Arianna 1
Brand Information
| Brand name | Arianna 1 |
| Active ingredient | Anastrozole |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Arianna 1.
Summary CMI
ARIANNA® 1
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about taking this medicine, speak to your doctor or pharmacist.
1. Why am I taking ARIANNA 1?
ARIANNA 1 contains the active ingredient anastrozole. ARIANNA 1 is used to treat breast cancer in women who no longer have their menstrual periods either naturally, due to their age or after surgery, radiotherapy or chemotherapy.
For more information, see Section 1. Why am I taking ARIANNA 1? in the full CMI.
2. What should I know before I take ARIANNA 1?
Do not take if you have ever had an allergic reaction to ARIANNA 1 or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take ARIANNA 1? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ARIANNA 1 and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take ARIANNA 1?
- The usual dose is one tablet every day.
- Swallow ARIANNA 1 tablets whole with plenty of water.
More instructions can be found in Section 4. How do I take ARIANNA 1? in the full CMI.
5. What should I know while taking ARIANNA 1?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking ARIANNA 1? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, some may be minor and temporary. However, some side effects may be serious. Tell your doctor immediately if you have sudden signs of allergy such as shortness of breath, wheezing or difficulty in breathing, swelling of the face, lips, tongue or any other parts of the body, rash, itching or hives on the skin, severe skin reactions with lesions, ulcers or blisters, liver pain or swelling and/or a general feeling of unwell with or without jaundice (yellowing of the skin and eyes).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ARIANNA® 1
Active ingredient(s): anastrozole
Consumer Medicine Information (CMI)
This leaflet provides important information about taking ARIANNA 1. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about taking ARIANNA 1.
Where to find information in this leaflet:
1. Why am I taking ARIANNA 1?
2. What should I know before I take ARIANNA 1?
3. What if I am taking other medicines?
4. How do I take ARIANNA 1?
5. What should I know while taking ARIANNA 1?
6. Are there any side effects?
7. Product details
1. Why am I taking ARIANNA 1?
ARIANNA 1 contains the active ingredient anastrozole.
ARIANNA 1 is a non-steroidal aromatase inhibitor, which reduces the amount of estrogen (female sex hormone) made by the body. In some types of breast cancer, estrogen can help the cancer cells grow. By blocking estrogen, ARIANNA 1 may slow or stop the growth of cancer.
ARIANNA 1 is used to treat breast cancer in women who no longer have their menstrual periods either naturally, due to their age or after surgery, radiotherapy or chemotherapy.
Your doctor may have prescribed ARIANNA 1 for another reason.
ARIANNA 1 is not addictive.
2. What should I know before I take ARIANNA 1?
Warnings
Do not take ARIANNA 1 if:
- you are allergic to:
- anastrozole, the active ingredient of ARIANNA 1
- any of the other ingredients in ARIANNA 1, as listed at the end of this leaflet
- other anti-estrogen medicines. - Some of the symptoms of an allergic reaction may include:
- skin rash, itching or hives;
- swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing;
- wheezing or shortness of breath. - you are still having menstrual periods
ARIANNA 1 should only be taken by women who are no longer having menstrual periods. - you are a man
Men are not normally treated with ARIANNA 1.
Check with your doctor if you:
- have or have had any other medical conditions
- have liver problems
- have kidney problems
- take any medicines for any other condition
- have allergies to any medicines or any other substances, such as foods, preservatives or dyes
- have osteoporosis, a family history of osteoporosis or risk factors for developing osteoporosis (such as smoking, a diet low in calcium, poor mobility, a slight build or treatment with steroid medicines).
Aromatase inhibitors may decrease bone mineral density (BMD) in women who have been through menopause, with a possible increased risk of fractures. Your doctor should discuss with you your treatment options for managing this possible increased risk of fractures.
Aromatase inhibitors may cause inflammation in tendons (see Section 6. Are there any side effects). At any sign of tendon pain or swelling - rest the painful area and contact your doctor.
Your doctor may want to take special care if you have any of these conditions.
If you have not told your doctor about any of the above, tell him/her before you start taking ARIANNA 1.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take ARIANNA 1 if you are pregnant or intend to become pregnant.
There is a possibility of harm to the developing baby as the medicine may be absorbed within the womb.
Do not breastfeed while taking ARIANNA 1.
Your baby can take in ARIANNA 1 from breast milk if you are breastfeeding.
Children
Do not give ARIANNA 1 to a child.
ARIANNA 1 is not recommended for use in children.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines should not be taken with ARIANNA 1. These include:
- tamoxifen, a medicine used to treat breast cancer
- any medicine that contains estrogen such as medicines used in Hormone Replacement Therapy (HRT) or oral contraceptives
- any health food products that contain natural estrogens used for post-menopausal symptoms
- medicines from a class called "Luteinising Hormone Releasing (LHRH) agonists", such as goserelin or leuprorelin.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ARIANNA 1.
4. How do I take ARIANNA 1?
How much to take
- The usual dose is one tablet every day.
- Swallow ARIANNA 1 tablets whole, with a glass of water.
