ADT Booster
Brand Information
| Brand name | ADT Booster |
| Active ingredient | Diphtheria toxoid + Tetanus toxoid |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the ADT Booster.
Summary CMI
ADT™ Booster
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor, nurse or pharmacist.
1. Why are my child or I being given ADT™ Booster?
ADT™ Booster contains the active ingredients diphtheria toxoid and tetanus toxoid. ADT™ Booster is used to prevent the diseases diphtheria and tetanus in adults and children aged 5 years and over who have been previously vaccinated against these diseases.
For more information, see Section 1. Why are my child or I being given ADT™ Booster? in the full CMI.
2. What should I know before my child or I are given ADT™ Booster?
Do not use if you or your child have ever had an allergic reaction to ADT™ Booster, formaldehyde or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you or your child have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before my child or I am given ADT™ Booster? in the full CMI.
3. What if my child or I are taking other medicines?
Some medicines may interfere with ADT™ Booster and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is ADT™ Booster given?
- ADT™ Booster is an injection into a muscle and will be given to you by a trained health professional.
More instructions can be found in Section 4. How is ADT™ Booster given? in the full CMI.
5. What should I know while my child or I are being given ADT™ Booster?
| Things you should do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while my child or I are being given ADT™ Booster? in the full CMI.
6. Are there any side effects?
The most common side effects include reaction at the injection site such as temporary redness, pain, itching or swelling. Severe allergic reactions are possible but rare.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ADT™ Booster (ay-dee-tee boo-ster)
Active ingredients: Diphtheria toxoid and Tetanus toxoid
Consumer Medicine Information (CMI)
This leaflet provides important information about using ADT™ Booster. You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about ADT™ Booster.
Where to find information in this leaflet:
1. Why are my child or I being given ADT™ Booster?
2. What should I know before my child or I are given ADT™ Booster?
3. What if my child or I are taking other medicines?
4. How is ADT™ Booster given?
5. What should I know while my child or I are being given ADT™ Booster?
6. Are there any side effects?
7. Product details
1. Why are my child or I being given ADT™ Booster?
ADT™ Booster contains the active ingredients diphtheria toxoid and tetanus toxoid.
ADT™ Booster is a “combination” vaccine. It helps prevent two diseases, each caused by infection with a different germ (bacteria). The diseases are
- diphtheria and
- tetanus.
Both of these infections are serious and can be life-threatening.
ADT™ Booster is used to vaccinate children 5 years of age or older and adults who have previously received at least three doses of a vaccine for primary immunisation against diphtheria and tetanus. ADT™ Booster is not intended for primary immunisation against diphtheria and tetanus.
ADT™ Booster is given as one additional dose (booster dose) with intervals according to national recommendations.
How ADT™ Booster works
ADT™ Booster works by getting your body to produce its own protection against the two types of germs. The germs are those that cause two different and serious infections
- diphtheria and
- tetanus.
The vaccine does not contain live germs and cannot give you these illnesses.
After you have ADT™ Booster, your body makes substances called antibodies. These antibodies fight both the diphtheria and the tetanus germs. When you come into contact with these germs, your body is usually ready to destroy them.
Most people who receive the booster dose (suitable only if in the past they have had the full primary course against diphtheria and tetanus) will produce enough antibodies to protect against both the diphtheria and tetanus diseases. However, as with all vaccines, 100% protection cannot be guaranteed.
2. What should I know before my child or I are given ADT™ Booster?
Warnings
You should not be given ADT™ Booster if:
- You or your child has had had a serious reaction (including severe allergy) to ADT™ Booster before
- you or your child are allergic to diphtheria toxoid, tetanus toxoid or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
ADT™ Booster is not suitable for children under 5 years of age.
Check with your doctor if you or your child:
- Have an acute illness, with or without fever (your doctor may decide to delay vaccination until the illness has passed)
- are allergic to formaldehyde. Formaldehyde is used during the manufacturing process and trace amounts may therefore be present in the vaccine
- have any other medical conditions
- take any medicines for any other condition
- have low immunity due to disease or drug treatment
During treatment, you or your child may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the possible risks and benefits of having ADT™ Booster during pregnancy or while you are breastfeeding.
3. What if my child or I are taking other medicines?
Tell your doctor, nurse or pharmacist if you or your child are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
The following medicines may interfere with ADT Booster™ and affect how it works:
- medicines that suppress the immune system
Check with your doctor, nurse or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ADT™ Booster.
4. How is ADT™ Booster given?
ADT™ Booster is given by a trained health professional, as an injection into the muscle.
How much is given and when
For the booster dose, one dose of 0.5 mL is given.
Ask your doctor, nurse or pharmacist to answer any questions you may have.
