Atacand Plus
Brand Information
| Brand name | Atacand Plus |
| Active ingredient | Candesartan cilexetil + Hydrochlorothiazide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Atacand Plus.
Summary CMI
ATACAND PLUS®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ATACAND PLUS?
ATACAND PLUS contains the active ingredients candesartan cilexetil and hydrochlorothiazide. ATACAND PLUS is used to treat high blood pressure also called hypertension. For more information, see Section 1. Why am I using ATACAND PLUS? in the full CMI.
2. What should I know before I use ATACAND PLUS?
Do not use if you have ever had an allergic reaction to ATACAND PLUS or any of the ingredients listed at the end of the CMI, have severe kidney or liver disease and/or conditions associated with impaired bile flow (cholestasis), or are taking blood pressure medicine containing aliskiren and you have diabetes mellitus or kidney problems.
Talk to your doctor if you have any other medical conditions, take any other medicines, are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ATACAND PLUS? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ATACAND PLUS and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ATACAND PLUS?
- ATACAND PLUS should be taken once daily, at about the same time each day.
- It does not matter whether you take ATACAND PLUS with food or on an empty stomach.
More instructions can be found in Section 4. How do I use ATACAND PLUS? in the full CMI.
5. What should I know while using ATACAND PLUS?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ATACAND PLUS? in the full CMI.
6. Are there any side effects?
Side effects may include headache, dizziness, weariness, chest or throat infection, flu-like symptoms, feeling sick (nausea, vomiting), back pain. Side effects that require urgent medical attention include: Aching muscles, tenderness or weakness in the muscle; swelling of the face, lips, tongue, throat, hands, feet or ankles; harsh sounds when breathing; rash, itching or hives; yellowing of the skin and/or eyes; easy bruising or bleeding more easily than normal; extreme fatigue, tiredness, weakness; signs of frequent infections such as fever, severe chills, sore throat or mouth ulcers; passing little or no urine; awareness of heartbeat; decrease in vision or pain in eyes; new and/or suspicious skin lesions.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ATACAND PLUS®
Active ingredient: candesartan cilexetil and hydrochlorothiazide
Consumer Medicine Information (CMI)
This leaflet provides important information about using ATACAND PLUS. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ATACAND PLUS.
Where to find information in this leaflet:
1. Why am I using ATACAND PLUS?
2. What should I know before I use ATACAND PLUS?
3. What if I am taking other medicines?
4. How do I use ATACAND PLUS?
5. What should I know while using ATACAND PLUS?
6. Are there any side effects?
7. Product details
1. Why am I using ATACAND PLUS?
ATACAND PLUS contains the active ingredient candesartan cilexetil and hydrochlorothiazide. Candesartan cilexetil is a type of medicine called an angiotensin II receptor antagonist (or blocker). Hydrochlorothiazide is a type of medicine called a diuretic.
ATACAND PLUS is used to treat high blood pressure (also called hypertension).
ATACAND PLUS lowers blood pressure by dilating (expanding) small blood vessels from the heart and reducing the amount of excess fluid in the body, letting the blood be pumped around the body more easily.
2. What should I know before I use ATACAND PLUS?
Warnings
Do not use ATACAND PLUS if:
- you are allergic to candesartan cilexetil, hydrochlorothiazide, or any of the ingredients listed at the end of this leaflet.
- you are allergic to any sulphur drugs (sulphonamides) such as some antibiotics or some medicines to treat diabetes.
- you have severe kidney or liver disease and/or conditions associated with impaired bile flow (cholestasis).
- you are taking blood pressure medicines containing aliskiren and you have diabetes mellitus (type I or II) or kidney problems.
- you have gout.
Check with your doctor if you:
Have any other medical condition, especially:
- kidney, liver or heart problems.
- a narrowing of the valves which allow blood into or out of the heart (aortic valve stenosis or mitral valve stenosis) or a genetic condition which causes thickening of the walls in the heart called Obstructive Hypertrophic Cardiomyopathy.
- a condition where your adrenal glands produce too much aldosterone, called primary hyperaldosteronism.
- recent excessive vomiting or diarrhoea.
- skin cancers, including non-melanoma skin cancer (NMSC), basal cell carcinoma (BCC) or squamous cell carcinoma (SCC).
- Systemic Lupus Erythematosus (SLE), a disease affecting the skin, joints and kidneys.
Check with your doctor if you are taking any medicines for any other conditions.
Check with your doctor if you:
- are on a salt restricted diet.
- had a past operation known as sympathectomy (a procedure on the sympathetic nerve chain sometimes used to treat excessive sweating, chronic pain, or poor blood flow to the legs).
- have any planned surgeries that may need a general anaesthetic (including dental surgery).
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use ATACAND PLUS if you are pregnant or planning to become pregnant.
Discuss with your doctor if you are pregnant or intend to become pregnant. ATACAND PLUS may affect your baby if you take it during pregnancy.
