Acular
Brand Information
| Brand name | Acular |
| Active ingredient | Ketorolac trometamol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Acular.
Summary CMI
ACULAR®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ACULAR®?
ACULAR® eye drops contain the active ingredient ketorolac trometamol. ACULAR® is used in adults:
- to treat itchy eyes occurring with the condition called seasonal allergic conjunctivitis, and
- to prevent and relieve swelling and redness after cataract surgery.
For more information, see Section 1. Why am I using ACULAR®? in the full CMI.
2. What should I know before I use ACULAR®?
Check the list of ingredients at the end of the CMI. Do not use ACULAR® if you have ever had an allergic reaction to any of them.
Talk to your doctor before you use this medicine if he/she is not aware that you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ACULAR®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ACULAR® and affect how it works, or ACULAR® may interfere with other medicines and affect how they work. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ACULAR®?
- Your doctor will advise how many drops to use depending on your condition.
- Use the drops four times per day.
- Your doctor will advise when to start using the drops and when to finish.
More instructions can be found in Section 4. How do I use ACULAR®? in the full CMI.
5. What should I know while using ACULAR®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ACULAR®? in the full CMI.
6. Are there any side effects?
Stinging and burning of the eye(s) when applying the eye drops (eye pain), blurred vision, infection, swelling at the front of the eye or the iris, red itchy eye(s), irritation of the surface of the eye, drooping or swollen lid, sensitivity to light, dry eye, lesion on the front of the eye, increased eye pressure, white spots on eye surface, headache, bleeding or swelling of retina, asthma, bronchospasm, severe allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ACULAR® (ack-yoo-lar)
Active ingredient(s): ketorolac trometamol (keet-oh-roll-ack troh-met-am-ol)
Consumer Medicine Information (CMI)
This leaflet provides important information about using ACULAR® eye drops. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ACULAR®.
Where to find information in this leaflet:
1. Why am I using ACULAR®?
2. What should I know before I use ACULAR®?
3. What if I am taking other medicines?
4. How do I use ACULAR®?
5. What should I know while using ACULAR®?
6. Are there any side effects?
7. Product details
1. Why am I using ACULAR®?
ACULAR® contains the active ingredient ketorolac trometamol.
Ketorolac trometamol works by decreasing prostaglandin E2 levels in the eye. Prostaglandins are normally produced by the body and are thought to be associated with pain, itching and inflammation.
ACULAR® is used in adults:
- to treat itchy eyes occurring with the condition called seasonal allergic conjunctivitis, and
- to prevent and relieve swelling and redness after cataract surgery.
2. What should I know before I use ACULAR®?
Warnings
Do not use ACULAR® if :
- you are allergic to ketorolac trometamol, or any of the ingredients listed at the end of this leaflet.
- you have ever had an allergic reaction to aspirin or other similar medicines used for arthritis or pain relief (non-steroidal anti-inflammatory drugs (NSAIDs)).
- the seal around the cap is broken.
- the bottle/packaging shows signs of tampering.
- the product does not look quite right.
- the expiry date on the bottle has passed.
Check with your doctor if you:
- have or have had a stomach ulcer
- have or have had problems with your lungs, such as asthma, bronchospasm
- have or have had problems with the surface of your eye (cornea)
- have or have had other eye conditions, such as dry eye syndrome
- have had recent eye surgery
- have diabetes
- have rheumatoid arthritis
- are using topical NSAIDs for any other condition
- have a condition that causes you to bleed or bruise easily
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed.
ACULAR® is not recommended in pregnant or breastfeeding women.
Children
It is not known if ACULAR® is safe and effective for use in children younger than 12 years of age.
3. What if I am taking other medicines?
Tell your optical practitioner, doctor or pharmacist if you are using any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket, or health food store.
Some medicines may interfere with ACULAR® and affect how it works; while ACULAR® may affect how other medicines work.
Using ACULAR® at the same time as other eye medicines, including similar medicines (NSAIDs) and steroids, may slow down the healing process, after surgery.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ACULAR®.
