Aczone Gel
Brand Information
| Brand name | Aczone Gel |
| Active ingredient | Dapsone |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Aczone Gel.
Summary CMI
ACZONE™ gel
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ACZONE™ gel?
ACZONE™ gel contains the active ingredient dapsone. ACZONE™ gel is used to treat acne.
For more information, see Section 1. Why am I using ACZONE™ gel? in the full CMI.
2. What should I know before I use ACZONE™ gel?
Check the list of ingredients at the end of the CMI. Do not use ACZONE™ gel if you have ever had an allergic reaction to any of them.
Talk to your doctor before you take this medicine if he/she is not aware that you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ACZONE™ gel? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ACZONE™ gel and affect how it works, or ACZONE™ gel may interfere with other medicines and affect how they work.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ACZONE™ gel?
- Use ACZONE™ gel once every day.
- Your doctor will tell you how much to use and where and how to apply it.
- Continue to use ACZONE™ gel for as long as your doctor tells you.
More instructions can be found in Section 4. How do I use ACZONE™ gel? in the full CMI.
5. What should I know while using ACZONE™ gel?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ACZONE™ gel? in the full CMI.
6. Are there any side effects?
Dryness, itching, pain, redness, stinging/burning, scaling of treated skin, methaemoblobinemia (shortness of breath, bluish colouring of the skin, headache, tiredness/lack of energy, dizziness).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ACZONE™ gel
Active ingredient: dapsone 7.5% w/w
Consumer Medicine Information (CMI)
This leaflet provides important information about using ACZONE™ gel. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ACZONE™ gel.
Where to find information in this leaflet:
1. Why am I using ACZONE™ gel?
2. What should I know before I use ACZONE™ gel?
3. What if I am taking other medicines?
4. How do I use ACZONE™ gel?
5. What should I know while using ACZONE™ gel?
6. Are there any side effects?
7. Product details
1. Why am I using ACZONE™ gel?
ACZONE™ gel 7.5% w/w (75 mg per gram) contains the active ingredient dapsone which has anti-inflammatory and antimicrobial properties. The exact mechanisms of action of dapsone gel for treating acne are not known. It is thought to act by helping to eliminate microbes as well as having an anti-inflammatory effect in acne.
ACZONE™ gel is used to treat acne. Acne is a characterised by inflamed pimples and non-inflamed pimples on the skin.
Your doctor may have prescribed ACZONE™ gel for another reason. Ask your doctor if you have any questions about why ACZONE™ gel has been prescribed for you.
2. What should I know before I use ACZONE™ gel?
Warnings
ACZONE™ gel is for topical use only.
Do not use ACZONE™ gel:
- if you are allergic to dapsone or any of the ingredients listed at the end of this leaflet.
- if you have a blood condition affecting red blood cells known as methaemoglobinaemia, which causes a reduction in the amount of oxygen being transported around the body by the blood.
- on broken skin.
- if the pump/packaging shows signs of tampering and if the product does not look quite right.
- If the expiry date on the tube has passed.
Tell your doctor if you:
- have been diagnosed with a shortage of the enzyme G6PD (glucose-6-phosphate dehydrogenase).
- have been diagnosed with a blood disease which causes the breakdown of red blood cells, such as haemolytic anaemia.
- using any other medicines applied to the skin, such as acne medicines with benzoyl peroxide.
- take any medicines for any other condition.
- are taking oral dapsone and/or antimalarial medications.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed. It is not known if ACZONE™ gel may harm your unborn baby. The use of ACZONE™ gel is not recommended during pregnancy. You and your doctor will need to decide whether to discontinue nursing.
Children
ACZONE™ gel is for use in patients 12 years and over. It is not known whether ACZONE™ gel is safe and useful for children under 12 years for acne.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket, or health food shop.
Some medicines may interfere with ACZONE™ gel and affect how it works, while ACZONE™ gel may affect how other medicines work.
Medicines that may increase the effect of ACZONE™ gel include:
- dapsone when taken by mouth (i.e. orally)
- medicines to treat or prevent malaria
- trimethoprim and sulfamethoxazole (antibiotic)
If ACZONE™ gel is used with benzoyl peroxide, there may be a yellow or orange discolouring of the skin and/or hair of the treated area. This effect is temporary.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ACZONE™ gel.
4. How do I use ACZONE™ gel?
How much to use
ACZONE™ gel is for topical use only. Your doctor will prescribe you how much to use and where to apply it.
