Amcal Nicotine Chewing Gum
Brand Information
| Brand name | Amcal Nicotine Chewing Gum |
| Active ingredient | Nicotine |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Amcal Nicotine Chewing Gum.
Full CMI
1. Why am I using Amcal Nicotine Chewing Gum?
Amcal Nicotine Chewing Gum contains the active ingredient nicotine [2mg or 4mg]. Amcal Nicotine Chewing Gum can help you stop smoking over a few months. You can also use it to help you stop smoking gradually over a longer period of time, if you cannot stop smoking immediately.
Whilst giving up isn't going to happen immediately, it is a habit you can break. But it's important to understand that there are two sides to quitting smoking.
Firstly, there is the psychological dependence on cigarettes. The smoking habit may have become an important part of your life, especially if you have smoked for many years.
Secondly, there is the physical addiction to nicotine to consider. Cigarettes contain nicotine and your body has become dependent on nicotine.
Amcal Nicotine Chewing Gum works on reducing your desire to smoke by providing some of the nicotine that you previously inhaled from cigarettes. This is what helps you to resist smoking.
When chewed, nicotine is released slowly from the gum and absorbed through the lining of the mouth. The amount of nicotine in the gum can also reduce some of the unpleasant effects that often happen when giving up smoking, such as feeling ill or irritable. Amcal Nicotine Chewing Gum does not act as quickly as smoking.
Amcal Nicotine Chewing Gum works most effectively when you have a strong personal commitment to stop smoking. You cannot rely on Amcal Nicotine Chewing Gum alone to break the habit. You are more likely to quit smoking when using the product with help from your pharmacist, doctor, a trained counsellor or a support program. Counselling is available from various groups and to get the best out of Amcal Nicotine Chewing Gum, we encourage you to enroll in a supportive group.
2. What should I know before I use Amcal Nicotine Chewing Gum?
Warnings
Do not use Amcal Nicotine Chewing Gum if:
- your mouth or throat is sore
- you are under 12 years old
- you are a non-smoker or an occasional smoker
- you are allergic or hypersensitive to nicotine or any of the other ingredients in the gum
- the expiry date printed on the carton or blister platform has passed
- the packaging is torn or shows signs of tampering
There are no health benefits to smoking. It is always better to give up smoking, and using Amcal Nicotine Chewing Gum can help. In general, any possible side effects associated with nicotine replacement therapy (NRT) are far outweighed by the well-established health dangers of continuing to smoke.
If you are in hospital because of a heart attack, severe heart rhythm disorders or a stroke, you should try to quit smoking without using NRT unless your doctor tells you to use it. Once you are discharged from the hospital, you may use NRT in consultation with your doctor.
If you have had allergic reactions that involve swelling of the lips, face and throat (angioedema) or itchy skin rash (urticaria), using NRT can sometimes trigger this type of reaction.
Check with your doctor or pharmacist if you:
- have had a recent heart attack or stroke
- have angina (chest pain) that is not well controlled or is getting worse
- have severe arrhythmia (irregular heartbeat)
- have a type of heart disease, including angina
- have had a stroke anytime in the past
- have high blood pressure or any circulation disorder
- have kidney or liver problems
- have a stomach ulcer or persistent stomach upset
- have hyperthyroidism (a disorder of the thyroid gland)
- have diabetes, as you should monitor your blood sugar levels more often than usual when starting Amcal Nicotine Chewing gum you may find your insulin or other medication requirements alter
- have phaeochromocytoma (a tumour of the adrenal gland)
- have an inability to tolerate fructose (as the gums contain sorbitol, which is a source of fructose)
- are pregnant or breastfeeding
- are aged 12 to 17 years
- have a history of epilepsy (fits)
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Ideally you should not use this program while you are pregnant, and you should stop smoking without using nicotine replacement therapy. Nicotine in any form may cause harm to your unborn baby. However, if you are unable to quit without the use of nicotine replacement therapy, seek advice from your pharmacist or doctor before starting a program. The decision to use NRT should be made as early on in your pregnancy as possible and you should aim to use it for only 2-3 months. Products that are taken intermittently, such as gum, are preferable to nicotine patches. However, patches may be preferred if you have nausea or sickness.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Nicotine is excreted in breast milk in quantities that may affect the child even in therapeutic doses. Like smoking, NRT should be avoided during breastfeeding. However, if you are unable to quit without the use of NRT, seek advice from your pharmacist or doctor before starting a program.