- Follow the directions given to you by your doctor or pharmacist carefully, they may differ from the information contained in the leaflet.
When to take ARIANNA 1
Take ARIANNA 1 at about the same time each day.
Taking your tablet(s) at about the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take ARIANNA 1 before, with or after food.
How to long to take ARIANNA
Keep taking ARIANNA 1 for as long as your doctor or pharmacist tells you.
ARIANNA 1 helps to control your condition, but does not cure it. Therefore, you must take ARIANNA 1 every day. Do not stop taking it unless your doctor tells you to - even if you feel better.
If you forget to take ARIANNA 1
ARIANNA 1 should be taken regularly at the same time each day.
If you miss your dose at the usual time, take it as soon as you remember, as long as it is 12 hours before the next dose is due.
If it is less than 12 hours to the next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you miss more than one dose, or are not sure what to do, check with your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist or doctor for some hints.
If you take too much ARIANNA 1
If you think that you or anyone else has taken too much ARIANNA 1, urgent medical attention may be needed.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking ARIANNA 1?
Things you should do
- Be sure to keep all your appointments with your doctor so your progress can be checked.
- If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are taking ARIANNA 1.
- If you go into hospital, please let the medical staff know that you are taking ARIANNA 1.
- If you become pregnant while taking ARIANNA 1, tell your doctor immediately.
Your doctor needs to know immediately so that ARIANNA 1 can be replaced by another medicine.
Remind any doctor, dentist or pharmacist you visit that you are taking ARIANNA 1.
Things you should not do
- Do not give ARIANNA 1 to anyone else, even if they have the same condition as you.
- Do not take ARIANNA 1 to treat any other complaints unless your doctor tells you to.
- Do not stop taking ARIANNA 1 unless you have discussed it with your doctor.
- Do not take ARIANNA 1 if the packaging is torn or shows signs of tampering.
- Do not take ARIANNA 1 for a longer time than your doctor has prescribed.
- Do not let yourself run out of ARIANNA 1 over the weekend or on holidays.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ARIANNA 1 affects you.
ARIANNA 1 may occasionally cause drowsiness, dizziness or other symptoms such as feeling weak or sleepy which could affect your ability to drive or operate machinery.
Looking after your medicine
- Keep your tablets in the original pack until it is time to take them.
If you take tablets out of the blister pack they will not keep well. - Keep it where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to take this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not take this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. Side effects may happen at the start of treatment or they may happen after you have been taking your medicine for some time. Some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
ARIANNA 1 helps most postmenopausal women with breast cancer, but it may have unwanted side effects in a some people.
If you get any side effects do not stop taking ARIANNA 1 without first talking to your doctor or pharmacist.
Do not be alarmed by this list of possible side effects.
You may not experience any of them.
You may experience the following more common side effects of ARIANNA 1:
Less serious side effects
| Very common (may affect more than 1 in 10 people) | What to do |
| Speak to your doctor if you have any of these very common side effects and they worry you. |
| Common (may affect up to 1 in 10 people) | What to do |
| Speak to your doctor if you have any of these common side effects and they worry you. |
These side effects are mostly mild to moderate in nature.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Serious side effects are uncommon or rare.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
ARIANNA 1 may be associated with changes in your blood, urine or liver. Your doctor may want to perform tests from time to time to check on your progress and detect any unwanted side effects.
Uncommon side effects can include trigger finger which is a condition in which one of your fingers or your thumb catches in a bent position.
Cases of inflammation of a tendon or tendonitis (connective tissues that connect muscles to bone) have been observed during the use of similar class of medicines, which might also occur during treatment with ARIANNA 1.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ARIANNA 1 contains
| Active ingredient (main ingredient) | Anastrozole 1 mg |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Lactose |
Do not take this medicine if you are allergic to any of these ingredients.
What ARIANNA 1 looks like
ARIANNA 1 are white to off white, round, biconvex, film-coated tablets debossed with "AHI" on one side and plain on other side (AUST R 259991).
ARIANNA 1 comes in blister packs containing 30 tablets.
Who distributes ARIANNA 1
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in September 2025.
ARIANNA® is a Viatris company trade mark
ARIANNA 1_cmi\Sep25/00
Brand Information
| Brand name | Arianna 1 |
| Active ingredient | Anastrozole |
| Schedule | S4 |
MIMS Revision Date: 01 September 2024
1 Name of Medicine
Anastrozole.
2 Qualitative and Quantitative Composition
Each tablet contains 1 mg of anastrozole as the active ingredient.
Excipients with known effect. Lactose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Arianna 1 tablets are white to off white, round, biconvex, film-coated tablets debossed with "AHI" on one side and plain on other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Early breast cancer. Adjuvant treatment of early breast cancer in postmenopausal women with estrogen/ progesterone-receptor-positive disease.
Advanced breast cancer. First line treatment of advanced breast cancer in postmenopausal women with estrogen/ progesterone-receptor-positive disease.
Treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy. Patients with estrogen-receptor-negative disease and patients who have not responded to previous tamoxifen therapy rarely respond to anastrozole.
