Adrenaline Juno 1:10,000
Brand Information
| Brand name | Adrenaline Juno 1:10,000 |
| Active ingredient | Adrenaline (epinephrine) |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Adrenaline Juno 1:10,000.
Summary CMI
Adrenaline Juno 1:10,000
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Adrenaline Juno 1:10,000?
Adrenaline Juno 1:10,000 contains the active ingredient adrenaline (epinephrine) (as acid tartrate). In some countries adrenaline is known as epinephrine. Adrenaline Juno 1:10,000 is usually only given in cases of extreme emergency. It may be used following a heart attack, or to make the heart beat if it has stopped. In heart conditions it can help to restart the heart and stimulates it to beat more strongly.
For more information, see Section 1. Why am I using Adrenaline Juno 1:10,000? in the full CMI.
2. What should I know before I use Adrenaline Juno 1:10,000?
Adrenaline is an emergency life saving product. However, tell your doctor if you have ever had an allergic reaction to adrenaline or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Adrenaline Juno 1:10,000? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with adrenaline and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Adrenaline Juno 1:10,000?
- Adrenaline Juno 1:10,000 will normally be given to you by a doctor, paramedic or specially trained nurse.
- The injection will be given directly into the vein (intravenously).
More instructions can be found in Section 4. How do I use Adrenaline Juno 1:10,000? in the full CMI.
5. What should I know while using Adrenaline Juno 1:10,000?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Adrenaline Juno 1:10,000? in the full CMI.
6. Are there any side effects?
Side effects may include fear, anxiety, confusion, tenseness, restlessness, headache, tremor, weakness, dizziness, cold hands and feet, nausea and vomiting, difficulty passing urine, fast or noticeable heart beat, chest pain, injection site reactions, difficulty breathing.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using Adrenaline Juno 1:10,000?
Adrenaline Juno 1:10,000 contains the active ingredient adrenaline (epinephrine) (as acid tartrate).
Adrenaline Juno 1:10,000 is usually only given in cases of extreme emergency. It may be used following a heart attack, or to make the heart beat if it has stopped. In heart conditions it can help to restart the heart and stimulates it to beat more strongly.
2. What should I know before I use Adrenaline Juno 1:10,000?
Warnings
Adrenaline Juno 1:10,000 is an emergency life-saving product. If possible, tell your doctor if:
- You are allergic to adrenaline, or any of the ingredients listed at the end of this leaflet.
- You have a history of medical conditions such as high blood pressure, heart conditions (ischemic heart disease, problems with the heart rhythm), diabetes mellitus, thyroid conditions, narrow angle glaucoma (high pressure in the eyes), brain damage, Phaeochromocytoma (a rare tumor of the adrenal gland).
In addition, Adrenaline Juno 1:10,000 should not be used:
- in shock (other than anaphylactic shock)
- during general anaesthesia
- with local anaesthesia
- during labour
- if the solution is discoloured.
Check with your doctor if you:
- Have any other medical conditions such as heart or vascular conditions, high blood pressure, kidney problems, diabetes, lung problems, thyroid problems, prostate problems, neurosis, glaucoma or parkinsonian syndrome.
- Have low potassium or high calcium levels in your blood.
- Take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Adrenaline should not be used in labor, as it can inhibit contractions.
Talk to your doctor if you are breastfeeding or intend to breastfeed. Adrenaline passes into breastmilk.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and adrenaline may interfere with each other. These include:
- some medicines used to treat coughs and colds
- medicines used to treat depression such as monoamine oxidase inhibitors and tricyclic antidepressants
- some antihistamines, medicines used to treat allergies
- thyroid hormones
- medicines which affect potassium levels such as diuretics, corticosteroids and theophylline
- some medicines used for high blood pressure or heart conditions, such as alpha-blockers, beta-blockers, quinidine and digoxin
- medicines to treat high blood sugar
- some medicines used to treat Parkinson's disease such as levodopa and catechol-O-methyl transferase inhibitors (COMT inhibitors)
- medicines known as parasympatholytics, used to treat asthma, urinary tract issues and gastrointestinal disorders
- general anaesthetics
- oxytocin, a medicine used to trigger uterine contractions for labour and delivery
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect adrenaline.
