Amoxycillin Sandoz Capsules and Suspension
Brand Information
| Brand name | Amoxycillin Sandoz Capsules and Suspension |
| Active ingredient | Amoxicillin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Amoxycillin Sandoz Capsules and Suspension.
Summary CMI
Amoxycillin Sandoz®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Amoxycillin Sandoz?
Amoxycillin Sandoz contains the active ingredient amoxicillin (as amoxicillin trihydrate). Amoxycillin Sandoz is used to treat a range of infections caused by bacteria.
For more information, see Section 1. Why am I using Amoxycillin Sandoz? in the full CMI.
2. What should I know before I use Amoxycillin Sandoz?
Do not use if you have ever had an allergic reaction to Amoxycillin Sandoz or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Amoxycillin Sandoz? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Amoxycillin Sandoz and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Amoxycillin Sandoz?
- The usual dose of Amoxycillin Sandoz is one dose taken three times a day.
- Amoxycillin Sandoz doses should be spaced evenly throughout the day e.g. every eight hours if taking it three times a day.
- Swallow Amoxycillin Sandoz 250 mg and 500 mg capsules whole with a glass of water. It can be taken with or without food.
- Keep taking Amoxycillin Sandoz until the course is finished or for as long as your doctor tells you.
More instructions can be found in Section 4. How do I use Amoxycillin Sandoz? in the full CMI.
5. What should I know while using Amoxycillin Sandoz?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Amoxycillin Sandoz? in the full CMI.
6. Are there any side effects?
Mild side effects of taking Amoxycillin Sandoz include diarrhoea, soreness of the mouth or tongue, or overgrowth of yeast infections (thrush).
Serious side effects include itching; rash; unusual bleeding or bruising; yellowing of the skin or eyes; dark urine or pale stools; difficulty or pain on passing urine; severe diarrhoea; excessive abnormal muscle movements; dizziness or convulsions; or a red rash commonly seen on both sides of buttocks, upper inner thighs, armpits, or neck. Other serious side effects include an allergic reaction. Signs of this include wheezing, swelling of the lips/mouth, difficulty in breathing, hayfever, lumpy rash (hives), or fainting.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Amoxycillin Sandoz®
Active ingredient(s): amoxicillin trihydrate capsules and suspension
Consumer Medicine Information (CMI)
This leaflet provides important information about using Amoxycillin Sandoz. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Amoxycillin Sandoz.
Where to find information in this leaflet:
1. Why am I using Amoxycillin Sandoz?
2. What should I know before I use Amoxycillin Sandoz?
3. What if I am taking other medicines?
4. How do I use Amoxycillin Sandoz?
5. What should I know while using Amoxycillin Sandoz?
6. Are there any side effects?
7. Product details
1. Why am I using Amoxycillin Sandoz?
Amoxycillin Sandoz contains the active ingredient amoxicillin (as amoxicillin trihydrate). Amoxycillin Sandoz belongs to a group of antibiotic medicines called penicillins.
Amoxycillin Sandoz is used to treat a range of infections caused by bacteria. These may be infections of the chest (pneumonia), tonsils (tonsillitis), sinuses (sinusitis), urinary and genital tract, skin and fleshy tissues.
Amoxycillin Sandoz works by killing the bacteria that cause these infections.
Amoxycillin Sandoz can also be used to prevent infection.
Ask your doctor if you have any questions about why Amoxycillin Sandoz has been prescribed for you.
Your doctor may have prescribed this medicine for another reason.
2. What should I know before I use Amoxycillin Sandoz?
Warnings
Do not use Amoxycillin Sandoz if:
- you are allergic to penicillin or similar types of antibiotics such as cephalosporins. If you have ever had an allergic reaction (such as rash) when taking an antibiotic, you should tell your doctor before you take Amoxycillin Sandoz.
- you are allergic to amoxicillin or any of the ingredients listed at the end of this leaflet. Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin.
- the expiry date (EXP) printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have any other medical conditions, especially the following:
- glandular fever (mononucleosis)
- blood disorders
- kidney or liver problems
(your dose may need to be changed or you may need to be given an alternate medicine) - take any medicines for any other condition.
- have ever had an allergic reaction (such as a rash) to any antibiotics in the past.
- have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Amoxycillin Sandoz may be used during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Amoxycillin Sandoz can pass to your baby from breast milk.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Amoxycillin Sandoz and affect how it works. These include:
- medicines used to treat gout e.g. probenecid or allopurinol.
