Alvesco
Brand Information
| Brand name | Alvesco |
| Active ingredient | Ciclesonide |
| Schedule | S4 |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Alvesco
Summary CMI
ALVESCO®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ALVESCO?
ALVESCO contains the active ingredient ciclesonide, a corticosteroid. When inhaled, ALVESCO helps to prevent asthma attacks in people aged 6 years and older.
For more information, see Section 1. Why am I using ALVESCO? in the full CMI.
2. What should I know before I use ALVESCO?
Do not use it if you have ever had an allergic reaction to ciclesonide, other steroids or ALVESCO or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you:
- have any other medical conditions (e.g., immune compromised, not vaccinated for chickenpox or measles or have any type of infection), damage to or recent surgery on the nose, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding or plan to breastfeed
- are switching from steroids that you take internally (e.g., by injection or by mouth), to ALVESCO
For more information, see Section 2. What should I know before I use ALVESCO? in the full CMI.
3. What if I am taking other medicines?
Tell your doctor about any other medicines you are taking. Some medicines may interfere with ALVESCO and affect how it works.
For more information, see Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ALVESCO?
Inhale ALVESCO into the lungs through your mouth for as long as the doctor tells you. Do not suddenly stop using it.
More information can be found in Section 4. How do I use ALVESCO? in the full CMI.
5. What should I know while using ALVESCO?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ALVESCO? in the full CMI.
6. Are there any side effects?
Common side effects may include: headache, fever, redness, swelling, pain or infection in the ear, nose throat or lungs. Other common side effects in children include: feeling or being sick, diarrhoea, upper abdominal pain, toothache. Serious side effects may include: frequent nosebleed, asthma, wheezing or problems breathing and hypersensitivity (allergic) reactions (rash, itching, hives on the skin; swollen face, tongue, throat or other parts of the body; shortness of breath, wheezing or problems breathing).
For more information including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using ALVESCO?
ALVESCO contains the active ingredient ciclesonide, a corticosteroid which, when inhaled into your lungs through your mouth, helps to prevent asthma attacks in people aged 6 years and older.
Asthma is a disease where the muscles around the airways become tight (bronchoconstriction) and the lining of the lungs becomes inflamed (swollen and irritated), making it hard to breathe.
When inhaled, ciclesonide acts directly on your air passages to reduce inflammation and help keep the air passages open. In doing so, it helps to improve your condition and to prevent asthma attacks from occurring. It is a “preventer” and needs to be used regularly.
If you have a sudden severe or continuous asthma attack you should use a different fast acting reliever inhaler, which contains a different medicine, to control wheezing or breathlessness, because ALVESCO works as a preventer rather than a reliever inhaler.
ALVESCO will NOT give you immediate relief of wheezing or breathlessness during sudden, severe or continuous asthma attacks. In this situation you should use a fast acting reliever inhaler, which contains a different medicine.
2. What should I know before I use ALVESCO?
Warnings
Do not use ALVESCO if:
- you are allergic to ciclesonide or any of the ingredients listed at the end of this leaflet
- you are having an acute asthma attack
- for children under 6 years of age
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions
- have had or are currently suffering from any liver problems
- are being treated for any infections
- have or have had tuberculosis (TB)
- are switching from steroids that you take internally (e.g., by injection or by mouth), to ALVESCO
- take any medicines for any other condition
While having ALVESCO, you may be at risk of getting certain side effects. It is important you understand these risks and how to watch for them. See extra information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Your doctor will talk to you about whether the benefits of having ALVESCO outweigh any possible effects on your unborn baby.
Talk to your doctor if you are breastfeeding or intend to breastfeed. Your doctor will talk to you about whether the benefits of having ALVESCO during breastfeeding outweigh any possible effects on your baby.
Having an operation
If you are going to have surgery, tell the surgeon or anaesthetist that you are using this medicine.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ALVESCO and affect how it works.
Medicines that may interfere with ALVESCO include:
- some antifungal medicines (e.g., ketoconazole, itraconazole)
- some antiviral medicines (e.g., ritonavir, nelfinavir)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ALVESCO.
4. How do I use ALVESCO?
How much to use
- ALVESCO is for use by inhaling into the lungs, through your mouth.
- Your doctor will normally start you on 80 micrograms once daily if you are not currently using a preventer medication. Otherwise your starting dose will depend on your current medication and how well your asthma is controlled.
- There are 2 strengths of ALVESCO and your doctor will have chosen the one which best suits your asthma.
- The number of puffs you are required to take daily will depend on the strength of ALVESCO you are using.
- Your doctor may adjust your dose so that you are taking the lowest dose of ALVESCO that will control your symptoms.
How to use the inhaler
Whenever possible, stand or sit in an upright position when inhaling.
If this is a new inhaler or if you have not used your inhaler for one week or more, it must be tested before use by removing the mouthpiece cover and pressing down on the canister inside the inhaler. Release 3 puffs into the air, away from you.
You do not need to shake your inhaler before using your medicine.
The medicine is already in a very fine solution, mixed to ensure you receive the correct dose with each puff.
Follow these instructions carefully and use the pictures to help you.
- Remove the mouthpiece cover and check inside and outside to make sure that the mouthpiece is clean and dry.