You are also recommended to use Amcal Nicotine Chewing Gum immediately after breastfeeding, to ensure that the baby gets the smallest amount of nicotine possible from breast milk.
Take special care
- If you have dentures, dental caps or partial bridges. As with other gums, Amcal Nicotine Chewing Gum may stick to your dentures, dental caps or partial bridges and may damage them. You should stop using the gum if it sticks to your dental work and discuss further use with your dentist. Patients who wear dentures or have temporomandibular joint disease (difficulty chewing) may have trouble chewing Amcal Nicotine Chewing Gum.
- Do not drink coffee or soft drinks for 15 minutes before chewing the gum. They may keep the nicotine in the gum from being absorbed properly.
- When taking this medicine there is a risk of dependence.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop. They will be able to advise you if stopping smoking may affect the way these medicines work.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Amcal Nicotine Chewing Gum.
4. How do I use Amcal Nicotine Chewing Gum?
How much to take / use
- Follow the instructions provided with the medicine.
- Do not exceed the recommended dosage.
How to get started
Amcal Nicotine Chewing Gum is not like ordinary chewing gum. You must stop smoking completely while you are using it.
It is probably best to nominate a specific day that you will stop smoking.
It may be helpful for you to:
- Try to choose a day when you will not be where others are smoking.
- A day with as little stress as possible.
- Choose a day not too far in the future.
Tell your family and friends that you have set this target "quit day". This is the day you take control of your habit and become a non-smoker.
Prepare to stop smoking by trying to reduce the number of cigarettes you smoke each day.
How to use Amcal Nicotine Chewing Gum
At the beginning of the "quit day", start to use Amcal Nicotine Chewing Gum before any cigarettes have been smoked.
The appropriate dose will depend on your previous smoking habits.
Use 2mg gum if:
- you were smoking less than 20 cigarettes a day
If, while using the 2mg gum, your withdrawal symptoms remain so strong as to threaten relapse, then you should use the 4mg gum.
Use 4mg gum if:
- you were smoking 20 or more cigarettes a day
- you have previously failed to stop smoking with 2mg gum
Chew one piece of gum when you feel the urge to smoke.
Follow these 4 easy steps:
- Chew one piece of gum slowly until the taste becomes strong.
- Rest the piece of chewing gum between your gum and cheek.
- Chew again when the taste has faded.
- Repeat the chewing routine for about 30 minutes.
Chew only one piece of gum at a time.
Chew slowly
The nicotine in Amcal Nicotine Chewing Gum is released a little at a time and absorbed through the lining of your mouth into your bloodstream. If you chew too quickly or for too long, you will swallow the nicotine with your saliva. It will be broken down in your stomach and wasted.
Do not use more than 20 pieces of the 2mg gum or 10 pieces of the 4mg gum in a day.
Most people use 8-12 pieces of the 2mg gum or 4-6 pieces of the 4mg gum in a day.
Usage Program for adults
There are two usage programs you can follow:
Program A. If you would like to stop smoking completely before starting the program:
- After about 3 months, gradually cut down the amount of gum you chew each day.
- When you are down to 1-2 pieces a day, you can stop using Amcal Nicotine Chewing Gum.
- You should be able to stop completely within 6 months from the start of the treatment.
Program B. If you cannot stop smoking completely before starting the program:
- Gradually increase gum use, while cutting down smoking.
- When you have cut down the number of cigarettes you smoke to a level you feel you can quit completely, follow Program A.
- If you have not cut down the number of cigarettes you smoke each day after 6 weeks, see your doctor or pharmacist.
Combination therapy
If you have relapsed in the past or if you experience cravings while using a single form of nicotine replacement therapy (NRT), you can combine the use of Amcal Nicotine Patch with Amcal Nicotine Chewing Gum 2mg.
The combination is more effective than either product alone in people who have been unable to quit smoking using a single NRT method, increasing your chances of successfully quitting.
When using Amcal Step 1 Nicotine Patch, chew one piece of Amcal Nicotine Chewing Gum 2mg if you get a craving. Use at least 4 pieces of gum and not more than 12 pieces in a day. Continue for 12 weeks.
