Artige
Brand Information
| Brand name | Artige |
| Active ingredient | Methylphenidate hydrochloride |
| Schedule | S8 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Artige.
Summary CMI
Artige®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I taking Artige?
Artige contains the active ingredient methylphenidate hydrochloride. Artige is used to treat attention deficit hyperactivity disorder (ADHD). It is also used to treat narcolepsy, a sleep-attack disorder. For more information, see Section 1. Why am I taking Artige? in the full CMI.
2. What should I know before I take Artige?
Do not use if you have ever had an allergic reaction to Artige or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take Artige? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Artige and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take Artige?
- ADHD (children only): Take 5mg once or twice daily at breakfast or lunch as per your doctor's instruction.
- Narcolepsy (adults only): Take 20 to 30 mg a day, depending on your doctor's instructions.
- Swallow whole or half tablet orally with water.
More instructions can be found in Section 4. How do I take Artige? in the full CMI.
5. What should I know while taking Artige?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking Artige? in the full CMI.
6. Are there any side effects?
Common side effects include headache, drowsiness, dizziness, sleepiness, shaking of the body, vomiting, mood changes, restlessness.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
WARNING: DRUG DEPENDENCE: Artige should be given cautiously to patients with a history of drug dependence or alcoholism. Chronic abusive use can lead to marked tolerance and psychological dependence with varying degrees of abnormal behaviour. Frank psychotic episodes can occur, especially with parenteral abuse. Careful supervision is required during withdrawal from abusive use since severe depression may occur. Withdrawal following chronic therapeutic use may unmask symptoms of the underlying disorder that may require follow up.
Artige®
Active ingredient: methylphenidate hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using Artige. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Artige.
Where to find information in this leaflet:
1. Why am I taking Artige?
2. What should I know before I take Artige?
3. What if I am taking other medicines?
4. How do I take Artige?
5. What should I know while taking Artige?
6. Are there any side effects?
7. Product details
1. Why am I taking Artige?
Artige contains the active ingredient methylphenidate hydrochloride. Artige is a class of medicine that increases the activity of the brain.
Artige is used to treat Attention Deficit Hyperactivity Disorder (ADHD). It is also used to treat narcolepsy, a sleep-attack disorder.
2. What should I know before I take Artige?
Warnings
Do not take Artige if:
- you are allergic to methylphenidate hydrochloride, or any of the ingredients listed at the end of this leaflet.
- you have anxiety, tension or agitation
- you have Tourette's Syndrome (uncontrolled speech, body movements or tics) or a family history of Tourette's
- you have Glaucoma (increased pressure in the eye)
- you have an overactive thyroid or other thyroid problems
- you have heart problems - heart attack, irregular heartbeat, chest pain (angina), heart failure, heart disease or if you were born with a heart problem
- you have very high blood pressure (hypertension) or narrowing of the blood vessels (arterial occlusive disease, which can cause pain in the arms and legs)
- you have severe depression, eating disorder (anorexia) or other mental illness
- you have a tumour in the adrenal gland which can cause high blood pressure and headaches.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions such as depression, suicidal thoughts or behaviour, seeing or feeling things that are not there (hallucinations), unusual active or depressed mood (bipolar illness), aggressive behaviour
- have abnormal thoughts or are over-excited, over-active or un-inhibited
- have motor or verbal tics, uncontrolled speech or movements, repeated twitching or make repeated sounds or words, or if a family member has any of these
- have had fits/convulsions
- experience all of these problems: restlessness, tremor, sudden muscle contractions, abnormal high temperature, nausea, vomiting
- have high blood pressure (headaches, blurred vision, chest pain)
- have any other medical conditions such as current or previous heart problems or serious heart rhythm problems.
- have any other medical conditions such as weakening of the blood vessels in the brain, inflammation of the blood vessels, have had a stroke
- have a history of drug or alcohol abuse
- have eye problems, including increased pressure in your eye, glaucoma or trouble seeing close-up
- take any medicines for any other condition
- have painful or long-lasting erections
- have serious heart problems. You should be examined by a Heart Specialist before you take Artige.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Artige should not be taken in the first 3 months of pregnancy.
Do not breast feed if you are taking Artige.
Children under 6 years
- Do not use Artige in children under 6 years of age.
Children under 18 years of age
- Your doctor needs to take a detailed history and evaluation before you take Artige.
- Children may have slower growth if taking Artige for a long time.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Do not take Artige if:
- you are taking monoamine oxidase inhibitors (MAOI) or have taken this medicine in the last 2 weeks.
Tell your doctor or pharmacist if you are taking any of the following medicines:
- medicines that increase blood pressure;
- tricyclic antidepressants (treats depression);
- alpha 2 agonists (treat high blood pressure);
- oral anticoagulants (prevent blood clots);
- anticonvulsants (treat fits/seizures);
- medicines that treat mental illnesses (antipsychotics)
- phenylbutazone (treats pain or fever);
- that influence dopamine levels e.g. disease which affects movement (Parkinson's disease);
- that change the level of serotonin in the body such as sertraline or venlafaxine that are used to treat depression.
Tell your doctor or pharmacist if you are having an operation. Artige cannot be taken with anesthetics due to an increased risk of blood pressure.
Artige may give false positives in drug testing.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Artige.
4. How do I take Artige?
How much to take
- ADHD (children only): Start with 5 mg once or twice daily with gradual increases of 5 mg or 10 mg weekly, depending on your doctor's instructions.