Discuss with your doctor if you are breastfeeding or intend to breastfeed. It is not known if ATACAND PLUS passes into breast milk. Because of its potential to effect the baby, it is not recommended to breastfeed while taking ATACAND PLUS.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and ATACAND PLUS may interfere with each other. These include:
- Any medicines containing potassium, including salt substitutes.
- Diuretics (fluid tablets).
- Lithium, a medicine used to treat psychiatric conditions such as bipolar disorder or schizoaffective disorder.
- Non-steroidal anti-inflammatory drugs (NSAIDs), medicines used to relieve pain, swelling and other symptoms of inflammation, including arthritis, such as aspirin, etoricoxib, and celecoxib.
- Medicines used to treat blood pressure, including aliskiren-containing medicines, angiotensin-converting-enzyme (ACE) inhibitors such as enalapril, lisinopril, ramipril, and captopril, and beta-blockers such as metoprolol and propranolol.
- Digoxin or mineralocorticoid receptor antagonists (MRAs), such as spironolactone and eplerenone, medicines used to treat heart failure.
- Colestipol and cholestyramine, medicines used to treat high blood cholesterol levels.
- Heparin, a medicine used to stop blood from clotting.
- Some antibiotics such as trimethoprim/ sulfamethoxazole and some anti-fungals such as amphotericin.
- Strong pain killers such as codeine, morphine, oxycodone and dextropropoxyphene.
- Barbiturates, such as phenobarbitone, which are used to prevent seizures.
- Medicines used to treat diabetes, including insulin.
- Calcium supplements or medicines containing calcium.
- Vitamin D supplements.
- Medicines called bile acid sequestrants used to treat high cholesterol (cholestyramine and colestipol hydrochloride).
- Medicines to treat irregular heartbeats such as digoxin.
- Corticosteroids such as prednisone, prednisolone, dexamethasone.
- Laxatives.
- Medicines used to treat cancer such as cyclophosphamide.
- Methotrexate, a medicine used to treat arthritis and some cancers.
- Medicines used for the treatment of Parkinson's disease including amantadine and biperiden.
- Ciclosporin, a medicine used to suppress the immune system.
- Diazoxide, a medicine used to treat low blood sugar.
- Medicines that contain atropine, such as for the treatment of diarrhoea.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ATACAND PLUS.
4. How do I use ATACAND PLUS?
How much to take
- The usual dose is one tablet once daily.
- Follow the instructions provided and use ATACAND PLUS until your doctor tells you to stop.
When to take ATACAND PLUS
- ATACAND PLUS should be taken once a day, at the same time each day. Keeping a regular time for taking ATACAND PLUS will help to remind you to take it.
- It does not matter whether you take ATACAND PLUS with food or on an empty stomach.
If you forget to use ATACAND PLUS
ATACAND PLUS should be used regularly at the same time each day. If you miss your dose at the usual time, take it as soon as you remember, as long as it is at least 12 hours before your next dose is due. Then go back to taking it as you normally would.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much ATACAND PLUS
If you think that you or anyone else may have taken too much ATACAND PLUS, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ATACAND PLUS?
Things you should do
Be sure to keep all of your doctor's appointments so that your progress can be checked. Your doctor will check your progress and may want to take some tests (e.g. blood tests, blood pressure) from time to time. These tests may help to prevent side effects.
Remind any doctor, dentist or pharmacist you visit that you are using ATACAND PLUS.
Things you should not do
- Do not stop using this medicine unless you have discussed it with your doctor.
Things to be careful of
- Move slowly when getting out of bed or standing up if you feel faint, dizzy or light-headed.
- Tell your doctor if you have excessive vomiting or diarrhoea while taking ATACAND PLUS.
- Make sure you drink enough water while you are taking ATACAND PLUS, especially during exercise and hot weather, or if you sweat a lot.
- Tell your doctor if you plan to have an examination such as an X-ray or a scan requiring and injection of iodinated contrast (dye) that you are taking ATACAND PLUS.
- If you have had skin cancer or if you develop a suspicious skin lesion during treatment with ATACAND PLUS, tell your doctor or pharmacist.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ATACAND PLUS affects you.
ATACAND PLUS may cause dizziness or weariness in some people during treatment.
Drinking alcohol
Tell your doctor if you drink alcohol.
Drinking alcohol while taking ATACAND PLUS may increase dizziness and light-headedness.
Looking after your medicine
Store ATACAND PLUS below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General disorders
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Joint or muscle related
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Infection related
| Make an appointment to speak to your doctor. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ATACAND PLUS contains
| Active ingredient (main ingredient) | candesartan cilexetil hydrochlorothiazide |
| Other ingredients (inactive ingredients) | Carmellose calcium Hyprolose Iron oxide red Iron oxide yellow Lactose monohydrate Magnesium stearate Maize starch Macrogol 8000 |
Do not take this medicine if you are allergic to any of these ingredients.
What ATACAND PLUS looks like
ATACAND PLUS 16/12.5 tablets are peach, oval, biconvex tablets with a score on both sides and engraved A/CS.
