Alphagan P 1.5
Brand Information
| Brand name | Alphagan P 1.5 |
| Active ingredient | Brimonidine tartrate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Alphagan P 1.5.
Summary CMI
ALPHAGAN® P 1.5
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ALPHAGAN® P 1.5?
ALPHAGAN® P 1.5 eye drops contains the active ingredient brimonidine tartrate. ALPHAGAN® P 1.5 is used to lower raised pressure in the eye(s) and to treat glaucoma.
For more information, see Section 1. Why am I using ALPHAGAN® P 1.5? in the full CMI.
2. What should I know before I use ALPHAGAN® P 1.5?
Check the list of ingredients at the end of the CMI. Do not use ALPHAGAN® P 1.5 if you have ever had an allergic reaction to any of them.
Talk to your doctor before you use this medicine if he/she is not aware that you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ALPHAGAN® P 1.5? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ALPHAGAN® P 1.5 eye drops and affect how it works, or ALPHAGAN® P 1.5 may interfere with other medicines and affect how they work.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ALPHAGAN® P 1.5?
Your doctor will tell you how many drops you need to use each day. The usual dosage of eye drops is one drop in the eye(s) two times a day.
More instructions can be found in Section 4. How do I use ALPHAGAN® P 1.5? in the full CMI.
5. What should I know while using ALPHAGAN® P 1.5?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ALPHAGAN® P 1.5? in the full CMI.
6. Are there any side effects?
The most common side effect is redness of the eye or eyelids.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ALPHAGAN® P 1.5 (al-fah gan pee 1.5)
Active ingredient: brimonidine tartrate (brim-on-id-deen tar-trate)
Consumer Medicine Information (CMI)
This leaflet provides important information about using ALPHAGAN® P 1.5. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ALPHAGAN® P 1.5.
Where to find information in this leaflet:
1. Why am I using ALPHAGAN® P 1.5?
2. What should I know before I use ALPHAGAN® P 1.5?
3. What if I am taking other medicines?
4. How do I use ALPHAGAN® P 1.5?
5. What should I know while using ALPHAGAN® P 1.5?
6. Are there any side effects?
7. Product details
1. Why am I using ALPHAGAN® P 1.5?
ALPHAGAN® P 1.5 contains the active ingredient brimonidine tartrate.
ALPHAGAN® P 1.5 is used to lower raised pressure in the eye and to treat glaucoma.
Glaucoma is a condition in which the pressure of fluid in the eye may be high. However, some people with glaucoma may have normal eye pressure.
Glaucoma is usually caused by a build up of the fluid which flows through the eye. This build up occurs because the fluid drains out of your eye more slowly than it is being pumped in. Since new fluid continues to enter the eye joining the fluid already there, the pressure continues to rise. This raised pressure may damage the back of the eye resulting in gradual loss of sight.
Eye damage can progress so slowly that the person is not aware of this gradual loss of sight. Sometimes even normal eye pressure is associated with damage to the back of the eye.
There are usually no symptoms of glaucoma. The only way of knowing that you have glaucoma is to have your eye pressure, optic nerve and visual field checked by an eye specialist or optometrist.
If glaucoma is not treated it can lead to serious problems, including total blindness. In fact, untreated glaucoma is one of the most common causes of blindness.
ALPHAGAN P 1.5 eye drops lower the pressure in the eye by decreasing the fluid produced and helping the flow of fluid out of the eye chamber.
Although ALPHAGAN P 1.5 eye drops help control your glaucoma, it does not cure it.
ALPHAGAN P 1.5 eye drops are used, either alone or together with other eye drops/medicines, to lower raised pressure within your eye(s).
Your doctor may have prescribed ALPHAGAN P 1.5 eye drops for you instead of ALPHAGAN eye drops. The main difference between ALPHAGAN P 1.5 and ALPHAGAN is the preservative; ALPHAGAN P 1.5 contains 'Purite' (hence the 'P') whereas ALPHAGAN contains benzalkonium chloride.
