Albey Venom
Brand Information
| Brand name | Albey Venom |
| Active ingredient | Honey bee venom |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Albey Venom.
Summary CMI
ALBEY VENOM
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given ALBEY VENOM?
ALBEY HONEY BEE VENOM contains freeze dried allergen extracts of honey bee venom (known by species name apis millifera).
ALBEY YELLOW JACKET VENOM contains freeze dried allergen extracts of Vespula spp (yellow jacket wasp) venom (commonly known as European Wasp or Yellow Jacket).
ALBEY WASP VENOM contains freeze dried allergen extracts of paper wasp venom known by the species name polistes spp.
ALBEY VENOM Products are used to diagnose and treat allergic reactions to honey bee stings, wasp stings, and yellow jacket stings
For more information, see Section 1. Why am I being given ALBEY VENOM? in the full CMI.
2. What should I know before being given ALBEY VENOM?
Do not use if you have ever had an allergic reaction to ALBEY HONEY BEE /YELLOW JACKET/WASP VENOM or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before being given ALBEY VENOM? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ALBEY VENOM and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is ALBEY VENOM given?
- ALBEY VENOM is given as a subcutaneous injection (under the skin). It must only be given by a nurse or doctor.
- Your doctor will administer the correct dose as appropriate depend on the type of therapy decided upon by your doctor.
- Stay under medical observation for 60 min after injection. More instructions can be found in Section 4. How is ALBEY VENOM given? in the full CMI.
5. What should I know while being given ALBEY VENOM?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given ALBEY VENOM? in the full CMI
6. Are there any side effects?
Some of the less serious side effects include nausea, diarrhoea, dizziness, headache, rash or itching, pain or redness at the injection site, and feeling weak or unwell. Serious side effects include: signs of an allergic reaction (such as trouble breathing, wheezing, swelling of the face or throat, or hives), severe breathlessness, fast, slow or irregular heartbeat, and chest pain. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I being given ALBEY VENOM?
ALBEY VENOMs contains active ingredients which are freeze dried extracts of venom protein known as allergen extracts. There are three types of venom – Paper Wasp Venom (Pollistes spp); Vespula spp (commonly known as European Wasp or Yellow Jacket) and Honey Bee Venom (Apis Millefera). ALBEY VENOMs belong to a group of medicines called Anti-allergy preparations.
ALBEY VENOMS are used for both diagnosis and treatment of potential allergic reactions to the relevant insect stings. It is one type of immunotherapy your doctor may choose to administer to reduce the risk of a severe allergic reaction.
The exact mechanism by which ALBEY VENOM Products work is not completely understood. Your doctor, however, may have prescribed the ALBEY VENOM product for another purpose. ALBEY VENOMs are not addictive.
Ask your doctor if you have any questions why the ALBEY VENOM has been prescribed for you.
2. What should I know before being given ALBEY VENOM?
Warnings
Do not use ALBEY VENOM if:
- you are hypersensitive (allergic) to any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- If in addition to the allergic reaction, you have had any unusual kidney, muscle, joint, skin, nervous system effects, blood related reactions after a Honey bee, Yellow Jacket or Paper Wasp sting.
- If you have an illness which affects the immune system
- If you suffer from severe and/or unstable asthma or have experienced severe asthma exacerbation
- Children under the age of 2 years
- If you are pregnant
Check with your doctor if you:
- have severe allergic symptoms.
- If you have had recently an asthma attack or your asthma symptoms, get noticeably worse than normal. It may be necessary to postpone the injection to a later date.
- You have cardiovascular and/or bronchopulmonary disease.
- If you suffer from mast cell disorder (mastocytosis) and/or increased serum tryptase
- You have cancer or immunodeficiency or if you are taking medicines which affect your immune system.
- Autoimmune disease in remission.
- take any medicines for any other condition. Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking ALBEY VENOM.(refer to section 3. What if I am taking other medicines?
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. It is not known whether ALBEY VENOMs are harmful to an unborn baby when taken by a pregnant woman.
Talk to your doctor if you are breastfeeding or intend to breastfeed. It is not known whether ALBEY VENOMs pass into breast milk. Your doctor will discuss the risks and benefits of using ALBEY VENOMs if you are breast-feeding
Carrying out diagnostic skin tests with ALBEY VENOM products is not recommended during pregnancy. Venom allergy treatment with ALBEY VENOMS must not be initiated during pregnancy. If you find out that you are pregnant while you are having this treatment, talk to your doctor who will decide with you if you can continue treatment with ALBEY or not.
Relevant condition-specific or medicine-specific
Before you start to take it:
Severe local or systemic reactions to venom administration can occur immediately (within one hour) or as delayed reactions. Patients should be kept under direct observation for at least one hour following skin testing and/or therapeutic injections and should be instructed to contact the doctor promptly if symptoms of an allergic reaction or shock occur. Patients should be instructed in the use of, and have available, an emergency anaphylaxis kit for self-administration of adrenaline.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with the ALBEY VENOM Product and affect how it works.
Tell your doctor or pharmacist if you are taking any of the below medicines.
- You are taking a beta blocker (i.e., a class of drugs often prescribed for heart conditions and high blood pressure but also present in some eye drops and ointments), as this drug may decrease the effectiveness of adrenaline, a medicine used to treat serious allergic reactions.