- The highest recommended dose of Artige for children is 60 mg per day.
- Narcolepsy (adults only): the average dose is 20 to 30 mg a day, depending on your doctor's instructions.
- The highest recommended dose of Artige for adults is 60 mg per day.
- Follow the instructions provided and use Artige until your doctor tells you to stop.
When to take Artige
- Artige should be taken once or twice a day (for example at breakfast or lunch) as per your doctor's instructions.
- Doses should be taken 1 to 2 hours before the maximum effect is needed.
- Swallow tablet with water.
- Artige should be taken before 6pm as it can cause trouble sleeping if the dose is taken late.
- In some cases, sleeplessness occurs as the medicine wears off. Talk to your doctor about doses in the evenings.
If you forget to take Artige
Artige should be used regularly at the same time each day. If you miss your dose at the usual time, take it as soon as possible.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
- Do not take more medicine than your doctor tells you to; do not take it more often and do not use it for longer than your doctor tells you - this medicine can be addictive.
If you use too much Artige
If you think that you have used too much Artige, you may need urgent medical attention. You could experience vomiting, feeling uneasy or tense (agitation), headache, shaking (tremor), muscle twitching, irregular heartbeat, flushing, fever, sweating, dilated pupils, breathing problems, confusion, fits, muscle spasms, or red-brown urine (muscle breakdown).
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking Artige?
Things you should do
Take your medicine as directed by your doctor.
Make sure you keep any doctor's or specialist appointments as your doctor needs to assess your treatment periodically.
Call your doctor straight away if you:
- Have any chest pain, fainting or other possible heart problems.
- Have any abnormal thoughts, over-active/excited or uninhibited actions, aggressive behaviour, are seeing things.
- Have fits or tics
- Have eye problems
- Have slurred speech, confusion, loss of coordination, sudden numbness
- Have painful or long-lasting erections
Remind any doctor, dentist or pharmacist you visit that you are taking Artige.
Things you should not do
- Do not stop using this medicine suddenly.
- Do not change your dose without speaking to your doctor first.
- Keep all doctors and specialists' appointments.
Growth in children
- Some children taking Artige for a long time can have a slower than normal growth.
Medicines that raise serotonin
- For example sertraline, venlafaxine used to treat depression.
- If you feel restless, have tremor, abnormal high temperature, nausea, vomiting - stop using Artige.
Driving or using machines
Be careful before you drive or use any machines or tools or ride a bike until you know how Artige affects you.
Artige may cause dizziness, drowsiness, blurred vision, or hallucinations (seeing or hearing things that aren't there) in some people.
Drinking alcohol
Do not drink alcohol while taking Artige.
Alcohol can make the side effects worse. Check to make sure food and other medicines taken do not contain alcohol.
Sudden withdrawal
Do not suddenly stop taking Artige.
Stopping Artige suddenly can cause withdrawal effects - patients can feel depressed or long lasting over-activity.
Looking after your medicine
- Store below 25°C.
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Problems of the brain, spinal cord and nerves:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Less serious side effects (continued)
| Less serious side effects | What to do |
Gut problems:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Problems of the brain, spinal cord and nerves:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Artige contains
| Active ingredient (main ingredient) | Methylphenidate hydrochloride |
| Other ingredients (inactive ingredients) | lactose, calcium phosphate, gelatin, magnesium stearate, purified talc and wheat starch |
| Potential allergens | Lactose, gluten, animal product |
Do not take this medicine if you are allergic to any of these ingredients.
What Artige looks like
Artige tablets are immediate release white tablets that are scored and marked AB and CG on the reverse; packs of 100 tablets.
Australian Registration Number:
Artige tablet: AUST R 117306
Who distributes Artige
NOVARTIS Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1 800 671 203
Web site: www.novartis.com.au
® = Registered Trademark
This leaflet was prepared in November 2025.
Internal document code:
(art300124c_v3 based on PI art300124i)
Brand Information
| Brand name | Artige |
| Active ingredient | Methylphenidate hydrochloride |
| Schedule | S8 |
MIMS Revision Date: 01 March 2024
1 Name of Medicine
Methylphenidate hydrochloride.
2 Qualitative and Quantitative Composition
Artige contain methylphenidate hydrochloride 10 mg.
Artige contains lactose, sugars, milk products, lactitol, gluten and sulfur dioxide.
For the full list of the excipients, see Section 6.1.
3 Pharmaceutical Form
Artige tablets are immediate release white tablets that are scored and marked AB and CG on the reverse.
4 Clinical Particulars
4.1 Therapeutic Indications
Attention-deficit hyperactivity disorder (ADHD). ADHD was previously known as attention-deficit disorder. Other terms used to describe this behavioural syndrome include: minimal brain dysfunction in children, hyperkinetic child syndrome, minimal brain damage, minimal cerebral dysfunction, minor cerebral dysfunction and psycho-organic syndrome of children.
Artige are indicated as an integral part of a total treatment program for ADHD that may include other measures (psychological, educational and social) for patients with this syndrome. Stimulants are not intended for use in the patient who exhibits symptoms secondary to environmental factors and/or other primary psychiatric disorders, including psychosis.
Special diagnostic considerations for ADHD in children. The aetiology of this syndrome is unknown and there is no single diagnostic test. Adequate diagnosis requires the use, not only of medical, but also of psychological, educational and social resources. Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, moderate to severe hyperactivity, minor neurological signs and an abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics.
Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in children who exhibit symptoms secondary to environmental factors (e.g. child abuse in particular) or primary psychiatric disorders. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medicine will depend upon the physician's assessment of the chronicity and severity of the child's symptoms.
Continuation of treatment in adolescent and special diagnostic considerations for ADHD in adults. There is limited information to guide clinicians about how long older adolescents should continue to receive treatment with drugs for attention deficit hyperactivity disorder (ADHD). The decision should be based on the extent to which symptoms of ADHD and social functioning have improved to a point that medication is no longer needed. If older adolescents have been largely symptom-free for a year and are functioning well, a trial without medication is warranted. This should be undertaken at times of low stress such as during holidays or in a period when a school routine is well established.
ADHD needs to be considered in adults who present with longstanding symptoms suggestive of ADHD (inattention, impulsivity, disorganisation) that appear to have started in childhood and are persisting into adult life. Further, people with personality disorder and/or problems with drug use accompanied by a significant level of impulsivity and inattention should be referred for evaluation by a psychiatrist with the training and skills required to assess and treat ADHD. This expertise is necessary due to the overlap of ADHD symptoms with anxiety, mood and personality disorders.
Narcolepsy. The symptoms include daytime sleepiness, inappropriate sleep episodes and rapidly occurring loss of voluntary muscle tone. Artige is effective for symptoms of sleepiness but not for loss of voluntary muscle tone.
4.2 Dose and Method of Administration
Pre-treatment screening. Treatment should only be initiated by specialist physicians with experience in the use of the drug. Before initiating Artige treatment, patients should be assessed for pre-existing cardiovascular and psychiatric disorders and a family history of sudden death, ventricular arrhythmia and psychiatric disorders. Weight and height should also be measured before treatment and documented on a growth chart (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Dosage. The dosage of Artige should be individualised according to the patient's clinical needs and responses.
Treatment with Artige should be initiated at a low dose, with increments at weekly intervals.
ADHD. In the treatment of ADHD, an attempt should be made to time administration of the drug to coincide with periods of greatest academic, behavioural or social difficulties for the patient.
If symptoms do not improve after dose titration over a one-month period, the drug should be discontinued.
If symptoms worsen or other adverse effects occur, the dosage should be reduced or, if necessary, the drug discontinued.
If the effect of the drug wears off too early in the evening, disturbed behaviour and/or inability to go to sleep may recur.
A small evening dose of Artige 10 mg tablet may help solve this problem.
Periodic assessment of the treatment in ADHD. Drug treatment does not need to be indefinite. Physicians should periodically re-evaluate the treatment with trial periods off medication to assess the patient's functioning without pharmacotherapy. Improvement may be sustained when the drug is either temporarily or permanently discontinued. When used in children with ADHD, treatment can usually be discontinued during or after puberty.
If therapy is interrupted for reasons other than those stated above, it should not be restarted at the dose that had been reached prior to treatment interruption, but should be re-titrated.
Children and adolescents (6 years and over). Start with 5 mg once or twice daily (e.g. at breakfast and at lunch) with gradual increments of 5 or 10 mg weekly. The total daily dosage should be administered in divided doses.
In some children with ADHD, sleeplessness may occur as the effect of the drug wears off. On rare occasions, an additional dose at about 8.00 p.m. may help; a trial dose may help to clarify the issue in an individual case, if the symptom warrants treatment.
Narcolepsy. Adults. Administer the tablets in divided doses 2 or 3 times daily. The average dose is 20 to 30 mg daily. Some patients may require 40 mg to 60 mg daily. In others, 10 mg to 15 mg daily will be adequate. Patients who are unable to sleep if Artige tablets are taken late in the day should take the last dose before 6 p.m.
Dosing for each patient requires titration to control symptoms. Single doses greater than 20 mg are associated with sympathomimetic side effects. Therefore, the average single dose should be less than 20 mg. A maximum total dose of 60 mg/day may be required.
Maximum daily doses. A maximum daily dose of 60 mg should not be exceeded for the treatment of narcolepsy.
Administration. The rate of absorption and, therefore, onset of action is faster when Artige tablets are taken with food. Dosage should, therefore, be standardised in relation to food to ensure consistency of effect.
Doses should be administered 1 to 2 hours before the maximum effect is required.
4.3 Contraindications
Artige is contraindicated in patients with the following:
anxiety and tension states;
agitation;
a family history or diagnosis of Tourette's syndrome;
glaucoma;
hyperthyroidism;
pre-existing cardiovascular disorders including uncontrolled hypertension, angina pectoris, arterial occlusive disease especially coronary arteries; heart failure, haemodynamically significant congenital heart disease, cardiomyopathies, myocardial infarction, cardiac arrhythmia and channelopathies (disorders caused by the dysfunction of ion channels);
treatment with monoamine oxidase inhibitors, and also within a minimum of 14 days following discontinuation of a monoamine oxidase (MAO) inhibitor (hypertensive crises may result);
phaeochromocytoma;
known drug dependence or alcohol abuse;
severe depression, anorexia nervosa, psychotic symptoms or suicidal tendency, since Artige might worsen these conditions;
known hypersensitivity to methylphenidate or to any component of the formulation.
4.4 Special Warnings and Precautions for Use
General. Treatment with methylphenidate is not indicated in all cases of ADHD and should be considered only after detailed history taking and evaluation of the patient. The decision to prescribe methylphenidate should depend on the physician's assessment of the chronicity and severity of the symptoms and in paediatric patients, the appropriateness to the child's age. Prescription should not depend solely on the presence of isolated behavioural characteristics. When the symptoms are associated with acute stress reactions, treatment with methylphenidate is usually not indicated.