ALPHAGAN P 1.5 belongs to a family of medicines called alpha-adrenergic agonists.
Your doctor may have prescribed ALPHAGAN P 1.5 eye drops for another reason. Ask your doctor if you have any questions about why ALPHAGAN P 1.5 eye drops have been prescribed for you.
2. What should I know before I use ALPHAGAN® P 1.5?
Warnings
Do not use ALPHAGAN® P 1.5 if:
- You are allergic to brimonidine tartrate, or any of the ingredients listed at the end of this leaflet.
- You are taking monoamine oxidase antidepressant medication
- Do not use in children younger than 2 years of age.
- The seal around the cap is broken
- The bottle/packaging shows signs of tampering
- The product does not look quite right
- The expiry date on the bottle or carton has passed. If you use this medicine after the expiry date has passed, it may not work.
If you are not sure whether you should start using ALPHAGAN P 1.5 eye drops, talk to your doctor.
Check with your doctor if you:
Have had an allergy to any other medicine or any other medicines or any other foods, preservatives, or dyes.
Have or have had any medical conditions, especially the following:
- liver or kidney disease
- severe, uncontrolled heart disease or poor blood flow to the heart
- poor blood flow to the brain
- depression
- Raynaud's syndrome. This is a condition marked by numbness, tingling and colour change (white, blue, then red) in the fingers when they are exposed to cold
- Dizziness on standing up, especially when getting up from a sitting or lying position
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed. Your doctor will discuss the possible risks and benefits of using ALPHAGAN® P 1.5 when pregnant or breast-feeding.
Use in children
- Children 2 years of age and above, especially those weighing ≤ 20 kg, should be treated with caution and closely monitored. Safety and effectiveness in paediatric patients have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ALPHAGAN® P 1.5 and affect how it works, while ALPHAGAN® P 1.5 may affect how other medicines work.
Do not take ALPHAGAN® P 1.5 if you are taking any of the following medicines:
- specific medicines to treat depression called "monoamine oxidase (MAO) inhibitors", such as phenelzine, tranylcypromine
Medicines where ALPHAGAN® P 1.5 may increase their effect include:
- barbiturates, traditionally used to treat epilepsy (seizures, fits)
- medicines used to relieve strong pain (such as opiates)
- medicines used to calm or put you to sleep
- anesthetics
- medicines used to treat high blood pressure, including beta blockers
- certain medicines used to treat asthma, severe headaches or coughs and colds, known as sympathomimetics.
- alcohol
Other medicines that are broken down by the liver may interact with ALPHAGAN® P 1.5.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ALPHAGAN® P 1.5.
4. How do I use ALPHAGAN® P 1.5?
How much to use
- Your doctor will tell you how many drops you need to use each day.
- The usual dosage of eye drops is one drop in the eye(s) two times a day (approximately every 12 hours).
Follow all directions given to you by your doctor carefully. The directions may differ from the information contained in this leaflet
When to use ALPHAGAN® P 1.5
- Use ALPHAGAN P 1.5 eye drops every day, at about the same time each day, unless your doctor tells you otherwise. Using your eye drops at the same time each day will have the best effect on your eye pressure. It will also help you remember when to use the eye drops.
In some instances, ALPHAGAN® P 1.5 needs to be used with other eye drops. Your doctor will let you know which drops, how to use them, and how long to use them.
Wait at least 5 to 10 minutes after using ALPHAGAN® P 1.5 before using other eye drops.
How to use ALPHAGAN® P 1.5
You may find it easier to put drops in your eye while you are sitting or lying down.
To open a new bottle of ALPHAGAN P 1.5 eye drops, first tear off the protective seal from the bottle. The contents are sterile if seal is intact. The seal will break and you can pull it off and then throw it away.
- Wash your hands well with soap and water.
- Shake the bottle gently.