4. How do I use Adrenaline Juno 1:10,000?
How much to use
Adrenaline Juno 1:10,000 will normally be given to you by your doctor or by a specially trained nurse.
If you have to give Adrenaline Juno 1:10,000, your doctor will have told you what dose to use. Follow all directions given to you by your doctor carefully.
This may differ from the information contained in this leaflet.
The injection will be given straight into the vein. Adrenaline Juno 1:10,000 should not be given into the buttocks.
Children will be given a lower dose of Adrenaline Juno 1:10,000 depending on their weight.
This medicine is for injection only. If you will be giving yourself or anybody else injections make sure you know how to give them. Ask your doctor if you are unsure. If you have to use Adrenaline Juno 1:10,000 in an emergency tell your doctor immediately, or go to Accident and Emergency at your nearest hospital. You may need further medical treatment
If you use too much Adrenaline Juno 1:10,000
The doctor or nurse giving you Adrenaline Juno 1:10, 000 will be experienced in its use, so it is extremely unlikely that you will be given too much.
However, if you think that you have used too much Adrenaline Juno 1:10,000, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Adrenaline Juno 1:10,000?
Things you should not do
Do not use Adrenaline Juno 1:10,000 to treat any other complaints unless your doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how adrenaline affects you.
Adrenaline Juno 1:10,000 may cause side effects such as disorientation, confusion or dizziness in some people.
Looking after your medicine
This medicine will generally be stored in the pharmacy or on the ward.
If you are storing Adrenaline Juno 1:10,000 at home, it should be stored in the original pack in a cool dark place where the temperature stays below 25°C. Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Adrenaline Juno 1:10,000 contains
| Active ingredient (main ingredient) | Adrenaline (Epinephrine) acid tartrate |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Adrenaline Juno 1:10,000 looks like
Adrenaline Juno 1:10,000 is a clear, colourless solution in packs of 10 pre-filled syringes. … (Aust R 282377).
Who distributes Adrenaline Juno 1:10,000
Juno Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
www.junopharm.com.au
This leaflet was prepared in February 2026.
Brand Information
| Brand name | Adrenaline Juno 1:10,000 |
| Active ingredient | Adrenaline (epinephrine) |
| Schedule | Unscheduled |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Adrenaline (epinephrine) acid tartrate.
2 Qualitative and Quantitative Composition
Each prefilled syringe contains adrenaline (epinephrine) acid tartrate equivalent to 1 mg adrenaline in 10 mL solution. Each prefilled syringe also contains 90 mg of sodium chloride and water for injections. The pH of the solution is adjusted between 3.0 and 3.4 with hydrochloric acid and/or sodium hydroxide.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Adrenaline Juno 1:10,000 is a clear, colourless, practically free from visible particles, sterile and pyrogen-free solution for injection in a 10 mL polypropylene pre-filled syringe. Adrenaline Juno 1:10,000 injection contains no antimicrobial agent. It should be used only once and any residue discarded. It is a clear, colourless solution and should not be used if it is coloured.
4 Clinical Particulars
4.1 Therapeutic Indications
Adrenaline Juno 1:10,000 is used as an adjunct in the management of cardiac arrest.
4.2 Dose and Method of Administration
Adrenaline (epinephrine) injection contains no antimicrobial agent. It should be used only once and any residue discarded. Adrenaline (epinephrine) injection should not be used if it is coloured.
Cardiac arrest. Adults. The recommended dose is 1 mg intravenously, using 10 mL of the 1:10,000 solution. This may be repeated every 3-5 minutes. If given through a peripheral line, each dose should be followed by a flush of 20 mL of IV fluid to ensure delivery of the drug to the central compartment.
Intracardiac administration is no longer recommended.
Children. The recommended dose is 10 microgram (0.1 mL) per kg bodyweight administered intravenously. This may be repeated every 3-5 minutes.