Sudden death and pre-existing structural cardiac abnormalities or other serious heart problems. It is essential that patients with pre-existing structural cardiac abnormalities or other serious heart problems being considered for treatment are assessed by a cardiologist before initiating treatment. Ongoing cardiological supervision should be maintained throughout treatment in these patients.
Children and adolescents. Sudden death has been reported in association with CNS stimulant treatment at usual doses in children and adolescents with structural cardiac abnormalities or other serious heart problems.
Stimulant products, including methylphenidate, generally should not be used in patients with known serious structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, or other serious cardiac problems that may increase the risk of sudden death due to the sympathomimetic effects of a stimulant drug. Before initiating methylphenidate treatment, patients should be assessed for pre-existing cardiovascular disorders and a family history of sudden death and ventricular arrhythmia (see Section 4.2 Dose and Method of Administration).
Adults. Sudden death, stroke, and myocardial infarction have been reported in adults taking stimulant drugs at usual doses for ADHD. Although the role of stimulants in these adult cases is also unknown, adults have a greater likelihood than children of having serious structural cardiac abnormalities, cardiomyopathy, serious heart rhythm abnormalities, coronary artery disease, or other serious cardiac problems. Adults with such abnormalities should also generally not be treated with stimulant drugs.
Cardiovascular conditions. Artige is contraindicated in patients with severe hypertension. Artige increases heart rate and systolic and diastolic blood pressure. Therefore, caution is indicated in treating patients whose underlying medical conditions might be compromised by increases in blood pressure or heart rate, e.g. those with pre-existing hypertension. Severe cardiovascular disorders are contraindicated (see Section 4.3 Contraindications).
Methylphenidate should be used cautiously in patients with hypertension. Blood pressure should be monitored at appropriate intervals in all patients taking methylphenidate, especially in those with hypertension. Patients who develop symptoms suggestive of cardiac disease during methylphenidate treatment should undergo a prompt cardiac evaluation.
Children, adolescents, or adults who are being considered for treatment with stimulant medicine should have a careful history (including assessment for a family history of sudden death or ventricular arrhythmia) and physical exam to assess for the presence of cardiac disease, and should receive further cardiac evaluation if findings suggest such disease. Patients who develop symptoms such as exertional chest pain, unexplained syncope, or other symptoms suggestive of cardiac disease during stimulant treatment should undergo a prompt cardiac evaluation.
Misuse and cardiovascular events. Misuse of stimulants of the central nervous system, including methylphenidate may be associated with sudden death and other serious cardiovascular adverse events.
Cerebrovascular. Cerebrovascular conditions. Patients with pre-existing central nervous system (CNS) abnormalities, e.g. cerebral aneurysm and/or other vascular abnormalities such as vasculitis or pre-existing stroke should not be treated with Artige. Patients with additional risk factors (history of cardiovascular disease, concomitant medicine that elevates blood pressure) should be assessed regularly for neurological/psychiatric signs and symptoms after initiating treatment with Artige (see Section 4.4 Special Warnings and Precautions for Use, Cardiovascular conditions; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Psychiatric conditions. Artige should not be used to treat severe depression or for the prevention or treatment of normal fatigue states. In psychotic patients administration of methylphenidate may exacerbate symptoms of behaviour disturbance and thought disorder.
Co-morbidity of psychiatric disorders in ADHD is common and should be taken into account when prescribing stimulant products. Prior to initiating treatment with methylphenidate, patients should be assessed for pre-existing psychiatric disorders and a family history of psychiatric disorders (see Section 4.2 Dose and Method of Administration).
Treatment of ADHD with stimulant products including Artige should not be initiated in patients with acute psychosis, acute mania, or acute suicidality. These acute conditions should be treated and controlled before ADHD treatment is considered. Methylphenidate should not be used as treatment for severe depression of either exogenous or endogenous origin.
In the case of emergent psychiatric symptoms or exacerbation of pre-existing psychiatric symptoms, Artige should not be given to patients unless the benefit outweighs the potential risk.
Psychotic symptoms. Psychotic symptoms, including visual and tactile hallucinations or mania have been reported in patients administered usual prescribed doses of stimulant products, including Artige (see Section 4.8 Adverse Effects (Undesirable Effects)). Physicians should consider treatment discontinuation if psychotic symptoms occur.
Bipolar illness. Particular care should be taken in using stimulants to treat ADHD in patients with comorbid bipolar disorder because of concern for possible induction of a mixed/manic episode in such patients.
Aggressive behaviour. Artige has been causally associated with aggression. Emergent aggressive behaviour or an exacerbation of baseline aggressive behaviour has been reported during stimulant therapy, including Artige. Physicians should evaluate the need for adjustment of treatment regimen in patients experiencing these behavioural changes, bearing in mind that upwards or downwards titration may be appropriate. Treatment interruption can be considered.
Suicidal tendency. Patients and caregivers of patients should be alerted about the need to monitor for clinical worsening, suicidal behaviour or thoughts or unusual changes in behaviour and to seek medical advice immediately if these symptoms appear. The physician should initiate appropriate treatment of the underlying psychiatric condition and consider a possible discontinuation or change in the ADHD treatment.