4.2 Dose and Method of Administration
Adults including the elderly. One tablet (1 mg) to be taken orally once a day.
For early breast cancer, the recommended total duration of hormonal therapy is 5 years. For patients being switched to anastrozole from tamoxifen, the switch should occur after completion of 2 to 3 years of tamoxifen therapy. There are no data to support switching at earlier or later time points.
Children. Not recommended for use in children.
Use in adults with renal impairment. No dose change is recommended.
Use in adults with hepatic impairment. No dose change is recommended.
4.3 Contraindications
Anastrozole must not be administered during pregnancy or lactation.
Known hypersensitivity to the active ingredient or to any of the excipients of this product.
4.4 Special Warnings and Precautions for Use
Use in renal and hepatic impairment. The apparent oral clearance of anastrozole in volunteers with stable hepatic cirrhosis or renal impairment (creatinine clearance less than 30 mL/min/1.73 m2) was in the range observed in healthy volunteers. Dosage adjustment is therefore not necessary. Anastrozole have not been investigated in patients with severe hepatic or severe renal impairment. The potential risk/benefit to such patients should be carefully considered before administration of anastrozole.
Bone mineral density. As anastrozole lowers circulating estrogen levels it may cause a reduction in bone mineral density with a possible consequent increased risk of fracture. Women with osteoporosis or at risk of osteoporosis should have their bone mineral density formally assessed by bone densitometry at the commencement of treatment and at regular intervals thereafter. Treatment or prophylaxis for osteoporosis should be initiated and monitored as appropriate.
In the phase III/IV SABRE study, 234 postmenopausal women with hormone receptor positive early breast cancer scheduled for treatment with anastrozole were stratified to low, moderate and high-risk groups according to their existing risk of fragility fracture. All patients received treatment with vitamin D and calcium. Patients in the low risk group received anastrozole alone, those in the moderate group were randomised to anastrozole plus bisphosphonate or anastrozole plus placebo and those in the high risk group received anastrozole plus bisphosphonate.
The 12-month main analysis has shown that patients already at moderate to high risk of fragility fracture had their bone health (assessed by bone mineral density and bone formation and resorption markers) successfully managed by using anastrozole in combination with a bisphosphonate. These findings were mirrored in the secondary efficacy variable of change from baseline in total hip BMD at 12 months.
Tendon disorders. The use of third generation aromatase inhibitors, including anastrozole, was found to be associated with tendonitis and tenosynovitis in randomised controlled trials. Tendon rupture was found to be a potential risk. Tendonitis and tenosynovitis were estimated to be of uncommon occurrence, and tendon rupture of rare occurrence. Monitor patients for signs and symptoms of tendon disorders during treatment with anastrozole.
Combination with LHRH agonists. There are no data available for the use of anastrozole with LHRH agonists. This combination should not be used outside clinical trials.
Use in the elderly. Anastrozole pharmacokinetics have been investigated in postmenopausal female volunteers and patients with breast cancer. No age related effects were seen over the range < 50 to > 80 years.
Paediatric use. Anastrozole is not recommended for use in children as safety and efficacy have not been established in this group of patients.
Use in pre-menopausal women. Anastrozole is not recommended for use in pre-menopausal women as safety and efficacy have not been established in this group of patients.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Anastrozole inhibited reactions catalysed by cytochrome P450 1A2, 2C8/9, and 3A4 in vitro with Ki values which were approximately 30 times higher than the mean steady-state Cmax values observed following a 1 mg daily dose. Anastrozole had no inhibitory effect on reactions catalysed by cytochrome P450 2A6 or 2D6 in vitro. Based on these in vitro and the in vivo results with antipyrine and cimetidine, it is unlikely that co-administration of anastrozole 1 mg with other drugs will result in clinically significant inhibition of cytochrome P450-mediated metabolism.
Other medicines that affect anastrozole. Demonstrated interactions. On the basis of clinical and pharmacokinetic data from the ATAC trial, tamoxifen must not be administered with anastrozole. Co-administration of anastrozole and tamoxifen resulted in a reduction of anastrozole plasma levels by 27% compared with those achieved with anastrozole alone.
Theoretical interactions. Estrogen-containing therapies should not be co-administered with anastrozole as they would negate its pharmacological action.
Potential interactions that have been excluded. A review of the clinical trial safety database did not reveal evidence of any clinically significant interaction in patients treated with anastrozole who also received commonly prescribed drugs. There were no clinically significant interactions with bisphosphonates (see Section 4.4 Special Warnings and Precautions for Use, Bone mineral density).
Effects of anastrozole on other medicines. Potential interactions that have been excluded. Antipyrine. Administration of a single 30 mg/kg or multiple 10 mg/kg doses of anastrozole to subjects had no effect on the clearance of antipyrine or urinary recovery of antipyrine metabolites.
Cimetidine. Pre-treatment with cimetidine, at a dose of 300 mg every six hours for four days, in normal postmenopausal women had no effect on the single dose pharmacokinetics of anastrozole (10 mg).