- Hold the inhaler upright with your forefinger on the top of the canister and your thumb on the base, below the mouthpiece.
- Breathe out as far as is comfortable.
- Place the mouthpiece in your mouth and close your lips firmly around it.
- Just after starting to breathe in through your mouth, press down on the top of the inhaler to release a puff while still breathing in slowly and deeply.
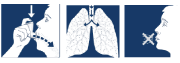
- Hold your breath, take the inhaler from your mouth and remove your finger from the top of the inhaler.
Continue holding your breath for approximately ten seconds or as long as is comfortable.
- Breathe out slowly through your mouth.

- If you have been instructed to take another puff, wait about half a minute and repeat steps 3 to 7.
- After use, always replace the mouthpiece cover to keep out dust.

Replace firmly and snap into position.
It is important that you do not rush steps 3 to 7.
A correct technique will ensure the right amount of ALVESCO is getting into your lungs every time you use your inhaler.
Care and Cleaning instructions
The mouthpiece of your inhaler should be cleaned weekly with a dry tissue or cloth.
DO NOT WASH OR PUT ANY PART OF THE ALVESCO INHALER IN WATER.
You should practice in front of the mirror for the first couple of times until you are confident that you are operating your inhaler properly. Make sure that none of your medicine is escaping from the top of inhaler or sides of your mouth.
If you find it difficult to use the inhaler with one hand, try using both hands. Put your two forefingers on top of the inhaler and both thumbs on the base below the mouthpiece. Tell your doctor, nurse or pharmacist if you are having difficulties.
When the canister is completely empty you will not feel or hear any of the propellant being discharged.
Use of inhaler with a spacer device
If you have problems with your inhaler technique, your doctor may recommend the use of a spacer device in combination with your inhaler. Different brands of spacers may affect the amount of medicine delivered to the lungs, so it is important that you be monitored for any loss of asthma control.
Before use, the spacer should be removed from the inhaler and washed in warm water and detergent and allowed to dry without rinsing or drying with a cloth. Wash the spacer at least monthly after use.
When using a spacer, you should inhale immediately after you press down on the top of the inhaler to release a puff.
Why would your Inhaler have stopped working?
Your inhaler may not work if:
- the mouthpiece is clogged and dirty
- the inhaler is empty (check by shaking the canister)
- the canister is wrongly positioned
If you forget to use ALVESCO
ALVESCO should be used every day at the same time.
If you miss your dose at the usual time, have it as soon as you remember.
If it is almost time for your next dose, skip the dose you missed and have your next dose when you are meant to.
Do not have a double dose to make up for the dose you missed.
If you have too much ALVESCO
If you think that you have used too much ALVESCO, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ALVESCO?
Things you should do
Tell your doctor straight away if:
- Your asthma symptoms suddenly get worse just after using ALVESCO. Use your fast acting reliever to manage your breathing in the meantime.
- You have severe wheezing or other problems breathing that are not being fixed by your fast-acting reliever inhaler.
- Your eyesight becomes blurred or you have other problems seeing. You may need to have your eyes checked by an eye specialist.
- You become pregnant while you are using ALVESCO.
Other things you should do:
- Keep using ALVESCO every day, even if your asthma is under control.
- Always keep a separate “fast-acting reliever” inhaler handy in case your breathing gets worse. ALVESCO is only used for preventing asthma, not treating it.
- Visit your doctor regularly. This is so they can watch your progress, test your lungs, change the dose if needed, and check any effects on your general health.
- Children and younger adults especially should see their doctor regularly so that they can be watched carefully for any effects on their growth.
- Tell your doctor if your asthma symptoms are getting worse - e.g., if you need to use more of your “fast-acting reliever” inhaler or the effect of your reliever puffers does not last as long as usual.
- Clean your inhaler once a week using a dry cloth or tissue.
Remind any doctor, dentist or pharmacist you visit that you are using ALVESCO.
Things you should not do
- Do not stop using this medicine unless your doctor tells you to. ALVESCO helps control your asthma. Therefore, you must use ALVESCO every day.
- Do not use ALVESCO to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
Do not change the amount you use without speaking to your doctor first.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ALVESCO affects you. ALVESCO is unlikely to affect your ability to drive and use machines.
ALVESCO contains alcohol
ALVESCO contains alcohol (ethanol). The amount in two puffs of this medicine is equivalent to less than 1 ml of wine or beer. The small amount of alcohol in this medicine will not have any noticeable effects, however talk to your doctor if you have been told to avoid alcohol for any reason.
Looking after your medicine
- Store the pack at room temperature, below 25°C. Do not refrigerate or freeze.
- Do not puncture or incinerate the canister even when it is empty as it may explode.
Follow the instructions above on how to look after your medicine.
When storing at room temperature, keep it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills.
Keep it where young children cannot reach it.
When to discard your medicine
- If your doctor or pharmacist tells you to stop using this medicine.
- If it is damaged.
- If the expiry date has passed.
- When you have used all of the doses.
Getting rid of any unwanted medicine
Take it to any pharmacy where it will be disposed of safely.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, nurse or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Digestive system
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Hypersensitivity (allergic) reaction:
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed may occur in some people.