When to use ACZONE™ gel
For acne: apply a thin film of ACZONE™ gel once a day to the entire face as well as other areas of the body affected by acne. Treatment with ACZONE™ gel is usually recommended for 12 weeks (3 months). Your doctor will decide whether treatment should be continued at the end of this time period.
How to apply ACZONE™ gel
- Wash your face and gently pat dry. Ensure any make-up is completely removed.
- Dispense a pea-sized amount of gel onto your hand.
- Apply a thin layer of gel to the entire face.
- Use ACZONE™ gel on other affected areas of the skin where the acne lesions appear once daily as well. Rub the gel in gently to ensure it is completely absorbed. All skin prone to acne should be treated.
- Carefully avoid application of the gel to other areas that are not affected by acne.
- If the gel is applied to areas that are not required to be treated, wash the gel off.
- Wash your hands thoroughly after applying ACZONE™ gel.
If you forget to use ACZONE™ gel
It is important that you use ACZONE™ gel at the same time each day.
If you miss applying the gel, make sure you apply it at your next scheduled time.
Do not use double the amount to make up for the application you missed.
If you use too much ACZONE™ gel
- If you accidentally put ACZONE™ gel in your eye(s), immediately rinse your eye(s) with large amounts of cool water. If you have any concerns, contact your doctor for advice.
If ACZONE™ gel is accidentally swallowed, you should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia or 0800 POISON or 0800 764 766 in New Zealand), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there appears to be no signs of discomfort or poisoning.
5. What should I know while using ACZONE™ gel?
Things you should do
- Carefully follow your doctor's instructions.
- Keep ACZONE™ gel away from your eyes and mouth. If the gel gets in your eyes, wash them with large amounts of cool water. Contact your doctor if eye irritation continues. Do not use it on broken skin. Follow your doctor's directions for other routine skin care and the use of make-up. You should talk to your doctor about the use of sunscreens, moisturisers and cosmetics. Remind any doctor, dentist or pharmacist you visit that you are using ACZONE™ gel.
- Do not leave the top/lid off the container for any length of time to avoid contaminating the gel
- If you become pregnant while using ACZONE™ gel, stop using it and tell your doctor immediately.
- Tell your doctor if your condition gets worse or does not get better while using ACZONE™ gel.
Things you should not do
Looking after your medicine
- Do not refrigerate or freeze ACZONE™ gel. Keep your ACZONE™ gel in a cool place where the temperature stays below 25°C. Keep the container where children cannot reach it. Do not leave the top/lid off the container for any length of time to avoid contaminating the gel.
Follow the instructions on the bottle or carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight, for example:
- do not store it in the bathroom or near a sink, or
- do not store it in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard any remaining gel after 14 weeks from the date you opened it.
Getting rid of any unwanted medicine
If your doctor advises that you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine if the expiry date on the tube has passed. If you use this medicine after the expiry date has passed, it may not work effectively.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Check with your doctor as soon as possible if you have any problems while using ACZONE™ gel even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
- When the active ingredient of ACZONE™ gel, dapsone, is taken orally as a tablet, it has been related to the abnormal breakdown of red blood cells (haemolytic anaemia, which may present as back pain, breathlessness, weakness with daily activities, dark-brown urine, high fever, and yellow or pale skin). If you have glucose-6phosphate dehydrogenase (G6PD) deficiency, you may have a greater risk for lowering your hemoglobin level. Please note that using ACZONE™ gel on the skin is not expected to put enough dapsone in the blood to cause clinical symptoms of haemolytic anaemia, however you should be aware of the signs and symptoms suggestive of this type of anaemia.
Serious side effects
| Serious side effects | What to do |
Dapsone has been known to cause methaemoglobinaemia which is a blood disorder of red blood cells that carry oxygen to the body. Signs and symptoms suggestive of this blood disorder are
| Discontinue ACZONE™ gel and call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Less serious side effects
| Less serious side effects | What to do |
Effects on the skin treated:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems in Australia or to nzphvc.otago.ac.nz/reporting in New Zealand. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ACZONE™ gel contains
| Active ingredient (main ingredient) | Dapsone 7.5% w/w (Each gram contains 75mg of dapsone) |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What ACZONE™ gel looks like
ACZONE™ gel is an off-white to yellow gel supplied in an airless pump polypropylene bottle containing pack sizes 3g, 30 g, 60 g or 90 g of gel.
It is also supplied in a 3 g tube as a physician sample.
(Aust R 266267).
