Aramine
Brand Information
| Brand name | Aramine |
| Active ingredient | Metaraminol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Aramine.
Summary CMI
Aramine®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about being given this medicine, speak to your doctor or pharmacist.
1. Why am I being given Aramine?
Aramine contains the active ingredient metaraminol tartrate. Aramine is used to increase your blood pressure which can drop during spinal anaesthesia or can drop as a reaction to medications or surgical complications.
For more information, see Section 1. Why am I being given Aramine? in the full CMI.
2. What should I know before being given Aramine?
You should not be given Aramine if you have ever had an allergic reaction to Aramine or any of the ingredients listed at the end of the CMI in particular, sodium metabisulfite.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before being given Aramine? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Aramine and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How will Aramine be given?
- Aramine must only be given by a doctor or nurse.
- It will be given in a hospital by direct injection into a vein or diluted and given as an infusion fluid into a vein.
More information can be found in Section 4. How will Aramine be given? in the full CMI.
5. What should I know when being given Aramine?
| Things you should do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know when being given Aramine? in the full CMI.
6. Are there any side effects?
Possible side effects include fast or pounding heartbeat, swelling of the face, lips, mouth or throat which may cause difficulty in swallowing or breathing and breathlessness. If these occur tell your doctor or nurse immediately.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Aramine®
Active ingredient: metaraminol tartrate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Aramine. You should also speak to your doctor if you would like further information or if you have any concerns or questions about using Aramine.
Where to find information in this leaflet:
1. Why am I being given Aramine?
2. What should I know before being given Aramine?
3. What if I am taking other medicines?
4. How will Aramine be given?
5. What should I know when being given Aramine?
6. Are there any side effects?
7. Product details
1. Why am I being given Aramine?
Aramine contains the active ingredient metaraminol tartrate. Aramine belongs to a group of medicines known as sympathomimetic amines which work by strengthening the contraction of the heart muscle and constricting blood vessels to help increase the blood pressure.
Aramine is used to increase your blood pressure which can drop during spinal anaesthesia or can drop as a reaction to medications or surgical complications.
2. What should I know before being given Aramine?
Warnings
You should not be given Aramine if:
- you are allergic to metaraminol tartrate, or any of the ingredients listed at the end of this leaflet. Aramine contains sodium metabisulfite. Sulfites may cause an allergic-type reaction or an asthmatic episode in certain susceptible people.
Symptoms of an allergic reaction may include: shortness of breath; wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
Always check the ingredients to make sure you can be given this medicine. - it is being given with cyclopropane or halothane anaesthesia (unless clinical circumstances demand their use).
- the solution is discoloured, cloudy, turbid, or a precipitate is present. The solution is normally clear and colourless.
Check with your doctor if you:
- have or have had any of the following medical conditions:
- liver disease
- heart or thyroid disease
- high blood pressure
- diabetes
- malaria - take any medicines for any other condition
- have allergies to any other medicines, sulfites or any other substances including foods, preservatives or dyes.
During treatment, you may be at risk of developing certain side effects. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Children
Aramine is not recommended for use in children.
3. What if I am taking other medicines?
Tell your doctor if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Aramine and affect how it works.
These include:
- monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs), medicines used to treat depression
- digoxin, a medicine used to treat heart failure.
Check with your doctor if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Aramine.
4. How will Aramine be given?
Aramine must only be given by a doctor or nurse.
How much is given
Your doctor will decide what dose of Aramine you will receive and how long you will receive it. This depends on your medical condition and other factors such as your weight.
How Aramine is given
Aramine will be given in a hospital by direct injection into a vein or diluted and given as an infusion fluid into a vein.
If you are given too much Aramine
As Aramine is always given to you in a hospital under the supervision of a doctor, it is unlikely that you will receive an overdose.
However, you should tell your doctor or nurse immediately if you feel unwell at all whilst you are being given Aramine. You may need urgent medical attention.
5. What should I know when being given Aramine?
Things you should do
- Always follow your doctor's instructions carefully.
Your doctor or nurse will monitor your blood pressure.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Aramine affects you.
Looking after your medicine
- Aramine should only be given to you in a hospital.
- It should be kept in the original packaging until it has been given to you.
- Aramine must be stored below 25°C, protected from light.
Getting rid of any unwanted medicine
Aramine should only be given to you in a hospital therefore the doctor or nurse will dispose of any leftover medicine.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or nurse if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
Heart-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects after being given Aramine. |
Tell your doctor if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What Aramine contains
| Active ingredient (main ingredient) | metaraminol tartrate |
| Other ingredients (inactive ingredients) | sodium chloride sodium metabisulfite tartaric acid sodium hydroxide water for injections |
| Potential allergens | sodium metabisulfite |
Do not take this medicine if you are allergic to any of these ingredients.
What Aramine looks like
Aramine is a clear, colourless to slightly yellow/pink solution in a clear glass vial closed with a rubber stopper, aluminium seal and a plastic flip off cap.