4. How do I use ACULAR®?
How much to use
Your doctor will tell you how many drops you need to use each day. Use ACULAR® as prescribed by your doctor.
Seasonal allergic conjunctivitis
- Use one drop in your eye(s) four times each day.
Cataract surgery
- Use one to two drops in your eye(s) four times each day.
When to use ACULAR®
Seasonal allergic conjunctivitis
- Continue to use the drops as instructed for up to four weeks.
Cataract surgery
- Use the drops as instructed starting 24 hours before surgery.
- Continue to use the drops for as long as your doctor advises, which may be up to 4 weeks.
In some instances, ACULAR® needs to be used with other eye drops. Your doctor will let you know which drops, how to use them and how long to use them.
How to use ACULAR®
These drops are for topical use only.
- Wash your hands well with soap and water.
- If you wear soft contact lenses, remove them before using ACULAR®.
- Tear off the protective seal on any new bottle and throw the seal away. Remove the lid/cap.
- Hold the bottle upside down in one hand between your thumb and index finger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up. This might be easier if you are sitting or lying down.
- Put the tip of the bottle close to your lower eyelid. Do not let the tip touch your eye or eyelid.
- Release one drop into the pouch or pocket formed between your eye and eyelid by gently squeezing the bottle.
- If a drop misses your eye, try again, and wipe off any excess that runs down your cheek.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Replace the lid/cap, sealing it tightly.
- Wash your hands again with soap and water.
- If you wear soft contact lenses, wait 15 minutes before replacing your contact lenses.
If you forget to use ACULAR®
It is important that you use ACULAR® at the same times each day.
If you forget to use ACULAR® and it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use the drops as soon as you remember, and then go back to using them as you would normally. If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not use double the number of drops to make up for the dose you missed.
If you use too much ACULAR®
Immediately rinse your eyes with warm water.
If ACULAR® is accidentally swallowed, you should immediately:
- phone the Poisons Information Centre
by calling 13 11 26, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there appear to be no signs of discomfort or poisoning.
5. What should I know while using ACULAR®?
Things you should do
- If you wear soft contact lenses, remove them before using ACULAR® eye drops, and wait at least 15 minutes before inserting them again. The preservative in ACULAR® eye drops (benzalkonium chloride) may be absorbed and cause discolouration of soft contact lenses.
- Tell your doctor if you think your eye(s) is/are not improving while using ACULAR®.
Call your doctor straight away if you:
- develop problems with your eyes, such as trauma, surgery, infection in the eye(s) or eyelid(s)
- become pregnant while using ACULAR®
Remind any doctor, dentist, pharmacist, or optometrist you visit that you are using ACULAR®.
Things you should not do
- Do not touch the dropper tip against your eye, eyelid, or anything else, to avoid contaminating the eye drops and to avoid injury to the eye caused by the tip.
- Do not stop using ACULAR® earlier than instructed or use it for longer than instructed without checking with your doctor.
Driving or using machines
Wait for your vision to clear after using ACULAR® before you drive or use machines.
Looking after your medicine
Follow the instructions on the label and carton on how to take care of your medicine properly.
Store it in a cool place below 30°C away from heat or sunlight; for example:
- do not store it in the bathroom or near a sink, or
- do not store it in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard the bottle 4 weeks after first opening it.
Getting rid of any unwanted medicine
If your doctor advises that you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your optical practitioner, doctor or pharmacist if you have any further questions about side effects.
Side effects listed below could include the symptoms such as eye pain, difficulty reading, sensitivity to light, seeing halos around lights, sudden vision loss, vision getting noticeably worse or blurry, blind or dark spots in vision, seeing flashes or floaters, headache, red or bloodshot eye, eye itching and discharge, and white patch on the front layer of the eyeball.
Less serious side effects
| Less serious side effects | What to do |
Effects on the eye:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Effects on the eye:
| Call your doctor straight away, if you notice any of these serious side effects. |
Body as a whole:
| Go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor, nurse, or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems in Australia. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop using any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ACULAR® contains
| Active ingredient (main ingredient) | ketorolac trometamol |
| Preservative |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What ACULAR® looks like
ACULAR® is a clear, colourless to pale-yellow solution. It is supplied in a white opaque plastic dropper bottles with a dropper tip containing 3 mL, 5 mL or 10 mL of liquid which are enclosed in a carton.
