APOHEALTH Sinus Nasal Decongestant Tablets
Brand Information
| Brand name | APOHEALTH Sinus Nasal Decongestant Tablets |
| Active ingredient | Pseudoephedrine hydrochloride |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the APOHEALTH Sinus Nasal Decongestant Tablets.
Summary CMI
APOHEALTH SINUS Nasal Decongestant Tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using APOHEALTH Sinus Nasal Decongestant tablets?
APOHEALTH Sinus Nasal Decongestant tablets contains the active ingredient pseudoephedrine hydrochloride. APOHEALTH Sinus Nasal Decongestant tablets is used to clear nasal congestion and dry runny nose, making breathing easier.
For more information, see Section 1. Why am I using APOHEALTH Sinus Nasal Decongestant tablets? in the full CMI.
2. What should I know before I use APOHEALTH Sinus Nasal Decongestant tablets?
Do not use if you have ever had an allergic reaction to pseudoephedrine hydrochloride or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use APOHEALTH Sinus Nasal Decongestant tablets? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with APOHEALTH Sinus Nasal Decongestant tablets and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use APOHEALTH Sinus Nasal Decongestant tablets?
- Adults and children over 12 years of age: 1 tablet 3 to 4 times a day, when necessary. Do not exceed 4 tablets in 24 hours.
- Not suitable for use in children under 12 years.
More instructions can be found in Section 4. How do I use APOHEALTH Sinus Nasal Decongestant tablets? in the full CMI.
5. What should I know while using APOHEALTH Sinus Nasal Decongestant tablets?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using APOHEALTH Sinus Nasal Decongestant tablets? in the full CMI.
6. Are there any side effects?
The common side effects includes raised blood pressure, difficulty sleeping, nervousness, tremor (shaking),dizziness, headache, fast or irregular heartbeat, palpitations, sweating, flushing, anxiety, restlessness, skin rash, hallucinations, dry mouth or loss of appetite.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
APOHEALTH SINUS Nasal Decongestant Tablets
Active ingredient(s): Pseudoephedrine hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using APOHEALTH Sinus Nasal Decongestant tablets. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using APOHEALTH Sinus Nasal Decongestant tablets.
Where to find information in this leaflet:
1. Why am I using APOHEALTH Sinus Nasal Decongestant tablets?
2. What should I know before I use APOHEALTH Sinus Nasal Decongestant tablets?
3. What if I am taking other medicines?
4. How do I use APOHEALTH Sinus Nasal Decongestant tablets?
5. What should I know while using APOHEALTH Sinus Nasal Decongestant tablets?
6. Are there any side effects?
7. Product details
1. Why am I using APOHEALTH Sinus Nasal Decongestant tablets?
APOHEALTH Sinus Nasal Decongestant tablets contains the active ingredient pseudoephedrine hydrochloride. APOHEALTH Sinus Nasal Decongestant tablets contains pseudoephedrine hydrochloride which acts as a decongestant.
APOHEALTH Sinus Nasal Decongestant tablets is used to clear nasal congestion and dry runny nose, making breathing easier.
2. What should I know before I use APOHEALTH Sinus Nasal Decongestant tablets?
Warnings
Do not use APOHEALTH Sinus Nasal Decongestant tablets if:
- you are allergic to pseudoephedrine, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- Symptoms of an allergic reaction may include: cough, shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue, throat or other parts of the body; rash, itching or hives on the skin; fainting; or hay fever-like symptoms.
If you think you are having an allergic reaction, do not take any more of the medicine and contact your doctor immediately or go to the Accident and Emergency department at the nearest hospital. - You are taking, or within 14 days after stopping, monoamine oxidase inhibitors (MAOIs), a type of medicine used to treat depression.
- You have very high blood pressure (severe hypertension) or hypertension not controlled by your medication.
- You have severe acute (sudden) or chronic (long-term) kidney disease or kidney failure.
- Do not give to children under 12 years.
- The expiry date (EXP) printed on the pack has passed.
- The packaging is torn, shows signs of tampering or it does not look quite right.
If you are not sure whether you should take this medicine, talk to your doctor or pharmacist.