After 12 weeks, you can wean yourself off therapy by either of the following methods:
- Stop use of Amcal Nicotine Patch and gradually reduce the number of Amcal Nicotine Chewing Gum 2mg you use until you no longer need them.
- a. Use Amcal Step 2 Nicotine Patch for 3-4 weeks, while using the same number of pieces of Amcal Nicotine Chewing Gum 2mg in a day that you have routinely used.
b. Then use Amcal Step 3 Nicotine Patch for a further 3-4 weeks, while using the same number of pieces of Amcal Nicotine Chewing Gum 2mg in a day that you have routinely used.
c. When patch use is no longer needed, gradually reduce the number of gums you use until you no longer need them.
Children 12 to 17 years old
Do not use for longer than 12 weeks. If you think you need to use for longer than 12 weeks, talk to your doctor or pharmacist.
Chew one piece of gum when you feel the urge to smoke. Follow the directions under 'How to use Amcal Nicotine Chewing Gum'. Use every 2 to 3 hours. Do not use more than 20 pieces of the 2mg gum, or 10 pieces of the 4mg gum in one day.
After about 8 weeks, gradually reduce to 1 to 2 pieces a day and then stop completely.
Do not use for children under 12 years.
If you use too much Amcal Nicotine Chewing Gum
If you think that you have used too much Amcal Nicotine Chewing Gum, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you smoke or use any other nicotine containing product while you are using Amcal Nicotine Chewing Gum, you may suffer an overdose of nicotine. However, if used correctly, nicotine overdose is unlikely.
The general symptoms of nicotine overdosage include: pallor, sweating, burning throat, nausea, salivation, vomiting, stomach upset or abdominal pain, diarrhoea, headache, dizziness, hearing and vision disturbances, tremor, confusion, weakness, seizures (fits), fast or irregular heartbeat, circulatory problems, fainting and breathing difficulties.
The risk of overdose is small as nausea and vomiting usually occurs at an early stage with excessive nicotine intake.
5. What should I know while using Amcal Nicotine Chewing Gum?
Things you should do
- Use Amcal Nicotine Chewing Gum exactly as instructed.
- If you follow these recommendations, you should get the full benefit of the Amcal Nicotine Chewing Gum program.
Tell all the doctors, dentists and pharmacists who are treating you that you are using Amcal Nicotine Chewing Gum.
Things you should not do
- Do not stop using Amcal Nicotine Chewing Gum suddenly. You may get side effects similar to those you would get if you stopped smoking suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Amcal Nicotine Chewing Gum affects you.
There is no evidence of any risk associated with driving or operating machinery if Amcal Nicotine Chewing Gum is used according to the recommended dose but remember that smoking cessation itself can cause behavioural changes
Looking after your medicine
Keep the gum in the blister pack inside the carton until you are ready to use it.
If you take the gum out of its blister pack, it may not keep well.
Store the gums in a cool, dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on windowsills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least 1.5 meters above the ground is a good place to store medicines.
Children: It is very important to keep Amcal Nicotine Chewing Gum out of reach and sight of children, as doses of nicotine that are tolerated by adult smokers can cause severe poisoning in small children and can be fatal.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
Like all medicines Amcal Nicotine Chewing Gum can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious /common side effects
| Less serious side effects | What to do |
Gastrointestinal:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. These are mostly mild and short-lived. You may experience these side effects as you decrease the number of gums that you use. |
Serious/rare side effects
| Serious side effects | What to do |
Cardiac disorders:
| Stop using Amcal Chewing Gum. Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
Amcal Nicotine Chewing Gum is available in two nicotine dosage strengths - Amcal Nicotine 2mg Chewing Gum and Amcal Nicotine 4mg Chewing Gum.
What Amcal Nicotine Chewing Gum contains
| Active ingredient (main ingredient) | Nicotine |
| Other ingredients (inactive ingredients) | Acesulfame potassium |
All Amcal Nicotine Chewing Gums are sugar-free.
Each gum contains sorbitol, xylitol and mannitol with a combined total of 0.4g per piece. For the 2mg strength, this is equivalent to 8g per maximum dose of 20 pieces. For the 4mg strength, this is equivalent to 4g per maximum dose of 10 pieces. Please note that products containing these ingredients may have a laxative effect or cause diarrhoea.