Always speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What ALVESCO contains
| Active ingredient (main ingredient) | ciclesonide |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | potassium sorbate |
Do not take this medicine if you are allergic to any of these ingredients.
ALVESCO does not contain gluten, sucrose, lactose, tartrazine or any other azo dyes.
What ALVESCO looks like
ALVESCO is a colourless solution in an aluminium canister with a metering valve.
There are two strengths and two sizes each:
ALVESCO 80 (80 micrograms per puff): 60 or 120 inhalations
ALVESCO 160 (160 micrograms per puff): 60 or 120 inhalations
Not all pack sizes or strengths may be available.
ARTG Numbers:
ALVESCO 80: AUST R 93724
ALVESCO 160: AUST R 93725
Who supplies ALVESCO
Chiesi Australia Pty Ltd
Level 7, Suite 1, 500 Bourke Street,
Melbourne, VIC 3000
Email: medinfo.au@chiesi.com
Website: www.chiesi.com.au
This leaflet was prepared in September 2025.
Brand Information
| Brand name | Alvesco |
| Active ingredient | Ciclesonide |
| Schedule | S4 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Ciclesonide.
2 Qualitative and Quantitative Composition
Alvesco is available in the following strengths:
Alvesco 80: 80 microgram ciclesonide per actuation.
Alvesco 160: 160 microgram ciclesonide per actuation.
Excipient(s) with known effect. Ethanol.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Inhalation, pressurised.
The drug is dissolved in a non-halogenated solution and delivered via a pMDI, resulting in an extra fine aerosol. The main particle fraction ranges from 1.1 microns to 2.1 microns, which ensures high lung deposition (> 50% of the ex-actuator dose), and less deposition in the oropharynx than marketed suspension formulations of other ICS.
4 Clinical Particulars
4.1 Therapeutic Indications
Alvesco is indicated as prophylactic treatment of asthma in adults, adolescents and in children 6 years of age and older.
4.2 Dose and Method of Administration
Alvesco is for oral inhalation use only.
Symptoms start to improve with Alvesco within 24 hours of treatment. However, due to its prophylactic nature, Alvesco should be taken regularly even when patients are asymptomatic.
Dosing recommendation for children (6-11 years). The recommended dose of Alvesco for children is 80 microgram or 160 microgram once daily. Alvesco can be administered as 1 or 2 puffs once daily either in the morning or evening. The use of a spacer is recommended for children 6-11 years (see Method of administration). The dose should be adjusted to the lowest dose at which effective control of asthma is maintained.
Dosing recommendation for adults and adolescents aged 12 years and older. The recommended dose range is 80 to 320 microgram per day in adult and adolescent patients. In certain circumstances the dosage may be increased in adults (see Adult patients - higher doses in certain circumstances). Patients should be given a starting dose of Alvesco which is appropriate to the severity of their disease. Typical starting doses in patients either newly diagnosed or not previously treated with inhaled corticosteroids (ICS) are provided in Table 1.

Adult patients - higher doses in certain circumstances. Adults with severe asthma may have their daily dose increased from 320 microgram once daily to 320 microgram twice daily. However, the superiority of this higher dose versus 320 microgram once daily has not been unequivocally established (see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.4 Special Warnings and Precautions for Use). The dose should be adjusted to the lowest dose at which effective control of asthma is maintained.
When transferring a patient from an oral steroid to ciclesonide, the patient should be in a relatively stable phase. A high dose of ciclesonide should be given in combination with the oral steroid for about 10 days. The oral steroid should be gradually reduced to the lowest possible level.
Special patient populations. Renal impairment. There is no need to adjust the dose in patients with renal impairment.
Hepatic impairment. There is no need to adjust the dose in patients with hepatic impairment.
Systemic exposure to the active metabolite (M1; 21-des-isobutyryl-ciclesonide) is increased in patients with hepatic impairment (see Section 5.2 Pharmacokinetic Properties).
Use in the elderly. There is no need to adjust the dose in elderly patients.
Use in paediatric patients. To date, there is insufficient data available in the treatment of children of 5 years and younger with Alvesco.
Method of administration. For detailed instructions see the Patient Instruction Leaflet.
The mouthpiece should be cleaned with a dry tissue or cloth weekly, do not wash the inhaler or put any part of the inhaler in water.
Because of the already high lung deposition and low deposition of active corticosteroid in the oropharynx, the use of a spacer with Alvesco is not routinely recommended for all patients. However, some patients may benefit from the consistent use of a spacer device in conjunction with their metered dose inhaler, particularly those with poor inhaler technique and children (6-11 years) as mentioned above. If a spacer is considered necessary, the AeroChamber Plus is a suitable device for using with Alvesco. The patient should be instructed to inhale after each actuation of drug into the spacer. Any delay between actuation and inhalation should be kept to a minimum.
Electrostatic charge on the walls of the spacer may cause variability in drug delivery. Patients should be instructed to wash the spacer in warm water and detergent and allow it to dry without rinsing or drying with a cloth. This should be performed before initial use of the spacer and at least monthly thereafter. In those patients using a spacer, a change in the make of spacer may be associated with an alteration in the amount of drug delivered to the lungs. The clinical significance of such alterations is uncertain. However, in these situations, the patient should be monitored for any loss of asthma control.