Check with your doctor if you:
- Have allergies to:
- Any other medicines
- Any other substances, such as foods, preservatives or dyes. - take any medicines for any other condition
- You have or had any medical condition, especially the following:
- High blood pressure
- Heart disease
- Depression
- Lung, liver or kidney disorders overactive thyroid gland
- Prostate problems such as enlarged prostate
- Bladder problems
- Diabetes mellitus (high blood sugar)
- Increased pressure in the eye (glaucoma)
- Any other medical problems - You are currently pregnant or you plan to become pregnant. Do not take this medicine whilst pregnant until you and your doctor have discussed the risks and benefits involved
- You are currently breastfeeding or you plan to breast-feed. Do not take this medicine whilst breastfeeding.
- Small amounts of pseudoephedrine may be excreted in breast milk.
- You are planning to have surgery or an anaesthetic.
- You are currently receiving or are planning to receive dental treatment.
- You are taking or are planning to take any other medicines. This includes vitamins and supplements that are available from your pharmacy, supermarket or health food shop.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with APOHEALTH Sinus Nasal Decongestant tablets and affect how it works.
- medicines for high blood pressure
- medicines used for arrhythmia (for e.g. digoxin, quinidine)
- medicines that suppress appetite and for slimming
- other nasal decongestants (e.g. medicines used to treat symptoms of colds and flu) medicines for depression
- medicines used to relieve the burning sensation associated with urinary tract infections
- levodopa (a medicine used to treat Parkinson's disease)
If you are taking any of these you may need a different dose or you may need to take different medicines.
Other medicines not listed above may also interact with pseudoephedrine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect APOHEALTH Sinus Nasal Decongestant tablets.
4. How do I use APOHEALTH Sinus Nasal Decongestant tablets?
How much to take / use
- Adults and children over 12 years of age:
1 tablet 3 to 4 times a day, when necessary. Do not exceed 4 tablets in 24 hours.
Not suitable for use in children under 12 years. - If you are unsure what dose to take, ask your pharmacist.
When to take / use APOHEALTH Sinus Nasal Decongestant tablets
- APOHEALTH Sinus Nasal Decongestant tablets should be used for the relief of minor and temporary ailments and should be used strictly as directed. Prolonged use may be harmful. If symptoms persist, seek medical advice.
If you forget to use APOHEALTH Sinus Nasal Decongestant tablets
APOHEALTH Sinus Nasal Decongestant tablets should be used regularly at the same time each day.
If you miss a dose, do not take a double dose to make up.
This medicine should only be used when necessary.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much APOHEALTH Sinus Nasal Decongestant tablets
If you think that you have used too much APOHEALTH Sinus Nasal Decongestant tablets, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using APOHEALTH Sinus Nasal Decongestant tablets?
Things you should do
Take APOHEALTH Sinus Nasal Decongestant tablets as described on the label and in this leaflet. If your symptoms do not improve within 7 days, you should contact your doctor for further advice.
Call your doctor straight away if you:
- you are about to be started on any new medicine
- you are pregnant or are planning to become pregnant
- you are breastfeeding or are planning to breast-feed
- you are about to have any blood tests
- you are going to have surgery or an anaesthetic or are going into hospital.
Remind any doctor, dentist or pharmacist you visit that you are using APOHEALTH Sinus Nasal Decongestant tablets.
Things you should not do
- Give this medicine to anyone else, even if their symptoms seem similar to yours.
- Take your medicine to treat any other condition unless your doctor or pharmacist tells you to.
- Change the dosage, without first checking with your doctor or pharmacist.
Things to be careful of
This product may cause sleeplessness if it is taken up to several hours before going to bed.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how APOHEALTH Sinus Nasal Decongestant tablets affects you.
APOHEALTH Sinus Nasal Decongestant tablets may cause dizziness in some people
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol may have effects while taking the medicine.
Looking after your medicine
Keep your medicine in its original packaging until it is time to take it.
If you take your medicine out of its original packaging it may not keep well.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep your medicine in a cool dry place where the temperature will stay below 30°C.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
When to discard your medicine (as relevant)
The expiry date (EXP) printed on the pack has passed.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Children and the elderly may have an increased chance of getting side effects
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Allergic reactions
If you think you are having an allergic reaction to APOHEALTH Sinus Nasal Decongestant tablets, do not take any more of this medicine and tell your doctor immediately or go to the Accident and Emergency department at your nearest hospital.
