line are at a high risk of developing hepatotoxicity.
The middle line (25% below the solid line) is included to allow for possible errors in plasma assays and estimated time from ingestion and should be used as a guide to treatment.
Patients who have a history of chronic alcohol abuse or are receiving enzyme inducing drugs are at greater risk of developing hepatotoxicity and if their plasma paracetamol level is up to 50% below the solid line, they may require treatment with acetylcysteine.
Patients presenting more than 15 hours after ingestion. Plasma paracetamol, bilirubin and AST levels should be determined on an urgent basis, and a clinical decision made on how to proceed. Acetylcysteine may be beneficial in patients presenting later than 15 hours after ingestion. The use of acetylcysteine should therefore be considered in high risk patients who present late, but only after discussion with doctors with substantial experience in the management of patients with paracetamol poisoning.
General management. Acetylcysteine should be administered if appropriate (see above). Acetylcysteine should be diluted in glucose 5% solution or 0.9% sodium chloride solution and administered by intravenous infusion. Nausea should be treated.
Daily liver function tests and measurements of plasma urea, electrolytes, haemoglobin levels, white blood cell counts, platelets and prothrombin time should be made. Patients should be monitored for coagulation disorders, hepatic encephalopathy, renal failure and cardiac toxicity (minor ST changes are common). There is usually a mild metabolic acidosis. Hepatic encephalopathy is likely if bilirubin is above 60 mmol/L on days 3 to 5, or if the prothrombin time is prolonged.
Adults. Acetylcysteine is infused in three intravenous infusions containing different doses. This will give a total dose of acetylcysteine 300 mg/kg infused over 20 hours.
Initial infusion. An initial dose of 150 mg/kg diluted in 200 mL of 5% glucose or 0.9% sodium chloride and infused over 15 to 60 minutes.
Second infusion. The initial infusion is followed by a continuous infusion of 50 mg/kg acetylcysteine diluted in 500 mL of 5% glucose or 0.9% sodium chloride over the next four hours.
Third infusion. The second infusion is followed by a continuous infusion of 100 mg/kg acetylcysteine diluted in 1,000 mL of 5% glucose or 0.9% sodium chloride over the next 16 hours.
Acetylcysteine intravenous infusion dosage guide. Acetylcysteine is supplied in ampoules containing 10 mL of 200 mg/mL for intravenous administration.
Table 1 is intended as a guide on the volume (mL) of acetylcysteine 200 mg/mL that is required to be added to glucose 5% or 0.9% sodium chloride to prepare the initial, second and third infusion solutions. In order to use this table, the patient's weight in kg should be determined. The volume (mL) of acetylcysteine 200 mg/mL that should be added to glucose 5% or 0.9% sodium chloride to prepare the initial, second and third infusion solutions is shown in the three columns next to the patient's weight.
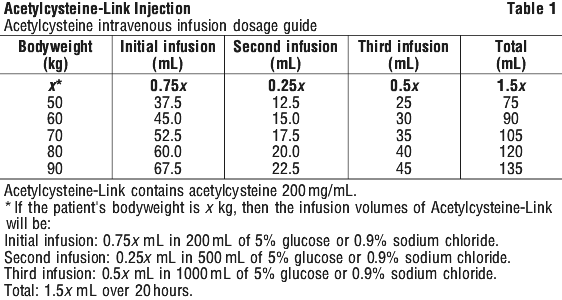
Product is for single use in one patient only. Discard any residue.
Children. Children should be treated with the same doses and regimens as adults. However, the quantity of intravenous fluid should be modified to take into account age and weight, as fluid overload is a potential danger.
4.3 Contraindications
Hypersensitivity or previous anaphylactic reaction to acetylcysteine or any component of the preparation.
4.4 Special Warnings and Precautions for Use
Acetylcysteine should be used with caution in asthma or where there is a history of bronchospasm. It should also be used with caution in patients with a past history of oesophageal varices and peptic ulceration (acetylcysteine induced vomiting may increase the risk of haemorrhage).
Acetylcysteine is not compatible with rubber and some metals, particularly iron, copper and nickel.
Acetylcysteine can be used satisfactorily with silicone rubber and plastic.
Patients with body weight less than 40 kg. For patients weighing less than 40 kg, adjustment of total volume is recommended when administering acetylcysteine, to minimise the risk of hyponatraemia, seizure and death.
Patients on fluid restriction. For patients on fluid restriction, adjustment of total volume is recommended when administering acetylcysteine, to minimise the risk of of hyponatraemia, seizure and death.
Use in renal/hepatic impairment. Caution should be taken when administering acetylcysteine in patients with hepatic or renal failure, since there is little data relating to the effects of acetylcysteine in impaired renal and/or hepatic function. The decision to administer should be passed on a risk/benefit assessment for the individual subject.