4.3 Contraindications
Alvesco should not be used in case of known hypersensitivity to any of the ingredients.
4.4 Special Warnings and Precautions for Use
Immunosuppression and risk of infections. As with all ICS, Alvesco should be administered with caution in patients with active or quiescent pulmonary tuberculosis, fungal, bacterial or viral infections, and only if these patients are adequately treated.
Acute asthma episodes. As with all ICS, Alvesco is not indicated in the treatment of status asthmaticus or other acute episodes of asthma where intensive measures are required.
As with all ICS, Alvesco is not designed to relieve acute asthma symptoms for which an inhaled short-acting bronchodilator is required. Patients should be advised to have such rescue medication available.
Patients with severe asthma are at risk of acute attacks and should have regular assessments of their asthma control including pulmonary function tests. Increasing use of short acting bronchodilators to relieve asthma symptoms indicate deterioration of asthma control. If patients find that short acting relief bronchodilator treatment becomes less effective, or they need more inhalations than usual, medical attention must be sought. In this situation, patients should be reassessed and consideration given to the need for increased anti-inflammatory treatment therapy (e.g. higher doses of ICS or a course of oral corticosteroids). The maximal daily dose is 640 microgram/day (given as 320 microgram twice a day but the superiority of this dose over 320 microgram/day has not been unequivocally demonstrated (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Severe asthma exacerbations should be managed according to standard medical practice.
Systemic effects. Inhaled steroid products are designed to direct glucocorticoid delivery to the lungs in order to reduce overall systemic glucocorticoid exposure and side effects. In sufficient doses however, all ICS can have adverse effects, notably depression of the hypothalamic pituitary adrenal (HPA) axis, reduction of bone density, cataract, glaucoma and retardation of growth rate in children and adolescents, and more rarely, a range of psychological or behavioural effects including psychomotor hyperactivity, sleep disorders, anxiety, depression or aggression (particularly in children). In steroid dependent patients, prior systemic steroid usage may be a contributing factor, but such effect can occur amongst patients who use only ICS regularly.
HPA axis suppression and adrenal insufficiency. The lowest dose of ciclesonide that causes suppression of the HPA axis (as indicated by 24-hour urinary cortisol concentrations), effects on bone mineral density or growth retardation in patients has not yet been established.
A controlled study compared 24-hour plasma cortisol AUC in 26 adult asthmatic patients following 7 days of treatment. Compared to placebo, treatment with ciclesonide 320, 640 and 1280 microgram/day did not statistically lower the 24-hour time averages of plasma cortisol (AUC(0-24)/24 hours) nor was a dose dependent effect seen. Hence, at therapeutic doses, no significant difference was detected between inhaled ciclesonide and placebo on HPA function and serum cortisol levels. However, potential effects on the HPA axis may occur in individual patients particularly at times of physiological stress (e.g. hot climate, illness or surgery). Similar results were seen in other studies in asthmatic children aged 4 to 12 years.
Growth. It is recommended that the height of children and adolescents receiving prolonged treatment with ICS is regularly monitored. In addition, consideration should be given to referring the patient to a paediatric respiratory specialist.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Transfer from oral corticosteroids. The benefits of inhaled ciclesonide should minimise the need for oral corticosteroids. However, patients transferred from oral steroids may remain at risk of impaired adrenal reserve for a considerable time after transferring to inhaled ciclesonide. The possibility of adverse effects may persist for some time. These patients may require specialised advice to determine the extent of adrenal impairment before elective procedures. The possibility of residual impaired adrenal response should always be considered in emergency (medical or surgical) and elective situations likely to produce stress, and appropriate corticosteroid treatment considered. Transfer of patients from systemic corticosteroid therapy to Alvesco may unmask pre-existing allergic conditions such as allergic rhinitis or eczema, previously suppressed by systemic corticosteroid therapy.
General. Paradoxical bronchospasm with an immediate increase of wheezing or other symptoms of bronchoconstriction after dosing should be treated with an inhaled short-acting bronchodilator which usually results in quick relief. If the patients find short-acting bronchodilator treatment ineffective, or they need more inhalations than usual, medical attention must be sought. This indicates a worsening of the underlying conditions, and warrants a reassessment of the therapy.
The patient should be assessed and therapy with Alvesco should only be continued if, after careful consideration, the expected benefit is greater than the possible risk. Correlation between severity of asthma and general susceptibility for acute bronchial reactions should be kept in mind (see Section 4.8 Adverse Effects (Undesirable Effects)).
The patient should be advised against abrupt discontinuation of therapy with Alvesco.
Patient inhaler technique should be checked regularly to make sure that inhaler actuation is synchronised with inhalation to ensure optimum delivery to the lungs (see Section 4.2 Dose and Method of Administration).
Concomitant treatment with ketoconazole or other potent CYP3A4 inhibitors should be avoided unless the benefit outweighs the increased risk of systemic side effects of corticosteroids (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in hepatic impairment. Systemic exposure to the active metabolite (M1) is increased in patients with hepatic impairment (see Section 5.2 Pharmacokinetic Properties, Pharmacokinetic characteristics in special patient populations, Hepatic impairment). Although no dosage reduction is necessary, prescribers should be aware of the possibility of an increased risk of systemic adverse effects (see Section 4.2 Dose and Method of Administration, Hepatic impairment).