Symptoms of an allergic reaction may include some or all of the following:
- cough, shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- rash, itching or hives on the skin
- fainting
- hay fever-like symptoms.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What APOHEALTH Sinus Nasal Decongestant tablets contains
| Active ingredient (main ingredient) | Pseudoephedrine hydrochloride 60mg |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | N/A |
This medicine contains no lactose, sugar, gluten, yeast, artificial colouring, preservatives, starch, dairy products, glucose or alcohol.
Do not take this medicine if you are allergic to any of these ingredients.
What APOHEALTH Sinus Nasal Decongestant tablets looks like
APOHEALTH Sinus Nasal Decongestant tablets come as white round-shaped tablets.
Blister pack of 12 tablets
Aust R 265772
Who distributes APOHEALTH Sinus Nasal Decongestant tablets
Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne, VIC 3121
www.arrotex.com.au
This leaflet was prepared in December 2025.
Brand Information
| Brand name | APOHEALTH Sinus Nasal Decongestant Tablets |
| Active ingredient | Pseudoephedrine hydrochloride |
| Schedule | S3 |
MIMS Revision Date: 01 February 2026
1 Name of Medicine
Pseudoephedrine hydrochloride.
2 Qualitative and Quantitative Composition
Active ingredient is pseudoephedrine hydrochloride 60 mg.
For the full list of excipients see Section 6.1 List of Excipients.
3 Pharmaceutical Form
60 mg tablet. White, round.
AUST R 265772.
4 Clinical Particulars
4.1 Therapeutic Indications
Nasal and sinus decongestant for symptomatic relief of runny noses and sinus congestion.
4.2 Dose and Method of Administration
Adults and children over 12 years. 1 tablet 3 to 4 times a day, when necessary.
Do not exceed 4 tablets per day.
Do not give to children under 12 years.
4.3 Contraindications
Known hypersensitivity to pseudoephedrine or to any other ingredient in the product (see Section 2 Qualitative and Quantitative Composition).
Patients receiving monoamine oxidase (MAO) inhibitor antidepressants and within 14 days of ceasing treatment with a MAO inhibitor.
Patients with severe or uncontrolled hypertension.
Patients with severe coronary artery disease.
Patients with severe acute or chronic kidney disease/renal failure.
Children under 12 years.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
4.4 Special Warnings and Precautions for Use
This medicine should be administered with caution in patients with: hypertension, hyperthyroidism, heart disease, patients with diabetes mellitus, patients with prostatic enlargement, elevated intraocular pressure, bladder dysfunction.
Ischaemic colitis: Some cases of ischaemic colitis have been reported with pseudoephedrine. Pseudoephedrine should be discontinued and medical advice sought if sudden abdominal pain, rectal bleeding or other symptoms of ischaemic colitis develop.
Pseudoephedrine may cause sleeplessness if taken up to several hours before going to bed.
Posterior reversible encephalopathy syndrome (PRES) and reversible cerebral vasoconstriction syndrome (RCVS). Cases of PRES and RCVS have been reported with the use of pseudoephedrine-containing products (see Section 4.8 Adverse Effects (Undesirable Effects)). The risk is increased in patients with severe or uncontrolled hypertension, or with severe acute or chronic kidney disease/renal failure (see Section 4.3 Contraindications).
Pseudoephedrine should be discontinued and immediate medical assistance sought if the following symptoms occur: sudden severe headache or thunderclap headache, nausea, vomiting, confusion, seizures and/or visual disturbances. Most reported cases of PRES and RCVS resolved following discontinuation and appropriate treatment.
Use in hepatic impairment. Pseudoephedrine should be used with caution in patients with severe hepatic dysfunction.
Use in renal impairment. Pseudoephedrine is contraindicated for use in patients with severe acute or chronic kidney disease/renal failure (see Section 4.3 Contraindications).
Use in the elderly. No data available.
Paediatric use. See Section 4.3 Contraindications.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The effects of pseudoephedrine may be enhanced by acetazolamide, disodium hydrogen citrate, sodium acid citrotartrate and sodium bicarbonate. Acetazolamide may reduce the glomerular filtration rate in the kidney, and decrease the rate of elimination of pseudoephedrine. Citrates and sodium bicarbonate could increase urinary pH, and decrease the rate of elimination of pseudoephedrine.