- contraceptive pill (as with other antibiotics, you may need to use extra birth control methods e.g. condoms)
- other antibiotics
- anticoagulants (used to prevent blood clots) such as warfarin
- methotrexate, a medicine used to treat arthritis and some types of cancers
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Amoxycillin Sandoz.
Amoxycillin Sandoz may influence the results of some laboratory tests.
4. How do I use Amoxycillin Sandoz?
How much to take/give
Take/Give Amoxycillin Sandoz as directed by your doctor.
Your doctor will tell you the dose of Amoxycillin Sandoz you will need to use each day. This depends on the condition being treated and whether any other medicines are being used.
The usual dose is one dose taken three times a day. The doses should be spaced evenly throughout the day e.g. every eight hours if taking it three times a day.
How to take/give Amoxycillin Sandoz
Amoxycillin Sandoz Capsules:
Swallow Amoxycillin Sandoz capsules whole with a glass of water.
Amoxycillin Sandoz can be taken with or without food.
Amoxycillin Sandoz Suspension:
The suspension will be prepared by your pharmacist who checks the seal prior to reconstitution. Shake the bottle well and accurately measure the dose with a measuring spoon. Your pharmacist will explain how many millilitres of suspension will be needed to receive the correct dose.
Do not use the reconstituted suspension if the colour is not off-white.
Shaking the bottle and using a measuring spoon will make sure that you get the correct dose.
How long to take/give Amoxycillin Sandoz for
Keep taking/giving Amoxycillin Sandoz until the course is finished or for as long as your doctor tells you.
Do not stop taking/giving Amoxycillin Sandoz just because you or your child feels better as the infection may return.
Do not stop taking/giving Amoxycillin Sandoz or change the dose without first checking with your doctor.
If you forget to use Amoxycillin Sandoz
If you miss your/your child's dose at the usual time, take/give it as soon as you remember. Then go back to taking/giving it as directed by your doctor.
If it is almost time for your/your child's next dose, skip the dose you/your child missed and take/give the next dose when you are meant to.
Do not take/give a double dose to make up for the dose you missed.
Do not take/give two doses within an hour or so of each other.
Taking more than the prescribed dose can increase the chance of unwanted side effects.
If you use too much Amoxycillin Sandoz
If you think that you have used too much Amoxycillin Sandoz, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Amoxycillin Sandoz?
Things you should do
Tell your doctor if, for any reason, you have not taken your medicine exactly as directed.
Otherwise, your doctor may think that it was not working as it should and change your treatment unnecessarily.
Tell your doctor or pharmacist you are taking Amoxycillin Sandoz before starting any other prescribed medicine.
Some medicines may affect the way other medicines work.
Call your doctor straight away if you:
- develop itching, swelling or a skin rash when you are taking Amoxycillin Sandoz. Do not take any more Amoxycillin Sandoz.
- develop severe diarrhoea when taking Amoxycillin Sandoz. Do not take any medication to stop the diarrhoea (e.g. Lomotil or Imodium).
Remind any doctor, dentist or pharmacist you visit that you are using Amoxycillin Sandoz.
Things you should not do
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Do not use Amoxycillin Sandoz to treat any other complaints unless your doctor says to.
Looking after your medicine
Keep your medicine in its original container, in a safe place out of reach of children.
Keep Amoxycillin Sandoz capsules or the suspension in a cool dry place, away from moisture or heat, where the temperature stays below 25°C.
For example, do not store it:
- in the bathroom or near a sink, or
- in the car on a hot day.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
Discard any unused suspension after 14 days or after the expiry date placed by your pharmacist on the label of the bottle during the preparation of the suspension.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Tell your doctor immediately if you experience any of these serious side effects. |
Signs of allergic reaction:
| STOP taking Amoxycillin Sandoz and contact your doctor immediately or go to the emergency department of your nearest hospital if you experience any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Amoxycillin Sandoz contains
Amoxycillin Sandoz Capsules:
| Active ingredient (main ingredient) | amoxicillin (as amoxicillin trihydrate) |
| Other ingredients (inactive ingredients) | magnesium stearate microcrystalline cellulose gelatin iron oxide yellow titanium dioxide. |
Amoxycillin Sandoz Suspension:
| Active ingredient (main ingredient) | amoxicillin (as amoxicillin trihydrate) |
| Other ingredients (inactive ingredients) | anhydrous citric acid sodium benzoate aspartame purified talc guar gum sodium citrate anhydrous silicon dioxide lemon flavouring orange flavouring peach-apricot flavouring. |
Do not take this medicine if you are allergic to any of these ingredients.