Warfarin. An interaction study with warfarin showed no clinically significant effect of anastrozole on warfarin pharmacokinetics or anticoagulant activity.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. In female rats treated orally with anastrozole for 14 days prior to mating up to day 7 of gestation, the fertility index (pregnancies/ matings) was reduced after oral doses of 1 mg/kg and above [9 times the maximum recommended clinical dose, based on body surface area (BSA)]. Pre-implantation loss was increased, and the number of implantations decreased, at doses of 0.02 mg/kg and above (0.2 times the maximum recommended clinical dose, based on BSA). It is not known whether anastrozole impairs fertility in humans.
Use in pregnancy. (Category C)
Anastrozole is contraindicated in pregnant women.
After oral administration of anastrozole to pregnant rats and rabbits, the drug was shown to cross the placenta and was detectable in fetal tissues at concentrations approximately 40% of corresponding maternal plasma drug concentrations. Anastrozole showed no evidence for teratogenic activity and had no effects on pregnancy parameters at oral doses of up to 1 mg/kg/day in rats and up to 0.2 mg/kg/day in rabbits (9 and 3 times the maximum recommended clinical dose, based on BSA, respectively). However, enlargement of the placenta was seen in rats and treatment of rabbits with anastrozole at doses greater than 0.2 mg/kg/day caused abortion in 100% of animals. These effects are consistent with disruption of estrogen dependent events during pregnancy and are not unexpected with a drug of this class.
In a peri-postnatal study (administration from day 17 of gestation to day 21 postpartum) in rats, increased resorption was observed at 0.5 mg/kg/day. Increased stillbirths and evidence for dystocia (increased variability in the length of gestation and/or vaginal bleeding at birth) were reported at doses of 0.1 mg/kg/day or greater. Pup survival was reduced at all doses tested (0.02 mg/kg/day and above, 0.2 times the maximum recommended clinical dose, based on BSA). There was no evidence of adverse effects on behaviour or reproductive performance of the first generation offspring attributable to maternal treatment with anastrozole.
Use in lactation. Anastrozole is contraindicated in breastfeeding women.
4.7 Effects on Ability to Drive and Use Machines
Anastrozole is unlikely to impair the ability of patients to drive and operate machinery. However, asthenia and somnolence have been reported with the use of anastrozole and caution should be observed when driving or operating machinery while such symptoms persist.
4.8 Adverse Effects (Undesirable Effects)
Anastrozole has generally been well tolerated. Adverse events have usually been mild to moderate with only few withdrawals from treatment due to undesirable events. Unless specified, the following frequency categories were calculated from the number of adverse events reported in a large phase III study conducted in 9366 postmenopausal women with operable breast cancer treated for 5 years and unless specified, no account was taken of the frequency within the comparative treatment group or whether the investigator considered it to be related to study medication. See Table 1.
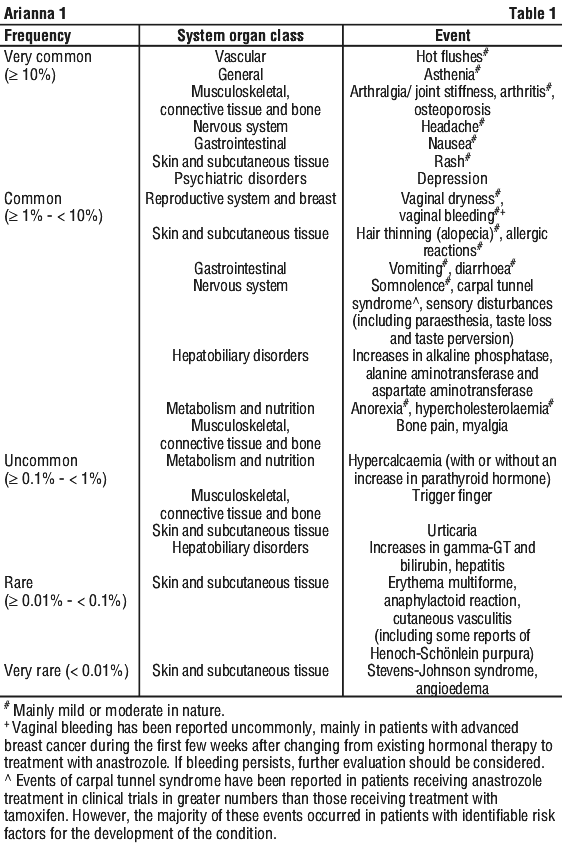
In studies in the adjuvant setting, anastrozole have been associated with an increased incidence of fractures compared to tamoxifen treatment during the active treatment phase. At the 100 month analysis of a large phase III study the off treatment fracture episode rate was no different between the anastrozole and tamoxifen treatment arms (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Post-marketing experience. Musculoskeletal and connective tissue disorders: tendonitis, tenosynovitis and tendon rupture (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is limited clinical experience of overdose of anastrozole. There are no reports where a patient has taken a dose exceeding 60 mg. No toxicity was observed and no clinically relevant adverse effects have been seen.