Concurrent use of pseudoephedrine with monoamine oxidase (MAO) inhibitor antidepressants may cause severe hypertension and headaches. Pseudoephedrine should not be taken within 14 days of ceasing treatment with a MAO inhibitor (see Section 4.3 Contraindications).
Concomitant use of pseudoephedrine and other sympathomimetic agents such as decongestants, appetite suppressants and amphetamine-like psychostimulants may lead to an increase in blood pressure and increase the potential for other side effects.
Administration with tricyclic antidepressants may increase blood pressure or increase the risk of arrhythmias.
Administration with digitalis glycosides, quinidine or levodopa may increase the risk of arrhythmias.
The antibacterial agent, furazolidone, is known to cause a progressive inhibition of monoamine oxidase. Although there are no reports of a hypertensive crisis caused by the concurrent administration of pseudoephedrine and furazolidone, they should not be taken together.
Pseudoephedrine may inhibit the effects of antihypertensive medication, including methyldopa or beta-adrenergic blocking agents.
Administration with other CNS stimulants may lead to increased CNS stimulation.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with pseudoephedrine. It is also not known whether pseudoephedrine can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. This medicine should be given to a pregnant woman only if the potential benefits to the patient are weighed against the possible risk to the foetus.
Australian categorisation definition of Category B2 drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed. Studies in animals are adequate or may be lacking, but available data show no evidence of an increased occurrence of foetal damage.
Use in lactation. Pseudoephedrine is excreted in breast milk in small amounts but the effect of this on breastfed infants is not known.
Use of pseudoephedrine while breastfeeding is not recommended.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Pseudoephedrine may cause insomnia, urinary retention or dose dependent CNS excitation. Rarely, sleep disturbances and hallucinations have been reported. Other symptoms may occur, such as nervousness, tremor, vertigo, headache, elevated blood pressure, tachycardia, arrhythmias, palpitations, sweating or flushing, anxiety, restlessness, dry mouth, anorexia, skin rashes and tension.
Gastrointestinal disorders (frequency unknown): Ischaemic colitis.
Children and the elderly are more likely to experience adverse effects than other age groups.
Nervous system disorders. Posterior reversible encephalopathy syndrome (PRES) (see Section 4.4 Special Warnings and Precautions for Use): frequency not known.
Reversible cerebral vasoconstriction syndrome (RCVS) (see Section 4.4 Special Warnings and Precautions for Use): frequency not known.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms include convulsions, irritability, palpitations, hypertension and difficulty passing urine.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Pseudoephedrine acts as a decongestant of the mucosa of the nose and sinuses to clear nasal congestion and dry nasal secretions.
Pseudoephedrine is a direct and indirect acting sympathomimetic agent, with similar actions to ephedrine, but with less cardiovascular or CNS effects.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Pseudoephedrine is readily absorbed from the gastrointestinal tract after oral administration. Peak concentrations are reached between 0.5 and 2 hours after administration.
Distribution. Distribution into extravascular sites is extensive with the apparent volume of distribution between 2.6 and 5 L/kg.
Metabolism. Pseudoephedrine is not substantially metabolised and is excreted primarily unchanged in the urine. Less than 1% of pseudoephedrine is metabolised in the liver, by N-demethylation, to norpseudoephedrine.
Excretion. As pseudoephedrine is a weak base with pKa of 9.4, the elimination half-life varies from 3-6 hours at a urine pH of 5 to 9; 16 hours at pH of 8, with 5-8 hours at pH 5.8. At high urine pH (greater than 7), the drug is extensively reabsorbed in the renal tubules and the rate of excretion is therefore dependent on pH and urine flow rate. This does not appear to be the case at low urine pH.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Inactive ingredients are magnesium stearate, microcrystalline cellulose, and calcium hydrogen phosphate dihydrate.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Blister pack of 12 tablets.
6.6 Special Precautions for Disposal
In Australia, any used medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
A white, crystalline powder or colourless crystals, freely soluble in water and in alcohol, sparingly soluble in methylene chloride. It melts at about 184°C.
Chemical structure.
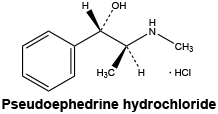
Molecular weight: 201.7.
CAS number. 345-78-8.
7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist Only Medicine.
Date of First Approval
10 December 2015
Date of Revision
03 December 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.