- If you are taking MAOIs, tricyclic antidepressants and COMT inhibitors. Allergen immunotherapy in patients treated with mono amine oxidase inhibitors (MAOIs) or tricyclic antidepressants or for Parkinson's disease with COMT inhibitors should be considered carefully as these treatments could potentiate the effect of adrenaline (epinephrine).
- If you are taking Angiotensin converting enzyme inhibitors (ACE inhibitors) as this treatment may increase the risk of serious reactions to insect stings or hymenoptera venom immunotherapy.
If you have not told your doctor about any of the above, tell them before you start taking ALBEY VENOM.
In addition tell your doctor if you are taking other allergy medicines such as antihistamines, asthma relief medication or steroids or a medication that blocks a substance called immunoglobulin E (IgE), or if you are being treated with other allergens, or if you recently had or if you plan to have a vaccination soon.
Patients should also avoid high-intensity physical exercise, hot baths, heavy meals and excessive alcohol consumption for several hours after taking medication.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ALBEY VENOM Products.
4. How is ALBEY VENOM given?
How much is given
- Your doctor will administer the correct dose as appropriate. You should stay under medical observation for at least 60 min after injection. The dose administered will depend on the type of therapy decided upon by your doctor.
- The dose schedule for children is the same as for adults, and 100 microgram per venom should still be considered the maintenance dose. Because of the smaller size of the child, the larger volumes of solution may produce excessive discomfort. Therefore, in order to achieve the total dose required, the volume of the dose may need to be divided into more than one injection per visit.
- You will be given ALBEY VENOM according to your prescription. The duration of your treatment will be decided by your doctor. The longer the course of treatment the better the therapeutic effect of the drug. Therefore, the course of treatment may last from three to five years. Once a maintenance dose is reached the frequency of administration should decrease.
How ALBEY VENOM is given
- This medicine is available only with a doctor's prescription and is usually administered under a doctor's supervision.
- Albey venom immunotherapy will be given to you by your doctor.as a subcutaneous injection (under the skin).
- Immunotherapy injections should never be given intravenously.
If you are given too much ALBEY VENOM
If you think that you are given too much ALBEY VENOM, you may need urgent medical attention. In the event of overdose there is a higher risk of side effects, in particular for systemic or local reactions as described in 6.Are there any side effects? you must remain under medical supervision.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while being given ALBEY VENOM?
Things you should do
Tell all doctors, dentists and pharmacists who are treating you that you are being given ALBEY VENOM.
Tell your doctor if you become pregnant while being given ALBEY VENOM.
Be sure to keep all of your appointments with your doctor so that your progress can be checked.
Call your doctor straight away if you do not feel well while you are taking ALBEY VENOM.
In cases of anaphylactic shock that can cause a fall in blood pressure and suddenly feeling generally unwell, emergency treatment will be required. If this happens your doctor will have an emergency kit available with a 'ready for use' adrenaline syringe. This is the reason why you are kept under medical supervision for at least one hour after having your injection.
Things you should not do
- Do not take any other medicines, whether they require a prescription or not, without first telling your doctor or consulting a pharmacist.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how the ALBEY VENOM product affects you.
ALBEY VENOM products are not expected to affect your ability to drive a car or operate machinery.
Drinking alcohol
Tell your doctor if you drink alcohol. Patients should also avoid alcohol consumption on the day after taking Albey Venom injection.
Alcohol may affect the way ALBEY VENOM works in your body.
Looking after your medicine
Your medicine will generally be stored in the doctor's surgery or clinic, or at the pharmacy. It should be kept refrigerated between 2° to 8°C. It should not be frozen.
At the time of reconstitution, your doctor will record the date of reconstitution and expiration date of reconstituted product in the space provided (day, month, year) on the product label and store the reconstituted vial in the refrigerator at between 2° to 8°C.
6. Are There Any Side Effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. ALBEY VENOMs generally cause few side effects and help most people with hypersensitivity to insect stings, however they may have unwanted side effects in a few people.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects. Tell your doctor or pharmacist as soon as possible if you do not feel well while you are being given ALBEY VENOM.
Less serious side effects
| Less serious side effects | What to do |
General disorders
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General disorders
| Call your doctor straight away, tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the serious side effects that are listed here. |
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell, even if it is not on this list.
Ask your doctor or pharmacist if you don't understand anything in this list.
This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known. Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects in Australia to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ALBEY VENOM contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) | In all venom products
|
| Albumin saline diluent | Human albumin Phenol Sodium chloride |
Advise your doctor if you are allergic to any of these ingredients
What ALBEY VENOMs look like
ALBEY VENOMs are available in composite packs containing a single vial of freeze dried powder of venom allergenic extract, with 1 x 9 mL vial of Albumin saline diluent and 3 vials of 1.8 mL Albumin saline diluent.
The powder is white to slightly yellow; the diluent is without color or exogenous particles.
The presence of aggregates or flocs may be observed.
The powder is to be reconstituted and diluted only with Albumin-Saline diluent
Albey Bee Venom
Honey bee venom extract 550 micrograms per vial
Albey Paper Wasp Venom
Paper wasp venom (Polistes spp.) 550 micrograms per vial.