If you or your child are given too much ADT™ Booster
Because each ADT™ Booster contains only one dose, overdosage is unlikely.
If you think that you or your child have been given too much ADT™ Booster, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while my child or I are being given ADT™ Booster?
Things you should do
Keep an updated record of your or your child's vaccinations.
Call your doctor, nurse or pharmacist straight away, or go straight away to the Emergency Department at your nearest hospital if you:
- have sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body
- have shortness of breath
- collapse.
Remind any doctor, dentist, nurse or pharmacist you visit that you or your child has been given ADT™ Booster.
Driving or using machines
Do not drive or use machines if you are feeling unwell after vaccination. Wait until any effects of the vaccine have worn off before you drive or use machines.
Looking after your medicine
ADT™ Booster is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store ADT™ Booster:
- Keep it where children cannot reach it
- Keep it in the original pack until it is time for it to be injected
- Keep it in the refrigerator, between 2°C and 8°C. DO NOT FREEZE ADT™ Booster. Freezing destroys the vaccine
- Do not use ADT™ Booster after the expiry date printed on the pack
- Do not use ADT™ Booster if the packaging is torn, shows signs of tampering, or does not look quite right.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines, including vaccines, can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, nurse or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Very common side effects (may affect more than 1 in 10 people):
| Speak to your doctor, nurse or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic Reactions:
Other reactions:
| Call your doctor, nurse or pharmacist straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor, nurse or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
After you have received medical advice for any side effects that you or your child experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor, nurse or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ADT™ Booster contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
The tip cap of the ADT™ Booster syringe contains latex (natural rubber). The ADT™ Booster syringe barrel, plunger rod and plunger stopper do not contain latex.
The ADT™ Booster vial and vial stopper do not contain latex.
The manufacture of this product includes exposure to cattle derived materials. There is no evidence that variant Creutzfeldt-Jakob disease, a brain condition linked to the consumption of cattle with “mad cow disease” has resulted from the administration of any vaccine product.
ADT™ Booster does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes, or preservatives.
What ADT™ Booster looks like
ADT™ Booster is supplied as a single dose (0.5 mL) in a needle-less pre-filled glass syringe (AUST R 130919) or vial (AUST R 130906). The vaccine should appear as a suspension of white or grey particles in a colourless or light yellow liquid.
Who distributes ADT™ Booster
In Australia:
Seqirus Pty Ltd
ABN 26 160 735 035
63 Poplar Road
Parkville VIC 3052
Telephone: 1800 642 865
www.cslseqirus.com.au
ADT™ is a trademark of Seqirus UK Limited or its affiliates.
This leaflet was prepared in November 2025
Brand Information
| Brand name | ADT Booster |
| Active ingredient | Diphtheria toxoid + Tetanus toxoid |
| Schedule | S4 |
MIMS Revision Date: 01 June 2022
1 Name of Medicine
Diphtheria and tetanus toxoids (adsorbed) (diphtheria and tetanus vaccine).
2 Qualitative and Quantitative Composition
ADT Booster is a suspension for intramuscular injection, containing aluminium hydroxide-adsorbed diphtheria and tetanus toxoids.
Each 0.5 mL dose contains no less than 2 International Units (IU) of purified diphtheria toxoid and no less than 20 IU of purified tetanus toxoid.
Each dose of ADT Booster also contains the following excipients: aluminium hydroxide hydrate corresponding to 0.5 mg aluminium, sodium chloride (4 mg), sodium hydroxide q.s. to pH 7, and water for injections.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalitis) has resulted from the administration of any vaccine product.
3 Pharmaceutical Form
Suspension for intramuscular injection. The vaccine should appear as a suspension of white or grey particles in a colourless or light yellow liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
Vaccination of children (≥ 5 years of age) and adults who have previously received at least 3 doses of a vaccine for primary immunisation against diphtheria and tetanus. ADT Booster is not intended for primary immunisation against diphtheria and tetanus.
Use of ADT Booster should be scheduled in accordance with official national recommendations.
4.2 Dose and Method of Administration
The dose of ADT Booster is 0.5 mL. Injections should be given by the intramuscular route.
For details of recommended vaccination schedules, including for tetanus prone wounds, refer to The Australian Immunisation Handbook of the NHMRC in Australia or the New Zealand Immunisation Handbook in New Zealand.
ADT Booster is recommended for re-vaccination after an initial primary course of vaccination.
The vaccine should be thoroughly shaken before use to ensure adequate dispersion when it is injected. The vaccine should appear as a suspension of white or grey particles in a colourless or light yellow liquid.
ADT Booster is for single use in one patient only. Discard any residue.
4.3 Contraindications
ADT Booster should not be administered to subjects who have previously experienced a serious reaction (e.g. anaphylaxis) to this vaccine or who are known to be hypersensitive to any of the vaccine components.