In the presence of hepatic failure due to paracetamol overdose the degree of existing liver damage and the possible risk associated with the administration of acetylcysteine should be considered.
Use in the elderly. There are no adequate or well controlled studies in elderly patients. For this reason, the safety and effectiveness of acetylcysteine in the elderly has not been established.
Paediatric use. While the safety and effectiveness of acetylcysteine Concentrated Injection in children has not been established, there is some limited clinical data in the use of acetylcysteine injection in children (also see Section 5.1 Pharmacodynamic Properties, Clinical trials).
No paediatric specific adverse reactions have been documented.
Effects on laboratory tests. Acetylcysteine may cause a false positive reaction with reagent dipstick tests for urinary ketones.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No information is available on the interaction of acetylcysteine with other drugs.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There was evidence of effects on fertility in male rats given acetylcysteine at doses up to 60% of the maximum clinical dose, on a body surface area basis. No effects were observed at doses 15% the maximum clinical dose, on a body surface area basis.
Use in pregnancy. (Category B2)
There was no evidence of teratogenicity in limited studies in rats and rabbits following administration of acetylcysteine during the period of gestation at doses up to 1.2 times the maximum clinical dose, on a body surface area basis. There are no well controlled studies in pregnant women but experience does not include any positive evidence of adverse effects to the fetus.
Category B2: Drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals are inadequate or may be lacking, but available data show no evidence of an increased occurrence of fetal damage.
Use in lactation. There was no evidence of adverse effects in a limited study in rats following administration of acetylcysteine during late gestation and lactation at 60% of the maximum clinical dose, on a body surface area basis. It is not known whether acetylcysteine and/or its metabolites are excreted in milk. There are no data on the use of acetylcysteine in lactating women and therefore breastfeeding is not recommended during treatment.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Intravenous administration of acetylcysteine, especially in the large doses needed to treat paracetamol overdose, may result in nausea, vomiting and other gastrointestinal symptoms. Hypersensitivity reactions have been reported following intravenous administration of acetylcysteine. Bronchospasm may occur in conjunction with a generalised anaphylactoid reaction. The symptoms of the anaphylactic-like reaction to acetylcysteine include airway obstruction (bronchospasm), angioedema, dyspnoea, hypotension, shock, tachycardia, urticaria, and injection site reaction (including rash). These reactions occur most commonly either during, or at the end of the period of the loading dose infusion, and may be dose related. Since these anaphylactic-like reactions usually occur following the loading dose, careful monitoring is recommended.
There have been rare instances of death associated with the use of intravenous acetylcysteine.
Adverse reactions reported are summarised in Table 2.
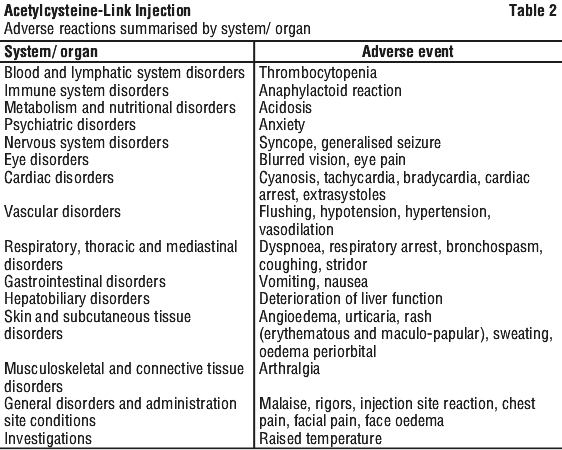
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdosage symptoms. Symptoms following overdosage with acetylcysteine have been similar to those of anaphylactoid reactions noted (see Section 4.8 Adverse Effects (Undesirable Effects)), but they may be more severe. Hypotension appears to be especially prominent. There is also a theoretical risk of hepatic encephalopathy.
Treatment. There is no specific treatment. General supportive measures should be carried out. It has been suggested that generalised reactions to acetylcysteine can be treated with intravenous injection of an antihistamine, and infusion of acetylcysteine should be temporarily stopped but can be restarted at a slower rate without further reaction.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Paracetamol is metabolised in the liver, mainly by conjugation with glucuronide and sulfate. It is also metabolised by cytochrome P450 to form a reactive, potentially toxic, metabolite. This metabolite is normally detoxified by conjugation with hepatic glutathione, to form nontoxic derivatives. In paracetamol overdosage, the glucuronide and sulfate conjugation pathways are saturated, so that more of the toxic metabolite is formed. As hepatic glutathione stores are depleted, this toxic metabolite may bind to hepatocyte proteins, leading to liver cell damage and necrosis. Acetylcysteine is a sulfhydryl (SH) group donor, and may protect the liver from damage by restoring depleted hepatic reduced glutathione levels, or by acting as an alternative substrate for conjugation with, and thus detoxification of, the toxic paracetamol metabolite.