Albey Yellow Jacket Venom Injection (Injection)
Vespula spp. venom extract 550 micrograms per vial.
Albumin-Saline Diluent - available in 9 mL and 1.8 mL vials
To be used only for the dilution of the above Albey Venoms.
AUST R: 161298 (Albey Bee Venom)
AUST R: 18770 (Albey Yellow Jacket Venom)
AUST R: 18695 (Albey Paper Wasp Venom)
AUST R : 32486 (Albumin-Saline Diluent)
Who distributes ALBEY VENOM
NAME AND ADDRESS OF THE SPONSOR IN AUSTRALIA:
Stallergenes Australia Pty Ltd
Suite 2408, 4 Daydream St.,
Warriewood, NSW 2102
Ph: 1800 824 166
NAME AND ADDRESS OF THE SPONSOR IN NEW ZEALAND:
Stallergenes Greer New Zealand Limited
Level 1, 24 Manukau Road,
Epsom, Auckland 1023
New Zealand
Ph: 0800 824 166
This leaflet was prepared in January 2026
Brand Information
| Brand name | Albey Venom |
| Active ingredient | Honey bee venom |
| Schedule | S4 |
MIMS Revision Date: 01 February 2025
1 Name of Medicine
Albey Bee Venom (Apis mellifera), Albey Yellow Jacket Venom (Vespula spp. venom), Albey Paper Wasp Venom (Polistes spp. venom).
2 Qualitative and Quantitative Composition
The freeze dried honey bee venom consists of 550 microgram protein/vial.
The freeze dried wasp venom consists of 550 microgram protein/vial.
The freeze dried yellow jacket consists of 550 microgram protein/vial.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Powder and solvent for solution for injection.
4 Clinical Particulars
4.1 Therapeutic Indications
Albey Honey Bee Venom, Wasp Venom and Yellow Jacket Venom are indicated for the diagnosis and treatment of hypersensitivity to honey bee venom, wasp venom and yellow jacket venom respectively.
4.2 Dose and Method of Administration
Dosage schedules. Sensitivity to venom differs from patient to patient. Thus it is not possible to provide a dosage schedule suitable for all patients. The suggested dose schedule shown below gives an injection once per week and was used in clinical trials and should be suitable for a majority of patients.
Venom immunotherapy with Albey Honey Bee/ Yellow Jacket/ Wasp Venom consists of an initial phase (stepwise dose increase) followed by a maintenance phase.
Initial phase of treatment. The following alternative methods can also be used.
a. Rush immunotherapy with injections every two hours (in this case the patient must be hospitalised).
b. Modified rush immunotherapy with 2 to 3 injections per day once per week (given on an outpatient basis).
In extremely sensitive patients, however, an individualised dose schedule must be employed which will be dictated by the patient's sensitivity. This individualised schedule will probably include weaker dilutions and smaller increments between doses in progressing to the maintenance level (100 microgram-per venom).
In identifying those patients to be classified as extremely sensitive, individuals reacting with significant skin test (weal greater than 5 mm and erythema greater than 20 mm) at intradermal skin test concentrations of 0.01 microgram/mL or less, or those patients experiencing a systemic reaction to any venom skin test concentration, should be considered highly sensitive.
The suggested dose schedule for immunotherapy with a single venom is shown in Table 1.
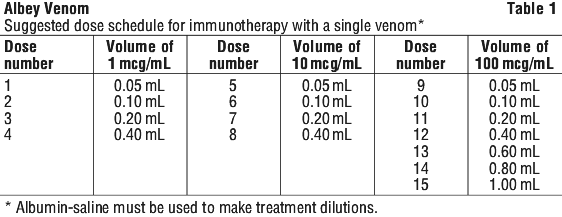

Maintenance phase of treatment. The recommended maintenance dose is 100 microgram of venom extract (1.0 mL of the 100 microgram/mL concentration).
This dose is given on an outpatient basis at intervals which are gradually increased to four weeks. Injections thereafter are given once per month.
In a previous clinical study with venom products, injections (using this suggested dose schedule) were given once per week at one study centre and twice or more per week at another centre. (For further discussions, see below). It must be considered important to achieve the 100 microgram per venom maintenance dose, since there is no data on effectiveness of maintenance levels below 100 microgram per venom.
In deciding the criteria for proceeding from dose to dose of the suggested dose schedule (see above), the results of a 1978-79 clinical study should be considered. A study centre 'A' reporting the least number of systemic reactions during pre-maintenance treatment held the dose constant in most of the cases where significant local reactions occurred. With the systemic reactions reported, this centre held the dose the same in approximately 80% of the incidences. The treatment injections were given at this centre usually once per week, and if a patient missed an appointment, the next dose was often the same as the preceding dose (depending on the previous reactivity of the patient). Patients treated at this centre reached maintenance in an average of 17 to 19 visits.
Another study centre 'B' reporting a higher incidence of systemic reactions, was more regimented in following the suggested dose schedule. This centre reduced or held the dose the same in less than 10% of the cases reporting significant local reactions. With the systemic reactions reported, this centre held the dose the same or reduced the dosage in approximately 20% of the cases. This centre gave more than one injection per week at the outset as circumstances and sensitivity allowed. Patients treated at this centre reached maintenance in an average of 14 visits.