Use in the elderly. Systemic exposure to M1 is also increased in elderly patients (see Section 5.2 Pharmacokinetic Properties, Pharmacokinetic characteristics in special patient populations, Use in the elderly). Although no dosage reduction is necessary, prescribers should be aware of the possibility of an increased risk of systemic adverse effects in such patients (see Section 4.2 Dose and Method of Administration, Use in the elderly).
Paediatric use. See Section 5.1 Pharmacodynamic Properties, Clinical trials.
To date, there is insufficient data available in the treatment of children of 5 years and younger with Alvesco.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In a drug-drug interaction study at steady state with ciclesonide and ketoconazole as a potent CYP3A4 inhibitor, the exposure to the active metabolite M1 increased approximately 3.5-fold, whereas the exposure to ciclesonide was not affected. Therefore, the concomitant administration of potent inhibitors of CYP3A4 (e.g. ketoconazole, itraconazole and ritonavir or nelfinavir) should be avoided unless the benefit outweighs the increased risk of systemic side effects of corticosteroids.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility was not affected in rats given 900 microgram/kg/day ciclesonide given by oral gavage.
Use in pregnancy. (Category B3)
There are no adequate and well controlled studies in pregnant women.
In animal studies glucocorticoids have been shown to induce malformations. Corticosteroids are known to induce foetotoxic and teratogenic effects in rodent and rabbit studies.
Embryofoetal development studies with daily SC dosing of ciclesonide in rabbits, abnormal foetal development (cleft palate, hind paw flexure, enlarged fontanelle, parchment like skin) was observed at systemic exposure levels (based on plasma AUC) ranging from about 3 to 12 times that anticipated clinically at the maximum recommended human dose.
Embryofoetal development studies in rats showed reduced foetal weight, skeletal anomalies, hydronephrosis and maternotoxicity at oral doses of 300-900 microgram/kg/day.
Similar studies with these doses extended until weaning revealed maternotoxicity, reduced pup weight gain, changes in pup organ weight and changes in behavioural development tests. The systemic exposure of dams relative to human exposure in these studies is not known, but doses represented 2-6 times the maximum recommended human dose on a body surface area basis.
As with other ICS preparations, Alvesco is not to be used during pregnancy unless the potential benefit to the mother justifies the potential risk to the mother or foetus. The lowest effective dose of ciclesonide needed to maintain adequate asthma control should be used. Infants born of mothers who received corticosteroids during pregnancy are to be observed carefully for hypoadrenalism.
Use in lactation. The excretion of ciclesonide or its metabolites into human milk has not been investigated.
There was limited excretion of ciclesonide and/or its metabolites into milk in lactating rats after intravenous or oral administration (respective maxima of 0.23% and 0.03% of dose/g tissues). Oral administration of ciclesonide to rats from early pregnancy until weaning was associated with adverse effects on dams and pups (see Use in pregnancy).
In breastfeeding mothers, the therapeutic benefits of the drug should be weighed against the potential hazards to mother and baby.
4.7 Effects on Ability to Drive and Use Machines
Alvesco has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial data in adults and adolescents. Approximately 5% of patients experienced adverse reactions in clinical trials with Alvesco given in the dose range 80 to 1280 microgram per day. In the majority of cases, these were mild and did not require discontinuation of treatment with Alvesco.
Table 2 shows the adverse events reported with a frequency of ≥ 2% from participants in studies of up to 1-year duration.
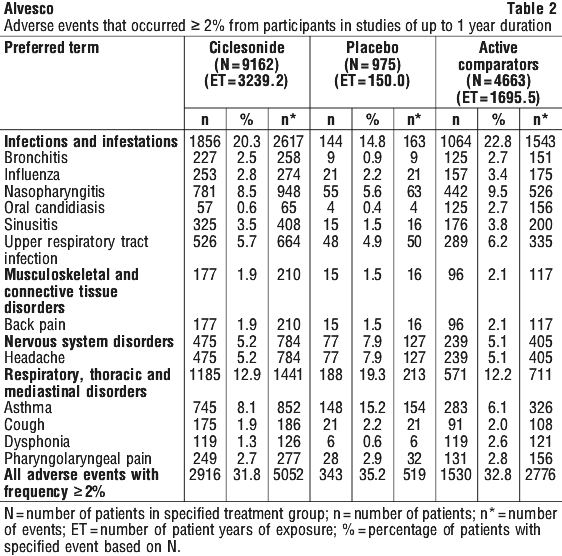

Clinical trial data in children. In children, the overall frequency of treatment emergent adverse events seen with Alvesco treatment was lower than that seen with placebo treatment. There was no evidence of any negative effect of Alvesco on short or long-term growth velocity.
The following adverse reactions were recorded during clinical trials in children (N=2827) with Alvesco, regardless of causality. (See Table 4.)

There have been very rare reports of psychiatric symptoms such as agitation, insomnia, depression, anxiety and behavioural changes with ciclesonide as well as with other ICS. Other reported psychiatric symptoms for which the incidence is unknown include psychomotor hyperactivity, sleep disorders and aggression.