ATACAND PLUS 32/12.5 tablets are yellow, oval, biconvex tablets with a score and engraved A/CJ on one side and a pressure sensitive bisect on the reverse side.
ATACAND PLUS 32/25 tablets are pink, oval, biconvex tablets with a score and engraved A/CD on one side and a pressure sensitive bisect on the reverse side.
ATACAND PLUS is registered in blister packs* of 7 or 30 tablets.
*not all presentations might be available in Australia.
Australian Registration Numbers:
ATACAND PLUS 16/12.5 – AUST R 76646
ATACAND PLUS 32/12.5 – AUST R 154247
ATACAND PLUS 32/25 – AUST R 154251
Who distributes ATACAND PLUS
Link Medical Products Pty Ltd
5 Apollo Street
Warriewood, NSW, 2102
Australia
1800 181 060
linkhealthcare.com.au
This leaflet was prepared in October 2025.
Brand Information
| Brand name | Atacand Plus |
| Active ingredient | Candesartan cilexetil + Hydrochlorothiazide |
| Schedule | S4 |
MIMS Revision Date: 01 August 2024
1 Name of Medicine
Candesartan cilexetil and hydrochlorothiazide.
2 Qualitative and Quantitative Composition
Atacand Plus 16/12.5 contains 16 mg candesartan cilexetil and 12.5 mg of hydrochlorothiazide.
Atacand Plus 32/12.5 contains 32 mg candesartan cilexetil and 12.5 mg of hydrochlorothiazide.
Atacand Plus 32/25 contains 32 mg candesartan cilexetil and 25 mg of hydrochlorothiazide.
Excipients with known effect. Sugars as lactose monohydrate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Atacand Plus 16/12.5 tablets are peach, oval biconvex tablets with a score on both sides and engraved A/CS.
Atacand Plus 32/12.5 tablets are yellow, oval, biconvex tablets with a score and engraved A/CJ on one side and a pressure sensitive bisect on the reverse side.
Atacand Plus 32/25 tablets are pink, oval, biconvex tablets with a score and engraved A/CD on one side and a pressure sensitive bisect on the reverse side.
4 Clinical Particulars
4.1 Therapeutic Indications
The treatment of hypertension. Treatment should not be initiated with these fixed dose combinations.
4.2 Dose and Method of Administration
The dose of Atacand Plus must be determined by careful titration of the dose of each of the individual components.
The recommended dose of Atacand Plus is 1 tablet once daily. Atacand Plus may be taken with or without food. Atacand Plus tablets should not be divided.
Atacand Plus 16/12.5 may be administered in patients whose blood pressure is not optimally controlled with hydrochlorothiazide alone or Atacand 16 mg monotherapy.
Atacand Plus 32/12.5 or 32/25 may be administered in patients whose blood pressure is not optimally controlled with hydrochlorothiazide alone or Atacand 32 mg monotherapy, or at a lower dose of Atacand Plus. Dose titration of candesartan cilexetil is recommended when adding on to hydrochlorothiazide monotherapy.
Most of the antihypertensive effect is usually attained within 4 weeks of initiation of treatment.
Atacand Plus should not be used to initiate treatment.
Special patient populations. Paediatric use. The safety and efficacy of Atacand Plus has not been established in children.
Use in the elderly. Dose titration of candesartan cilexetil is recommended before treatment with Atacand Plus.
Hepatic impairment. Dose titration of candesartan cilexetil is recommended before treatment with Atacand Plus in patients with mild to moderate chronic liver disease.
Atacand Plus should not be used in patients with severe hepatic impairment and/or cholestasis (see Section 4.3 Contraindications).
Renal impairment. In patients with mild to moderate renal impairment (i.e. creatinine clearance 30-80 mL/min/1.73 m2 BSA) a dose titration is recommended.
Atacand Plus should not be used in patients with severe renal impairment (i.e. creatinine clearance < 30 mL/min/1.73 m2 BSA).
Intravascular volume depletion. Patients who are severely volume and/or sodium depleted should have this corrected before being treated with Atacand Plus.
4.3 Contraindications
Hypersensitivity to any component of Atacand Plus or to sulfonamide derived drugs.
Pregnancy and lactation.
Severe renal impairment (creatinine clearance < 30 mL/min/1.73 m2 BSA).
Severe hepatic impairment and/or cholestasis.
Gout.
The use of Atacand Plus in combination with aliskiren containing medicines in patients with diabetes mellitus (type I or II) or with moderate to severe renal impairment (GFR < 60 mL/min/1.73 m2).
4.4 Special Warnings and Precautions for Use
General. In patients whose vascular tone and renal function depend predominantly on the activity of the renin angiotensin aldosterone system (e.g. patients with severe congestive heart failure or underlying renal disease, including renal artery stenosis), treatment with drugs that affect this system has been associated with acute hypotension, azotaemia, oliguria or, rarely, acute renal failure. As with any antihypertensive agent, excessive blood pressure decrease in patients with ischaemic heart disease or atherosclerotic cerebrovascular disease could result in a myocardial infarction or stroke.