Therefore, in proceeding with the suggested dose schedule, or modified schedules (for highly sensitive patients), it is suggested that if a systemic, extremely large local (10 cm or more induration, or other severe local symptoms), or persistent and severe delayed local reaction occurs, the dose at the next visit should be held constant (or reduced, depending on judgment on severity of the reaction) as was done at study centre 'A' which reported the least number of systemic reactions during the course of therapy.
Following the achievement of maintenance level (100 microgram per venom), approximately 80% or more patients were given a second maintenance injection at a one week interval. The third maintenance injection was usually (in approximately 60% of the patients) at a two week interval. The next injection was usually within three weeks, and the patients were then injected for ongoing maintenance at approximately monthly intervals. It is suggested that if a systemic, extremely large local (10 cm or more induration, or other severe local symptoms), or persistent and severe delayed local reaction occurs following a maintenance injection, the dose at the next visit should still remain constant.
The optimum duration for hyposensitisation therapy is not known, so current recommendations are that maintenance injections be continued indefinitely, year around, particularly in patients experiencing life-threatening anaphylaxis after insect stings.
Interruption of treatment or exceeding of the recommended interval between two injections (unrelated to the occurrence of side effects). Venom immunotherapy requires strict adherence to the protocol.
If the treatment is interrupted, there is no specific protocol for resumption of treatment.
The prescribing doctor will adapt the modalities for resuming treatment according to each clinical case and the time since the last injection. A gradual resumption phase may be necessary to reach the maintenance dose.
Patients should be kept under close observation in a hospital setting when resuming treatment.
Adjustment when adverse reactions occur. In case of a systemic adverse reaction occurring during the initial phase with stepwise dose escalation, it may be necessary to reevaluate the treatment regimen. For example, it is possible to resume the protocol with the injection of the last or second last well tolerated venom dose by increasing the subsequent doses more slowly. Premedication with H1 antihistamines can also be established. In case of a large local injection site reaction, the dose may be split between two injections or injection sites. These injection site reactions are common but are not predictive of a systemic reaction. Antihistamine treatment taken a few hours before injection can help reduce their occurrence.
Paediatric population. The recommended dosage schedules are identical in adults, adolescents and children.
Venom immunotherapy is contraindicated in children younger than 2 years of age (see Section 4.3 Contraindications).
Few data are available in children younger than 5 years of age.
The benefit-risk balance should be carefully assessed before initiating bee/ yellow jacket/ wasp venom immunotherapy in children aged 2 to 5 years.
Because of the smaller size of the child, the larger volumes of solution may produce excessive discomfort. Therefore, in order to achieve the total dose required, the volume of the dose may need to be divided into more than one injection per visit.
Method of administration. The use of Albey Honey Bee/ Yellow Jacket/ Wasp Venom should be supervised by a doctor experienced in allergology and venom immunotherapy under strict control in hospital (see Section 4.4 Special Warnings and Precautions for Use).
Use for the diagnostic test prick testing. Prick testing should be done before intradermal testing. In both the prick and intradermal tests, a negative control test with diluent alone must be performed.
The flexor surface of the forearm is the usual location for skin testing. It is important that a separate sterile syringe and needle be used for each extract and each patient.
Prick tests are accomplished using a solution of 1 microgram/mL venom protein. See instructions on reconstituting and diluting the venom in Tables 1 and 2. One drop of the 1 microgram/mL venom protein solution is applied to the forearm, and the skin is pricked through the surface of the drop with a sterile 27 gauge needle. The prick is superficial and should not draw blood.
For prick tests, a positive reaction (reaction greater than diluent control) at the 1 microgram/mL concentration indicates a high level of sensitivity to the test venom. Patients showing a positive reaction to the prick test at this concentration should begin intradermal tests at concentrations of not more than 0.0001 to 0.001 microgram/mL. Patients with negative prick tests may begin intradermal tests at a concentration of 0.001 microgram/mL.
Intradermal reaction test. A volume of 0.02-0.03 mL should be used for intradermal testing. Introduce the needle into the superficial skin layers until the bevel is completely buried, then slowly inject 0.02-0.03 mL aliquot of the venom dilution, making a small bleb.
Start intradermal tests with the most dilute solution. If after 20 minutes no skin reaction is obtained, continue the intradermal testing using tenfold increments in the concentration until a reaction of 5 to 10 mm weal and 11 to 20 mm erythema is obtained, or until a concentration of 1 microgram/mL has been tested, whichever occurs first.
A patient should be considered sensitive to the test venom when a skin response of 5 to 10 mm weal, 11 to 20 mm erythema (or greater) occurs at a concentration of 1 microgram/mL or less, providing that this reaction is greater than that of the diluent control.
There is a refractory period of at least 2 weeks following a systemic allergic reaction to a hymenoptera sting during which a false negative skin test can be observed. As the duration of the refractory period may be longer, it may be preferred to perform the diagnostic test at least 4 to 6 weeks after the allergic reaction to a sting to avoid the possibility of a false negative.
Use in venom immunotherapy treatment. Subcutaneous route only.
Do not inject by the intravenous route.
The product should be administered by slow subcutaneous injection on the outer side of the arm or in the deltoid region, taking care to avoid intravenous injection.