What Amoxycillin Sandoz looks like
Amoxycillin Sandoz 250 mg Capsules - opaque yellow hard gelatin capsules. Available in blisters packs of 20 capsules (Aust R 63332).
Amoxycillin Sandoz 500 mg Capsules - opaque yellow hard gelatin capsules. Available in blisters packs of 20 capsules (Aust R 63333).
Amoxycillin Sandoz 125 mg/5 mL Suspension - After mixing, the suspension is white to slightly yellowish. Available in 100 mL bottle (Aust R 93721).
Amoxycillin Sandoz 250 mg/5 mL Suspension - After mixing, the suspension is white to slightly yellowish. Available in 100 mL bottle (Aust R 93720).
Who distributes Amoxycillin Sandoz
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
This leaflet was prepared in November 2025.
® Registered Trade Mark. The trade marks mentioned in this material are the property of their respective owners.
Brand Information
| Brand name | Amoxycillin Sandoz Capsules and Suspension |
| Active ingredient | Amoxicillin |
| Schedule | S4 |
MIMS Revision Date: 01 October 2024
1 Name of Medicine
Amoxicillin trihydrate.
2 Qualitative and Quantitative Composition
Active ingredient: amoxicillin trihydrate.
Amoxycillin Sandoz Powder for Suspension contains 125 mg/5 mL or 250 mg/5 mL amoxicillin (as trihydrate).
Each Amoxycillin Sandoz Capsules contains 250 mg or 500 mg amoxicillin (as trihydrate).
Amoxicillin trihydrate is a white or almost white, crystalline powder.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Amoxycillin Sandoz Powder for Suspension is a white to slightly yellowish powder.
Amoxycillin Sandoz Capsules are white to cream powder in an opaque yellow hard gelatin capsule.
4 Clinical Particulars
4.1 Therapeutic Indications
Treatment of the following infections due to susceptible strains of sensitive organisms.
Note. Therapy should be guided by bacteriological studies, including sensitivity tests, and by clinical response. Amoxicillin alone or in combination with another antibiotic, may be used in an emergency where the causative has not been identified.
Respiratory tract infections (acute and chronic): H. influenzae; Streptococcus; S. pneumoniae; Staphylococcus, nonpenicillinase producing; E. coli (see Section 5.1 Pharmacodynamic Properties, Mechanism of action, Microbiology).
Urogenital infections (complicated and uncomplicated, acute and chronic): E. coli (see Section 5.1 Pharmacodynamic Properties, Mechanism of action, Microbiology), P. mirabilis and Strep. faecalis.
Gonorrhoea: N. gonorrhoea (nonpencillinase producing).
Skin and skin structure infections: Staphylococcus, nonpenicillinase producing; Streptococcus; E. coli (see Section 5.1 Pharmacodynamic Properties, Mechanism of action, Microbiology).
Prophylaxis of endocarditis: amoxicillin may be used for the prophylaxis of bacterial endocarditis in individuals at particular risk, such as those with a prosthetic heart valve or those who have previously had endocarditis.
Infections caused by pathogens with established penicillin G susceptibility should preferentially be treated with penicillin G.
4.2 Dose and Method of Administration
Dosage. Normal renal function. Upper respiratory tract infections; genitourinary tract infections; skin and soft tissue infections. Adults. 250 mg every eight hours.
Children (under 20 kg). 20 mg/kg/day in equally divided doses, every 8 hours.
In severe infections or those caused by less susceptible organisms, 500 mg every eight hours for adults and 40 mg/kg/day in equally divided doses every 8 hours for children may be needed.
Lower respiratory tract infections. Adults. 500 mg every eight hours.
Children (under 20 kg). 40 mg/kg/day every 8 hours in equally divided doses.
Urethritis, gonococcal. Adults. 3 g as a single dose.
Cases of gonorrhoea with a suspected lesion of syphilis should have darkfield examinations before receiving amoxicillin and monthly serological tests for a minimum of 4 months.
Acute, uncomplicated lower urinary tract infections in non-pregnant adult females. 3 g as a single dose.
Chronic urinary tract infections. Frequent biological and clinical appraisals are recommended for patients under treatment for chronic urinary tract infections. Doses smaller than those recommended should not be used. Therapy for stubborn infections may have to be extended for several weeks.