- Remove the cap.
- Hold the bottle upside down in one hand between your thumb and forefinger or index finger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up.
- Put the tip of the bottle close to your lower eyelid. Do not let the tip touch your eye or eye lid.
- Release one drop into the pouch or pocket formed between your eye and eyelid by gently squeezing the bottle.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body. Ask your doctor for more specific instructions on this technique.
- Replace the cap, sealing it tightly.
- Wash your hands again with soap and water to remove any residue.
Be careful not to touch the dropper tip against your eye, eyelid or anything else to avoid contaminating the eye drops.
Contaminated eye drops may give you an eye infection.
ALPHAGAN® P 1.5 eye drops are for use in eyes only.
You may feel a slight burning sensation in the eye shortly after using the eye drops. If this persists, or is very uncomfortable, contact your doctor or pharmacist.
If you forget to use ALPHAGAN® P 1.5
It is important that you use ALPHAGAN® P 1.5 at the same time each day.
If you forget to use ALPHAGAN® P 1.5 and it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use the drops as soon as you remember, and then go back to using them as you would normally.
Do not take a double dose to make up for the dose you missed.
If you use too much ALPHAGAN® P 1.5
Immediately rinse your eyes with warm water.
If ALPHAGAN® P 1.5 is accidentally swallowed
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there appear to be no signs of discomfort or poisoning.
5. What should I know while using ALPHAGAN® P 1.5?
Things you should do
- Continue to use ALPHAGAN® P 1.5 as instructed by your doctor. ALPHAGAN® P 1.5 helps to control your condition but does not cure it.
- Use all the medication prescribed to treat your eye condition.
- Keep all your doctor's appointments so that your condition can be monitored, and your eye pressure can be checked.
- If you develop an eye infection, receive an eye injury, or have eye surgery tell your doctor. Your doctor may tell you to use a new container of ALPHAGAN P 1.5 eye drops because of possible contamination of the old one or may advise you to stop your treatment with ALPHAGAN P 1.5 eye drops.
Call your doctor straight away if you:
- suspect an eye infection (e.g. redness, discharge, pain)
- become pregnant while using ALPHAGAN® P 1.5.
Remind any doctor, dentist, pharmacist, or optometrist you visit that you are using ALPHAGAN® P 1.5.
Things you should not do
- Do not touch the dropper tip against your eye, eyelid or anything else, to avoid contaminating the eye drops.
- Do not stop using ALPHAGAN® P 1.5 without checking with your doctor. Your eye pressure can rise again and may cause damage to your eye.
Driving or using machines
- Wait for your vision to clear after using ALPHAGAN® P 1.5 before you drive or use machines.
- Do not drive or operate machines if you feel fatigued or drowsy while using ALPHAGAN® P 1.5.
Drinking alcohol
ALPHAGAN® P 1.5 may cause drowsiness and this can be made worse by drinking alcohol.
Looking after your medicine
Store below 25°C.
To avoid contamination of the solution, keep container tightly closed. Do not touch the dropper tip to any surface. Contents are sterile if seal is intact.
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example:
- do not store it in the bathroom or near a sink, or
- do not store it in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard the bottle 4 weeks after first opening it.
Write the date on the bottle when you open the eye drops and throw out the remaining solution after four weeks.
Eye drops contain a preservative which helps prevent germs growing in the solution for the first four weeks after opening the bottle. After this time there is a greater risk that the drops may become contaminated and cause an eye infection. A new bottle should be opened.
Getting rid of any unwanted medicine
If your doctor advises that you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not open the bottle to use this medicine after the expiry date has passed.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Effects on the eye(s)
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop using any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ALPHAGAN® P 1.5 contains
| Active ingredient (main ingredient) |
|
| Preservative |
|
| Other ingredients (inactive ingredients) |
|
Do not USE this medicine if you are allergic to any of these ingredients.