(Not all presentations may be available)
Who distributes ACZONE™ gel?
AbbVie Pty Ltd
241 O'Riordan Street
Mascot NSW 2020
AUSTRALIA
Toll free: 1800 252 224 (AU)
ACZONE™ gel is distributed in New Zealand by:
AbbVie Limited
Wellington 6011
NEW ZEALAND
Freephone: 0800 659 912
This leaflet was prepared in December 2025.
© 2026 AbbVie. All rights reserved.
ACZONE and its design are trademarks of Allergan, Inc., an AbbVie company.
Brand Information
| Brand name | Aczone Gel |
| Active ingredient | Dapsone |
| Schedule | S4 |
MIMS Revision Date: 01 October 2023
1 Name of Medicine
Dapsone.
2 Qualitative and Quantitative Composition
Each gram of Aczone gel, 7.5% w/w, contains 75 mg of dapsone.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Aczone is a topical gel.
4 Clinical Particulars
4.1 Therapeutic Indications
For the topical treatment of acne vulgaris in patients 12 years of age or older.
4.2 Dose and Method of Administration
For dermatological (topical) use only.
Aczone 7.5% w/w gel should only be applied to affected areas. For external use only. Not for oral, ophthalmic or intravaginal use. Not for use on broken skin. If contact with eyes occurs, rinse thoroughly with water.
After the skin is gently washed and patted dry, approximately a pea-sized amount of Aczone 7.5% w/w gel, should be applied in a thin layer to the entire face once daily. In addition, a thin layer may be applied to other affected areas once daily. Aczone 7.5% w/w gel should be rubbed in gently and completely.
Patients should be instructed to wash their hands after application of Aczone 7.5% w/w gel.
If there is no improvement after 12 weeks, treatment with Aczone 7.5% w/w gel should be reassessed.
4.3 Contraindications
Aczone 7.5% w/w gel is contraindicated in individuals who have shown hypersensitivity to any of its components.
Aczone 7.5% w/w gel is contraindicated in individuals with congenital or idiopathic methaemoglobinaemia.
4.4 Special Warnings and Precautions for Use
Haematological effects. Oral dapsone treatment has produced dose-related haemolysis and haemolytic anaemia. Individuals with glucose-6-phosphate dehydrogenase (G6PD) deficiency are more prone to haemolysis with the use of certain drugs. G6PD deficiency is the most prevalent in populations of African, South Asian, Middle Eastern and Mediterranean ancestry.
In clinical studies, there was no evidence of clinically relevant haemolysis or haemolytic anaemia in patients treated with topical dapsone.
Aczone 7.5% w/w gel, should be discontinued if signs and symptoms suggestive of haemolytic anaemia occur. Avoid use of Aczone 7.5% w/w gel in patients who are taking oral dapsone or antimalarial medications because of the potential for haemolytic reactions.
The combination of topical dapsone with trimethoprim/ sulfamethoxazole (TMP/SMX) may increase the likelihood of haemolysis in patients with G6PD deficiency.
Methaemoglobinaemia has been reported for oral dapsone and has been reported in postmarketing cases for topical dapsone. Patients with G6PD deficiency or congenital or idiopathic methaemoglobinaemia are more susceptible to drug-induced methaemoglobinaemia. Use of Aczone 7.5% w/w gel should be avoided in those patients with congenital or idiopathic methaemoglobinaemia.
Signs and symptoms of methaemoglobinaemia may be delayed for some hours after exposure. Initial signs and symptoms of methaemoglobinaemia are characterised by a slate grey cyanosis seen in, for example, buccal mucous membranes, lips and nail beds. Patients should be advised to discontinue Aczone 7.5% w/w gel and seek immediate medical attention in the event of cyanosis.
Dapsone can cause elevated methaemoglobin levels particularly in conjunction with methaemoglobin-inducing agents.
Peripheral neuropathy. Peripheral neuropathy (motor loss and muscle weakness) has been reported with oral dapsone treatment. No events of peripheral neuropathy were observed in clinical studies with topical dapsone treatment.
Skin. Skin reactions (toxic epidermal necrolysis, erythema multiforme, morbilliform and scarlatiniform reactions, bullous and exfoliative dermatitis, erythema nodosum, and urticaria) have been reported with oral dapsone treatment. With the exception of urticaria, these types of skin reactions were not observed in clinical studies with topical dapsone treatment.