Bacteriological and clinical appraisals may have to be continued for several months following cessation of treatment.
Prophylaxis of endocarditis. Dental procedures. Prophylaxis for patients undergoing extraction, scaling or surgery involving gingival tissues, who are not having general anaesthetic, and who have not received a penicillin in the previous month. (Note: Patients with prosthetic heart valves should be referred to hospital).
Adults (including elderly). 3 g orally, 1 hour before procedure. A second does may be given 6 hours later if considered necessary.
Children. Under 10 years: half-adult dose; under 5 years: quarter adult dose.
(Note: Prophylaxis with alternative antibiotics should be considered if the patient has received penicillin within the previous month, or is allergic to penicillin).
Duration of treatment. Treatment should be continued for a minimum of 48 to 72 hours beyond the time when patients become asymptomatic or evidence of bacterial eradication has been obtained. It is recommended that there be a minimum of 10 days treatment for any infection caused by haemolytic streptococci to prevent occurrence of acute rheumatic fever or glomerulonephritis.
Method of administration. For oral administration.
Dosage adjustment. Renal impairment. In renal impairment the excretion of the antibiotic will be delayed, and depending on the degree of impairment, it may be necessary to reduce the total daily doses.
In patients receiving peritoneal dialysis, the maximum recommended dose is 500 mg/day. Amoxicillin may be removed from the circulation by haemodialysis.
Neonates. Experience in neonates is too limited to make any recommendations regarding dosage or the appropriateness of the oral route.
Children. The children's dosage is intended for individuals whose weight will not cause dosage greater than that recommended for adults. Children receiving amoxicillin every 8 hours and weighing more than 20 kg should receive the adult recommended doses.
4.3 Contraindications
Amoxicillin is a penicillin and should not be given to patients with a history of hypersensitivity to β-lactam antibiotics (e.g. penicillins, cephalosporins, carbapenem or monobactam). Potential cross allergy to other beta-lactams such as cephalosporins should be taken into account.
Known and suspected hypersensitivity to the active substance, to any of the penicillins or known hypersensitivity to any of the excipients.
Antibiotics have no place in trivial infections.
4.4 Special Warnings and Precautions for Use
Hypersensitivity reactions. Serious, and occasionally fatal, hypersensitivity reactions (including anaphylaxis, anaphylactoid, and severe cutaneous reactions) have been reported in patients receiving beta-lactam antibiotics. Hypersensitivity reactions can also progress to Kounis syndrome, a serious allergic reaction that can result in myocardial infarction (see Section 4.8 Adverse Effects (Undesirable Effects)). These reactions are more likely to occur in individuals with a history of penicillin hypersensitivity and in atopic individuals. Although anaphylaxis is more frequent following parenteral therapy, it has occurred in patients on oral therapy. Before commencing therapy with any penicillin careful enquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins or other allergens. If an allergic reaction occurs, appropriate therapy should be instituted and amoxicillin therapy discontinued. Patients should be told about the potential occurrence of allergic reactions and instructed to report them.
Serious anaphylactic reactions require immediate emergency treatment with adrenaline. Oxygen, intravenous steroids and airways management, including intubation should be administered as necessary.
Special caution should be exercised in patients with allergic diatheses or bronchial asthma and hay fever.
Drug-induced enterocolitis syndrome (DIES) has been reported mainly in children receiving amoxicillin (see Section 4.8 Adverse Effects (Undesirable Effects)). DIES is an allergic reaction with the leading symptom of protracted vomiting (1-4 hours after administration of amoxicillin) in the absence of allergic skin or respiratory symptoms. Further symptoms could comprise abdominal pain, diarrhoea, hypotension or leucocytosis with neutrophilia. There have been severe cases including progression to shock.
Skin reactions. The occurrence at the treatment initiation of a feverish generalised erythema associated with pustula may be a symptom of acute generalised exanthemous pustulosis (AGEP). This reaction requires amoxicillin discontinuation and contra-indicates any subsequent administration.
Amoxicillin should be avoided if infectious mononucleosis is suspected since the occurrence of a morbilliform rash has been associated with this condition following the use of amoxicillin.
Lymphatic leukaemia. Amoxicillin should be given with caution to patients with lymphatic leukaemia as they are susceptible to amoxicillin induced skin rashes.