What ALPHAGAN® P 1.5 looks like
ALPHAGAN® P 1.5 is a clear, greenish-yellow solution. It is supplied in a plastic dropper bottle containing 5 mL of liquid.
(AUST R 158888).
Who distributes ALPHAGAN® P 1.5
ALPHAGAN® P 1.5 is distributed in Australia by:
AbbVie Pty Ltd
Mascot NSW 2020
Australia
Toll-free: 1800 252 224
For more information about glaucoma, contact Glaucoma Australia on 1800 500 880.
This leaflet was prepared in November 2025.
Version 1
© 2025 AbbVie. All rights reserved.
ALPHAGAN and its design are trademarks of Allergan, Inc., an AbbVie company.
Brand Information
| Brand name | Alphagan P 1.5 |
| Active ingredient | Brimonidine tartrate |
| Schedule | S4 |
MIMS Revision Date: 01 November 2023
1 Name of Medicine
Brimonidine tartrate.
2 Qualitative and Quantitative Composition
Each mL of Alphagan P eye drops contains brimonidine tartrate 1.5 mg (equivalent to 0.99 mg as brimonidine free base).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Eye drops, solution.
Alphagan P 1.5 0.15% is a sterile ophthalmic solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Alphagan P 1.5 eye drops are effective in lowering elevated intraocular pressure in patients with chronic open angle glaucoma or ocular hypertension. Alphagan P 1.5 eye drops can be used in the treatment of glaucoma as either monotherapy or in combination with topical beta-blockers.
4.2 Dose and Method of Administration
The recommended dose is one drop of Alphagan P 1.5 eye drops in the affected eye(s) twice daily, approximately 12 hours apart.
If more than one topical ophthalmic medicine is to be used, other eye drops should not be used within five to ten minutes of using Alphagan P 1.5 eye drops.
In order to minimise systemic absorption of Alphagan P 1.5 eye drops, apply pressure to the tear duct immediately following administration.
4.3 Contraindications
Alphagan P 1.5 eye drops are contraindicated in patients with hypersensitivity to brimonidine tartrate or any component of this medication. This product is also contraindicated in patients receiving monoamine oxidase (MAO) inhibitor therapy.
Alphagan P 1.5 eye drops are contraindicated in infants and children < 2 years of age.
4.4 Special Warnings and Precautions for Use
Identified precautions. Alphagan P 1.5 eye drops should be used with caution in patients with depression, cerebral or coronary insufficiency, Raynaud's phenomenon, orthostatic hypotension or thromboangiitis obliterans.
During the studies there was a loss of effect in some patients. The IOP-lowering efficacy observed with brimonidine eye drops during the first month of therapy may not always reflect the long-term level of IOP reduction. Patients prescribed IOP-lowering medication should be routinely monitored for IOP.
Cardiovascular disease. Although Alphagan P 1.5 eye drops had minimal effect on blood pressure and heart rate of patients in clinical studies, caution should be observed in treating patients receiving Alphagan P 1.5 with severe, uncontrolled cardiovascular disease.
Hypersensitivity. Delayed ocular hypersensitivity reactions have been reported with Alphagan, with some reported be associated with an increase in IOP.
Use in hepatic and renal impairment. Alphagan P 1.5 eye drops have not been studied in patients with hepatic or renal impairment; caution should be used in treating such patients.
Use in the elderly. No data available.
Paediatric use. Safety and effectiveness of Alphagan P 1.5 eye drops in children has not been established; however, during post-marketing surveillance, apnoea, bradycardia, coma, hypotension, hypothermia, hypotonia, lethargy, pallor, respiratory depression, and somnolence have been reported in neonates, infants, and children receiving brimonidine either for congenital glaucoma or by accidental oral ingestion. [See Section 4.3 Contraindications.]
Children 2 years of age and above, especially those weighing ≤ 20 kg, should be treated with caution and closely monitored due to the high incidence and severity of somnolence.