There is no clinical experience of accidental overdosage. In animal studies, anastrozole demonstrated low acute toxicity. Clinical trials have been conducted with various dosages of anastrozole, up to 60 mg in a single dose given to healthy male volunteers and up to 10 mg daily given to postmenopausal women with advanced breast cancer; these dosages were well tolerated. A single dose of anastrozole that results in life-threatening symptoms has not been established.
There is no specific antidote to overdosage and treatment must be symptomatic. In the management of an overdose, consideration should be given to the possibility that multiple agents may have been taken. Vomiting may be induced if the patient is alert.
Dialysis may be helpful because anastrozole are not highly protein bound. General supportive care, including frequent monitoring of vital signs and close observation of the patient, is indicated.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Anastrozole is a potent and highly selective non-steroidal aromatase inhibitor. It significantly lowers serum estradiol concentrations and has no detectable effect on formation of adrenal corticosteroids or aldosterone.
Many breast cancers have estrogen receptors and growth of these tumours can be stimulated by estrogen. In postmenopausal women, estradiol is produced primarily from the conversion of androstenedione to estrone through the aromatase enzyme complex in peripheral tissues. Estrone is subsequently converted to estradiol. Many breast cancers also contain aromatase; the importance of tumour-generated estrogens is uncertain.
Reducing circulating estradiol levels has been shown to produce a beneficial effect in women with breast cancer. In postmenopausal women, anastrozole at a daily dose of 1 mg produced estradiol suppression of greater than 80% using a highly sensitive assay.
Anastrozole does not possess any progestogenic, androgenic or estrogenic activity.
Daily doses of anastrozole up to 10 mg do not have any effect on cortisol or aldosterone secretion, measured before or after standard ACTH challenge testing. Corticoid supplements are therefore not needed.
In a phase III/IV study there was a neutral effect on plasma lipids in those patients treated with anastrozole.
Clinical trials. Switching in treatment of early breast cancer. A prospectively planned, combined analysis of 2 multicentre, open-label, randomised controlled trials (ABCSG trial 8 and ARNO 95) was conducted to examine the efficacy of switching postmenopausal patients with hormone-receptor positive early breast cancer receiving tamoxifen (20 or 30 mg daily), to anastrozole (1 mg daily). A total of 3224 patients who had completed 2 years adjuvant treatment with tamoxifen and had remained disease-free, were randomised to receive anastrozole for 3 years (n = 1618) or to continue on tamoxifen for 3 years (20 to 30 mg daily; n = 1606). The total duration of hormonal therapy was 5 years. Patients did not receive adjuvant chemotherapy. 74% of patients had lymph-node negative disease at commencement of hormonal therapy.
The primary endpoint was event-free survival, with an event being defined as loco-regional or distant recurrence or the development of contralateral breast cancer. Overall survival was a secondary end-point. Median follow-up after randomisation was 28 months, and 55% of patients in each group had completed the planned 5 years of hormonal therapy. Results are presented in Table 2.

Another open-label, randomised controlled trial (the ITA study) enrolled 448 postmenopausal patients with estrogen-receptor positive early breast cancer. All patients had lymph node involvement. Patients who remained disease-free after receiving 2 to 3 years of tamoxifen therapy were randomly assigned to receive anastrozole (1 mg daily; n = 233) or to continue therapy with tamoxifen (20 mg daily, n = 225) for a total of 5 years hormonal therapy in each arm. 67% of patients in each arm received adjuvant chemotherapy.
The primary endpoint was disease recurrence, with a recurrence being defined as loco-regional or distant recurrence. Event-free survival was a secondary end-point with an event being defined as loco-regional or distant recurrence, the development of contralateral breast cancer, the development of a second primary cancer, or death occurring without disease recurrence. Overall survival was also a secondary end-point. Median follow-up after randomisation was 36 months. Results are presented in Table 3.

Adjuvant treatment of early breast cancer in postmenopausal women. In a multicentre, double-blind trial (ATAC; trial 0029) 9,366 postmenopausal women aged 33 to 95 years old with early breast cancer were randomised to receive adjuvant treatment with anastrozole 1 mg daily, tamoxifen 20 mg daily, or a combination of the two treatments for five years or until recurrence of disease.
The primary endpoint was disease-free survival (i.e. time to occurrence of a distant or local recurrence, new contralateral breast cancer or death from any cause). Secondary and additional prospectively defined endpoints included time to distant recurrence, the incidence of contralateral breast cancer, overall survival, time to recurrence and time to death following recurrence. See Figures 3-4.
Demographic and other baseline characteristics were similar among the two treatment arms, with approximately 84% of patients with hormone receptor positive disease. The median follow-up was 100 months.
Treatment with anastrozole was superior to tamoxifen in the intention-to-treat (ITT) group, with statistically significant risk reductions in disease-free survival and time to recurrence of 10% and 19% respectively (see Table 4). In the clinically relevant hormone-receptor-positive sub-group, statistically significant benefits of anastrozole compared to tamoxifen were also observed for disease-free survival and time to recurrence, with risk reductions of 15 and 24% respectively (see Figures 1-2 and Table 4). The absolute benefit in recurrence rates of anastrozole over tamoxifen increased over the entire 100 month follow up period. The gain in absolute benefit during the off-treatment period demonstrates a superior carryover effect to that already demonstrated with 5 years of tamoxifen treatment (see Figure 2).