Non-susceptible microorganisms. Amoxicillin is not suitable for the treatment of some types of infection unless the pathogen is already documented and known to be susceptible or there is a very high likelihood that the pathogen would be suitable for treatment with amoxicillin. This particularly applies when considering the treatment of patients with urinary tract infections and severe infections of the ear, nose and throat.
Amoxicillin, an aminopenicillin, is not the treatment of choice in patients presenting with sore throat or pharyngitis because of the possibility that the underlying cause is infectious mononucleosis, in the presence of which there is a high incidence of rash if amoxicillin is used.
Overgrowth of non-susceptible microorganisms. Prolonged use may occasionally result in overgrowth of non-susceptible organisms.
Antibiotic associated pseudomembranous colitis has been reported with many antibiotics including amoxicillin. A toxin produced by Clostridium difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. Clostridium difficile associated diarrhoea (CDAD) has been reported with the use of nearly all antibacterial agents and may range in severity from mild diarrhoea to fatal colitis. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with antibiotic use (this may occur up to several weeks after cessation of antibiotic therapy). If prolonged or significant diarrhoea occurs or the patient experiences abdominal cramps, treatment should be discontinued immediately and the patient investigated further. Mild cases usually respond to drug discontinuation alone. However, in moderate to severe cases appropriate therapy with a suitable oral antibacterial agent effective against Clostridium difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated. Drugs which delay peristalsis e.g. opiates and diphenoxylate with atropine (Lomotil) may prolong and/or worsen the conditions and should not be used.
Convulsions. Convulsions may occur in patients with impaired renal function or in those receiving high doses or in patients with predisposing factors (e.g. history of seizures, treated epilepsy or meningeal disorders (see Section 4.8 Adverse Effects (Undesirable Effects)).
Anticoagulants. Abnormal prolongation of prothrombin time (increased INR) has been reported rarely in patients receiving amoxicillin and oral anticoagulants. Appropriate monitoring should be undertaken when anticoagulants are prescribed concurrently. Adjustments in the dose of oral anticoagulants may be necessary to maintain the desired level of anticoagulation.
Prolonged therapy. As with any potent drug, periodic assessment of renal, hepatic and haematopoietic function should be made during prolonged therapy. Elevated liver enzymes and changes in blood counts have been reported.
The possibility of superinfection with mycotic or bacterial pathogens should be kept in mind. If superinfection occurs (usually involving Aerobacter, Pseudomonas or Candida) discontinue the drug and/or institute appropriate therapy.
Jarisch-Herxheimer reaction. The Jarisch-Herxheimer reaction has been seen following amoxicillin treatment of Lyme disease. It results directly from the bactericidal activity of amoxicillin on the causative bacteria of Lyme disease, the spirochaete Borrelia burgdorferi. Patients should be reassured that this is a common and usually self-limiting consequence of antibiotic treatment of Lyme disease.
Urinary tract infections. Following single dose therapy of acute lower urinary tract infections, the urine should be cultured. A positive culture may be evidence of a complicated or upper urinary tract infection, and higher dose or prolonged course of treatment may be appropriate.
Crystalluria. In patients with reduced urine output, crystalluria has been observed very rarely, predominantly with parenteral therapy. In patients with bladder catheters, a regular check of patency should be maintained (see Section 4.8 Adverse Effects (Undesirable Effects); Section 4.9 Overdose). At high doses, adequate fluid intake and urinary output must be maintained to minimise the possibility of amoxicillin crystalluria.
Use in renal impairment. Dosage should be adjusted in patients with renal impairment (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use, Convulsions).
Use in the elderly. No data available.
Paediatric use. Precaution should be taken in premature children and during neonatal period: renal, hepatic and haematological functions should be monitored.
Effects on laboratory tests. Since high urine concentrations of amoxicillin may result in false positive reactions when testing for the presence of glucose in urine using Clinitest, Benedict's solution or Fehling's solution, it is recommended that glucose tests based on enzyme-based glucose oxidase reactions (such as Clinistix, or Testape) be used.
Following administration of ampicillin to pregnant women, a transient decrease in plasma concentration of total conjugated oestriol, oestriol-glucuronide, conjugated oestrone and oestradiol has been noted. This effect may also occur with amoxicillin.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Allopurinol. The concurrent administration of allopurinol and ampicillin increases substantially the incidence of rashes in patients receiving both drugs as compared to patients receiving ampicillin alone. It is not known whether this potentiation of ampicillin rashes is due to allopurinol or the hyperuricemia present in these patients. Similar reactions can be expected with amoxicillin.