Before each injection, carefully check the type of venom extract used, the dosage, volume and date of the previous injection (interval between each injection).
Patients who have multiple venom sensitivities should be given each specific venom injection in a separate site. Note which venom preparation is injected at a specific site, so that dosage of that venom preparation can be adjusted if an excessive local reaction occurs. In patients receiving more than one venom, there is theoretically a greater risk of systemic reactions.
The patient should be kept under medical observation for at least 60 minutes after each injection. Patients should be instructed to avoid factors that could trigger allergic reactions on the day of the injection such as: high-intensity physical exercise, hot baths, copious meals and alcohol consumption.
In case of febrile episode or signs of infection or inflammatory disease, it is recommended to suspend injections until the infection has resolved.
In cases of severe atopic dermatitis, it is recommended to treat the condition before initiating allergen immunotherapy. Injections should be suspended in case of acute exacerbations.
In asthmatic patients, the injection should be postponed in case of acute exacerbation until 24 to 48 hours after respiratory symptoms have resolved.
The conduct of venom immunotherapy must be regularly recorded on the treatment logbook that the patient must keep.
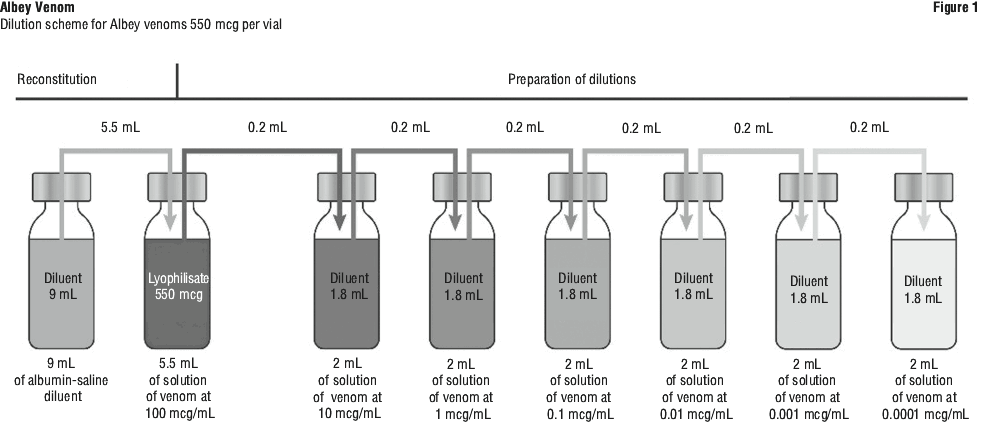
Reconstitution of the solution and dilutions. The solution is reconstituted by adding the adequate volume (5.5 mL in 550 microgram vial) of diluent in the vial of powder.
Mix gently until completely dissolved by turning the vial over several times. Do not shake to avoid foaming.
The resulting concentration in the vial is 100 microgram/mL.
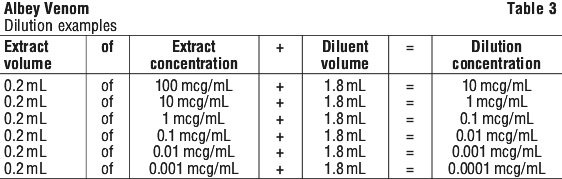
To obtain a concentration 10 times lower (10 microgram/mL), extract a volume of 0.2 mL of the reconstituted solution and transfer it into the vial containing 1.8 mL of solvent. This can be repeated to prepare successive 10-fold dilutions. Additional vials of solvent are provided separately if necessary, depending on the dilutions used.
Before taking the dose to be injected, be sure to turn the vial over several times in order to mix its contents carefully without shaking to avoid foaming.
For instructions on reconstitution and dilution of the medicinal product before administration, see Figure 1.
4.3 Contraindications
Hypersensitivity to any of the excipients listed in Section 6.1 List of Excipients.
Unusual reactions (renal, muscular, articular, cutaneous, neurological, haematological) after sting.
Active or poorly controlled auto-immune disorder.
Severe or uncontrolled asthma (FEV1 < 70% of predicted value).
Children under the age of 2 years.
The initiation of venom immunotherapy is contraindicated during pregnancy due to risk of the systemic effects.
4.4 Special Warnings and Precautions for Use
Hyposensitisation for insect sting allergy should be given to those patients who have experienced significant systemic reactions from insect stings and who demonstrate hypersensitivity by skin testing with these products.
Patients currently on whole body hymenoptera insect immunotherapy should be completely reevaluated by both history and venom skin testing before treatment with these venom products is initiated.
Risk of severe systemic reactions. Due to the risk of severe, potentially fatal systemic allergic reactions, any injection of this medicinal product must be carried out under the supervision of a doctor experienced in venom immunotherapy and in conditions where an emergency treatment is available (including adrenaline injection to manage a possible anaphylactic shock). Patients must be informed of the associated signs and symptoms requiring immediate medical attention. In the event of a systemic reaction, continuation of venom immunotherapy should be reconsidered by the doctor.
Asthmatic patients. As with any allergen immunotherapy, it is necessary to check, before each injection, that the asthma is well controlled.