Systemic effects of ICS may occur, particularly at doses higher than recommended. Possible systemic effects include Cushing's syndrome, Cushingoid features, adrenal suppression, growth retardation in children and adolescents and decrease in bone mineral density (see Section 4.4 Special Warnings and Precautions for Use, Systemic effects).
Eye disorders with frequency unknown, such as blurred vision, cataract or glaucoma, have been reported with systemic and topical corticosteroid use (see Section 4.4 Special Warnings and Precautions for Use, Systemic effects).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
Acute. Inhalation by healthy volunteers of a single dose of 2880 microgram of ciclesonide was well tolerated. The potential for acute toxic effects following overdose of inhaled Alvesco is low. After acute overdosage no specific treatment is necessary.
Chronic. After prolonged administration of 1280 microgram of ciclesonide no significant clinical signs of adrenal suppression were observed. However, if higher than recommended dosage is continued over prolonged periods, some degree of adrenal suppression cannot be excluded. Monitoring of adrenal reserve may be necessary. In cases of ciclesonide overdose, therapy may still be continued at a suitable dosage for symptom control.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Ciclesonide (pure R-epimer) belongs to a new class of on-site activated non-halogenated ICS. Ciclesonide is an ester pro-drug with approximately 100-fold lower affinity for the glucocorticoid receptor than its active metabolite (M1; 21-des-isobutyryl-ciclesonide) and budesonide and fluticasone. Endogenous activation occurs primarily via esterases located in the lung, to give M1.
Bronchial inflammation is known to be an important component in the pathogenesis of asthma. Inflammation occurs in both large and small airways and also causes an associated increase in airway responsiveness to a variety of inhaled stimuli. In clinical trials, ciclesonide has been shown to reduce airway reactivity to adenosine monophosphate in hyperreactive patients. Pre-treatment with ciclesonide for seven days significantly attenuated the early and late phase reactions following inhaled allergen challenge. Inhaled ciclesonide treatment was also shown to attenuate the increase in inflammatory cells (total eosinophils) and inflammatory mediators in induced sputum.
Clinical trials. Clinical trials in adults and adolescents. Forty-two studies with Alvesco pMDI were initiated in Europe, Canada, Japan, USA and South Africa. Over 14,000 patients were evaluated including adolescents (12-17 years), adults and elderly patients (65-75 years). Most studies were double blind, some were placebo controlled whereas others used beclomethasone dipropionate, budesonide or fluticasone propionate as an active control. Patients classified as mild, moderate or severe asthmatics were included.
Alvesco was demonstrated to be well tolerated and effective in treating asthma of varying disease severity in adults and adolescents. Safety and efficacy was maintained over 12 months.
Treatment with Alvesco in recommended doses did not cause HPA axis suppression as measured by 24-hour serum and urine cortisol concentrations or cosyntropin tests.
Tables 5, 6 and 7 present the outcome of primary endpoints in efficacy studies comparing Alvesco with placebo, budesonide and fluticasone propionate. All studies were a randomised (evenly) parallel group design of 12 weeks duration.
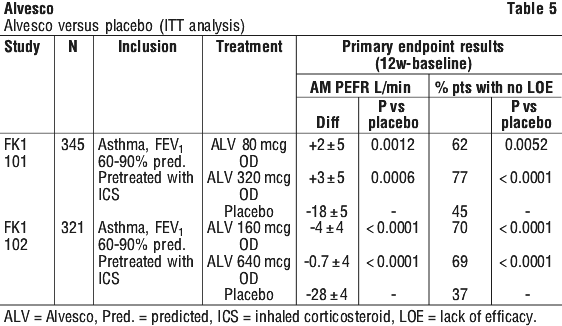


Late studies that examined dose-response of ciclesonide over 320 microgram/day were studies M1 140 (ciclesonide 160 microgram/day versus 640 microgram/day) and XRP 323/324 (640 and 320 microgram/day and fluticasone propionate 880 microgram/day).
Study M1 140. The objective of this randomised, parallel group study (n = 680 patients) was to show superiority of ciclesonide 320 microgram twice a day over ciclesonide 160 microgram/day. There were two primary variables over the 12-week treatment phase: time to first asthma exacerbation (loss of efficacy) and change in FEV1 from T0 to Tend/last. First asthma exacerbation/ loss of efficacy was defined as worsening asthma which required treatment with additional asthma medications other than increased use of rescue medication. After a 2-week baseline period when the patients received fluticasone propionate 250 microgram twice a day, randomization occurred if FEV1 were ≤ 70% predicted, asthma symptom scores ≥ 4 for the 4 of the last 7 days before randomization or ≥ puffs of rescue medication were used in the last 4 days before randomization. Six hundred and eighty patients were randomised to (ratio 1:1): 341 to ciclesonide 640 microgram/day and 339 to ciclesonide 160 microgram/day; 595 of them completed the study (ciclesonide 640 microgram/day 91.2% and ciclesonide 160 microgram/day 71.1%). Ciclesonide 640 microgram/day (320 microgram twice daily) was superior to ciclesonide 160 microgram/day with regard to time to occurrence of a first exacerbation (p = 0.005) 12.7% of patients in the ciclesonide 160 microgram/day group and 6.7% of patients in the ciclesonide 640 microgram/day experienced exacerbation. For the second primary efficacy variable, FEV1 (L/min) from T0 to Tend/last, this increased in both treatment groups; ciclesonide 160 microgram/day 0.269 and ciclesonide 320 microgram twice a day 0.332 (both p < 0.0001) but superiority of ciclesonide 320 microgram twice a day over ciclesonide 160 microgram/day was not shown.