Digoxin. An increase in the absorption of digoxin is possible on concurrent administration with amoxicillin. A dose adjustment of digoxin may be necessary.
Anticoagulants. Concomitant administration of amoxicillin and anticoagulants from the coumarin class, may prolong the bleeding time. A dose adjustment of anticoagulants may be necessary (see Section 4.4 Special Warnings and Precautions for Use). If coadministration is necessary, the prothrombin time or international normalised ratio should be carefully monitored with the addition or withdrawal of amoxicillin.
A large number of cases showing an increase of oral anticoagulant activity has been reported in patients receiving antibiotics. The infectious and inflammatory context, age and the general status of the patient appear as risk factors. In these circumstances, it is difficult to know the part of the responsibility between the infectious disease and its treatment in the occurrence of INR disorders. However, some classes of antibiotics are more involved, notably fluoroquinolones, macrolides, cyclines, cotrimoxazole and some cephalosporins.
Probenecid. Concomitant use of probenecid is not recommended. Probenecid decreases the renal tubular secretion of amoxicillin. Concurrent use with amoxicillin may result in increased and prolonged blood levels of amoxicillin.
Tetracyclines. Tetracyclines and other bacteriostatic drugs may interfere with the bactericidal effects of amoxicillin.
Methotrexate. Penicillin may reduce the excretion of methotrexate causing a potential increase in toxicity. Serum methotrexate levels should be closely monitored in patients who receive amoxicillin and methotrexate simultaneously (see Section 4.4 Special Warnings and Precautions for Use). Amoxicillin decreases the renal clearance of methotrexate, probably by competition at the common tubular secretion system.
Caution is recommended when amoxicillin is given concomitantly with:
Oral hormonal contraceptives. In common with other antibiotics, amoxicillin may affect the gut flora, leading to lower oestrogen reabsorption and reduced efficacy of combined oral contraceptives. Administration of amoxicillin can transiently decrease the plasma level of oestrogens and progesterone, and may reduce the efficacy of oral contraceptives. It is therefore recommended to take supplemental non-hormonal contraceptive measures.
Other forms of interactions. Forced diuresis leads to a reduction in blood concentrations by increased elimination of amoxicillin.
Amoxicillin may decrease the amount of urinary estriol in pregnant women.
At high concentrations, amoxicillin may diminish the results of serum glycemia levels.
Amoxicillin may interfere with protein testing when colorimetric methods are used.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no data on the effects of amoxicillin on fertility in humans. Reproductive studies in animals have shown no effects on fertility.
Use in pregnancy. (Category A)
Animal studies with amoxicillin have shown no teratogenic effects. The product has been in extensive clinical use since 1972 and its suitability in human pregnancy has been well documented in clinical studies.
Amoxicillin may be used in pregnancy when the potential benefits outweigh the potential risks associated with treatment.
Use in labour and delivery. Oral ampicillin class antibiotics are generally poorly absorbed during labour. Studies in guinea pigs have shown that intravenous administration of ampicillin decreased the uterine tone, frequency of contractions and duration of contractions. However, it is not known whether the use of amoxicillin in humans during labour or delivery has immediate or delayed adverse effects on the foetus, prolongs the duration of labour or increases the likelihood that forceps delivery or other obstetrical intervention or resuscitation of the newborn will be necessary.
Use in lactation. Ampicillin class antibiotics are excreted in breast milk; therefore caution should be exercised when amoxicillin is administered to a nursing woman. So far, no detrimental effects for the breast-fed infant have been reported after taking amoxicillin. Amoxicillin can be used during breast-feeding. However, breast-feeding must be stopped if gastrointestinal disorders (diarrhoea, candidosis or skin rash) occur in the new born.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. However, undesirable effects may occur (e.g. allergic reactions, dizziness, convulsions), which may influence the ability to drive and use machines (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.8 Adverse Effects (Undesirable Effects)
As with other penicillins, it may be expected that untoward reactions will be essentially limited to sensitivity phenomena. They are more likely to occur in individuals who have previously demonstrated hypersensitivity to penicillins.
The most commonly reported adverse drug reactions (ADRs) are diarrhoea, nausea and skin rash.