Information for patients. As with other alpha-agonists, brimonidine can potentially cause fatigue and/or drowsiness in some patients. Patients who engage in hazardous activities requiring mental alertness, such as driving and operating machinery, should be cautioned of the potential for a decrease in mental alertness.
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures to avoid eye injury and contamination of eye drops.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Although specific drug interaction studies have not been conducted with Alphagan P 1.5 eye drops, the possibility of an additive or potentiating effect with CNS depressants (alcohol, barbiturates, opiates, sedatives, or anesthetics) should be considered.
Because Alphagan P 1.5 eye drops may reduce blood pressure, caution using drugs such as beta-blockers (ophthalmic and systemic), antihypertensives and/or cardiac glycosides is advised.
Caution is advised when initiating or changing the dose of a concomitant systemic agent which may interact with alpha-adrenergic agonists or interfere with their activity (i.e. sympathomimetic agents, agonists or antagonists of the adrenergic receptor).
Tricyclic antidepressants have been reported to blunt the hypotensive effect of systemic clonidine. It is not known whether the concurrent use of these agents with Alphagan P 1.5 eye drops can lead to an interference in IOP lowering effect, although in rabbit experiments, tricyclic antidepressants did not alter the IOP response to brimonidine. No data on the level of circulating catecholamines after Alphagan P 1.5 eye drops are instilled are available. Caution, however, is advised in patients taking tricyclic antidepressants which can affect the metabolism and uptake of circulating amines.
As brimonidine is metabolised primarily by the liver, most likely by cytochrome P450 and aldehyde oxidase, this may affect the metabolism of other drugs that utilise the cytochrome P450 pathway.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Brimonidine did not have a significant effect on fertility in rats at oral doses of up to 0.66 mg/kg/day (ca 115 times the anticipated AUC in patients).
Use in pregnancy. (Category B3)
There are no studies of brimonidine in pregnant women. In rats, the drug crosses the placenta and enters the fetal circulation.
Because animal reproductive studies are not always predictive of human response, Alphagan P 1.5 should be used during pregnancy only if the potential benefit to the mother justifies the potential risk to the foetus.
In pregnant rats, brimonidine was associated with maternotoxicity and increased early resorptions/ post-implantation losses and decreased pup viability and body weights at estimated exposures (based on AUC) of 390 times the expected exposures in humans treated therapeutically. The drug was also maternotoxic in rabbits and caused abortions at exposures about 26 times greater than those expected in humans. In both rats and rabbits, brimonidine was not teratogenic.
Use in lactation. It is not known whether brimonidine is excreted in human milk. Therefore, a decision should be made whether to discontinue the drug, taking into account the importance of the drug to the mother. In lactating rats, levels of the drug in milk were up to 12 times higher than those in maternal plasma; and in a perinatal and postnatal study in rats, brimonidine was associated with decreased pup viability and pup weights during lactation at maternal plasma exposures of about 116 times greater than those expected in humans.
4.7 Effects on Ability to Drive and Use Machines
As with other alpha-agonists, brimonidine can potentially cause fatigue and/or drowsiness in some patients. Patients who engage in hazardous activities requiring mental alertness, such as driving and operating machinery, should be cautioned of the potential for a decrease in mental alertness.
Alphagan P 1.5 may also cause blurred vision or visual disturbance in some patients. The patient should wait until these symptoms have cleared before driving or using machinery.
4.8 Adverse Effects (Undesirable Effects)
The most commonly reported adverse reaction is conjunctival hyperaemia, occurring in 18.2% of patients. This is usually transient and does not normally require discontinuation of treatment.
Allergic conjunctivitis occurred in 9.2% of subjects (causing withdrawal in 7.4% of subjects) in clinical trials, with the onset between 3 and 9 months in the majority of patients.