Dual blockade of the renin angiotensin aldosterone system (RAAS). There is evidence that the concomitant use of ACE inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of Atacand Plus with ACE inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
If dual blockade therapy is considered necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure.
ACE inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. The use of Atacand Plus with aliskiren is contraindicated in patients with diabetes mellitus (type I or II) or moderate to severe renal impairment (GFR < 60 mL/min/1.73 m2) (see Section 4.3 Contraindications).
Renal artery stenosis. Other drugs that affect the renin angiotensin aldosterone system, i.e. angiotensin converting enzyme (ACE) inhibitors, may increase blood urea and serum creatinine in patients with bilateral renal artery stenosis or stenosis of the artery to a solitary kidney. A similar effect may be anticipated with angiotensin II receptor antagonists.
Aortic and mitral valve stenosis or obstructive hypertrophic cardiomyopathy. As with other vasodilators, special caution is indicated in patients suffering from haemodynamically relevant aortic or mitral valve stenosis, or obstructive hypertrophic cardiomyopathy.
Primary hyperaldosteronism. Patients with primary hyperaldosteronism will not generally respond to antihypertensive drugs acting through inhibition of the renin angiotensin aldosterone system. Therefore, the use of Atacand Plus in these patients is not recommended.
Fluid and electrolyte imbalance. As for any patient receiving diuretic therapy, periodic determination of serum electrolytes should be performed at appropriate intervals.
Thiazides, including hydrochlorothiazide, can cause fluid or electrolyte imbalance (hypercalcaemia, hypokalaemia, hyponatraemia, hypomagnesaemia and hypochloraemic alkalosis).
Hydrochlorothiazide dose dependently increases urinary potassium excretion which may result in hypokalaemia. This effect of hydrochlorothiazide seems to be less evident when combined with candesartan cilexetil. The risk of hypokalaemia may be increased in patients with cirrhosis of the liver, in patients experiencing brisk diuresis, in patients with an inadequate oral intake of electrolytes and in patients receiving concomitant therapy with corticosteroids or adrenocorticotropic hormone (ACTH).
Based on experience with the use of other drugs that affect the renin angiotensin aldosterone system, concomitant use of Atacand Plus and ACE inhibitors, aliskiren, potassium sparing diuretics, potassium supplements or salt substitutes or other drugs that may increase serum potassium levels may lead to increases in serum potassium.
Non-melanoma skin cancer. An increased risk of non-melanoma skin cancer (NMSC) [basal cell carcinoma (BCC) and squamous cell carcinoma (SCC)] with increasing cumulative dose of hydrochlorothiazide exposure has been observed in two epidemiological studies based on Danish National Cancer Registry (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Photosensitizing actions of hydrochlorothiazide could act as a possible mechanism for NMSC.
Patients taking hydrochlorothiazide should be informed of the risk of NMSC and advised to regularly check their skin for any new lesions and promptly report any suspicious skin lesions. Possible preventive measures such as limited exposure to sunlight and UV rays and, in case of exposure, adequate protection should be advised to the patients in order to minimize the risk of skin cancer. Suspicious skin lesions should be promptly examined potentially including histological examinations of biopsies. The use of hydrochlorothiazide may also need to be reconsidered in patients who have experienced previous NMSC (see Section 4.8 Adverse Effects (Undesirable Effects)).
Acute respiratory toxicity. Very rare severe cases of acute respiratory toxicity, including acute respiratory distress syndrome (ARDS) have been reported after taking hydrochlorothiazide. Pulmonary oedema typically develops within minutes to hours after hydrochlorothiazide intake. At the onset, symptoms include dyspnoea, fever, pulmonary deterioration, and hypotension. If diagnosis of ARDS is suspected, Atacand Plus should be withdrawn, and appropriate treatment given. Hydrochlorothiazide should not be administered to patients who previously experienced ARDS following hydrochlorothiazide intake.
Kidney transplantation. There is limited clinical experience regarding the administration of Atacand Plus in patients who have undergone renal transplantation.
Combination use of ACE inhibitors or angiotensin receptor antagonists, anti-inflammatory drugs and thiazide diuretics. The use of an ACE inhibiting drug (ACE inhibitor or angiotensin receptor antagonist), an anti-inflammatory drug (NSAID or COX-2 inhibitor) and a thiazide diuretic at the same time increases the risk of renal impairment. This includes use in fixed combination products containing more than one class of drug. Combined use of these medications should be accompanied by increased monitoring of serum creatinine, particularly at the institution of the combination. The combination of drugs from these three classes should be used with caution particularly in elderly patients or those with pre-existing renal impairment.
Metabolic and endocrine effects. Treatment with a thiazide diuretic may impair glucose tolerance. Dosage adjustment of anti-diabetic drugs, including insulin, may be required. Latent diabetes mellitus may become manifest during thiazide therapy. Increases in cholesterol and triglyceride levels have been associated with thiazide diuretic therapy. At the doses contained in Atacand Plus, only minimal effects were observed. Thiazide diuretics increase serum uric acid concentration and may precipitate gout in susceptible patients.