4.3 Contraindications
Known hypersensitivity to sympathomimetic amines.
Shock (other than anaphylactic shock).
Cardiac dilatation and coronary insufficiency.
Hypertension.
Ischaemic heart disease.
Arrhythmias.
Cerebral arteriosclerosis.
Diabetes mellitus.
Hyperthyroidism.
Narrow angle (congestive) glaucoma.
Organic brain damage.
Phaeochromocytoma.
During general anaesthesia with halogenated hydrocarbons or cyclopropane.
With local anaesthesia in fingers, toes, ears, nose or genitalia - there is a danger of vasoconstriction producing sloughing of tissues in these areas.
Labour - it may delay the second stage by inhibiting spontaneous or oxytocin-induced contractions of the pregnant human uterus.
Conditions in which vasopressor drugs may be contraindicated e.g. thyrotoxicosis.
In obstetrics when maternal blood pressure is in excess of 130/80 mmHg.
Do not use if solution is discoloured.
(Also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.)
4.4 Special Warnings and Precautions for Use
The patient/carer should be informed about the possibility of biphasic anaphylaxis which is characterised by initial resolution followed by recurrence of symptoms sometime later.
Other beta-agonist sympathomimetics. Allow sufficient time to elapse before or after administering another beta-agonist sympathomimetic agent to avoid additive effects.
Disease states. Use with extreme caution in the elderly, and in patients with cardiovascular disease, phenothiazine induced circulatory collapse, cerebrovascular insufficiency, diabetes, hypertension, chronic lung disease, angina pectoris, prostatic hypertrophy, psychoneurosis or hyperthyroidism.
Use with extreme caution in patients with long standing bronchial asthma and emphysema who have developed degenerative heart disease. Anginal pain may be induced when coronary insufficiency is present. Syncope has occurred following administration to asthmatic children. In patients with parkinsonian syndrome the drug increases rigidity and tremor.
In patients with high intraocular pressure in angle-closure glaucoma, adrenaline dilates the pupil (mydriasis) and produces an acute episode of angle-closure. Adrenaline reduces intraocular pressure in open-angle glaucoma, decreasing aqueous formation and increasing outflow facility.
The risk of toxicity is increased if the following conditions are pre-existing: structural cardiac disease, severe obstructive cardiomyopathy, hypokalaemia, hypercalcaemia, severe renal impairment, patients taking monoamine oxidase (MAO) inhibitors (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions), patients taking concomitant medication which results in additive effects, or sensitises the myocardium to the actions of sympathomimetic agents (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Prolonged use of adrenaline can result in severe metabolic acidosis because of elevated blood concentrations of lactic acid.
General anaesthesia. Concurrent use with cyclopropane, halogenated hydrocarbon or similar volatile anaesthetics may produce fatal ventricular arrhythmias.
Diabetic patients. A greater increase may be produced in heart rate, blood glucose, lactate, glycerol and free fatty acids when adrenaline (epinephrine) is administered to diabetic patients with autonomic neuropathy than in diabetics without neuropathy.
Circulatory support. When adrenaline (epinephrine) is used for circulatory support, correction of hypervolaemia, metabolic acidosis, and hypoxia or hypercapnia should be carried out beforehand or concomitantly.
Gangrene. Intra-arterial administration must be avoided as marked vasoconstriction may result in gangrene.
Local ischaemic necrosis can occur from repeated injections in one site.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Other sympathomimetic agents. Adrenaline (epinephrine) should not be administered concomitantly with other sympathomimetic agents because of the possibility of additive effects and increased toxicity.
Rapidly acting vasodilators. These can counteract the marked pressor effects of adrenaline (epinephrine).
General anaesthetics. Administration of adrenaline (epinephrine) in patients receiving cyclopropane, halogenated hydrocarbon or similar volatile general anaesthetics that increase cardiac irritability and seem to sensitise the myocardium to adrenaline (epinephrine), may result in arrhythmias including ventricular premature contractions, tachycardia or fibrillation and acute pulmonary oedema if hypoxia is present.