The following undesirable effects considered to be at least possibly related to treatment were reported during two 12-month clinical trial studies where Alphagan P 1.5 eye drops were administered three times daily (see Tables 1 and 2):


Post-marketing experience. The following adverse reactions have been identified during post-marketing use of Alphagan P 1.5 in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
Immune system disorders. Not known: hypersensitivity.
Eye disorders. Not known: vision blurred, conjunctivitis.
General disorders and administration site conditions. Not known: fatigue, dizziness.
Nervous system disorders. Somnolence.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Adults. Ophthalmic overdose. In those cases received, the events reported have generally been those already listed as adverse reactions.
Systemic overdose resulting from accidental ingestion. There is very limited information regarding accidental ingestion of brimonidine in adults. The only adverse event reported to date was hypotension.
Treatment of an oral overdose includes supportive and symptomatic therapy; a patent airway should be maintained.
Paediatric population. Symptoms of brimonidine overdose such as apnoea, bradycardia, coma, hypotension, hypothermia, hypotonia, lethargy, pallor, respiratory depression, and somnolence have been reported in neonates, infants, and children receiving Alphagan as part of medical treatment of congenital glaucoma or by accidental oral ingestion.
Oral overdoses of other α2-agonists have been reported to cause symptoms such as hypotension, asthenia, vomiting, lethargy, sedation, bradycardia, arrhythmias, miosis, apnoea, hypotonia, hypothermia, respiratory depression and seizure.
Treatment of an oral overdose includes supportive and symptomatic therapy; a patent airway should be maintained.
In the event of a topical overdosage, flush eye with a topical ocular irrigant.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Brimonidine tartrate is an alpha-2 adrenergic agonist that is 1000-fold more selective for the alpha-2 adrenoreceptor than the alpha-1 adrenergic receptor. Affinities at human alpha-1 and alpha-2 adrenoreceptors are ~2000 nanoM and ~2 nanoM, respectively. This selectivity results in no mydriasis and the absence of vasoconstriction in microvessels associated with human retinal xenografts.
Topical administration of brimonidine solution decreases intraocular pressure (IOP) in humans. When used as directed, brimonidine eye drops have the action of reducing elevated IOP with minimal effect on cardiovascular parameters.
Brimonidine has a rapid onset of action, with the peak ocular hypotensive effect occurring at two hours post-dosing. The duration of effect is 12 hours or greater.
Fluorophotometric studies in animals and humans suggest that brimonidine tartrate has a dual mechanism of action. Alphagan P 1.5 eye drops lower IOP by reducing aqueous humor production and enhancing uveoscleral outflow.
Clinical trials. Elevated IOP presents a major risk factor in glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss. Brimonidine has the action of lowering intraocular pressure with minimal effect on cardiovascular and pulmonary parameters.
Studies with Alphagan eye drops. Monotherapy. The efficacy of Alphagan eye drops was demonstrated in two multicentre studies comparative with timolol 0.5% lasting up to one year in subjects with glaucoma or ocular hypertension. A total of 513 subjects received Alphagan eye drops in the two studies.
The overall mean decrease (± SD) in IOP from baseline at 12 months, as measured at peak response, was 6.20 ± 4.08 mmHg for brimonidine monotherapy and 5.56 ± 3.65 mmHg for timolol monotherapy. At trough response, these figures were 3.74 ± 3.83 mmHg for brimonidine and 5.80 ± 3.35 mmHg for timolol.
These results represent approximately 16% - 26% mean reduction from baseline measurements. IOP decreases were maintained for up to one year; no tachyphylaxis was observed. 9.4% of subjects treated with Alphagan eye drops and 5.1% of subjects treated with timolol 0.5% were discontinued because of inadequately controlled intraocular pressure. 30% of these patients withdrew during the first month of therapy.
Adjunctive therapy. The ability of Alphagan eye drops to lower IOP when used in combination with other anti-glaucoma agents has been evaluated in two large scale multicentre, randomised studies, involving 321 patients, 150 of which received brimonidine.