(AUST R 63539).
(Not all presentations may be available.)
Who distributes ACULAR®?
ACULAR® is distributed in Australia by:
AbbVie Pty Ltd
Mascot NSW 2020
AUSTRALIA
Ph: 1800 252 224
This leaflet was prepared in December 2025.
©2025 AbbVie. All rights reserved.
ACULAR® and its design are trademarks of Allergan, Inc., an AbbVie company
Brand Information
| Brand name | Acular |
| Active ingredient | Ketorolac trometamol |
| Schedule | S4 |
MIMS Revision Date: 01 February 2026
1 Name of Medicine
Ketorolac trometamol.
2 Qualitative and Quantitative Composition
Acular contains ketorolac trometamol 5 mg/mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Acular Eye Drops is a clear, colourless to pale yellow, sterile, isotonic aqueous solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Acular Eye Drops are indicated for the short term (2-4 weeks) relief of symptoms of seasonal allergic conjunctivitis.
Acular Eye Drops are also indicated for the short term (2-4 weeks) prophylaxis and reduction in inflammation in patients undergoing cataract extraction.
4.2 Dose and Method of Administration
Seasonal allergic conjunctivitis. The recommended dose of Acular Eye Drops for the relief of symptoms of seasonal allergic conjunctivitis is one drop (0.25 mg) instilled in the eye four times daily. Treatment may be continued for up to four weeks.
Postoperative inflammation. The recommended dose of Acular Eye Drops for the prophylaxis and treatment of inflammation in patients who have undergone cataract extraction is one to two drops (0.25-0.5 mg) four times daily, starting 24 hours before surgery and continuing for 2-4 weeks.
4.3 Contraindications
Acular Eye Drops are contraindicated in patients hypersensitive to any of the components of the medication.
The potential exists for cross sensitivity to acetylsalicylic acid, phenylacetic acid derivatives and other nonsteroidal anti-inflammatory medicines. Acular Eye Drops are contraindicated in patients who have previously exhibited sensitivities to these drugs.
4.4 Special Warnings and Precautions for Use
Patients with bleeding tendencies. With some nonsteroidal anti-inflammatory drugs a potential exists for increased bleeding time due to interference with thrombocyte aggregation. There have been reports that ocularly applied nonsteroidal anti-inflammatory drugs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery. Acular Eye Drops should be used with care in patients with known bleeding tendencies, or in patients who are receiving other medications which may prolong bleeding time, or patients with a known history of peptic ulceration.
Cross-sensitivity. There exists the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other nonsteroidal anti-inflammatory agents. Therefore, caution should be used when treating individuals who have previously exhibited sensitivities to these drugs.
There have been post-marketing reports of bronchospasm or exacerbation of asthma in patients, who have either a known hypersensitivity to aspirin/ nonsteroidal anti-inflammatory drugs or a past medical history of asthma associated with the use of Acular, which may be contributory. Caution is recommended in the use of Acular in these individuals.
Delayed healing. All topical nonsteroidal anti-inflammatory drugs (NSAIDs) may slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
Corneal effects. Use of topical NSAIDs may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs and should be closely monitored for corneal health.
Topical NSAIDs should be used with caution in patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g. dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time as they may be at increased risk for corneal adverse events which may become sight threatening.
Post-marketing experience with topical NSAIDs also suggests that use more than 24 hours prior to surgery or use beyond 14 days post-surgery may increase patient risk for the occurrence and severity of corneal adverse events.
Masking of infections. In common with other anti-inflammatory drugs, Acular Eye Drops may mask the usual signs of infections.
Use in the elderly. No data available.
Paediatric use. Safety and efficacy have not been demonstrated in children aged less than 12 years.
Effects on laboratory tests. No data available.