Hypotension, volume depleted patients. Atacand Plus like all antihypertensive agents may cause symptomatic hypotension in some patients. Symptomatic hypotension may be expected to occur more frequently in patients who have been sodium and/or volume depleted by vigorous diuretic therapy and/or dietary salt restrictions, or vomiting and/or diarrhoea or haemodialysis. Sodium and/or volume depletion should be corrected prior to administration of Atacand Plus.
Postsympathectomy. The antihypertensive effects of thiazide diuretics may be increased in the postsympathectomy patient.
Systemic lupus erythematosus. Exacerbation or activation of systemic lupus erythematosus has been reported with the use of thiazide diuretics.
Anaesthesia and surgery. Hypotension may occur during anaesthesia and surgery in patients treated with angiotensin II antagonists due to blockade of the renin angiotensin aldosterone system. Very rarely, hypotension may be severe such that it may warrant the use of intravenous fluids and/or vasopressors.
Choroidal effusion, acute myopia and angle-closure glaucoma. Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction resulting in choroidal effusion with visual field defect, transient myopia and acute angle-closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue drug intake as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
Use in hepatic impairment. Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte balance may precipitate hepatic coma. There is no clinical experience with Atacand Plus in patients with hepatic impairment. Use in patients with severe hepatic impairment is contraindicated. Caution is advised in patients with mild to moderate hepatic impairment.
Use in renal impairment. As with other agents inhibiting the renin-angiotensin-aldosterone system, changes in renal function may be anticipated in susceptible patients treated with Atacand Plus (see Section 4.3 Contraindications).
Use in the elderly. For dosage recommendations for use of Atacand Plus in elderly patients please see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use, Combination use of ACE inhibitors or angiotensin receptor antagonists, anti-inflammatory drugs and thiazide diuretics; Section 5.2 Pharmacokinetic Properties, Pharmacokinetics in special populations.
Paediatric use. Safety and efficacy have not been established in children.
Effects on laboratory tests. In general, there were no clinically important influences of candesartan cilexetil/hydrochlorothiazide on routine laboratory variables. Increases in creatinine, urea, potassium, uric acid, glucose and ALAT (SGPT) and decreases in sodium have been observed. Minor decreases in haemoglobin and increases in ASAT (SGOT) have been observed in single patients.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The antihypertensive effect of Atacand Plus may be enhanced by other antihypertensives.
Dual blockade of the renin angiotensin aldosterone system (RAAS). Clinical trial data has shown that dual blockade of the RAAS through the combined use of ACE inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute renal failure) compared to the use of a single RAAS acting agent (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Candesartan cilexetil. Compounds which have been investigated include hydrochlorothiazide, warfarin, digoxin (see Hydrochlorothiazide below), oral contraceptives (i.e. ethinyloestradiol/ levonorgestrel), glibenclamide and nifedipine. No pharmacokinetic interactions of clinical significance were identified in these studies.
The antihypertensive effect of angiotensin II receptor antagonists, including candesartan, may be attenuated by NSAIDs including COX-2 inhibitors and acetylsalicylic acid.
As with ACE inhibitors, concomitant use of AIIRAs and NSAIDs may lead to an increased risk of worsening of renal function, including possible acute renal failure, and an increase in serum potassium, especially in patients with poor pre-existing renal function. The combination should be administered with caution, especially in older patients and in volume depleted patients. Patients should be adequately hydrated and consideration should be given to monitoring renal function after initiation of concomitant therapy and periodically thereafter.
Candesartan is eliminated only to a minor extent by hepatic metabolism (CYP2C9).
Interaction studies performed to date show no effect of candesartan on the metabolising capacity of CYP2C9 and CYP3A4. Based on in vitro data, no interaction would be expected to occur in vivo with drugs whose metabolism is dependent upon cytochrome P450 isoenzymes CYP1A2, CYP2A6, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4.
Hydrochlorothiazide. Alcohol, barbiturates, opioids and anaesthetics. Potentiation of thiazide diuretic induced orthostatic hypotension may occur.
Anti-diabetic agents (oral and insulin). Thiazides may increase blood glucose concentration and adjustment of anti-diabetic medication may be required.
Cardiac glycosides and other antiarrhythmics. Thiazide induced hypokalaemia and hypomagnesaemia predisposes to the potential cardiotoxic effects of digitalis glycosides and antiarrhythmics. Periodic monitoring of serum potassium is recommended when Atacand Plus is administered with such drugs.
Calcium salts and vitamin D. Thiazide diuretics may increase the serum calcium concentration due to decreased excretion. If calcium or vitamin D is prescribed, serum calcium levels should be monitored and calcium dosage adjusted accordingly.
Cholestyramine resin and colestipol hydrochloride. The absorption of thiazide may be delayed or decreased in the presence of bile acids sequestrants. Atacand Plus should be taken at least one hour before or after such drugs.