In the first study, brimonidine 0.2% twice daily as an adjunct to β-blocker therapy was compared with pilocarpine 2% administered three times daily, as an adjunct to β-blocker therapy. The overall mean decrease (± SD) in IOP from baseline at 3 months, as measured at peak response, was 4.92 ± 3.02 mmHg for brimonidine adjunctive therapy and 5.52 ± 3.08 mmHg for pilocarpine adjunctive therapy. At trough response, these figures were 3.95 ± 2.67 mmHg for brimonidine adjunctive therapy and 3.81 ± 2.75 mmHg for pilocarpine adjunctive therapy. These results represent a mean additional decrease in IOP for Alphagan adjunctive therapy of 17% - 22%.
The second study was an 8 month comparison of the additive IOP lowering effect to an already established β-blocker eye drop regimen, of Alphagan 0.2% eye drops or dipivefrine 0.1% eye drops. Adjunctive Alphagan eye drops was shown to be superior to adjunctive dipivefrine 0.1% at peak effect and equivalent in efficacy to adjunctive dipivefrine at trough at most time points.
The overall mean decrease (± SD) in IOP from baseline at 3 months, as measured at peak response, was 3.26 ± 3.16 mmHg for Alphagan adjunctive therapy and 2.33 ± 3.13 mmHg for dipivefrine adjunctive therapy. At trough response, these figures were 2.89 ± 3.14 mmHg for Alphagan adjunctive therapy and 3.31 ± 3.69 mmHg for dipivefrine adjunctive therapy. These results represent a mean additional decrease in IOP for brimonidine adjunctive therapy of 12% - 15%.
Studies with Alphagan P 1.5 eye drops. The efficacy and safety of Alphagan P 1.5 eye drops was demonstrated by comparison with that of Alphagan eye drops in a 3 month multicentre study involving 407 patients with glaucoma or ocular hypertension already controlled with Alphagan eye drops (study 017). Alphagan P eye drops used twice daily were found to provide non-inferior efficacy compared to Alphagan eye drops used twice daily, with the upper limit of the 95% confidence interval around the difference in mean IOP change from baseline between Alphagan P 1.5 and Alphagan being no more than 0.79 mm at any timepoint (NS). Alphagan P 1.5 eye drops also tended towards less overall adverse reactions than Alphagan eye drops (16.7% vs 22.1%) and less allergic conjunctivitis (3.9% vs 4.4%). The most frequently reported adverse reaction was conjunctival hyperaemia (7.9% vs 3.9%).
The long-term safety of Alphagan P 1.5 eye drops was confirmed by comparison with that of Alphagan eye drops in two multicentre studies of 12 months duration. In these studies, patients were randomised to brimonidine 0.15% (Alphagan P 1.5) eye drops three times daily, brimonidine-Purite 0.2% eye drops three times daily, or brimonidine 0.2% (Alphagan) eye drops three times daily. Pooled data from these studies demonstrated that Alphagan P 1.5 eye drops were associated with significantly less adverse reactions than Alphagan eye drops overall (49.7% vs 62.4%), as well as in terms of the following specific adverse reactions: allergic conjunctivitis (9.2% vs 15.7%), eye discharge (1.3% vs 3.9%), conjunctival hyperaemia (18.2% vs 25.6%) and oral dryness (5.3% vs 10.4%). Similarly, Alphagan P 1.5 eye drops were associated with significantly less adverse reactions than brimonidine-Purite 0.2% for allergic conjunctivitis (9.2% vs 14.6%) and oral dryness (5.3% vs 9.4%). Brimonidine-Purite 0.2% eye drops were also associated with less adverse reactions than Alphagan eye drops for allergic conjunctivitis (14.6% vs 15.7%) and oral dryness (9.4% vs 10.4%) suggesting a safety benefit from Purite substitution, even when brimonidine concentration was unchanged. These safety data support those of study 017, and demonstrate that Alphagan P 1.5 eye drops provide the most favourable safety profile with the lowest effective dose of brimonidine.