Each piece of gum also contains 11.5 mg (0.5 mmol) sodium which should be taken into account by those on a low sodium diet. For the 2 mg strength, this is equivalent to 230 mg (10 mmol) sodium per maximum dose of 20 pieces. For the 4 mg strength, this is equivalent to 115 mg (5mmol) sodium per maximum dose of 10 pieces.
Do not take this medicine if you are allergic to any of these ingredients.
What Amcal Nicotine Chewing Gum looks like
Amcal Nicotine Chewing Gum is a rectangular white chewing gum with polished surface.
- Amcal Nicotine Chewing Gum 2mg Mint
(Aust R 229432) - Amcal Nicotine Chewing Gum 4mg Mint
(Aust R 229433) - Amcal Nicotine Chewing Gum 2mg Fruit
(Aust R 229431)
Who distributes Amcal Nicotine Chewing Gum

Available only from Amcal
Allied Master Chemists of Australia Limited
3 Myer Place
Rowville Vic 3178 Australia
www.amcal.com.au
AUSTRALIAN OWNED
Amcal Nicotine Patches are supplied in Australia by:
Perrigo Australia
Suite 7, Level 1,
13a Narabung Way
Belrose NSW 2085
Customer Service: 1800 805 546
Date of Preparation: July 2014
Date of last amendment: October 2025
ACC003714P:4
Brand Information
| Brand name | Amcal Nicotine Chewing Gum |
| Active ingredient | Nicotine |
| Schedule | Unscheduled |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Nicotine.
2 Qualitative and Quantitative Composition
The active ingredient of Amcal Chewing Gum is nicotine.
Amcal Chewing Gum contains 2 mg or 4 mg nicotine per piece of gum. It is available in fruit and mint flavours and is sugar-free.
Each gum contains sorbitol, xylitol and mannitol with a combined total of 0.4 g per piece. For the 2 mg strength, this is equivalent to 8 g per maximum dose of 20 pieces. For the 4 mg strength, this is equivalent to 4 g per maximum dose of 10 pieces. Please note that products containing these ingredients may have a laxative effect or cause diarrhoea.
Each piece of gum also contains 11.5 mg (0.5 mmol) sodium which should be taken into account by those on a low sodium diet. For the 2 mg strength, this is equivalent to 230 mg (10 mmol) sodium per maximum dose of 20 pieces. For the 4 mg strength, this is equivalent to 115 mg (5 mmol) sodium per maximum dose of 10 pieces.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A rectangular, white chewing gum with polished surface.
4 Clinical Particulars
4.1 Therapeutic Indications
Source of nicotine as an aid for smoking cessation. May be used as part of a smoking reduction strategy by smokers who are unable or not ready to stop smoking abruptly as a step towards stopping completely.
4.2 Dose and Method of Administration
The strength of Amcal Chewing Gum should be chosen according to the smoker's tobacco dependence. Highly dependent smokers, as well as smokers who have failed to quit when using the 2 mg gum, should use the 4 mg strength. Otherwise, the 2 mg strength should be used.
One piece of gum should be chewed when the user feels the urge to smoke. The amount chewed should normally be 8-12 of the 2 mg pieces or 4-6 of the 4 mg pieces per day, up to a maximum of 20 of the 2 mg pieces or 10 of the 4 mg pieces per day.
Chewing technique. 1. One piece of gum should be chewed until the taste becomes strong.
2. The chewing gum should be rested between the gum and cheek.
3. When the taste fades, chewing should commence again.
4. The chewing routine should be repeated for 30 minutes.
Concomitant use of acidic beverages such as coffee or soft drinks may interfere with the buccal absorption of nicotine. Acidic beverages should be avoided for 15 minutes prior to chewing the gum.
The patient should be advised to make every effort to completely stop smoking during treatment with Amcal Chewing Gum.
After three months, users should gradually cut down the number of pieces chewed each day until only 1-2 pieces of gum per day are required, at which time they should stop using the product. This process may take 6 months from the start of treatment. Counselling may help smokers to quit. Those using NRT for more than 9 months should seek advice from a healthcare professional.
Gradual cessation of smoking. For smokers who are unwilling or unable to suddenly quit, Amcal Chewing Gum may be used whenever there is an intense desire to smoke to help reduce the number of cigarettes smoked, before stopping smoking completely. The smoker should attempt a reduction in cigarette consumption as soon as possible. Consult a healthcare professional if the number of cigarettes smoked has not been reduced in 6 weeks. Once the number of cigarettes has been reduced to a point where the smokers can quit completely, then the Amcal Chewing Gum program should be followed. Consult a healthcare professional if an attempt to stop smoking completely has not commenced within 6-9 months of beginning treatment.