Lithium. Reversible increases in serum lithium concentrations and toxicity have been reported during concomitant administration of lithium with ACE inhibitors or hydrochlorothiazide. A similar effect may occur with angiotensin II receptor antagonists (AIIRAs) and careful monitoring of serum lithium levels is recommended during concomitant use.
Nonsteroidal anti-inflammatory drugs (NSAIDs). The diuretic, natriuretic and antihypertensive effect of hydrochlorothiazide is blunted by NSAIDs.
Hypokalaemic agents. The potassium depleting effect of hydrochlorothiazide could be expected to be potentiated by other drugs associated with potassium loss and hypokalaemia (e.g. other kaliuretic diuretics, laxatives, amphotericin, carbenoxolone, salicylic acid derivatives).
Potassium sparing agents. Based on experience with the use of other drugs that affect the renin angiotensin aldosterone system, concomitant use of Atacand Plus and potassium sparing diuretics, potassium supplements or salt substitutes or other drugs that may increase serum potassium levels (e.g. heparin sodium, trimethoprim/ sulfamethoxazole) may lead to increases in serum potassium.
Nondepolarising muscle relaxants (e.g. tubocurarine). The effect of nondepolarising skeletal muscle relaxants (e.g. tubocurarine) may be potentiated by hydrochlorothiazide.
Pressor amines. Hydrochlorothiazide may cause the arterial response to pressor amines to decrease but not enough to exclude a pressor effect.
Iodinated contrast media. Hydrochlorothiazide may increase the risk of acute renal insufficiency especially with high doses of iodinated contrast media.
Corticosteroids, ACTH. The risk for hypokalaemia may be increased during concomitant use of steroids or adrenocorticotropic hormone (ACTH).
Amantadine. Thiazides may increase the risk of adverse effects caused by amantadine.
Beta-blockers and diazoxide. The hyperglycaemic effect of beta-blockers and diazoxide may be enhanced by thiazides.
Anticholinergic agents (e.g. atropine). Anticholinergic agents (e.g. atropine, biperiden) may increase the bioavailability of thiazide diuretics by decreasing gastrointestinal motility and stomach emptying rate.
Cytotoxic drugs (e.g. cyclophosphamide, methotrexate). Thiazides may reduce the renal excretion of cytotoxic drugs (e.g. cyclophosphamide, methotrexate) and potentiate their myelosuppressive effects.
Ciclosporin. Concomitant treatment with ciclosporin may increase the risk of hyperuricaemia and gout type complications.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The effects of hydrochlorothiazide alone or in combination with candesartan cilexetil on fertility have not been evaluated in animal studies. However, candesartan cilexetil alone had no adverse effects on the reproductive performance of male or female rats at oral doses up to 300 mg/kg/day.
Use in pregnancy. (Category D)
The use of Atacand Plus is contraindicated during pregnancy (see Section 4.3 Contraindications). Patients receiving Atacand Plus should be made aware of that before contemplating a possibility of becoming pregnant so that they can discuss appropriate options with their treating physician. When pregnancy is diagnosed, treatment with Atacand Plus must be stopped immediately and if appropriate, alternative therapy should be started.
The use of drugs that act directly on the renin angiotensin system has been associated with fetal and neonatal injury, including hypotension, neonatal skull hypoplasia, anuria, reversible or irreversible renal failure, and death. Exposure to angiotensin II receptor antagonist therapy is known to induce human fetotoxicity (decreased renal function, oligohydramnios, skull ossification retardation) and neonatal toxicity (renal failure, hypotension, hyperkalaemia).
There is limited experience with hydrochlorothiazide during pregnancy, especially during the first trimester. Animal studies are insufficient. Hydrochlorothiazide crosses the placenta. Based on the pharmacological mechanism of action of hydrochlorothiazide, its use during pregnancy may compromise foetoplacental perfusion and may cause foetal and neonatal effects like icterus, disturbance of electrolyte balance and thrombocytopenia.
Use in lactation. It is not known whether candesartan is excreted in human milk. However, candesartan is excreted in the milk of lactating rats. Hydrochlorothiazide passes into human milk. Because of the potential for adverse effects on the nursing infant, breastfeeding should be discontinued if the use of Atacand Plus is considered essential.
4.7 Effects on Ability to Drive and Use Machines
When driving vehicles or operating machines, it should be taken into account that dizziness or weariness may occur during treatment of hypertension.
4.8 Adverse Effects (Undesirable Effects)
Adverse events were mild and transient in controlled clinical studies with various doses of candesartan cilexetil/hydrochlorothiazide (candesartan cilexetil up to 32 mg and hydrochlorothiazide up to 25 mg). The overall incidence of adverse events showed no association with age or gender. Withdrawals from treatment due to adverse events were similar with candesartan cilexetil/hydrochlorothiazide (2.3-3.3%) and placebo (2.7-4.3%).