The following adverse reactions have been reported as associated with the use of amoxicillin:
Gastrointestinal. Nausea, vomiting, diarrhoea. Intestinal candidiasis, antibiotic associated colitis (including pseudomembranous colitis and haemorrhagic colitis), superficial discoloration of the teeth (especially with the suspension) have been reported rarely (see Section 4.4 Special Warnings and Precautions for Use). Usually the discoloration can be removed by teeth brushing. If severe and persistent diarrhoea occurs, the very rare possibility of pseudomembranous colitis should be considered. The administration of anti-peristaltic drug is contraindicated. Black hairy tongue and haemorrhagic colitis have been reported very rarely.
Drug-induced enterocolitis syndrome: not known (see Section 4.4 Special Warnings and Precautions for Use).
Cardiac disorders. Kounis syndrome: not known.
Hypersensitivity. Erythematous maculopapular rash, pruritus and urticaria have been reported occasionally. Rarely, skin reactions such as erythema multiforme exsudativum, acute generalised pustulosis, Lyell's syndrome, Stevens-Johnson syndrome, toxic epidermal necrolysis, bullous and exfoliative dermatitis, acute generalised exanthematous pustulosis (AGEP), Jarisch-Herxheimer reaction and drug reaction with eosinophilia and systemic symptoms (DRESS), and symmetrical drug-related intertriginous and flexural exanthema (SDRIFE) (baboon syndrome) have been reported. As with other antibiotics, severe allergic reactions including angioneurotic oedema, anaphylaxis, serum sickness, hypersensitivity vasculitis and interstitial nephritis (crystalluria) have been reported rarely. Whenever such reactions occur, amoxicillin should be discontinued.
Note. Urticaria, other skin rashes and serum sickness-like reactions may be controlled with antihistamines and, if necessary, systemic corticosteroids.
Anaphylaxis is the most serious reaction experienced (see Section 4.4 Special Warnings and Precautions for Use).
Hepatic. A moderate rise in AST and/or ALT has occasionally been noted, but the significance of this finding is unknown. As with other beta-lactam antibiotics, hepatitis and cholestatic jaundice have been reported rarely.
Haemic and lymphatic systems. Reactions such as anaemia, thrombocytopenia, thrombocytopenic purpura, eosinophilia and leucopenia (including severe neutropenia or agranulocytosis) have been reported during therapy with other penicillins. These reactions are usually reversible on discontinuation of therapy and are believed to be hypersensitivity phenomena. Prolongation of bleeding time and prothrombin time have also been reported rarely.
Central nervous system effects. CNS effects have been seen rarely. They include hyperkinesia, dizziness and convulsions. Convulsions may occur in patients with impaired renal function or in those receiving high doses. Aseptic meningitis has also been reported.
Infections and infestations. Mucocutaneous candidiasis have been reported very rarely.
Renal and urinary tract disorders. Interstitial nephritis, crystalluria (including acute renal injury) have been reported very rarely (see Section 4.9 Overdose).
Skin and subcutaneous tissue disorders. Linear IgA disease: not known.
Miscellaneous. Superficial tooth discolouration has been reported very rarely in children. Good oral hygiene may help to prevent tooth discolouration as it can usually be removed by brushing.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Signs of overdosage of amoxicillin would predominately be gastrointestinal related. The symptoms may include abdominal or stomach cramps and pain, severe nausea, vomiting or diarrhoea. Treatment of penicillin overdosage should be symptomatic and supportive. Haemodialysis may aid in the removal of penicillins from the blood. Amoxicillin crystalluria, in some cases leading to renal failure, has been observed (see Section 4.8 Adverse Effects (Undesirable Effects)).
Please see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects).
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Microbiology. Amoxicillin trihydrate is a broad-spectrum penicillin similar to ampicillin in its bactericidal action. It is believed to act through the inhibition of biosynthesis of cell wall mucopeptide. It is active against both Gram-positive and Gram-negative microorganisms. Amoxicillin is active in vitro against beta-lactamase negative strains of Proteus mirabilis, and Haemophilus influenzae. In vitro studies have also demonstrated activity against most strains of alpha- and beta-haemolytic streptococci. Streptococcus pneumoniae, and beta-lactamase negative strains of staphylococci, Neisseria gonorrhoeae, Neisseria meningitidis and Enterococcus faecalis. However, some of the organisms are sensitive to amoxicillin only at concentrations achieved in the urine. Strains of gonococci which are relatively resistant to benzyl penicillin may also be resistant to amoxicillin. Amoxicillin is not effective against penicillinase producing bacteria, particularly resistant staphylococci which now have a high prevalence. All strains of Pseudomonas, Klebsiella, Enterobacter, indole positive Proteus, Serratia marcescens, Citrobacter, penicillinase producing N. gonorrhoeae and penicillinase producing H. influenzae are also resistant. Escherichia coli isolates are becoming increasingly resistant to amoxicillin in vitro due to the presence of penicillinase-producing strains. See Table 1.