Motor and verbal tics. CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported (see Section 4.8 Adverse Effects (Undesirable Effects)). Therefore, clinical evaluation for tics in patients should precede use of stimulant medicine. Family history should be assessed and clinical evaluation for tics or Tourette's syndrome in patients should precede use of methylphenidate for ADHD treatment. Artige is contraindicated in case of diagnosis or family history of Tourette's syndrome (see Section 4.3 Contraindications). Patients should be regularly monitored for the emergence or worsening of tics during treatment with methylphenidate.
Serotonin syndrome. Serotonin syndrome has been reported following co-administration of methylphenidate with serotonergic drugs such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). The concomitant use of methylphenidate and serotonergic drugs is not recommended as this may lead to the development of serotonin syndrome. The symptoms of serotonin syndrome may include mental status changes (e.g. agitation, hallucinations, delirium, and coma), autonomic instability (e.g. tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g. tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g. nausea, vomiting, diarrhoea). Prompt recognition of these symptoms is important so that treatment with methylphenidate and serotonergic drugs can be immediately discontinued and appropriate treatment instituted (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Paediatric use. Methylphenidate should not be used in children under 6 years of age, since safety and efficacy in this age group have not been established. Medicines should be kept out of the reach of children.
Growth retardation. Moderately reduced weight gain and slight growth retardation have been reported with the long term use of stimulants, including methylphenidate, in children (see Section 4.8 Adverse Effects (Undesirable Effects)). Growth should be monitored as clinically necessary during treatment with methylphenidate and patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
Careful follow up of weight and height in children aged 7 to 10 years who were randomised to either methylphenidate or non-medicine treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medicine treated children over 36 months (to the ages of 10 to 13 years), suggests that consistently medicated children (i.e. treatment for 7 days per week throughout the year) have a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this period of development.
Published data are inadequate to determine whether chronic use of amphetamines may cause similar suppression of growth, however, it is anticipated that they likely have this effect as well. Therefore, growth should be monitored during treatment with stimulants, and patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
The retardation of growth referred to under Section 4.8 Adverse Effects (Undesirable Effects) is usually followed by catch-up growth when the medicine is discontinued. In order to minimise such complications, drug-free periods over weekends, school holidays and long vacations are advocated by some specialists.
Fatigue. Methylphenidate should not be employed for the prevention or treatment of normal fatigue states.
Seizures. There is some clinical evidence that methylphenidate may lower the convulsion threshold in patients with a history of seizures, with prior EEG abnormalities in the absence of seizures and, rarely, in the absence of a history of seizures and no prior EEG evidence of seizures. Safe concomitant use of anticonvulsants and methylphenidate has not been established. In the presence of seizures, the drug should be discontinued.
Acute angle closure glaucoma. There have been reports of acute angle closure glaucoma associated with methylphenidate treatment. Although the mechanism is not clear, Ritalin-treated patients considered at risk for acute angle closure glaucoma (e.g. patients with significant hyperopia) should be evaluated by an ophthalmologist.
Increased intraocular pressure and glaucoma. There have been reports of an elevation of intraocular pressure (IOP) and glaucoma (including open angle glaucoma and angle closure glaucoma) associated with methylphenidate treatment (see Section 4.8 Adverse Effects (Undesirable Effects)). Close monitoring of Ritalin-treated patients with a history of abnormally increased IOP or glaucoma is recommended.
Priapism. Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate products in both paediatric and adult patients. Priapism was not reported with drug initiation but developed after some time on the drug, often subsequent to an increase in dose. Priapism has also appeared during a period of drug withdrawal (drug holidays or discontinuation). Patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
Peripheral vasculopathy, including Raynaud's phenomenon. Stimulants, including Artige, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, have been observed in postmarketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g. rheumatology referral) may be appropriate for certain patients.
Drug abuse and dependence. Caution is called for in emotionally unstable patients, such as those with a history of drug or alcohol dependence, because they may increase the dosage on their own initiative. Chronic abuse can lead to marked tolerance and psychological dependence with varying degrees of abnormal behaviour. Frank psychotic episodes may occur, especially in response to parenteral abuse.
Clinical data indicate that children given Artige are not more likely to abuse drugs as adolescents or adults. Methylphenidate abuse or dependence does not appear to be a problem in adolescents or adults who were treated with methylphenidate for ADHD as children.
Use with alcohol. Alcohol may exacerbate the CNS adverse reactions of psychoactive drugs, including methylphenidate. Therefore, it is advisable for patients to abstain from alcohol during treatment.
Withdrawal. Careful supervision is required during drug withdrawal, since this may unmask depression as well as the effects of chronic over-activity. Some patients may require long-term follow-up.
Haematological effects. Data on safety and efficacy of long-term use of methylphenidate are not complete. Patients requiring long-term therapy should be carefully monitored and periodic complete blood counts, differential and platelet counts are advisable during prolonged therapy. In the event of haematological disorders appropriate medical intervention should be considered (see Section 4.8 Adverse Effects (Undesirable Effects)).
Effects on laboratory tests. Methylphenidate may induce false positive laboratory tests for amphetamines, particularly with immunoassays screen test.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Anti-hypertensive drugs. Artige may decrease the effectiveness of drugs used to treat hypertension.
Use with drugs that elevate blood pressure. Methylphenidate should be used with caution in patients being treated with drugs that elevate blood pressure due to the risk of severe hypertension (see Section 4.4 Special Warnings and Precautions for Use, Cerebrovascular conditions).