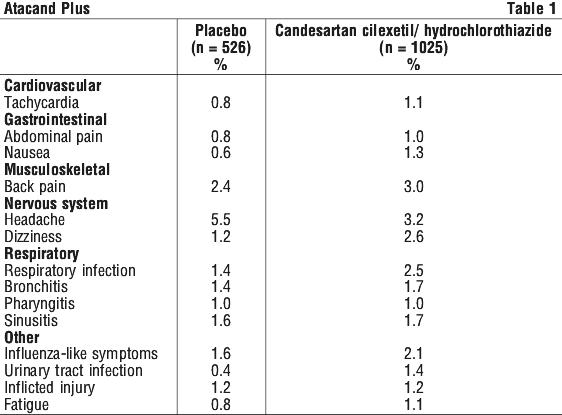
Clinical adverse events, regardless of causal relationship, with a cumulative 8-week incidence rate of ≥ 1% during treatment with candesartan cilexetil/hydrochlorothiazide up to 16/12.5 mg in double blind placebo-controlled trials are presented in Table 1.
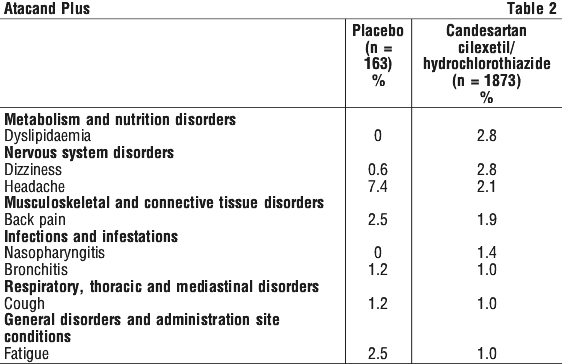
Clinical adverse events, regardless of causal relationship, occurring in ≥ 1% of the patients during 8-week randomised treatment with candesartan cilexetil/ hydrochlorothiazide 32/12.5 mg and 32/25 mg in double blind clinical trials are presented in Table 2.
Hydrochlorothiazide. The following clinical adverse events have been reported to occur with hydrochlorothiazide monotherapy: anorexia, loss of appetite, gastric irritation, diarrhoea, constipation, jaundice (intrahepatic cholestatic jaundice), pancreatitis, leucopenia, neutropenia/ agranulocytosis, thrombocytopenia, aplastic anaemia, hemolytic anaemia, bone marrow depression, photosensitivity reactions, fever, rash, cutaneous lupus erythematosus-like reactions, reactivation of cutaneous lupus erythematosus, urticaria, necrotising angiitis (vasculitis, cutaneous vasculitis), anaphylactic reactions, toxic epidermal necrolysis, respiratory distress (including pneumonitis, pulmonary oedema and acute respiratory distress syndrome (very rare) - see Section 4.4 Special Warnings and Precautions for Use), hyperglycaemia, glycosuria, hyperuricaemia, electrolyte imbalance (including hyponatraemia and hypokalaemia), increases in cholesterol and triglycerides, increases in BUN and serum creatinine, renal dysfunction, interstitial nephritis, muscle spasm, weakness, restlessness, transient blurred vision, lightheadedness, postural hypotension, vertigo, paraesthesia, cardiac arrhythmias, sleep disturbances, depression, choroidal effusion, acute myopia, acute angle closure glaucoma.
Post marketing. The following adverse reactions have been reported very rarely (< 0.01%) in post marketing experience.
Musculoskeletal, connective tissue and bone disorders. Myalgia.
Rare reports of rhabdomyolysis have been reported in patients receiving angiotensin II receptor blockers.
Although causality to candesartan has not been established, the following neuropsychiatric cardiovascular adverse reactions have been very rarely reported during post marketing surveillance. These were agitation, anxiety, depression, insomnia, somnolence, nervousness, nightmare, sleep disorder and palpitations.
The following adverse reactions have been reported post-marketing with hydrochlorothiazide, regardless of causality:
Neoplasms benign, malignant and unspecified (incl cysts and polyps). Frequency "not known": Non-melanoma skin cancer (Basal cell carcinoma and squamous cell carcinoma) (see Section 4.4 Special Warnings and Precautions for Use; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Based on pharmacological considerations, the main manifestation of an overdose of candesartan cilexetil is likely to be symptomatic hypotension and dizziness. In two case reports of overdose (160 mg and 432 mg candesartan cilexetil) patient recovery was uneventful.
The main manifestation of an overdose of hydrochlorothiazide is acute loss of fluid and electrolytes. Symptoms such as dizziness, hypotension, thirst, tachycardia, ventricular arrhythmias, sedation/ impairment of consciousness and muscle cramps can also be observed.
Management. No specific information is available on the treatment of overdosage with Atacand Plus. The following measures are, however, suggested in case of overdosage.
Administration of activated charcoal with or without gastric lavage. If symptomatic hypotension should occur, symptomatic treatment should be instituted and vital signs monitored. The patient should be placed supine with the legs elevated. If this is not sufficient, plasma volume should be increased by infusion of isotonic saline solution. Serum electrolyte and acid balance should be checked and corrected, if needed. Sympathomimetic drugs may be administered if the abovementioned measures are not sufficient.