Aramine is available as a 10 mg/mL solution supplied in a carton containing 5 vials:
10 mg/mL: Aust R 284785
Who distributes Aramine
Phebra Pty Ltd
17-19 Orion Road
Lane Cove West NSW 2066
Australia
Telephone: 1800 720 020

This leaflet was prepared in December 2025.
Aramine, Phebra and the Phi symbol are registered trademarks of Phebra Pty Ltd, 19 Orion Road, Lane Cove West, NSW 2066, Australia.
Brand Information
| Brand name | Aramine |
| Active ingredient | Metaraminol |
| Schedule | S4 |
MIMS Revision Date: 01 December 2020
1 Name of Medicine
Metaraminol tartrate.
2 Qualitative and Quantitative Composition
Aramine injection contains 10 mg/mL metaraminol (as tartrate) as the active ingredient.
Each mL of Aramine injection contains metaraminol (as tartrate) 10 mg and sodium metabisulfite 2.0 mg as antioxidant.
Metaraminol tartrate is a white, crystalline powder, which is freely soluble in water, slightly soluble in alcohol, and practically insoluble in chloroform and in ether.
List of excipients with known effect. Sodium chloride and sodium metabisulfite.
This medicine contains sodium as sodium chloride and sodium metabisulfite.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Aramine injection is a clear colourless to slightly yellow/pink sterile solution of metaraminol tartrate equivalent to 10 mg/mL metaraminol.
4 Clinical Particulars
4.1 Therapeutic Indications
Prevention and treatment of the acute hypotensive state occurring with spinal anaesthesia; adjunctive treatment of hypotension due to haemorrhage, reactions to medications, surgical complications, and shock associated with brain damage due to tumour or trauma.
It may also be useful as an adjunct in the treatment of hypotension due to cardiogenic shock or septicaemia.
4.2 Dose and Method of Administration
Aramine injection is for intravenous administration only (injection or infusion) and should be used in one patient on one occasion only. It contains no antimicrobial preservative. Unused solution should be discarded.
Because the maximum effect is not immediately apparent, at least ten minutes should elapse before increasing the dosage. As the effect tapers off when the vasopressor is discontinued, the patient should be carefully observed so that therapy can be reinitiated promptly if the blood pressure falls too rapidly. Patients with coexistent shock and acidosis may show a poor response to vasopressors. Established methods of shock management, such as blood or fluid replacement when indicated, and other measures directed to the specific cause of the shock also should be used.
Intravenous infusion (for adjunctive treatment of hypotension). The recommended dose is 15 to 100 mg (1.5 to 10 mL) in 500 mL of sodium chloride injection or glucose injection 5%, adjusting the rate of infusion to maintain the blood pressure at the desired level.
Higher concentrations of metaraminol tartrate (150 to 500 mg/500 mL of infusion fluid) have been used.
If the patient needs additional saline or glucose solution at a rate of flow that would provide an excessive dose of the vasopressor, the recommended volume (500 mL) of infusion fluid should be increased accordingly. Conversely, if a smaller volume of infusion fluid is desirable, the required dose of metaraminol tartrate may be added to less than 500 mL of diluent.
Compatibility. In addition to sodium chloride injection and glucose injection 5%, the following infusion solutions were found physically and chemically compatible with metaraminol tartrate when 5 mL of Aramine injection was added to 500 mL of infusion solution: Ringer's injection and lactated Ringer's injection.
When metaraminol injection is mixed with an infusion solution, sterile precautions should be observed. To reduce microbiological hazard, use as soon as practicable after preparation. If storage is necessary, hold at 2-8°C for not more than 24 hours.
The injection solution contains no antimicrobial preservative and is for single use in one patient only. Discard any residue.
Direct intravenous injection. In severe shock, when time is of great importance, it may be desirable to administer Aramine injection by direct intravenous injection. The suggested dose is 0.5 to 5 mg (0.05 to 0.5 mL), followed by an infusion of 15 to 100 mg in 500 mL of diluent.
Direct intravenous injection of undiluted solution should be employed only in instances of grave emergency when prompt action is imperative to save life. Extreme care must be exercised to give the proper dose.
For direct intravenous injection use of a lower strength, 0.5 mg/mL metaraminol injection is recommended. A lower strength 0.5 mg/mL metaraminol injection can be obtained by diluting this 10 mg/1 mL product to 20 mL with 0.9% sodium chloride or by using a pre-diluted solution available under other tradenames.
4.3 Contraindications
Use with cyclopropane or halothane anaesthesia should be avoided, unless clinical circumstances demand such use.
Hypersensitivity to any component of this product including sulfites.
4.4 Special Warnings and Precautions for Use
Aramine injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
Caution should be exercised to avoid an excessive blood pressure response. Rapidly induced hypertensive responses have been reported to cause acute pulmonary oedema, cardiac arrhythmias and arrest. Patients with cirrhosis should be treated with caution, with adequate restoration of electrolytes if diuresis ensues. A fatal ventricular arrhythmia has been reported in a patient with Laennec's cirrhosis while receiving metaraminol tartrate. In several instances, ventricular extrasystoles that appeared during infusion subsided promptly when the rate of flow was reduced.