In the event of a recent exacerbation of asthma, assessed clinically and/or by peak expiratory flow (PEF) and/or forced expiratory volume in 1 second (FEV1), treatment must be suspended and can only be resumed after improvement and on the opinion of the prescribing doctor.
Monitoring of patients with cardiovascular or bronchopulmonary risk factors. Due to an increased risk in case of systemic reactions, venom immunotherapy should be carried out under close observation in a hospital setting in patients with associated risk factors such as cardiovascular and/or bronchopulmonary disease. The underlying disease should be stabilized before venom immunotherapy initiation.
Mastocytosis. In patients suffering from mastocytosis and/or increased serum tryptase levels > 20 nanogram/mL, the risk of severe systemic reactions may be increased. In addition, venom immunotherapy may have less efficacy in these situations compared with the general allergic population. Venom immunotherapy in these patients should be carried out under close observation in a hospital setting.
Concomitant treatments. Tricyclic antidepressants, monoamine oxidase inhibitors (MAOIs) or COMT inhibitors. In the event of a severe allergic reaction, the use of adrenaline may be necessary. In patients treated with tricyclic antidepressants, monoamine oxidase inhibitors (MAOIs) or COMT inhibitors, which can increase plasma adrenaline concentrations, the risk of adverse effects related to adrenaline may be increased with a potentially fatal outcome. This risk should be taken into consideration before initiating venom immunotherapy.
Beta-blockers. Beta-blockers (including eye drops) interact with adrenaline which may then be less effective in treating possible anaphylactic reactions. This risk should be evaluated before initiating venom immunotherapy.
Angiotensin converting enzyme inhibitors (ACE inhibitors). Angiotensin converting enzyme inhibitors (ACE inhibitors) have been associated with an increased risk of serious reactions to insect stings or hymenoptera venom immunotherapy. The possibility of discontinuing ACE inhibitor therapy during hymenoptera venom immunotherapy should be considered. Concomitant administration of venom immunotherapy and an ACE inhibitor may be warranted in cases where there is no effective alternative to the ACE inhibitor and the individual benefit-risk balance is considered favourable.
When it is absolutely necessary to maintain these treatments in patients for whom hymenoptera venom immunotherapy is considered, the benefit-risk balance of the indication for this venom immunotherapy should be carefully assessed and the treatment can only be carried out under close supervision in a hospital.
Malignancies, immune defects, immunodeficiency, immunosuppression and auto-immune disease in remission. In the current state of knowledge, the effects of venom immunotherapy in subjects with cancer or acquired immunodeficiency are not clearly documented. Concomitant treatment with immunosuppressive agents may reduce the effectiveness of venom immunotherapy. Furthermore, caution should be exercised when prescribing venom immunotherapy in subjects with an auto-immune disease in remission.
Therefore, the benefits of the indication for venom immunotherapy should be carefully weighed against the risks in these situations.
Patients showing negative intradermal skin tests to specific venoms at 1 microgram/mL are not recommended for venom treatment.
Any injections, including immunotherapy, should be avoided in patients with a bleeding tendency.
Since routine immunisations have been suspected of exacerbating autoimmune diseases, immunotherapy should be given cautiously to patients with other immunological diseases and only if the risk from insect stings is greater than the risk of exacerbating the underlying disorder.
Venom sensitivity differs for individual patients, thus it is not possible to provide a dosage schedule that is universally suited to all patients. The dosage schedule shown under Section 4.2 Dose and Method of Administration is a summary of the schedule used in clinical trials of this product and found suitable for the majority of patients.
In highly sensitive patients, the doctor may be required to use a modified dose schedule, based on the patient's sensitivity to and tolerance of the injections.
Lower initial doses and smaller dosage increments than shown under Section 4.2 Dose and Method of Administration may be necessary.
Reconstitute the freeze dried venoms by adding 5.5 mL sterile diluent (albumin-saline) to the vial using a sterile syringe. Swirl or rock the container to dissolve the venom completely. Do not shake, since foaming leads to denaturation (inactivation) of protein.
Diluting fluid should be forcibly drawn into the sealed vial when the syringe needle penetrates the seal during reconstitution. Failure of this to occur for a particular vial indicates possible loss of vacuum. Discard vials without vacuum. In the event that an anaphylactic reaction occurs from overdose or inadvertent injection into the bloodstream, treat with adrenaline as directed under Section 4.8 Adverse Effects (Undesirable Effects).
A separate autoclave sterilised or disposable needle and syringe should be used for each patient to prevent transmission of homologous serum hepatitis and other infectious agents from one person to another.
Do not reinsert a needle into a diluent or into a vial containing a different venom, which has been previously inserted into a venom vial.