Topical application of Aczone 7.5% w/w gel followed by benzoyl peroxide in patients with acne vulgaris may result in a temporary local yellow or orange discolouration of the skin and facial hair.
Use in the elderly. The safety and efficacy of Aczone 7.5% w/w gel have not been established in patients above the age of 65 years.
Paediatric use. The safety and efficacy of Aczone 7.5% w/w gel have not been established in patients under the age of 12 years.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Systemic exposure is lower with once daily Aczone 7.5% w/w gel than with twice daily dapsone gel, 5% w/w. No formal drug-drug interaction studies were conducted with Aczone 7.5% w/w gel.
Trimethoprim-sulfamethoxazole. A drug-drug interaction study evaluated the effect of the use of dapsone gel, 5% w/w in combination with double strength (160 mg/800 mg) TMP/SMX. During co-administration, systemic levels of TMP and SMX were essentially unchanged, however, levels of dapsone and its metabolites increased in the presence of TMP/SMX. The systemic exposure from Aczone 7.5% w/w gel is expected to be about 1% of that from the 100 mg oral dose, even when co-administered with TMP/SMX.
Topical benzoyl peroxide. Topical application of Aczone 7.5% w/w gel followed by benzoyl peroxide in patients with acne vulgaris may result in a temporary local yellow or orange discolouration of the skin and facial hair.
Concomitant use with drugs that induce methaemoglobinaemia. Concomitant use of Aczone 7.5% w/w gel with drugs that induce methaemoglobinaemia such as sulfonamides, acetaminophen, acetanilide, aniline dyes, benzocaine, chloroquine, dapsone, naphthalene, nitrates and nitrites, nitrofurantoin, nitroglycerin, nitroprusside, pamaquine, para‐aminosalicylic acid, phenacetin, phenobarbital, phenytoin, primaquine and quinine may increase the risk for developing methaemoglobinaemia (see Section 4.4 Special Warnings and Precautions for Use).
There is currently limited data on effects of use with topical antibiotics or topical retinoids.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The effects of dapsone on fertility and general reproductive performance were assessed in male and female rats following oral (gavage) dosing. Sperm motility was decreased in male rats that received 2 mg/kg/day or more, with a reduction in sperm count and density also observed in rats treated with 3 mg/kg/day or more (approximately 16 and 24 times the systemic exposure observed in humans under maximal topical use conditions based on AUC comparisons). Reductions in the mean numbers of embryo implantations and viable embryos in untreated females mated with males that had been dosed at 12 mg/kg/day or greater were likely due to reduced numbers or effectiveness of sperm, indicating impairment of male fertility (approximately 95 times the systemic exposure observed in humans under maximal topical use conditions based on AUC comparisons).
Dapsone reduced the mean number of corpora lutea and implantations in female rats treated with 30 mg/kg/day or more from 15 days prior to mating and for 17 days thereafter (approximately 900 times the systemic exposure observed in humans under maximal topical use conditions based on AUC comparisons). Maternal toxicity also occurred at these dose levels. Doses of 12 mg/kg/day did not affect the number of corpora lutea or implantations, which was associated with relative systemic exposures approximately 350 times than that expected clinically.
Use in pregnancy. (Category B3)
Use in pregnancy is not recommended.
There are no adequate and well controlled studies in pregnant women. Dapsone has been shown to have an embryocidal effect (increased early resorptions and decreased live litter size) when administered orally in rats at 75 mg/kg/day and in rabbits at 150 mg/kg/day (approximately 1400 and 400 times the systemic exposure observed in humans under maximal topical use conditions, based on AUC comparisons, respectively). These effects were associated with maternal toxicity.
Dapsone was assessed for effects on perinatal/ postnatal pup development and postnatal maternal behaviour and function in a study in which dapsone was orally administered to female rats daily beginning around the time of implantation and continuing throughout lactation. Maternal toxicity (decreased bodyweight and food consumption) and developmental effects (increase in stillborn pups and decreased pup weight and growth during lactation) were seen at a dapsone dose of 30 mg/kg/day (approximately 900 times the systemic exposure observed in humans under maximal topical use conditions, based on AUC comparisons). In addition, oral dosing with the excipient diethylene glycol monoethyl ether (DGME) alone also increased the incidence of stillbirths in rats at a dose estimated to be only 2 times that given clinically based on body surface area. No effects were observed on the viability, maturation, behaviour, learning ability, or reproductive function of surviving pups following treatment with dapsone or DGME in pregnancy and lactation.