5.2 Pharmacokinetic Properties
Absorption. After ocular administration of a 0.1% and 0.2% solution of Alphagan P 1.5 eye drops three times daily for 7 days, plasma concentrations were low (mean Cmax was 0.03 nanogram/mL and 0.06 nanogram/mL for the 0.1% and 0.2% solutions, respectively). There was a slight accumulation in plasma after multiple instillations. The area under the plasma concentration time curve over 8 hours at steady state (AUC0-8h) was 0.14 nanogram.hr/mL and 0.25 nanogram.hr/mL for the 0.1% and 0.2% solutions, respectively. The mean apparent half-life in the systemic circulation was approximately 2 hours in humans after topical dosing.
Peak plasma brimonidine concentration (Cmax) is predicted to be 0.03 nanogram/mL when Alphagan P 1.5 is administered twice daily for 7 days.
Metabolism. In humans, brimonidine is primarily metabolised extensively in the liver.
Excretion. Urinary excretion is the major route of elimination of the drug and its metabolites. Approximately 87% of an orally-administered radioactive dose was eliminated within 120 hours, with 74% found in the urine.
The pharmacokinetics of Alphagan P 1.5 eye drops have not been specifically studied in patients with hepatic or renal disease (see Section 4.4 Special Warnings and Precautions for Use) or in paediatric patients (see Section 4.3 Contraindications; Section 4.2 Dose and Method of Administration).
5.3 Preclinical Safety Data
Genotoxicity. Brimonidine tartrate was non-genotoxic in assays for chromosomal damage (Chinese hamster cells in vitro, in vivo bone marrow cytogenetic assay and a dominant lethal assay). In assays for gene mutations in Salmonella typhimurium and Escherichia coli, brimonidine gave a positive response in one S. typhimurium strain without metabolic activation. Other strains gave negative results.
Carcinogenicity. No compound-related carcinogenic effects were observed in 21 month and 2 year studies in mice and rats given oral doses of 2.5 mg/kg/day and 1.0 mg/kg/day brimonidine respectively. Plasma concentrations of brimonidine in mice and rats in the high dose groups were at least 110 times greater than those expected in humans dosed therapeutically.
6 Pharmaceutical Particulars
6.1 List of Excipients
Active. Brimonidine tartrate 1.5 mg (equivalent to 0.99 mg as brimonidine free base).
Preservative. Sodium chlorite (as Purite) 1.8 microgram.
Inactives. Carmellose sodium, boric acid, borax, sodium chloride, potassium chloride, calcium chloride dihydrate, magnesium chloride hexahydrate, and purified water. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (6.6-7.4).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
18 months.
6.4 Special Precautions for Storage
Store below 25°C.
To avoid contamination of the solution, keep container tightly closed. Do not touch dropper tip to any surface. Contents are sterile if seal is intact.
6.5 Nature and Contents of Container
Alphagan P 1.5 (brimonidine tartrate ophthalmic solution) 0.15% sterile solution is supplied in plastic dropper bottles. Each bottle has a fill volume of 5 mL.
AUST R 158888.
6.6 Special Precautions for Disposal
Discard contents 4 weeks after opening the bottle.
6.7 Physicochemical Properties
Brimonidine tartrate is an off-white, pale yellow to pale pink powder and is soluble in water (34 mg/mL). In solution, brimonidine tartrate has a clear, greenish-yellow colour.
Chemical structure.
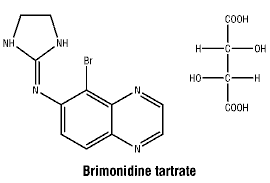
Molecular weight: 442.24 as the tartrate salt.
Empirical formula: C11H10BrN5, C4H6O6.
CAS number. 79570-19-7.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
25 May 2010
Date of Revision
19 September 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.