Combination therapy. If smokers have previously relapsed with use of one form of nicotine replacement therapy (NRT), combination therapy could be beneficial. Smokers who experience breakthrough cravings or have difficulty controlling cravings using one form of NRT alone could combine the use of Amcal Patch Step 1 with another form of NRT such as Amcal Chewing Gum 2 mg. Amcal Chewing Gum 4 mg should not be used with Amcal Patches.
When using Amcal Patch Step 1 in addition to Amcal Chewing Gum 2 mg, it is recommended that 4 to 12 pieces are used each day. Most people will use 5 to 6 pieces. Do not exceed 12 pieces a day.
Combination therapy should be used for 12 weeks, after which one of the two following programs should be followed:
1. Stop use of Amcal Patch and gradually reduce the number of gums used until they are no longer needed.
2. Continue with Amcal Patch Step 2 for 3-4 weeks, then Amcal Patch Step 3 for a further 3-4 weeks while maintaining the number of Amcal Chewing Gum 2 mg that is used each day. After use of patches is ceased, gradually reduce the number of gums used until they are no longer needed.
Use in children under 18 years. Children aged 12 to 17 years should only use Amcal Chewing Gum under the advice of a healthcare professional. Treatment should not exceed 12 weeks without consultation with a healthcare professional, who should reassess the person for their commitment to quitting smoking and the likely benefit of continued treatment, before recommending use of NRT in this age group beyond 12 weeks. Treatment should not be extended by more than a further 4 weeks in this case. Do not use in children under 12 years.
4.3 Contraindications
Amcal Chewing Gum should not be used by non-smokers, children under 12 years, occasional smokers or those with known hypersensitivity to nicotine or any of the excipients in the formulation.
4.4 Special Warnings and Precautions for Use
Nicotine is a toxic and addictive drug and milligram doses are potentially fatal if rapidly absorbed. For any smoker, with or without concomitant disease or pregnancy, the risk of nicotine replacement in a smoking cessation program should be weighed against the hazard of continued smoking and the likelihood of achieving cessation of smoking without nicotine replacement.
Treatment with Amcal Chewing Gum should be discontinued if symptoms of nicotine overdose appear. Mild intoxication produces nausea, vomiting, abdominal pain, diarrhoea, headache, sweating, and pallor (see Section 4.9 Overdose).
Amcal Chewing Gum must be kept out of reach of children at all times.
Occasional smokers are not expected to benefit from the use of Amcal Chewing Gum.
Underlying cardiovascular disease. Patients hospitalised for myocardial infarction, severe dysrhythmia or cerebrovascular accident who are considered to be haemodynamically unstable should be encouraged to stop smoking with non-pharmacological interventions. If this fails, Amcal Chewing Gums may be considered, but as data on safety in this patient group are limited, initiation should only be under close medical supervision.
Once patients are discharged from hospital, they can use NRT as normal. If there is a clinically significant increase in cardiovascular or other effects attributable to nicotine, the gum dose should be reduced or discontinued.
Dependent smokers with a recent myocardial infarction, unstable or worsening angina pectoris including Prinzmetal's angina, severe cardiac arrhythmias, uncontrolled hypertension or recent cerebrovascular accident should follow the same approach, prioritizing non-pharmacological interventions before considering NRT under close medical supervision.
Amcal Chewing Gum should be used with caution in patients with:
severe hypertension, stable angina pectoris, cerebrovascular disease, occlusive peripheral arterial disease, heart failure;
hyperthyroidism or pheochromocytoma, as nicotine causes release of catecholamines;
moderate to severe hepatic and/or severe renal impairment, as the clearance of nicotine or its metabolites may be decreased with the potential for increased adverse events.
Diabetes mellitus. Smokers with diabetes mellitus should be advised to monitor their blood sugar levels more closely than usual when NRT is initiated because catecholamine release can affect carbohydrate metabolism and vasoconstriction may delay or reduce insulin absorption.
Seizures. Potential risks and benefits of nicotine should be carefully evaluated before use in subjects taking anti-convulsant therapy or with a history of epilepsy as cases of convulsions have been reported in association with nicotine.