Ciclesonide 320 microgram twice a day - ciclesonide 160 microgram/day Δ 0.062 (p = 0.0639). This study did not use a comparator dose of ciclesonide 320 microgram/day. The added benefit of 320 microgram twice a day over 320 microgram/day was not examined.
Study XRP 323/324. Was a phase III double-blind, double dummy, parallel-group, multicentre, placebo-controlled, efficacy and safety study of ciclesonide pMDI 320 microgram/day, 640 microgram/day and Flovent pMDI (fluticasone propionate) 880 microgram/day (ex-actuator) administered twice daily for 12 weeks in the treatment of severe persistent asthma in adolescents and adults. Patients were required to have been on ≥ 500 microgram fluticasone propionate or equivalent for at least one month prior to baseline and used β2-agonist > twice per week. During baseline the patients took 50 or 25% of their usual ICS dose. At randomization, FEV1 was ≥ 40% and ≤ 65% predicted and a reduction of ≥ 10% from the actual FEV1 value at entry to baseline. The primary efficacy variable was change in FEV1 from baseline to week 12 and the primary efficacy analysis was the treatment difference between active treatments and placebo. The first comparison was between ciclesonide 640 microgram/day and placebo followed by the comparison between ciclesonide 320 microgram/day and placebo. The intent to treat population totalled 527 patients. FEV1, improved significantly from T0 to T12 for all treatments: 0.25 L/min for placebo, 0.36 L/min for ciclesonide 320 microgram, 0.43 L/min for ciclesonide 640 microgram, and 0.50 L/min for FP 880 microgram. There was a statistically significant difference in FEV1 for ciclesonide 640 microgram vs. placebo, (Δ 0.18, p = 0.0008), ciclesonide 320 microgram/day vs. placebo (Δ 0.11, p = 0.0374) and fluticasone 880 vs. placebo (0.24, p = 0.0001). The treatment differences between ciclesonide 640 microgram and 320 microgram and ciclesonide 640 microgram and fluticasone 880 microgram treatment groups were not significant clinically or statistically but some dose-related trends were seen.
Study FK1 102. Was a placebo controlled, parallel group study that compared 12 weeks treatment with 160 or 640 microgram/day ciclesonide or placebo; the primary efficacy variables were change in morning peak expiratory flow from initial to last observation and the fraction of patients with predefined loss of efficacy up to week 12. The adults enrolled had mild to moderate asthma. This was a superiority study of ciclesonide 640 microgram/day and then ciclesonide 160 microgram/day compared to placebo. The first comparison did not show superiority.
Note. This study did not use a comparator dose of ciclesonide 320 microgram/day. Added benefit of 640 microgram/day over ciclesonide 160 microgram/day was not shown in mild to moderate asthma.
Clinical trials in children (under 12 years of age). In four active-controlled studies of 12 weeks duration in children comparable efficacy to the respective active control was shown for lung function as measured by FEV1 and peak expiratory flow, asthma symptom scores, and need for inhaled β2-agonist. In two of these studies ciclesonide was administered with a spacer.
The effect on growth in 609 children aged 5 to 9 years was investigated in a placebo-controlled multi-centre, double-blind, randomised parallel-group study of 12 months duration. In the modified intention-to-treat (mITT) analysis, the mean growth velocities observed during the double-blind treatment period were 5.76 cm/year in the placebo group, 5.75 cm/year in the 40 microgram ciclesonide group, and 5.60 cm/year in the 160 microgram ciclesonide group. It can be concluded that doses of ciclesonide administered at 40 microgram or 160 microgram once daily were noninferior to placebo with respect to growth velocity. In addition, no significant difference was observed between ciclesonide and placebo as measured by 24-hour urinary free cortisol in 292 patients who were studied for HPA axis function.
Growth was also assessed by stadiometry in one of the double-blind, double-dummy, randomised parallel group 12-week studies in a subset of patients (ciclesonide 160 microgram od with spacer: N = 58, budesonide 400 microgram od administered by DPI: N = 26). Height increased by 1.2 cm in the ciclesonide group and by 0.7 cm in the budesonide group. A between treatment comparison showed superiority of ciclesonide over treatment with budesonide (p = 0.0025).
5.2 Pharmacokinetic Properties
Ciclesonide is dissolved in a non-halogenated solution which results in a finer aerosol and less oropharyngeal deposition than suspension formulations of other ICS. In addition, ciclesonide is delivered as a pro-drug, with a low level of conversion to the active metabolite (M1) in the oropharynx.
In the following all doses of Alvesco are given as ex-actuator. 160 microgram Alvesco ex-actuator corresponds to 200 microgram ex-valve. Doses for other ICS are given as ex-valve.