Use in lactation. Although systemic absorption of dapsone following topical application of Aczone 7.5% w/w gel is minimal relative to oral dapsone administration, it is known that dapsone is excreted in human milk. Because of the potential for oral dapsone to cause adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue Aczone 7.5% w/w gel taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
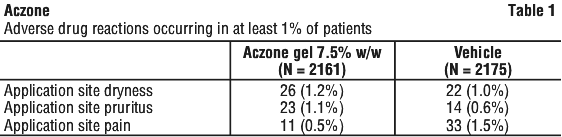
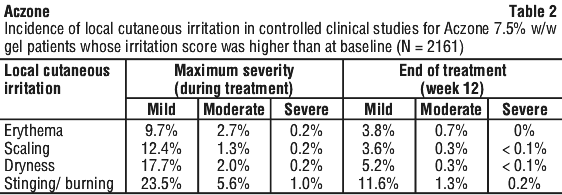
Pre-marketing clinical trials. A total of 2161 patients were treated with Aczone 7.5% w/w gel for 12 weeks in 2 controlled clinical studies. Adverse drug reactions that were reported in at least 1% of patients treated with either Aczone 7.5% w/w gel or vehicle appear in Table 1.
Most adverse drug reactions were mild in severity. One patient treated with Aczone 7.5% w/w gel discontinued the study due to application site pruritus, which resolved without sequelae.
Experience with oral use of dapsone. Although not observed in the clinical studies with topical dapsone, serious adverse reactions have been reported with oral use of dapsone, including agranulocytosis, haemolytic anaemia, peripheral neuropathy (motor loss and muscle weakness), and skin reactions (toxic epidermal necrolysis, erythema multiforme, morbilliform and scarlatiniform reactions, bullous and exfoliative dermatitis, erythema nodosum, and urticaria).
Post-marketing experience. Methaemoglobinaemia has been identified during postmarketing use of topical dapsone in clinical practice. Because it was reported voluntarily from a population of unknown size, estimates of frequency cannot be made (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Aczone 7.5% w/w gel is not for oral use. If oral ingestion occurs, medical advice should be sought.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The anti-inflammatory properties of dapsone result from inhibition of granulocyte cytotoxicity, via inhibition of peroxidases and scavenging of reactive oxygen species. The antimicrobial properties of dapsone result from competitive inhibition of dihydropteroate synthase, a bacterial enzyme necessary for synthesis of folic acid. The mechanism of action of dapsone gel in treating acne vulgaris is not known.
Clinical trials. The safety and efficacy of once daily use of Aczone 7.5% w/w gel was assessed in two large 12-week multicentre, randomised, double-blind, vehicle-controlled studies (studies 1 and 2). Efficacy was assessed in a total of 4340 patients 12 years of age and older with moderate acne vulgaris, 20 to 50 inflammatory and 30 to 100 non-inflammatory lesions at baseline, who were randomised to receive either Aczone 7.5% w/w gel or vehicle, where vehicle has the same formulation as Aczone 7.5% w/w gel without the active ingredient dapsone.
The co-primary efficacy variables for these studies were:
Proportion of patients with either 0 (none) or 1 (minimal) on the Global Acne Assessment Score at week 12.
Mean reduction in absolute lesion count from baseline at week 12 in: inflammatory lesions; non-inflammatory lesions.
Additional efficacy variables were:
Mean reduction in absolute total lesion count from baseline at week 12.
Percent reduction in lesion count from baseline at week 12 in: total lesions; inflammatory lesions; non-inflammatory lesions.
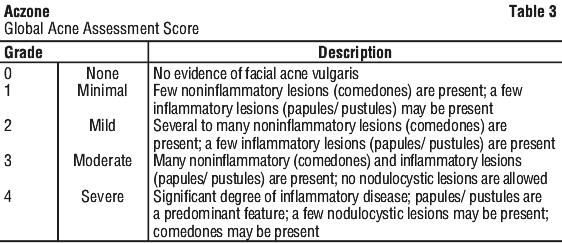
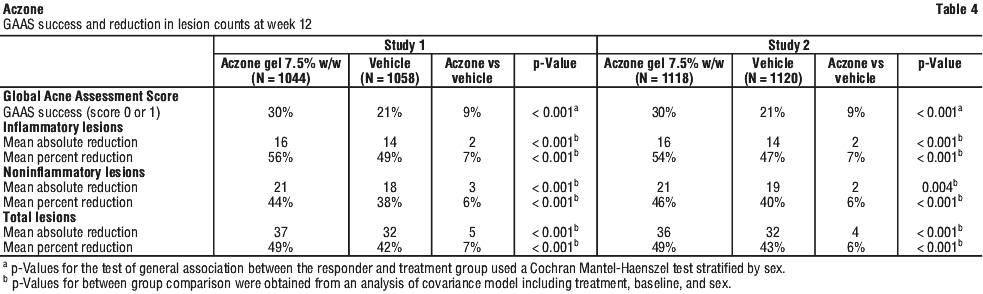
Success was defined as a score of "none" (score 0) or "minimal" (score 1) on the Global Acne Assessment Score (GAAS) at week 12. The GAAS used a 5-point scale as shown in Table 3. There was an improvement in GAAS and lesion count in both the Aczone 7.5% w/w and vehicle groups. There was a statistically significant treatment effect favouring Aczone 7.5% w/w gel compared to vehicle at week 12 (Table 4).