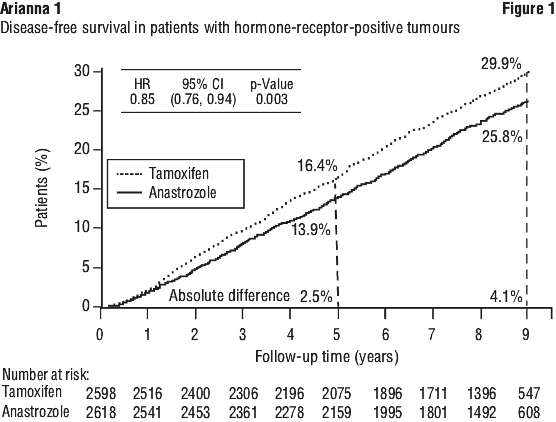
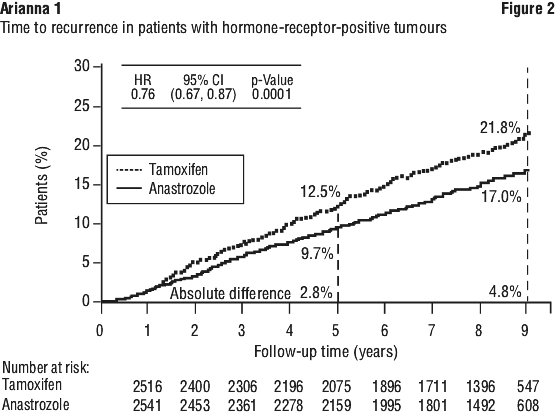
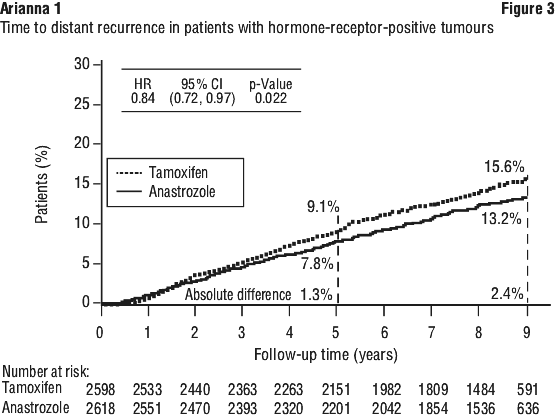
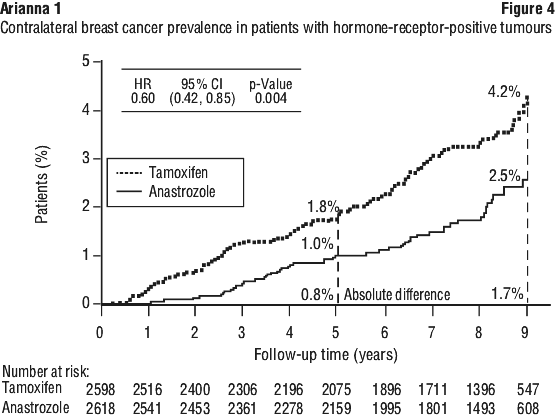
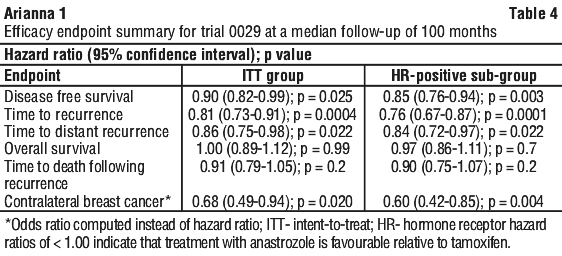
Other secondary and additional outcome variables were all either significantly in favour of anastrozole or with trends evident in favour of anastrozole when compared to tamoxifen (see Table 4).
Overall, anastrozole was well tolerated. Withdrawals due to medicine-related adverse events were less common with anastrozole compared to tamoxifen (6.5% vs 8.9%, odds ratio 0.71, 95% CI 0.59-0.86, p = 0.0004). The following adverse events were reported regardless of causality. Patients receiving anastrozole had a significant decrease in hot flushes, vaginal bleeding, vaginal discharge, endometrial cancer, venous thromboembolic events and ischaemic cerebrovascular events compared to patients receiving tamoxifen. Patients receiving anastrozole had an increase in joint disorders (including arthritis, arthrosis and arthralgia) and total number of fractures compared with patients receiving tamoxifen. Although the incidence of fractures (both serious and non-serious, occurring either during or after treatment) were higher in the anastrozole compared to the tamoxifen treatment group, the incidences of hip fractures were similar between the groups. The fracture rate for anastrozole whilst on treatment falls within the broad range of fracture rates reported in an age-matched postmenopausal population.