In a study comparing oropharyngeal deposition, the AUC (in nanomol x hr/L) of M1 recovered from the oropharynx after a 640 microgram dose of Alvesco was 4% of the AUC of budesonide recovered after an 800 microgram (ex-valve) dose of a suspension formulation of budesonide.
Similarly, the AUC of M1 recovered from the oropharynx after a 640 microgram dose of Alvesco was 8% of the AUC of fluticasone recovered after a 1000 microgram dose (ex-valve) of a suspension formulation of fluticasone.
Based upon a γ-scintigraphy experiment in healthy subjects, 52% of the dose leaving the Alvesco pMDI is deposited in the lungs. In line with this figure, the mean systemic bioavailability for M1 is ≥ 50%. Systemic exposure to M1 is approximately proportional to dose.
Studies with oral and intravenous dosing of radiolabelled drug have shown an incomplete extent of oral absorption (24.5%). With a powder capsule formulation of ciclesonide, the oral bioavailability of both ciclesonide and M1 is low (< 0.5% for ciclesonide, < 1% for M1). The swallowed portion of the inhaled drug is not expected to contribute significantly to systemic absorption.
Distribution. Following IV administration of ciclesonide the volume of distribution was estimated at 2.9 L/kg for ciclesonide and 12.1 L/kg for M1. Ciclesonide and M1 are highly bound to plasma proteins (98-99%).
Metabolism. Ciclesonide is primarily hydrolysed to M1 by esterase enzymes in the lung. Studies with human liver microsomes showed that M1 is metabolised predominantly by CYP3A4 catalysis to hydroxylated metabolites, which have a lower affinity (4-15 fold) than M1 for the glucocorticosteroid receptor. Furthermore, in studies in rats, lipophilic fatty acid ester conjugates of the M1 in the lung were detected. This could maintain levels of the active principle due to C21-ester hydrolysis in the lung.
Excretion. Following IV administration of ciclesonide, clearance of ciclesonide was 152 L/hr and clearance of M1 was estimated at 228 L/hr. Half-life was estimated at 0.94 hours for ciclesonide and 2.8 hours for M1.
Ciclesonide and its metabolites are predominantly excreted via the faeces, after oral and intravenous administration, indicating that biliary excretion is the major route of elimination.
Pharmacokinetic characteristics in special patient populations. Hepatic impairment. Following a single inhaled dose of 1280 microgram of Alvesco in patients with moderate or severe cirrhosis, plasma concentrations of M1 were increased approximately two-fold compared to healthy subjects.
Use in the elderly. Following a single inhaled dose of 1280 microgram of Alvesco in healthy elderly male subjects, plasma concentrations of M1 were increased approximately two-fold compared to healthy young male subjects.
5.3 Preclinical Safety Data
Genotoxicity. Ciclesonide did not induce gene mutations in bacterial or mammalian assays in vitro, nor induce chromosomal aberrations in CHO cells or human lymphocytes in vitro. However, ciclesonide induced micronuclei in mouse bone marrow in vivo in oral doses ≥ 75 mg/kg in females and > 1000 mg/kg in males. The estimated systemic exposure (plasma AUC) to the active metabolite at the no effect dose level was ≥ 6 times that expected in humans at the maximum clinical dose. Positive in vivo clastogenicity results have also been observed with other corticosteroids and may result from effects on erythrocyte differentiation. The clinical relevance of these clastogenicity findings is unknown.
Carcinogenicity. Carcinogenicity was investigated in a 2-year inhalation study in rats receiving up to 104 microgram/kg/day (females) or 90 microgram/kg/day (males) ciclesonide and in a 2-year oral study in mice receiving up to 900 microgram/kg/day ciclesonide respectively.
Gastric adenomas (benign tumour) were significantly increased in female mice receiving a 70-fold higher dose (on a mg/kg basis) compared to the amount of the maximal recommended clinical inhalation dose estimated to be swallowed per day. This effect may arise from a local action in the antrum.
There were no significant tumorigenic effects of low doses of ciclesonide in the rat 2 year inhalation study (systemic exposure based on plasma AUC that is similar to that expected in humans given the maximum daily dose).
6 Pharmaceutical Particulars
6.1 List of Excipients
Ethanol and norflurane ((HFA-134a) a propellant, which does not contain chlorofluorocarbons (CFCs)).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not refrigerate or freeze. Do not puncture or incinerate even when empty as canister may explode.
6.5 Nature and Contents of Container
Alvesco is registered* in the following pack sizes.
Alvesco 80. 60 or 120 inhalations.
Alvesco 160. 60 or 120 inhalations.
*Not all presentations may be available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical name: [11β,16α (R)]-16,17-[(Cyclohexylmethylene)bis(oxy)]-11-hydroxy-21-(2-methyl-1-oxopropoxy) pregna-1,4-diene-3,20-dione.
Chemical structure.
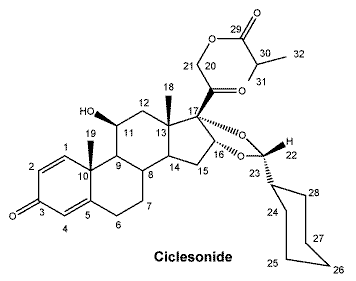
Molecular formula: C32H44O7.
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
25 February 2004
Date of Revision
04 September 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.