5.2 Pharmacokinetic Properties
Absorption. In a pharmacokinetic study, male and female subjects with acne vulgaris were randomised to receive either 2 gram of Aczone gel, 7.5% w/w, topically to the entire face, upper chest, upper back and shoulders once daily for 28 days (N = 19) or 2 gram of dapsone gel, 5%, topically to the same application area twice daily for 28 days (N = 18). Steady state for dapsone was reached within 7 days of dosing for both treatment groups. On day 28, the mean dapsone AUC0-24h was 282 ± 146 nanogram.h/mL for Aczone gel, 7.5% w/w, given once daily, whereas the mean dapsone AUC0-24h was 379 ± 142 nanogram.h/mL for dapsone gel, 5%, given twice daily. The daily systemic exposure following once daily application of Aczone gel, 7.5% w/w, was approximately 28.7% lower relative to dapsone gel, 5%, given twice daily. The systemic exposure from Aczone gel, 7.5% w/w is expected to be about 1% of that from a 100 mg oral dose.
Distribution. Following oral administration, approximately 74% of dapsone is bound to plasma proteins.
Metabolism. Following oral administration, dapsone is metabolised by two major pathways to form N-acetyl dapsone and dapsone hydroxylamine.
Excretion. Following oral administration, approximately 85% of the administered dapsone is recovered in urine, mainly as soluble metabolites, and only a small fraction (5% to 15%) is excreted as unchanged drug in humans.
5.3 Preclinical Safety Data
Genotoxicity. Dapsone was not mutagenic in a bacterial reverse mutation assay (Ames test) using S. typhimurium and E. coli, with and without metabolic activation. Dapsone increased both numerical and structural aberrations in a chromosome aberration assay conducted with Chinese hamster ovary (CHO) cells. Systemic exposure to dapsone did not induce chromosomal aberrations in in vivo micronucleus assays conducted in mice and rats.
Carcinogenicity. Dapsone was not carcinogenic to rats when orally administered to females for 92 weeks or males for 100 weeks at dose levels up to 15 mg/kg/day (associated with AUC values greater than 460 times the systemic exposure observed in humans under maximal topical use conditions). At higher doses, dapsone has been reported to induce mesenchymal and thyroid tumours in rats.
Topical dapsone at 5% w/w, did not increase the rate of formation of ultraviolet light-induced skin tumours when topically applied to hairless mice in a 12-month photocarcinogenicity study.
6 Pharmaceutical Particulars
6.1 List of Excipients
Diethylene glycol monoethyl ether (DGME); methyl hydroxybenzoate; acrylamide/sodium acryloyldimethyltaurate copolymer; isohexadecane; polysorbate 80 and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
24 months.
6.4 Special Precautions for Storage
Store below 25°C. Do not refrigerate or freeze.
Keep tube tightly closed when not in use.
6.5 Nature and Contents of Container
Gel: 7.5% w/w off-white to yellow gel supplied in 30 g, 60 g and 90 g airless pump polypropylene bottles. Also supplied as a 3 g physician sample in a tube.
6.6 Special Precautions for Disposal
Discard any remaining gel after 14 weeks from the date of opening.
6.7 Physicochemical Properties
Chemical structure.
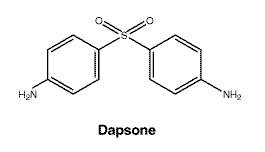
Empirical formula: C12H12N2O2S.
Molecular weight: 248.
CAS number. 80-08-0.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
10 January 2017
Date of Revision
22 August 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.