Cardiovascular drugs. Adrenaline (epinephrine) should not be used in patients receiving high dosage of other drugs, e.g. quinidine, digoxin and other cardiac glycosides, that can sensitise the heart to arrhythmias.
Antihypertensive therapy. Special care is advisable in patients receiving antihypertensive therapy as severe hypertension may result.
Alpha blockers. The vasoconstrictive and hypertensive effects of adrenaline are antagonised by alpha-adrenergic blocking agents such as ergot alkaloids and phentolamine. The administration of adrenaline (epinephrine) to patients receiving alpha blockers may result in both hypotension and cardiac-accelerating effects.
Beta blockers. The administration of adrenaline (epinephrine) to patients receiving non-selective beta blockers (e.g. propranolol) may result in severe hypertension, followed by a reflex bradycardia, due to stimulation of adrenergic receptors.
CNS and other drugs. Tricyclic antidepressants, some antidepressants, monoamine oxidase inhibitors (MAO inhibitors), catechol-O-methyl transferase inhibitors (COMT inhibitors), theophylline, oxytocin, parasympatholytics, some antihistamines, thyroid hormones, levodopa and alcohol may potentiate the effects of adrenaline (epinephrine), especially on heart rhythm and rate.
Patients on MAOIs should not receive sympathomimetic treatment.
Drugs causing potassium loss. The hypokalaemic effect of adrenaline (epinephrine) may be potentiated by other drugs that cause potassium loss, including corticosteroids, potassium-depleting diuretics and aminophylline or theophylline; patients receiving high doses of beta2-adrenergic agonists concomitantly should have their plasma-potassium concentration monitored.
Hypoglycaemic agents. Adrenaline (epinephrine) induced hyperglycaemia may lead to loss of blood sugar control in diabetic patients treated with hypoglycaemic agents. It may be necessary for diabetic patients receiving adrenaline to increase their dosage of insulin or oral hypoglycaemic drugs.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Adrenaline (epinephrine) has been administered to a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
However, the use of adrenaline (epinephrine) during labour is contraindicated because it may delay the second stage by inhibiting spontaneous or oxytocin-induced contractions of the pregnant human uterus.
Use in lactation. Adrenaline (epinephrine) is excreted in the breast milk. The use of adrenaline (epinephrine) in breast-feeding women is therefore not recommended.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of this registration.
4.8 Adverse Effects (Undesirable Effects)
Adrenaline (epinephrine) may cause reactions such as fear, anxiety, tenseness, restlessness, disorientation, impaired memory, confusion, irritability, hallucinations and psychotic states.
Headache, weakness, dizziness, anorexia, nausea and vomiting and difficulty in micturition with urinary retention may also occur.
Muscle tremor and hypokalaemia, psychomotor agitation, pallor, respiratory difficulty, hyperglycaemia, sweating, hypersalivation, cold extremities and insomnia have also been reported.
Palpitations, tachycardia (sometimes with anginal pain) and cardiac arrhythmias may also occur along with hypertension which in some instances may induce reflex bradycardia as can vasodilation with flushing and hypotension. Ventricular fibrillation may occur and severe hypertension may lead to cerebral haemorrhage and pulmonary oedema.
Overdosage or inadvertent IV injection of usual subcutaneous doses of adrenaline (epinephrine) may cause hypertension. Cerebrovascular or other haemorrhage and hemiplegia may result, especially in geriatric patients. Inadvertent IV injection of adrenaline (epinephrine) has also been reported to have caused convulsions, metabolic acidosis, and renal failure with anuria.
Repeated injections of adrenaline (epinephrine) can cause necrosis as a result of vascular constriction at the injection site. Prolonged use or overdosage of adrenaline (epinephrine) can result in severe metabolic acidosis.
Pulmonary oedema has been associated with excessive parenteral administration of adrenaline (epinephrine) and following topical aerosol application.