4.4 Special Warnings and Precautions for Use
As with other injectable vaccines, appropriate medical treatment and supervision should always be available in the event of anaphylactic reaction. Adrenaline should always be readily available whenever the injection is given.
ADT Booster is not intended for primary immunisation against diphtheria and tetanus.
Vaccination should normally be postponed in persons with moderate or severe acute illness, with or without fever.
Mild common illnesses are not contraindications to vaccination.
In children and adults with compromised immune response, the serological response may be impaired.
Vaccination of children and adults receiving immunosuppressive treatment can take place, but may result in a reduced immunological response.
Formaldehyde is used during the manufacturing process and trace amounts may be present in the final product. Caution should be taken in subjects with known hypersensitivity to formaldehyde.
Too frequent booster vaccination will increase the risk of adverse reactions.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No data available.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Pregnancy category A - Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
No relevant animal data are available.
No increase in frequency of malformations or other direct or indirect harmful effects on the foetus have been observed.
During pregnancy the possible risk of clinical infection following exposure should be weighed against the theoretical risks of vaccination.
Use in lactation. There is no evidence that vaccination of the breast-feeding mother with ADT Booster is harmful to the infant.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Following vaccination with ADT Booster, the most common adverse reactions are redness and swelling at the injection site and fever. These reactions most commonly start within 48 hours from the day of vaccination.
Systemic reactions reported for this type of vaccine include pruritus, rash, urticaria and peripheral oedema, anaphylactoid and hypersensitivity reactions, flu-like symptoms (including headache, rigors, asthenia, fatigue and myalgia), pyrexia, nausea, vomiting and dizziness. Postvaccinal neurologic disorders have been reported following the injection of almost all biological products and the possibility of their occurrence must be considered. Such disorders have included hypoaesthesia, paraesthesia and brachial radiculitis.
For the frequency of the adverse effects that have been reported for ADT Booster, see Table 1. The adverse reactions listed in Table 1 are based on data from clinical trials in children, adolescents and adults, and are classified according to MedDRA System Organ Class.
The safety evaluation of ADT Booster also includes adverse reactions from clinical trials and spontaneous reporting with vaccines containing the same or higher antigen content of diphtheria and tetanus than ADT Booster in combination with aluminium hydroxide and other vaccine antigens.
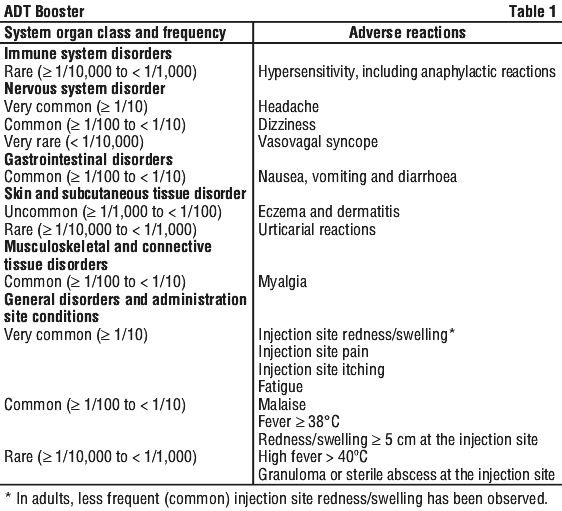
4.9 Overdose
There have been no cases of overdosage reported.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
In New Zealand, call the New Zealand Poisons Centre on 0800 POISON or 0800 764 766 for advice on overdosage management.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Following intramuscular injection, ADT Booster stimulates the immune system with the effect that antibodies are formed that protect against the diseases caused by exposure to Corynebacterium diphtheriae and Clostridium tetani. Protection against diphtheria and tetanus can be expected to last for up to 10 years.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
In the absence of compatibility studies, this vaccine must not be mixed with other vaccines or medicinal products.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
ADT Booster should be stored at 2°C to 8°C. It must not be frozen. Discard if vaccine has been frozen.
6.5 Nature and Contents of Container
ADT Booster can be supplied in a 0.5 mL needle-less pre-filled syringe or vial (Type 1 glass). Both these presentations may not necessarily be marketed.
Syringe and vial pack sizes: 1 x 0.5 mL and 5 x 0.5 mL.
ADT Booster does not contain preservatives or ingredients of human origin.
The tip cap of the ADT Booster syringe contains latex (natural rubber). The ADT Booster syringe barrel, plunger rod and plunger stopper do not contain latex.
The ADT Booster vial and vial stopper do not contain latex.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. No data available.
CAS number. No data available.
7 Medicine Schedule (Poisons Standard)
S4 Prescription Only Medicine.
Date of First Approval
29 January 2010
Date of Revision
08 April 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.