Aseptic techniques should always be employed when administering skin tests and/or treatment injections.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
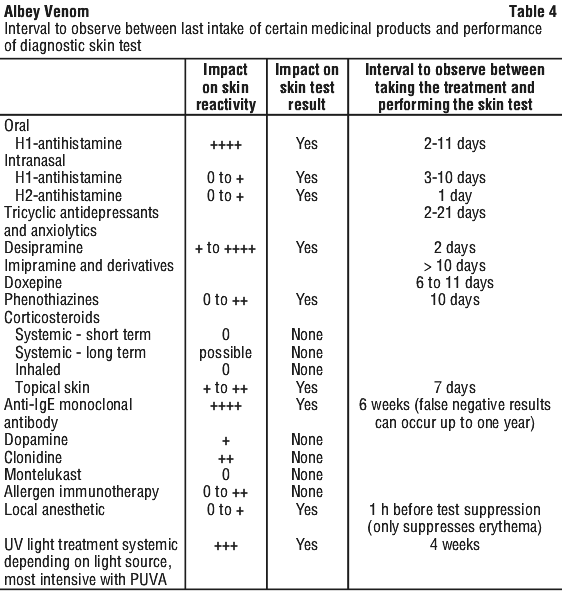
Medicinal products interfering with the diagnostic test. Some medicinal products inhibit skin reactivity. For this reason, it is necessary to observe an interval between intake of these medicinal products and performance of skin tests. This is particularly true for oral H1-antihistamines and anti-IgE monoclonal antibodies, but also for other drugs which are not necessarily used for the treatment of allergic disease such as anxiolytics. Topical skin corticoids may alter skin reactivity. Table 4 provides information for the interval to be observed between last intake of certain medicinal products and performance of diagnostic skin test.
Vaccine. There are no studies documenting the effect of allergen immunotherapy on vaccination. As a precaution, between 2 injections of Albey, it is recommended to observe an interval of 1 week before vaccination, and 2 weeks after vaccination, except in an emergency (see Section 4.4 Special Warnings and Precautions for Use).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No fertility studies were conducted with Albey hymenoptera venoms.
Use in pregnancy. Specific studies addressing risk to mother and fetus have not been done with venom products. Animal reproduction studies have not been conducted with Albey hymenoptera venoms.
There are no epidemiological studies on the use of Albey hymenoptera venoms in pregnant women.
Diagnostic skin tests with Albey hymenoptera venoms are not recommended during pregnancy so as not to expose the pregnant woman to the risk of a systemic allergic reaction.
Venom immunotherapy should not be initiated during pregnancy. As the risk of a systemic allergic reaction (anaphylactic shock) cannot be ruled out, even during the maintenance phase, the doctor should assess whether continuation of venom immunotherapy during pregnancy is warranted.
Use in lactation. There are no data on the excretion of Albey hymenoptera venoms in human milk.
No animal studies were conducted to investigate excretion of Albey hymenoptera venoms into milk.
The doctor should determine whether or not it is necessary to discontinue treatment with Albey hymenoptera venoms in light of the benefit of treatment for the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Doctors administering venom testing or treatment materials should be experienced in the treatment of severe systemic reactions (see Section 4.4 Special Warnings and Precautions for Use).
Patients should have available an emergency anaphylaxis kit containing adrenaline and be instructed in its use for emergency treatment of possible systemic reactions occurring at times after the patient has departed the treatment premises.
Treatment with Albey Honey Bee/ Yellow Jacket/ Wasp Venom can cause local allergic reactions at the injection site and/or systemic reactions. Local reactions can include large, painful or persistent local reactions. Large local reactions occurred in approximately 60% of patients given immunotherapy. None of the local reactions required specific treatment, however subsequent injections in many instances were held to the previous dose or a reduced dose. Some patients had repeated large local reactions that slowed the increase in the hyposensitisation dose.
Cases of anaphylactic shock with sudden cardiovascular collapse requiring immediate administration of adrenaline (epinephrine) have been reported.
The tolerance of a dose may vary over time according to the specific reactivity of the individual and their environment (depending on the patient's sensitivity, use of a modified dose schedule for immunotherapy treatment may be required; see Section 4.4 Special Warnings and Precautions for Use).
Systemic reactions may occur at any time after skin tests or hyposensitisation.
Severe allergic reactions including severe laryngopharyngeal disorder or systemic allergic reactions such as serious anaphylactic reactions (i.e. acute onset of an illness with involvement of the skin, mucosal tissue, or both, respiratory compromise, persistent gastrointestinal symptoms, or reduced blood pressure and/or associated symptoms) can occur.
Table 5 provides details of adverse reactions that have been observed in post-marketing reports.
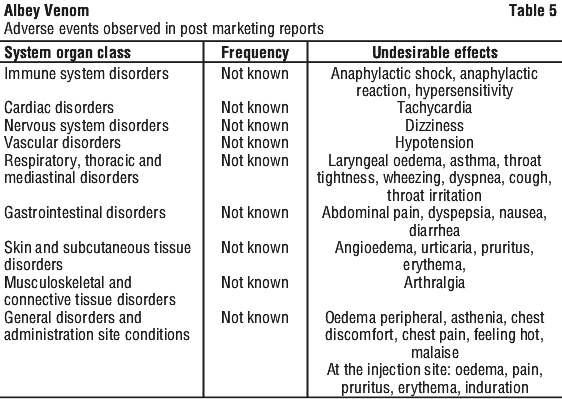
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Local reactions at the site of injection in the form of a weal or swelling occur frequently and are not cause for alarm, but if they persist, the dosage may need adjustment.
In case of injection of doses higher than those prescribed, the risk and severity of undesirable reaction may be increased leading to severe systemic reactions or local allergic reactions. See Section 4.8 Adverse Effects (Undesirable Effects).