Gas gangrene which can be fatal has been reported following intramuscular injection of adrenaline (epinephrine)into the buttock or thigh. This appears to have been due to clostridium organisms on the skin being deposited into muscle tissue during injection, with the vasoconstrictor properties of adrenaline (epinephrine) enhancing the effects of the infection (see Section 4.2 Dose and Method of Administration).
High doses may result in ventricular arrhythmias.
Rigidity and tremor may be exacerbated in patients with Parkinsonism.
Syncopal episodes have been reported in children.
Psychiatric disorders may be exacerbated.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Overdosage with adrenaline (epinephrine) produces a rapid rise in blood pressure resulting in cerebrovascular haemorrhage, cardiac arrhythmias leading to ventricular fibrillation and death. Pulmonary oedema may also lead to death because of the peripheral constriction and cardiac stimulation produced.
Treatment. To counteract the pressor effects of adrenaline (epinephrine), use rapidly acting vasodilators, for instance nitrates or α-blocking agents.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Adrenaline (epinephrine) acts on both alpha and beta adrenergic receptors of tissues innervated by sympathetic nerves, except the sweat glands and arteries of the face. It is the most potent alpha receptor activator. Adrenaline (epinephrine) stimulates the heart to increased output; raises the systolic blood pressure; lowers diastolic blood pressure; relaxes bronchial spasm and mobilises liver glycogen, resulting in hyperglycaemia and possibly glycosuria.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Parenterally administered adrenaline (epinephrine) has a rapid onset and short duration of action. The circulating drug is metabolised by the liver and other tissues. The majority is taken up and metabolised by sympathetic nerve endings. Adrenaline (epinephrine) is excreted in the urine, mainly in the form of metabolites.
Adrenaline (epinephrine) crosses the placenta but not the blood-brain barrier. It is also distributed into breast milk. (See Section 4.6 Fertility, Pregnancy and Lactation).
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, water for injections, hydrochloric acid and/or sodium hydroxide.
6.2 Incompatibilities
Adrenaline (epinephrine) is incompatible with oxidising agents, alkalis, copper, zinc, iron, silver and other metals.
Adrenaline (epinephrine) has been reported to be incompatible with solutions containing the following: aminophylline, ampicillin sodium, amylobarbitone sodium, ascorbic acid, benzylpenicillin potassium, calcium chloride, calcium gluconate, cephalothin sodium, chloramphenicol sodium succinate, chlortetracycline hydrochloride, corticotrophin, diazepam, digitoxin, ergometrine maleate, erythromycin gluceptate, frusemide, hyaluronidase, hydrocortisone sodium succinate, methicillin sodium, nitrofurantoin, noradrenaline acid tartrate, novobiocin sodium, pentobarbitone sodium, procaine, prochlorperazine edisylate, promazine hydrochloride, sodium bicarbonate, sulfadiazine sodium, suxamethonium chloride, tetracycline hydrochloride, vancomycin hydrochloride, vitamin B complex with ascorbic acid, warfarin sodium.
This list is not intended to be comprehensive. Refer to standard texts for further information.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in the aluminium pouch and blister to protect from light and oxygen. Do not open the Aluminium pouch and blister until use. After opening of the pouch the product must be used immediately. Do not freeze.
6.5 Nature and Contents of Container
Adrenaline Juno 1:10,000 is available as 1 mg/10 mL (1:10,000) 10 mL. Prefilled syringe in a pack of 1 or 10.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical name: (1R)-1-(3, 4- Dihydroxyphenyl)-2- (methylamino) ethanol hydrogen (2R, 3R)-2,3- dihydroxybutanedioate.
Chemical structure.
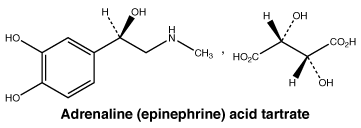
Molecular weight: 333.3.
Molecular formula: C13H19NO9 or C9H13NO3.C4H6NO6.
7 Medicine Schedule (Poisons Standard)
Unscheduled.
Date of First Approval
14 May 2018
Date of Revision
03 February 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.