Because of possible hypertensive crisis, Artige is contraindicated in patients being treated (currently or within the preceding 2 weeks) with non-selective, irreversible MAO-inhibitors (see Section 4.3 Contraindications).
Use with anaesthetics. There is a risk of sudden blood pressure and heart rate increase during surgery. If surgery is planned, Artige should not be taken on the day of surgery.
Use with centrally acting alpha-2 agonists (e.g. clonidine). Serious adverse events including sudden death, have been reported in concomitant use with clonidine, although no causality for the combination has been established.
Use with dopaminergic drugs. As an inhibitor of dopamine reuptake, Artige may be associated with pharmacodynamic interactions when coadministered with direct and indirect dopamine agonists (including DOPA and tricyclic antidepressants) as well as dopamine antagonists (antipsychotics, e.g. haloperidol).
Concomitant use of Artige with antipsychotics is not recommended due to its counteracting mechanism of action. If upon medical assessment the combination is deemed necessary, monitoring for extrapyramidal symptoms (EPS) is recommended, as the concomitant use of methylphenidate with antipsychotics may increase the risk of EPS when there is a change (increase or decrease) in dosage of either or both medications.
Use with serotonergic drugs. The concomitant use of methylphenidate and serotonergic drugs is not recommended as this may lead to the development of serotonin syndrome (see Section 4.4 Special Warnings and Precautions for Use). Methylphenidate has been shown to increase extracellular serotonin and noradrenaline and appears to have weak potency in binding serotonin transporter.
Pharmacokinetic interactions. Artige is not metabolized by cytochrome P450 to a clinically relevant extent. Inducers or inhibitors of cytochrome P450 are not expected to have any relevant impact on Artige pharmacokinetics. Conversely, the d- and l- enantiomers of methylphenidate did not relevantly inhibit in vitro cytochrome P450 1A2, 2C8, 2C9, 2C19, 2D6, 2E1 and 3A.
Artige coadministration did not increase plasma concentrations of the CYP2D6 substrate desipramine.
Case reports have shown that methylphenidate may inhibit the metabolism of coumarin anticoagulants, anticonvulsants (phenobarbitone, primidone, phenytoin), phenylbutazone and tricyclic antidepressants (imipramine, desipramine), but pharmacokinetic interactions were not confirmed when explored at higher sample sizes. Reduction in the dosage of these drugs may be required when they are given concomitantly with methylphenidate.
Serious adverse events have been reported in concomitant use with clonidine, although no causality for the combination has been established. The safety of using methylphenidate in combination with clonidine or other centrally acting alpha-2 agonists has not been systematically evaluated.
Other specific drug-drug interaction studies with Artige have not been performed in vivo.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No human data on the effect of methylphenidate on fertility are available.
Women of child-bearing potential. Methylphenidate should not be prescribed for women of childbearing age unless, in the opinion of the physician, the potential benefits outweigh the possible risks (see Use in pregnancy).
Use in pregnancy. (Category D)
The safety of methylphenidate for use during human pregnancy has not been established. Data from a cohort study of in total approximately 3,400 pregnancies exposed in the first trimester do not suggest an increased risk of overall birth defects. There was a small increased occurrence of cardiac malformations in women who receive methylphenidate during the first trimester of pregnancy, compared with non-exposed pregnancies. Methylphenidate should not be prescribed for pregnant women unless, in the opinion of the physician, the potential benefits outweigh the possible risks.
As a general rule no drugs should be taken during the first 3 months of pregnancy, and the benefits and risks of taking drugs should be carefully considered throughout the whole of the pregnancy.
Use in lactation. Case reports showed that methylphenidate was distributed into breast milk reaching a milk-to-plasma ratio of approximately 2.5. For safety reasons, mothers taking methylphenidate should refrain from breastfeeding their infants. A decision should be made by the prescriber whether the mother must abstain from breastfeeding or abstain from methylphenidate therapy, taking into account the benefit of breastfeeding to the child and the benefit of therapy to the mother.
4.7 Effects on Ability to Drive and Use Machines
Artige may cause dizziness, drowsiness, blurred vision, hallucinations or other CNS side effects (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients experiencing such side effects should refrain from driving, operating machinery, or engaging in other potentially hazardous activities.
4.8 Adverse Effects (Undesirable Effects)
Post-marketing experience. Nervousness and insomnia are very common adverse reactions which occur at the beginning of Artige treatment and are usually controlled by reducing the dosage and omitting the drug in the afternoon or evening.
Loss of appetite is very common but usually transient. Abdominal pain, insomnia and tachycardia are common, usually at the beginning of treatment and may be alleviated by concomitant food intake.
Tabulated summary of adverse drug reactions. Adverse drug reactions listed in Table 1 are listed by MedDRA system organ class. Within each system organ class, the adverse drug reactions are ranked by frequency, with the most frequent reactions first. Within each frequency grouping, adverse drug reactions are presented in order of decreasing seriousness. In addition, the corresponding frequency category for each adverse drug reaction is based on the following convention (CIOMS III): very common ≥ 10%; common ≥ 1% to < 10%; uncommon ≥ 0.1% to < 1%; rare ≥ 0.01% to < 0.1%; very rare < 0.01%.

Adverse events reported since market introduction in patients taking methylphenidate include suicide, suicide attempt and suicidal ideation. No causal relationship between methylphenidate and these events has been established.