A risk-benefit assessment should be made by an appropriate healthcare professional for patients with the following conditions:
Gastrointestinal disease. Swallowed nicotine may exacerbate symptoms in subjects suffering from active oesophagitis, oral or pharyngeal inflammation, gastritis or active peptic ulcer. Avoid use of Amcal Chewing Gum if oral or pharyngeal inflammation is present.
Transfer dependence. Nicotine transferred dependence can occur.
Smokers who wear dentures or who have temporomandibular joint disease may experience difficulty in chewing the medicated chewing gum. In this case, it is recommended that they use a different pharmaceutical form of nicotine replacement therapy.
There is the possibility that, as with other gums, Amcal Chewing Gum may stick to dentures, dental caps or partial bridges and may damage dental work.
Use in the elderly. No data available.
Paediatric use. Data on the use of NRT in treating adolescents under the age of 18 years are limited.
NRT should only be used in adolescents 12 to 17 years after consultation with a healthcare professional and use should be restricted to 12 weeks. If treatment is required for longer than 12 weeks, this should be discussed with a healthcare professional.
Do not use in children under 12 years.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No clinically relevant interactions between NRT and other drugs have definitely been established. However nicotine may enhance the haemodynamic effects of adenosine.
Smoking but not nicotine is associated with increased CYP1A2, and possibly CYP1A1, activity. After cessation of smoking there may be reduced clearance of substrates for these enzymes and increased plasma levels of some medicinal products. This is of potential clinical importance in products with a narrow therapeutic window e.g. theophylline, ropinirole, clozapine and olanzapine.
Cessation of smoking, with or without NRT, may alter the individual's response to concomitant medication and may require adjustment of dose. In particular, anticonvulsants may require special monitoring and/or dosage adjustment.
Dose reduction may be required for:
caffeine, oestrogens, imipramine, lignocaine, oxazepam, pentazocine, theophylline, warfarin, possibly due to reversal of hepatic enzyme induction on smoking cessation;
insulin, possibly due to increase in subcutaneous absorption on smoking cessation;
adrenergic antagonists (e.g. prazosin, labetalol), possibly due to reduction in circulating catecholamines on smoking cessation.
Dose increase may be required for:
adrenergic agonists (e.g. isoprenaline, phenylephrine), possibly due to reduction in circulating catecholamines on smoking cessation.
Smoking may lead to reduced analgesic effects of opioids (e.g. dextropropoxyphene, pentazocine), reduced diuretic response to furosemide, reduced effect of beta-adrenergic blockers (e.g. propranolol) on blood pressure and heart rate decrease and reduced responder rates in ulcer healing with H2-antagonists.
Both smoking and nicotine may raise the blood levels of cortisol and catecholamines. Dosages of nifedipine, adrenergic antagonists and adrenergic agonists may need to be adjusted.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category D)
In pregnant women, complete cessation of tobacco consumption should always be recommended without nicotine replacement therapy (NRT). However, for women unable to quit on their own, NRT may be recommended to assist a quit attempt. Nicotine is harmful to the foetus however, the risk for the foetus is probably less than to be expected with continued smoking due to:
Lower maximal plasma concentrations compared to inhaled nicotine, resulting in a nicotine exposure less or not more than associated with smoking.
No exposure to polycyclic hydrocarbons and carbon monoxide.
As nicotine does pass to the foetus, the decision to use NRT should be made as early on in pregnancy as possible with the aim of discontinuing after use for two to three months.
If NRT is used during pregnancy, Amcal Chewing Gum should preferably be used while pregnant as they usually provide a lower daily dose of nicotine than patches. However, if the woman suffers from nausea and/or vomiting, the patch may be preferred but should be removed before going to bed.
Due to an absence of specific studies, combination therapy with patches and oral forms is not recommended during pregnancy unless the healthcare professional considers it necessary to ensure abstinence.
Use in lactation. Nicotine is excreted in breast milk in quantities that may affect the child even in therapeutic doses. Like smoking, nicotine replacement therapy should be avoided during breast-feeding. However Amcal Chewing Gum may be used if necessary. Women should breastfeed just before they use the product to allow time between NRT use and feeding to be as long as possible.
Due to an absence of specific studies, combination therapy with patches and oral forms is not recommended during lactation unless the healthcare professional considers it necessary to ensure abstinence.