Management. Treatment must be adapted to the patient's clinical state. The patient must remain under observation until complete resolution of symptoms.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. As per current guidelines, venom immunotherapy is based on the combination of positive diagnostic tests and a case history of severe systemic reactions.
The mechanism by which immunotherapy is achieved is not known completely. IgG antibodies (blocking antibodies) appear in the serum of patients treated with injected venom. No direct relationship has been identified between the level of blocking antibody (or the ratio of blocking antibody to IgE antibody directed to the same venom antigens) and the degree of immunotherapy. However, patients who show protection from symptoms after stings have been found to have raised levels of specific blocking antibody.
Initially, after a period of immunotherapy with specific venom antigens, levels of IgE antibody may increase. However, from studies carried out with other venom preparations, these levels are reported to decline after a time. After maintenance level has been reached and maintained, symptoms after stings have been shown to decrease considerably.
It is not known if skin sensitising antibody can be eradicated or if the patient can be entirely cured, nor is it known how long immunotherapy must be continued.
Skin testing with insect venoms is useful to demonstrate the presence of IgE antibodies which account for the patient's hypersensitivity symptoms. Patients are seldom able to identify the insect which stung them, so skin testing is used to determine the insect which stung them. Dilutions of these venom products will help judge the sensitivity of the patient and whether the patient should be treated.
It is not absolutely known what levels of venom that elicit positive skin tests are diagnostic of clinical sensitivity. However, patients with a history of reactions (any of three types: generalised urticaria or angioedema; respiratory difficulty due either to laryngeal oedema or to bronchospasm; or vascular collapse, with or without loss of consciousness) to previous stings and a positive skin test to a venom intradermal injection of approximately 1 microgram/mL had about a 60% chance of reacting again when stung by the same insect. These patients should receive venom immunotherapy.
Patients with a history of reaction (any of the three reaction types described above) to previous stings, but who did not demonstrate a positive skin test reaction to venom, are not recommended for immunotherapy treatment. There is no data to determine whether a patient who might react to a higher concentration, e.g. 2 to 10 microgram/mL, is at risk from subsequent stings. Since it is not known if sting sensitive patients who subsequently lose their IgE antivenom antibody can be resensitised by further stings, it is advisable to retest these patients after any subsequent stings. However, since the level of venom specific IgE may fall to low levels briefly.
After a sting, patients should not be retested until 2 to 4 weeks after any sting.
Immunotherapy is indicated for those patients diagnosed as sensitive (see Diagnosis) and is accomplished by using graded dilutions of the appropriate insect venom or venoms to control the severity of the patient's symptoms from subsequent stings.
Increasing doses of venom are given at intervals, dependent on the patient's ability to tolerate the venoms, until a maintenance dosage (100 microgram per venom is recommended) is reached and maintained. It is considered important that the patient be able to reach this dosage since the efficacy of lower maintenance dosages has not been established.
Actions. This product may be used for both diagnosis and immunotherapy.
Diagnosis. Diluted solutions of stinging insect venoms injected intradermally will produce weal and erythema reactions in patients who have significant IgE mediated, type I immediate hypersensitivity to stings of these insects.
Treatment. Repeated injections of increasing doses of insect venom extracts have been shown to ameliorate the intensity of allergic symptoms upon subsequent insect stings.
Clinical trials. In a clinical trial, three patients at the maintenance dosage of bee venom (100 microgram per venom) showed no systemic reaction following an insect sting challenge. The remaining ten patients were not challenged. The patients in this study reached maintenance (100 microgram per venom) usually within 2 ½ to 3 ½ months after beginning therapy. Whether efficacy of therapy is influenced by the time required to reach maintenance has not yet been determined.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
The freeze dried honey bee venom consists of mannitol 42.3 mg/vial and sodium chloride 1.72 mg/vial. The freeze dried wasp venom consists of mannitol 42.3 mg/vial. The freeze dried yellow jacket consists of mannitol 42.3 mg / vial.
Albumin-saline contains sodium chloride 0.9%, phenol 0.4% and normal human serum albumin 0.03% (see Section 2 Qualitative and Quantitative Composition).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Maintain stock solutions and dilutions constantly at 2 to 8°C. Refrigerate. Do not freeze.
At the time of reconstitution, record date of reconstitution and expiration date of reconstituted product in the space provided (day, month, and year) on the product label. Expiration date of the reconstituted venom depends on the type of reconstituting fluid used. Products reconstituted in albumin-saline have an expiration date of 6 months from date of reconstitution. Date of expiration after reconstitution must not exceed Final Expiration Date indicated on the container label (see Table 6 for expiration dates, including dilutions).

6.5 Nature and Contents of Container
Vacuum sealed vial (10 mL capacity), 550 microgram: 1's (plus diluent).
Albey Bee Venom - AUST R 161298.
Albey Yellow Jacket Venom - AUST R 18770.
Albey Paper Wasp Venom - AUST R 18695.
The albumin-saline diluent (AUST R 32486) is available in cartons consisting of the following pack sizes: 20 vials of 9.0 mL, 5 vials of 1.8 mL.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
Discard vials without vacuum. Each vial is for single patient use only.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. No data available.
CAS number. No data available.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine - S4.
Date of First Approval
28 April 2009
Date of Revision
04 December 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.