Adverse drug reactions from spontaneous reports and literature cases (frequency not known). The following adverse drug reactions have been derived from post-marketing experience with methylphenidate via spontaneous case reports and literature cases (see Table 2). Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency which is therefore categorized as not known. Adverse drug reactions are listed according to system organ classes in MedDRA. Within each system organ class, ADRs are presented in order of decreasing seriousness.

Immune system disorders. Hypersensitivity reactions such as auricular swelling.
Renal and urinary disorders. Haematuria, incontinence.
General disorders and administration site conditions. Sudden cardiac death.
Investigations. Cardiac murmur.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Signs and symptoms of acute overdosage, mainly due to over-stimulation of the central nervous system and from excessive sympathomimetic effects, may include: vomiting, agitation, tremors, hyperreflexia, muscle twitching, convulsions (may be followed by coma), euphoria, confusion, hallucinations, delirium, sweating, flushing, headache, hyperpyrexia, tachycardia, palpitation, cardiac arrhythmias, hypertension, mydriasis, dryness of mucous membranes and rhabdomyolysis.
Treatment. Treatment consists of appropriate supportive measures and symptomatic treatment of life-threatening events, e.g. hypertensive crisis, cardiac arrhythmias, convulsions. For the most current guidance for treatment of symptoms of overdose, the practitioner should consult the Poisons Information Centre on 13 11 26 or current toxicological publication.
The patient must be protected against self-injury and against external stimuli that would aggravate overstimulation already present. If the signs and symptoms are not too severe and the patient is conscious, further absorption may be limited by administration of activated charcoal. In cases of marked agitation, intravenous doses of diazepam or haloperidol should be given. Hypertension may be controlled by alpha-adrenergic blocking agents or intravenous sodium nitroprusside.
Intensive care must be provided to maintain adequate circulation and respiratory exchange; external cooling procedures may be required for hyperpyrexia.
The efficacy of peritoneal dialysis or extracorporeal haemodialysis for methylphenidate overdosage has not been established. Clinical experience with overdose is limited. Patients who have received doses higher than those recommended should be carefully monitored. In the event of overdose leading to clinically significant hypocalcaemia, reversal may be achieved with supplemental oral calcium and/or infusion of calcium gluconate.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacodynamic Pharmacotherapeutic group: psychostimulants. ATC code: N06BA04.
Pharmacodynamics. Artige is a racemate consisting of a 1:1 mixture of d-threo methylphenidate (d-MPH) and l-threo methylphenidate (l-MPH).
Mechanism of action. Methylphenidate is a central nervous system (CNS) stimulant. Its mode of action in humans is not completely understood but methylphenidate presumably exerts its stimulant effect by an inhibition of dopamine and norepinephrine reuptake into presynaptic neurons and thereby increasing these neurotransmitters in the extraneuronal space.
There is neither specific evidence which clearly establishes the mechanism whereby methylphenidate produces its mental and behavioural effects in children, nor conclusive evidence as to how these effects relate to the condition of the central nervous system.
The l-enantiomer is thought to be pharmacologically inactive.
Repeated oral administration of methylphenidate to young rats was associated with decreased spontaneous locomotor activity at systemic exposures (plasma AUC) about 3-fold that at the maximum clinical dose, due to an exaggerated pharmacological activity of methylphenidate. A deficit in the acquisition of a specific learning task was also observed, only in females, at systemic exposures (plasma AUC) 8-fold that at the maximum clinical dose. The clinical relevance of these findings is unknown.
5.2 Pharmacokinetic Properties
Absorption. Following oral administration of Artige tablets, the active substance, methylphenidate hydrochloride, is rapidly and almost completely absorbed from the tablets. Owing to extensive first-pass metabolism, the absolute bioavailability was 22 ± 8% for the d-enantiomer and 5 ± 3% for the l-enantiomer. Ingestion with food increased both the Cmax (23%) and the AUC (15%) of methylphenidate, but had no effect on the rate of absorption. Peak plasma concentrations of approx. 40 nanomol/litre (11 nanogram/mL) are attained, on the average, 2 hours after administration. The peak plasma concentrations, however, vary markedly from one person to another. The area under the plasma concentration curve (AUC), as well as the peak plasma concentration, is proportional to the size of the dose administered.
Distribution. In the blood, methylphenidate and its metabolites become distributed in the plasma (57%) and the erythrocytes (43%). Methylphenidate and its metabolites have low plasma protein-binding (approximately 15%). The apparent volume of distribution (Vd) has been calculated at 13.1 L/kg after an oral dose. The volume of distribution was 2.65 ± 1.11 L/kg for d-MPH and 1.80 ± 0.91 L/kg for l-MPH, following intravenous administration of 10 mg MPH.
Methylphenidate excretion into breast milk has been noted in two case reports where the calculated relative infant dose was ≤ 0.2% of the weight adjusted maternal dose (see Section 4.6 Fertility, Pregnancy and Lactation, Use in lactation). Adverse events were not noted in either infant (6 months and 11 months of age).
Metabolism. Biotransformation of methylphenidate, primarily by the carboxyl esterase CES1A1, is rapid and extensive. Peak plasma concentrations of the main, deesterified, metabolite, α-phenyl-2-piperidine acetic-acid (ritalinic acid), are attained about 2 hours after administration and are 30 to 50 times higher than those of the unchanged substance. The half-life of α-phenyl-2-piperidine acetic acid is about twice that of methylphenidate. Only small amounts of hydroxylated metabolites (e.g. hydroxymethylphenidate and hydroxyritalinic acid) are detectable. Therapeutic activity seems to be principally due to the parent compound.