Information for patients. Acular should not be administered while wearing contact lenses. Acular contains the preservative benzalkonium chloride, which may be absorbed and cause discoloration to soft contact lenses. Contact lenses should be removed prior to administration of Acular and may be reinserted 15 minutes following administration.
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures to avoid injury and contamination of eye drops.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No specific clinical studies on interactions with other drugs have been conducted with Acular Eye Drops. There were no reports in the controlled studies of interactions of Acular Eye Drops with systemic and ophthalmic medications such as antibiotics, sedatives, beta blockers, carbonic anhydrase inhibitors, miotics, mydriatics, cycloplegics, local anaesthetics and corticosteroids.
Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems (see Section 4.4 Special Warnings and Precautions for Use, Delayed healing).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Impairment of fertility did not occur in male or female rats at oral doses of 9 mg/kg (1.2 times human parenteral AUC) and 16 mg/kg (2.1 times the human parenteral AUC) respectively.
Use in pregnancy. (Category C)
Ketorolac trometamol and its metabolites have been shown to pass into the foetus. Safety in human pregnancy has not been established. Not recommended in pregnancy.
Reproduction studies have been performed in rabbits and rats at oral doses of 3.6 and 10 mg/kg/day, respectively. The results from these studies did not reveal any significant evidence of harm to the foetus. However, studies in rabbits have shown a small increase in the incidence of major vessel anomalies.
Use in lactation. Ketorolac trometamol is excreted in the milk of lactating rats and rabbits, and has been detected in human breast milk. A peri/ postnatal study in rats showed reductions in postnatal growth and survival of the offspring when ketorolac trometamol was administered to lactating rats at oral dose levels greater than 4.8 mg/kg/day. Thus, use of ketorolac trometamol in lactating mothers is not recommended.
4.7 Effects on Ability to Drive and Use Machines
As Acular Eye Drops may cause transient blurring on instillation, caution is required with the use of hazardous machinery or driving, which are not recommended unless vision is clear. The patient should wait until their vision clears before driving or using machinery.
4.8 Adverse Effects (Undesirable Effects)
In controlled clinical studies, the most frequently reported adverse events with the use of Acular Eye Drops have been transient stinging and burning on instillation (eye pain). These events were reported by up to 40% of subjects treated with Acular. Other adverse events reported in controlled clinical studies (at an incidence of ≥ 1%) included conjunctivitis (scratching, foreign body sensation, itching, erythema), local allergic reactions, superficial keratitis, keratic precipitates (1%), haemorrhage retinal (1%), cystoid macular edema (1%), burning eye (1%), pruritus eye (1%), eye trauma (1%), intraocular pressure (2%), corneal oedema, eye infection, eye inflammation, eye irritation and hypersensitivity. Ptosis, blepharitis, photophobia, blurred vision, eye dryness, corneal lesion, iritis, and glaucoma were also reported in > 1% of patients in some studies. Uncommon (> 0.1% and < 1%) adverse events reported were eye dryness, corneal infiltrates, ulcerative keratitis, visual disturbance and headaches.
Note. The frequency of 1% represents 1 patient.
Post-marketing experience. The following adverse reactions have been identified during post-marketing use. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In post-marketing experience ocular burning and stinging, eye irritation, local allergic reactions, superficial ocular infections, superficial keratitis, eye oedema, ocular hyperaemia, conjunctival hyperaemia, eye swelling, eye pain, eye pruritus and ulcerative keratitis were the most frequently reported adverse reactions. Systemic allergic reactions have been reported very rarely. There have been post-marketing reports of bronchospasm or exacerbation of asthma in patients who have either a known hypersensitivity to aspirin/ nonsteroidal anti-inflammatory drugs or a past medical history of asthma, associated with the use of Acular which may be contributory (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is no experience of overdose by the ophthalmic route. If accidentally ingested, fluids should be taken to dilute the effects, if any.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Class. Ketorolac trometamol is a member of the pyrrolo-pyrolle group of nonsteroidal anti-inflammatory drugs for ophthalmic use.