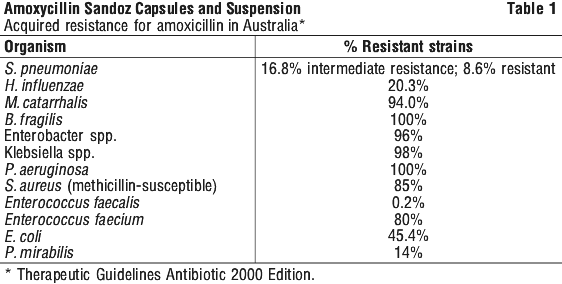
A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of "Intermediate" indicates that the result should be considered equivocal, and if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone, which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy may be selected.
Note. The prevalence of resistance may vary geographically for selected species and local information on resistance is desirable, particularly when treating severe infections.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Amoxicillin is stable in the presence of gastric acid and is rapidly and well absorbed after oral administration, even in the presence of food. Peak serum levels are reached within one or two hours after ingestion. Orally administered doses of amoxicillin 250 and 500 mg result in average peak serum levels one to two hours after administration of 5 microgram/mL and 10.25 microgram/mL respectively. Detectable serum levels of amoxicillin are present eight hours after ingestion of a single dose.
Distribution. Amoxicillin readily distributes in most body tissues and fluids with the exception of brain and spinal fluid except when the meninges are inflamed. Amoxicillin has been shown to diffuse into sputum and saliva. Amoxicillin is only 17% protein bound in serum.
Metabolism. Amoxicillin is excreted in the urine as unchanged drug and as penicilloic acid.
Excretion. Amoxicillin is excreted mainly via the urine where it exists in a high concentration. Concentrations in the bile may vary and are dependent upon normal biliary function. Amoxicillin is eliminated with a half-life of 61.3 minutes with normal renal function and up to 16-20 hours in the absence of renal function. Amoxicillin is excreted in the urine as unchanged drug and as penicilloic acid. Approximately 75% of a 1 g dose is excreted in the urine within six hours with normal renal function. However, there is a proportional difference in the amount excreted following different doses, due to lack of linearity in the rate of absorption with higher doses. Elimination of amoxicillin can be delayed by concurrent administration of probenecid.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Capsules. Magnesium stearate, microcrystalline cellulose, gelatin, iron oxide yellow and titanium dioxide.
Powder for oral suspension. Anhydrous citric acid, sodium benzoate, aspartame, purified talc, sodium citrate anhydrous, guar gum, silicon dioxide, lemon flavouring, peach-apricot flavouring and orange flavouring.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information on interactions with other medicines and other forms of interactions, see Section 4.5.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Amoxycillin Sandoz capsules. Store below 25°C, protect from moisture.
Amoxycillin Sandoz powder for oral suspension. Powder: store below 25°C. Keep the container tightly closed.
After mixing: store in a refrigerator (2°C-8°C).
6.5 Nature and Contents of Container
Amoxycillin Sandoz Powder for Suspension is available in 60 mL and 100 mL bottles.
Amoxycillin Sandoz Capsules are available in a PVC/PVDC/Al blister pack. Amoxycillin Sandoz 250 mg is available in packs of 20 capsules. Amoxycillin Sandoz 500 mg is available in packs of 20 and 28 capsules.
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
Discard unused suspension after 14 days.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Amoxicillin trihydrate is slightly soluble in water and in ethanol (96%), practically insoluble in chloroform, in ether and in fatty oils.
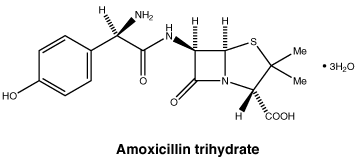
Chemical structure. The chemical name of Amoxicillin trihydrate is (2S,5R,6R)-6-[(R)-2-amino-2-(4-hydroxyphenyl)acetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid trihydrate. Its molecular formula is C16H19N3O5S.3H2O (Molecular Weight: 419.4) and its structural formula is:
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
22 May 2008
Date of Revision
05 August 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.