A plot of the annual first fracture rates, throughout the study, shows that following the end of treatment the annual first event rates were similar in the anastrozole and tamoxifen treatment groups and the increased first fracture rate seen during treatment was not continued in the post-treatment follow-up period (see Figure 5).
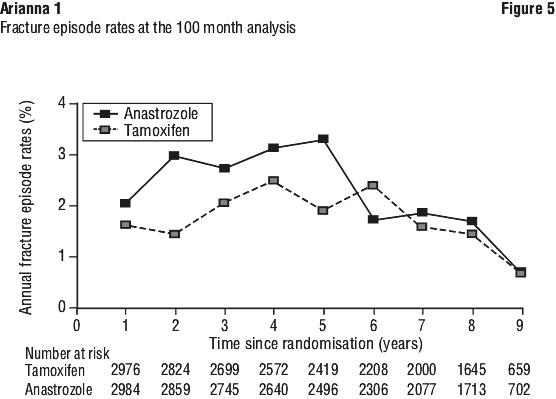
Ischaemic cardiovascular events (consisting mainly of angina pectoris) in the on-treatment period were reported more frequently in patients treated with anastrozole compared to those treated with tamoxifen (mainly associated with patients with pre-existing ischaemic heart disease), although the difference was not statistically significant (p = 0.1224).
At a median follow-up of 33 months, the combination arm did not demonstrate any efficacy benefit when compared with tamoxifen in either the ITT group or the hormone-receptor-positive sub-group. This treatment arm was discontinued from the trial.
First line therapy in postmenopausal women with advanced breast cancer. In two similar controlled trials (Trials 0027 and 0030), 1021 postmenopausal women aged 30 to 92 years old, with advanced breast cancer [stage IV (metastatic disease) and stage III (locally advanced disease)] were randomised to receive anastrozole 1 mg (n = 511) or tamoxifen 20 mg (n = 510) once daily as first line therapy.
The primary end points for both trials were time to progression, objective response rate and safety. The trials were designed to allow data to be pooled. The median duration of follow-up was 18.8 and 17.7 months in Trial 0027 and Trial 0030 respectively. The number of patients still on trial treatment at the end of the follow-up period was as follows (see Table 5):
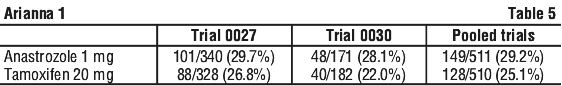
Anastrozole was at least as effective as tamoxifen for the primary endpoints of time to progression and objective-response rate. A comparison of the results for the primary endpoints, for both trials, is provided in Table 6. Positive estrogen/ progesterone receptor status had an impact on the primary efficacy parameters and this may partly explain the difference in results between the two trials.
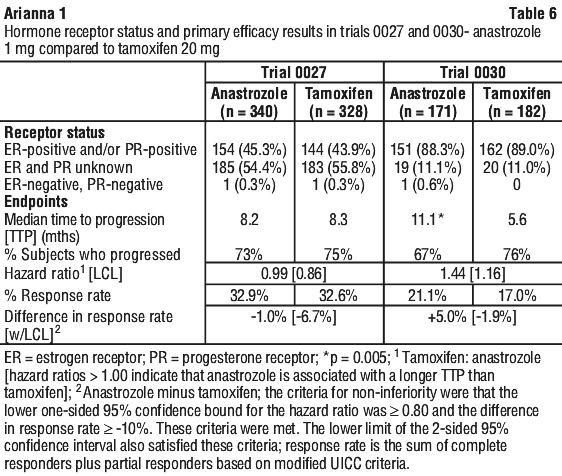
At a median follow-up of approximately 30 months and with approximately 60% of patients having died, the data from both studies combined demonstrated significant prolongation of survival with anastrozole 1 mg compared to megestrol acetate. The median time to death for anastrozole 1 mg was 26.7 months compared to 22.5 months for megestrol acetate, with a 2 year survival rate for anastrozole 1 mg of 56.1% compared to 46.3% for megestrol acetate. The hazard ratio of risk of death of patients on anastrozole 1 mg compared to megestrol acetate was 0.78, and there was a statistically significant difference in time to death (p < 0.025).
An open label, balanced, randomized, two-treatment, two period, two-sequence, single dose, two-way crossover, comparative oral bioavailability study of two formulations of anastrozole tablets 1 mg was conducted in 26 healthy, postmenopausal human female subjects under fasting conditions. The study compared anastrozole tablets 1 mg with reference product Arimidex 1 mg tablets.
Statistical comparisons of geometric means for Test vs reference for anastrozole Tmax, Cmax and AUC0-∞ were as follows (see Table 7):
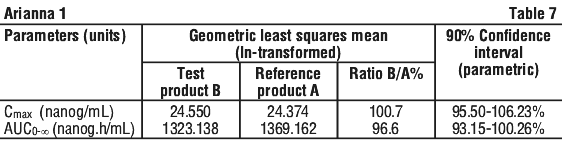
This comparison of test product with reference product anastrozole met the predefined criteria for bioequivalence, as the calculated 90% CI for all ratios of pre-specified In-Transformed PK parameters fell within the range 80.00%-125.00%.