Mechanism of action. Ketorolac trometamol is a nonsteroidal anti-inflammatory agent demonstrating analgesic and anti-inflammatory activity. It is believed to inhibit the cyclo-oxygenase enzyme essential for prostaglandin biosynthesis. Ocular administration of ketorolac trometamol reduces prostaglandin E2 levels in the aqueous humour. Ketorolac trometamol given systemically does not cause pupil constriction. Results from clinical studies indicate that Acular Eye Drops has no significant effect upon intraocular pressure.
Clinical trials. Allergic conjunctivitis. A total of 203 patients with seasonal allergic conjunctivitis were evaluable for efficacy in two randomised, controlled clinical trials of Acular (ketorolac) eye drops against placebo. The patients who received ketorolac demonstrated significant decreases in itching and conjunctival inflammation over the 7 day treatment period.
Post-operative inflammation. A total of 206 patients who had undergone cataract surgery participated in two randomised, controlled clinical trials of Acular (ketorolac) eye drops against placebo. The patients who received ketorolac showed significantly greater decreases in anterior chamber cells and anterior chamber flare over the two weeks of treatment. 39% of ketorolac patients achieved a zero score for anterior cells and flare after 2 weeks of treatment compared to 12% of placebo patients.
5.2 Pharmacokinetic Properties
Absorption. Two drops (0.1 mL) of 0.5% Acular Eye Drops instilled into the eyes of patients 12 hours and 1 hour prior to cataract extraction achieved measurable levels in 8 of 9 patients' eyes (mean ketorolac concentrations 95 nanogram/mL aqueous humour, range 40-170 nanogram/mL). One drop (0.05 mL) of 0.5% ketorolac trometamol solution was instilled into one eye and one drop of the vehicle into the other eye three times a day for 21 days in 26 normal subjects.
Distribution. Only 4 of 13 subjects had detectable amounts of ketorolac in their plasma (range 10.7 to 22.5 nanogram/mL) after 15 minutes at Day 10 during topical ocular treatment. Average peak plasma level following intramuscular administration of 30 mg ketorolac trometamol was 2.2 microgram/mL 50 minutes after administration.
5.3 Preclinical Safety Data
Genotoxicity. Ketorolac trometamol was not genotoxic in a series of assays for gene mutations and DNA damage. Ketorolac trometamol did not cause chromosome breakage in the mouse micronucleus test in vivo at 1590 microgram/mL, approximately 1000 times the average human plasma levels, but increased the incidence of chromosomal aberrations in Chinese hamster ovarian cells in vitro at higher concentrations.
Carcinogenicity. An 18-month study in mice at oral doses of ketorolac trometamol of 2 mg/kg/day (equal to 1.2 times the human systemic exposure at the maximum recommended IM dose of 90 mg/day, based on AUC) and a 24-month study in rats at oral doses of 5 mg/kg (0.7 times the human parenteral AUC) showed no evidence of tumours.
6 Pharmaceutical Particulars
6.1 List of Excipients
Benzalkonium chloride 0.01% (w/v), disodium edetate 0.1% (w/v), octoxinol 40, sodium chloride, sodium hydroxide/hydrochloric acid (to adjust pH) and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
24 months.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Acular (ketorolac trometamol) eye drops is supplied in white opaque plastic dropper bottles with dropper applicators.
Eye drops: 3 mL, 5 mL and 10 mL (3 mL and 10 mL pack sizes are not marketed).
6.6 Special Precautions for Disposal
Discard any unused contents 28 days after opening the bottle.
6.7 Physicochemical Properties
Chemical structure. Ketorolac trometamol is a white to off-white crystalline substance, which is a racemic mixture. It may exist in three crystalline forms, all of which are equally soluble in water.
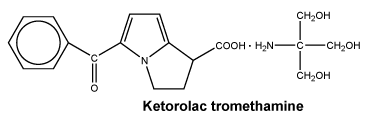
Empirical formula: C19H24N2O6.
Molecular weight: 376.41.
pKa: 3.54.
CAS number. 74103-07-4.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
06 March 1998
Date of Revision
03 December 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.