Clinical trials. Observational study - paediatric patients. An open-label, observational study conducted in the greater Newcastle area, New South Wales, Australia, documented treatment for paediatric patients who presented with a paracetamol overdose during a 16 year period from January 1987 to January 2003. This study was primarily considered a safety study.
Data from 148 paediatric patients, with an age range of 2 months to 15 years (this corresponds to 186 cases) were evaluated. Twenty three (23) out of 148 paediatric patients were given intravenous acetylcysteine treatment on at least one admission. Of these, 14 paediatric patients in the age group 12 to < 16 years (9.5% of the group), received acetylcysteine within 8 hours of ingesting paracetamol on at least one admission. There was a delay of at least 8 hours for 9 paediatric patients in this age group (6.1% of the group). One other paediatric patient in the age group 2 to < 5 years of age (0.7% of the group), received acetylcysteine with a delay of at least 8 hours on at least one admission.
Of the 23 patients who received intravenous acetylcysteine treatment, 3 patients (13%) experienced an adverse reaction (anaphylactoid reaction, rash and flushing, transient erythema). There were no deaths of paediatric patients. None of the paediatric patients receiving intravenous acetylcysteine developed hepatotoxicity, whilst two patients not receiving intravenous acetylcysteine did develop hepatotoxicity. The number of paediatric patients examined in this study is too small to provide a statistically significant finding for efficacy, however, the results appear to be consistent to those observed in adults.
Safety study. A randomised, open-label, multi-centre clinical study was conducted in Australia to compare the rates of anaphylactoid reactions between two rates of infusion for the intravenous acetylcysteine loading dose. One hundred and nine subjects were randomised to a 15 minute infusion rate and 71 subjects were randomised to a 60 minute infusion rate. The loading dose was 150 mg/kg, followed by a maintenance dose of 50 mg/kg over 4 hours and then 100 mg/kg over 16 hours. Of the 180 patients examined in this study, 27% were male and 73% were female. Ages ranged from 15 to 83 years, with the mean age being 29.9 years (+13.0).
Within the first 2 hours following intravenous acetylcysteine administration, 17% of all patients developed an anaphylactoid reaction (18% in the 15-minute treatment group; 14% in the 60-minute treatment group) (see Section 4.4 Special Warnings and Precautions for Use). A subgroup of 58 subjects (33 in the 15-minute treatment group; 25 in the 60-minute treatment group) was treated within 8 hours of paracetamol ingestion. No hepatotoxicity occurred within this subgroup; however with 95% confidence, the true hepatotoxicity rates could range from 0% to 9% for the 15-minute treatment group and from 0% to 12% for the 60-minute treatment group.
5.2 Pharmacokinetic Properties
Acetylcysteine is the N-acetyl derivative of the naturally occurring amino acid, L-cysteine, and is deacetylated in the liver to cysteine, or oxidised to other metabolites such as N-acetylcystine or N,N-diacetylcystine. The parent compound and metabolites may be present in the plasma either free or protein bound. Renal clearance accounts for about 30% of total body clearance. Following intravenous administration, mean terminal half-lives have been calculated to be 1.95 and 5.58 hours respectively for reduced and total acetylcysteine.
5.3 Preclinical Safety Data
Genotoxicity. No evidence of mutagenicity was obtained in limited gene mutation assays with acetylcysteine. The potential for acetylcysteine to cause chromosomal damage has not been investigated.
Carcinogenicity. Carcinogenicity assays have not been performed with acetylcysteine. In rats, no evidence of carcinogenicity was reported following 18 months of daily dietary administration of acetylcysteine at 60% of the maximum clinical dose, on a body surface area basis.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium hydroxide, disodium edetate, water for injections.
6.2 Incompatibilities
Acetylcysteine is not compatible with rubber and some metals, particularly iron, copper and nickel. Acetylcysteine can be used satisfactorily with silicone rubber and plastic.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Acetylcysteine-Link is supplied in 10 mL ampoules (Glass Type I Clear).
It is available in the pack size 10 x 10 mL ampoules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Acetylcysteine is a white, crystalline powder with a slight acetic odour. It is soluble in water and alcohol and practically insoluble in chloroform, dichloromethane and ether.
Acetylcysteine-Link has a pH of 7.0.
Chemical structure.
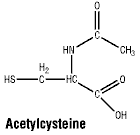
IUPAC name: (2R)-2-(Acetylamino)-3-sulfanylpropanoic acid.
Molecular formula: C5H9NO3S. Molecular weight: 163.2.
CAS number. 616-91-1.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
31 January 2012
Date of Revision
04 August 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.