5.2 Pharmacokinetic Properties
Absorption. Absorption of anastrozole is rapid and maximum plasma concentrations typically occur within two hours of dosing (under fasted conditions). Food slightly decreases the rate but not the extent of absorption. The small change in the rate of absorption is not expected to result in a clinically significant effect on steady-state plasma concentrations during once daily dosing of anastrozole.
Distribution. Anastrozole is 40% bound to plasma proteins. The pharmacokinetics of anastrozole are linear over the dose range of 1 mg to 20 mg and do not change with repeated dosing.
Approximately 90 to 95% of plasma anastrozole steady-state concentrations are attained after 7 daily doses. There is no evidence of time or dose-dependency of anastrozole pharmacokinetic parameters.
Anastrozole pharmacokinetics are independent of age in postmenopausal women.
Metabolism. Anastrozole is extensively metabolised by postmenopausal women with less than 10% of the dose excreted in the urine unchanged within 72 hours of dosing. Metabolism of anastrozole occurs by N-dealkylation, hydroxylation and glucuronidation. The metabolites are excreted primarily via the urine. Triazole, a major metabolite in plasma and urine, does not inhibit aromatase.
Excretion. Anastrozole is eliminated slowly with a plasma elimination half-life of 40 to 50 hours.
Paediatric pharmacokinetics. In boys with pubertal gynecomastia, anastrozole was rapidly absorbed, was widely distributed, and was eliminated slowly with a half-life of approximately 2 days. Pharmacokinetic parameters in boys were comparable to those of postmenopausal women. Clearance of anastrozole was lower in girls than in boys and exposure higher. Anastrozole in girls was widely distributed and slowly eliminated, with an estimated half-life of approximately 0.8 days.
5.3 Preclinical Safety Data
Genotoxicity. Anastrozole did not show evidence of genotoxicity in assays for gene mutations in vitro and chromosomal damage in vitro and in vivo.
Carcinogenicity. In a two year rat oncogenicity study, anastrozole caused an increase in incidence of hepatic adenomas and carcinomas and uterine stromal polyps in females and thyroid adenomas in males at the high dose (25 mg/kg/day), where exposure (AUC) was approximately 100-fold that which occurs at the maximum recommended clinical dose. At the no tumourigenic effect level (5 mg/kg/day), exposure (AUC) was approximately 20-fold that which occurs at the maximum recommended clinical dose.
In a two year mouse oncogenicity study, anastrozole induced benign ovarian tumours and a disturbance in the incidence of lymphoreticular neoplasms (fewer histiocytic sarcomas in females and more deaths as a result of lymphomas). The benign tumourigenic effect on the ovary occurred at all doses including the lowest dose tested (5 mg/kg/day) [exposure (AUC) was approximately 1 to 2-fold that which occurs at the maximum recommended clinical dose]. The clinical relevance of these findings in the mouse are not clear.
Preclinical chronic toxicity. Multiple dose toxicity studies utilized rats and dogs. No no-effect levels were established for anastrozole in the toxicity studies, but those effects that were observed at the low doses (1 mg/kg/day) and mid doses (dog 3 mg/kg/day; rat 5 mg/kg/day) were related to either the pharmacological or enzyme inducing properties of anastrozole and were unaccompanied by significant toxic or degenerative changes. Plasma levels of anastrozole at these doses in rats and dogs were at least 3 and 12 times greater, respectively, than those expected in human postmenopausal women during treatment with anastrozole. At higher doses of anastrozole, nephropathy was observed in rats, ECG changes were observed in dogs, and changes in cholesterol levels were observed in both animal species.
6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose monohydrate, povidone, sodium starch glycollate type A, magnesium stearate, hypromellose, macrogol 300 and titanium dioxide.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
PVC/PVDC blister/aluminium foil blister containing 30 tablets.
Australian register of therapeutic goods (ARTG). AUST R 259991 - Arianna 1 anastrozole 1 mg film-coated tablet blister pack.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Chemical name: 2,2'-[5-(1H-1,2,4-triazol-1-ylmethyl)- 1,3-phenylene]bis(2-methylpropiononitrile).
Structural formula:
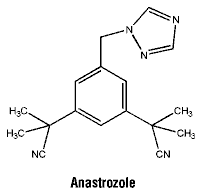
Molecular weight: 293.4.
Anastrozole is a fine white to off-white powder. Anastrozole has moderate aqueous solubility (0.53 mg/mL at 25°C) which is dependent on pH from pH 1 to 4 but independent of pH thereafter.
Solubility of anastrozole is independent of pH in the physiological range. Anastrozole is freely soluble in methanol, acetone, ethanol and tetrahydrofuran and very soluble in acetonitrile. It is also very soluble in chloroform and dimethylformamide, freely soluble in benzene and ethyl acetate and soluble in isopropyl alcohol and toluene.
CAS number. 120511-73-1.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
04 December 2015
Date of Revision
05 August 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.