4.7 Effects on Ability to Drive and Use Machines
Smoking cessation can cause behavioural changes. Any risks associated with driving vehicles or operating machinery are considered minimal when the gum is used according to the recommended dose.
4.8 Adverse Effects (Undesirable Effects)
In principle, Amcal Chewing Gum can cause adverse effects similar to those associated with nicotine administered by smoking. Many of the observed adverse reactions are consistent with the pharmacological effects of nicotine, which are dose dependent.
The following undesirable effects detailed in Tables 1 and 2 are nicotine related adverse events for all oral dosage forms.
Table 1 shows events which were identified from a double-blind, randomised, placebo controlled lozenge clinical study involving 1818 patients. Adverse events reported in this study have been considered for inclusion, where the incidence in the 2mg or 4mg nicotine arm was higher than the corresponding placebo arm. Frequencies calculated from the study safety data.


Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. In overdose, symptoms corresponding to heavy smoking may be seen. General symptoms of nicotine poisoning include: pallor, sweating, burning throat, salivation, nausea, vomiting, abdominal cramps, diarrhoea, headache, dizziness, hearing and vision disturbances, tremor, mental confusion, muscle weakness, palpitations, dilated pupils, tachycardia, cardiac arrhythmias, dyspnoea, circulatory disturbance, convulsions, prostration, absence of neurological reaction, respiratory failure.
Lethal doses may produce convulsions, and death follows as a result of peripheral or central respiratory paralysis or, less frequently, cardiac failure. The acute lethal oral dose is approximately 0.5-0.75 mg per kg body weight, corresponding in an adult to 40-60 mg. Doses of nicotine that are tolerated by adult smokers during treatment can produce severe symptoms of poisoning in small children and may prove fatal. If poisoning is suspected in a child, a doctor must be consulted immediately.
Overdose with Amcal Chewing Gum could occur if many pieces are chewed simultaneously. Risk of overdose is small as nausea and vomiting usually occurs at an early stage. Risk of poisoning by swallowing the gum is small. Since the release of nicotine from the gum is slow, very little nicotine is absorbed from the stomach and intestine. Any that is absorbed will be inactivated by the liver.
Treatment. For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Amcal Chewing Gum mimics the pharmacological effects of nicotine from smoking and may, therefore, be used to help provide relief from nicotine withdrawal symptoms. In addition to its effects on the central nervous system, nicotine produces haemodynamic effects such as increased heart rate and systolic blood pressure.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. When the gum is chewed, nicotine is steadily released into the mouth and is rapidly absorbed through the buccal mucosa. By the swallowing of nicotine-containing saliva, a proportion reaches the stomach and intestine where it is inactivated.
Distribution. The peak plasma concentration after a single dose of the 2 mg chewing gum is approximately 6.4 nanograms per mL. For the 4 mg chewing gum, it has been calculated that the nicotine peak plasma concentration after a single dose is approximately 9.3 nanograms per mL (after approximately 60 minutes). After 45 minutes average plasma concentration of nicotine when smoking a cigarette is 15-30 nanograms per mL.
Nicotine crosses the blood-brain barrier, the placenta and is detectable in breast milk.
Metabolism. The plasma half-life is approximately three hours.
Excretion. Nicotine is eliminated mainly via hepatic metabolism, small amounts being eliminated in unchanged form via the kidneys.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Amcal Chewing Gum contains chewing gum base (containing butylated hydroxytoluene), calcium carbonate, carnauba wax, gelatin, glycerol, mannitol, menthol, polacrilin, sodium bicarbonate, sodium carbonate, sorbitol, titanium dioxide, water - purified and xylitol.
Amcal Chewing Gums contain saccharin, saccharin sodium and acesulfame potassium.
Flavours. Fruit - fruit flavour. Mint - eucalyptus oil, peppermint oil and dementholised mint oil.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Amcal Chewing Gum is available in fruit and mint flavours, containing 2 mg or 4 mg nicotine per piece; packed in PVC/PVDC blister packs then boxed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
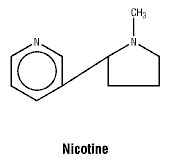
Chemical structure. Chemical name: 1-methyl-2-(3-pyridyl) pyrrolidine.
Molecular formula: C10H14N2.
Molecular weight: 162.26.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
16 October 2014
Date of Revision
28 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.