With the prolonged action of this drug, a cumulative effect is possible, and with an excessive vasopressor response there may be a prolonged elevation of blood pressure even when therapy with metaraminol tartrate is discontinued.
Because of its vasoconstrictor effect, metaraminol tartrate should be given with caution in the presence of heart or thyroid disease, hypertension, or diabetes. Sympathomimetic amines may provoke a relapse in patients with a history of malaria.
When vasopressor amines are used for long periods, the resulting vasoconstriction may prevent adequate expansion of the circulating volume and may cause perpetuation of the shock state. There is evidence that plasma volume may be reduced in all types of shock, and that the measurement of central venous pressure is useful in assessing the adequacy of the circulating blood volume. Therefore, blood or plasma volume expanders should be employed when the principal reason for hypotension or shock is decreased circulating volume.
In choosing the site of injection, it is important to avoid those areas recognised as unsuitable for the use of any pressor agent, and to discontinue the infusion immediately if infiltration or thrombosis occurs. Although the urgent nature of the patient's condition may force the choice of an unsuitable injection site, the preferred areas of injection should be used when possible. The larger veins of the antecubital fossa or thigh are preferred to the veins in the ankle or the dorsum of the hand, particularly in patients with peripheral vascular disease, diabetes mellitus, Buerger's disease, or conditions with coexistent hypercoagulability.
Use in the elderly. No data available.
Paediatric use. The effect of therapy with Aramine injection in children has not been established.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Aramine injection should be used with caution in digitalised patients, since the combination of digitalis and sympathomimetic amines is capable of causing ectopic arrhythmic activity.
MAOIs and tricyclic antidepressants have been reported to potentiate the action of sympathomimetic amines.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
There are no well controlled studies in pregnant women. Aramine injection may cause fetal hypoxia by constricting the uterine vessels thereby limiting placental perfusion.
Aramine injection should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Use in lactation. It is not known whether Aramine injection is secreted in human milk. Because many drugs are secreted in human milk, caution should be exercised if Aramine injection is given to a breastfeeding woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Abscess formation, tissue necrosis or sloughing rarely follow the use of metaraminol tartrate.
Sympathomimetic amines, including metaraminol tartrate, may cause sinus or ventricular tachycardia or other arrhythmias, especially in patients with myocardial infarction.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdosage may result in severe hypertension accompanied by headache, constricting sensation in the chest, nausea, vomiting, euphoria, diaphoresis, pulmonary oedema, tachycardia, bradycardia, sinus arrhythmia, atrial or ventricular arrhythmias, myocardial infarction, cardiac arrest or convulsions.
Should an excessive elevation of blood pressure occur, it may be immediately relieved by a sympatholytic agent, e.g. phentolamine. An appropriate antiarrhythmic agent may also be required.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
The oral LD50 in the rat and mouse is 240 mg/kg and 99 mg/kg, respectively.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Metaraminol is a potent sympathomimetic amine that increases both systolic and diastolic blood pressure. The pressor effect begins one to two minutes after intravenous injection, about 10 minutes after intramuscular injection, 5 to 20 minutes after subcutaneous injection, and lasts about 20 minutes to one hour. Metaraminol has a positive inotropic effect on the heart and has a peripheral vasoconstrictor action.
Renal, coronary, and cerebral blood flow are a function of perfusion pressure and regional resistance. In most instances of cardiogenic shock, the beneficial effect of sympathomimetic amines is attributable to their positive inotropic effect. In patients with insufficient or failing vasoconstriction, there is additional advantage to the peripheral action of metaraminol, but in most patients with shock, vasoconstriction is adequate and any further increase is unnecessary. Therefore, blood flow to vital organs may decrease with metaraminol if regional resistance increases excessively.
The pressor effect of metaraminol is decreased but not reversed by alpha-adrenergic blocking agents. A primary or secondary fall in blood pressure and a tachyphylactic response to repeated use are uncommon.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. No data available.
Distribution. No data available.
Metabolism. No data available.
Excretion. No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each mL of Aramine injection contains sodium chloride 8.5 mg, sodium metabisulfite 2.0 mg as antioxidant in water for injections to 1 mL. Tartaric acid and/or sodium hydroxide are added for pH adjustment.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the ARTG. The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Aramine injection is presented as 1 mL clear, colourless to slight yellow/pink solution in a 2 mL clear type I glass vial.
It is supplied in a carton containing 5 vials.
Phebra product code: INJ188.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
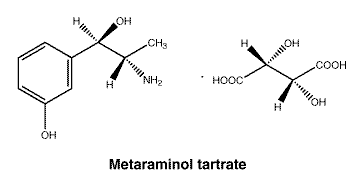
The molecular weight of the compound is 317.29. The molecular formula is C9H13NO2.C4H6O6.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
16 January 2018
Date of Revision
19 October 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.