Excretion. Methylphenidate is eliminated from the plasma with a mean half-life of 2 to 3 hours, and the calculated mean systemic clearance is 4 to 10 L/h/kg after an oral dose. The systemic clearance is 0.40 ± 0.12 L/h/kg for d-MPH and 0.73 ± 0.28 L/h/kg for l-MPH. Within 48 to 96 hours, 78 to 97% of the dose administered is excreted in the urine and 1 to 3% in the faeces in the form of metabolites. Unchanged methylphenidate appears in the urine only in small quantities (< 1%). Most of the dose is excreted in the urine as α-phenyl-2-piperidine acetic acid (60-86%).
Special populations. Effect of age. There are no apparent differences in the pharmacokinetic behaviour of methylphenidate in hyperactive children and healthy adult volunteers.
Patients with renal impairment. Elimination data from patients with normal renal function suggest that renal excretion of unchanged methylphenidate would hardly be diminished in the presence of impaired renal function. However, renal excretion of the metabolite α-phenyl-2-piperidine acetic acid may be reduced.
5.3 Preclinical Safety Data
Fertility. Methylphenidate did not impair fertility in male or female mice that were fed diets containing the drug in an 18 week Continuous Breeding study. The study was conducted at doses up to 160 mg/kg/day, approximately 11-fold the highest recommended human dose of methylphenidate on a mg/m2 basis.
Reproductive animal toxicity. Adequate animal reproduction studies to establish safe use of methylphenidate during pregnancy have not been conducted. Oral administration of methylphenidate to rabbits during the period of organogenesis has produced teratogenic effects at systemic exposures (plasma AUC) approximately 3 times clinical exposure at the maximum recommended human dose. The exposure at the no-effect dose was less than human exposure. In rats, teratogenic effects were not seen at systemic exposures (plasma AUC) approximately 12 times clinical exposure at the maximum recommended human dose.
Genotoxicity. Methylphenidate was not mutagenic in assays in vitro (Ames reverse mutation assay and the mouse lymphoma cell forward mutation assay). Methylphenidate showed evidence of a weak clastogenic response in vitro (Chinese Hamster Ovary cells) but was negative in vivo (mouse bone marrow micronucleus assay).
Carcinogenicity. In a lifetime carcinogenicity study carried out in B6C3F1 mice, methylphenidate caused an increase in hepatocellular adenomas and, in males only, an increase in hepatoblastomas at a daily dose of approximately 60 mg/kg/day. This dose is approximately 4 times the maximum recommended human dose of methylphenidate on a mg/m2 basis. Hepatoblastoma is a relatively rare rodent malignant tumour type. The mouse strain used is sensitive to the development of hepatic tumours, and the significance of these results to humans is unknown.
Methylphenidate did not cause any increases in tumours in a lifetime carcinogenicity study carried out in F344 rats; the highest dose used was approximately 50 mg/kg/day, which is approximately 7 times the maximum recommended human dose of methylphenidate on a mg/m2 basis.
In a 24-week carcinogenicity study in the transgenic mouse strain p53+/-, there was no evidence of carcinogenicity. Male and female mice were fed diets containing the same concentration of methylphenidate as in the lifetime carcinogenicity study; approximately 60 and 74 mg/kg/day of methylphenidate, respectively, which is approximately 4 and 5 times the maximum recommended human dose of methylphenidate on a mg/m2 basis, respectively.
Comment. The US Food and Drug Administration examined data from the Surveillance, Epidemiology and End Results (SEER) database for the years 1973 to 1991 and found that the estimated incidence of hepatoblastoma in the general population was not greater than 1 in 10 million person-years.
A total of 174 cases of hepatoblastoma were reported by the SEER for the period 1973 to 1995. The age-adjusted incidence rate is very low (IR=0.0382 per 100,000 person-years). The majority of cases (149 out of 174) were diagnosed among the age group 0 to 4 years old, which is in accordance with the natural history of the disease. For the age group 5 to 24 years old the rates of hepatoblastoma are very low with 14 cases reported. For the 0 to 4 years old age group, incidence rates of hepatoblastoma have risen slowly, ranging from 0.3032 per 100,000 in 1973 to 0.4889 per 100,000 in 1995. On the basis of experience since marketing Artige, there is no evidence that the incidence is higher in patients receiving Artige.
6 Pharmaceutical Particulars
6.1 List of Excipients
Artige tablet lactose, calcium phosphate, gelatin, magnesium stearate, purified talc and wheat starch.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from moisture.
6.5 Nature and Contents of Container
Blister packs.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
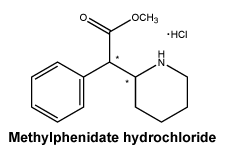
Chemical structure. Active ingredient: Methylphenidate hydrochloride.
Chemical names: Methyl (R*, R*)-(±)-α-phenyl-2-piperidine acetate hydrochloride; or 2-Piperidineacetic acid, α-phenyl-, methyl ester, hydrochloride; or Methyl α-phenyl-2-piperidineacetate hydrochloride.
Molecular formula: C14H19NO2.HCl.
Molecular weight: 269.8.
7 Medicine Schedule (Poisons Standard)
Controlled Drug (S8).
Date of First Approval
02 June 2005
Date of Revision
30 January 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.