Candesartan cannot be removed by haemodialysis. It is not known to what extent hydrochlorothiazide is removed by haemodialysis. Contact the Poisons Information Centre for advice on management.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Angiotensin II is the primary vasoactive hormone of the renin angiotensin aldosterone system and plays a significant role in the pathophysiology of hypertension and other cardiovascular disorders. It also has an important role in the pathogenesis of end organ hypertrophy and damage. The major physiological effects of angiotensin II, such as vasoconstriction, aldosterone stimulation, regulation of salt and water homeostasis and stimulation of cell growth, are mediated via the type 1 (AT1) receptor.
Candesartan cilexetil is a prodrug suitable for oral use. It is rapidly converted to the active drug, candesartan, by ester hydrolysis during absorption from the gastrointestinal tract. Candesartan is an angiotensin II receptor antagonist, selective for AT1 receptors, with tight binding to and slow dissociation from the receptor. It has no agonist activity.
Candesartan does not inhibit angiotensin converting enzyme (ACE), which converts angiotensin I to angiotensin II and degrades bradykinin. Since there is no effect on ACE and no potentiation of bradykinin or substance P, angiotensin II receptor antagonists are unlikely to be associated with cough. This has been confirmed in controlled clinical studies with candesartan. Candesartan does not bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation.
Hydrochlorothiazide inhibits the active reabsorption of sodium, mainly in the distal kidney tubules, and promotes the excretion of sodium, chloride and water. The renal excretion of potassium and magnesium increases dose dependently, while calcium is reabsorbed to a greater extent. Hydrochlorothiazide decreases plasma volume and extracellular fluid and reduces cardiac output and blood pressure. During long-term therapy, reduced peripheral resistance contributes to the blood pressure reduction.
Candesartan and hydrochlorothiazide have additive antihypertensive effects. In hypertensive patients, Atacand Plus results in dose dependent and long lasting reduction in arterial blood pressure without a reflex increase in heart rate. There is no indication of serious or exaggerated first dose hypotension or rebound effect after cessation of treatment.
After administration of a single dose of Atacand Plus, onset of the antihypertensive effect generally occurs within 2 hours. With continuous treatment, most of the reduction in blood pressure is attained within four weeks and is sustained during long-term treatment.
Atacand Plus once daily provides effective and smooth blood pressure reduction over 24 hours, with little difference between maximum and trough effects during the dosing interval. In double blind, randomised studies, the incidence of cough was lower during treatment with candesartan cilexetil/ hydrochlorothiazide than during treatment with combinations of ACE inhibitors and hydrochlorothiazide.
Age and gender have no influence on the efficacy of Atacand Plus.
In a variety of preclinical safety studies conducted in several species, expected exaggerated pharmacological effects (e.g. renal changes leading to juxtaglomerular cell hypertrophy, adrenal gland zona glomerulosa atrophy and reduced heart weight related to reduced afterload), due to modification of the renin angiotensin aldosterone system homeostasis, have been observed. The incidence and severity of the effects induced were dose and time related and have been shown to be reversible in adult animals. Addition of hydrochlorothiazide caused a potentiation of the nephrotoxicity seen with candesartan alone, however, without any qualitatively new findings.
Clinical trials. In a randomised, double blind, parallel group, 8-week clinical study, including 1975 randomised patients not adequately controlled on 32 mg candesartan cilexetil once daily, the addition of 12.5 mg or 25 mg hydrochlorothiazide resulted in additional blood pressure reductions of 7/3 mmHg and 9/4 mmHg respectively over 32 mg monotherapy. The candesartan cilexetil/ hydrochlorothiazide 32/12.5 mg and 32/25 mg combinations produced overall mean blood pressure reductions of 13/9 mmHg and 16/10 mmHg, respectively. This study also demonstrated that the 32/25 mg combination was significantly more effective than the 32/12.5 mg combination.
In two 8-week clinical studies (randomised, double blind, placebo controlled, parallel group) including 275 and 1524 randomised patients respectively, the candesartan cilexetil/ hydrochlorothiazide combinations 32/12.5 mg and 32/25 mg resulted in blood pressure reductions of 21/14 mmHg for the highest dose, and were significantly more effective than the respective monotherapy components.
Epidemiological studies have shown that long term treatment with hydrochlorothiazide reduces the risk for cardiovascular morbidity and mortality. There are no data regarding the effects of candesartan cilexetil and candesartan cilexetil/ hydrochlorothiazide on morbidity and mortality in hypertensive patients.
Non-melanoma skin cancer (NMSC). Based on available data from epidemiological studies, cumulative dose-dependent association between hydrochlorothiazide (HCTZ) and NMSC has been observed. One study included a population comprised of 71,553 cases of basal cell carcinoma (BCC) and of 8,629 cases of squamous cell carcinoma (SCC) matched to 1,430,883 and 172,462 population controls, respectively. High HCTZ use (≥ 50,000 mg cumulative) was associated with an adjusted OR of 1.29 (95% CI: 1.23-1.35) for BCC and 3.98 (95% CI: 3.68-4.31) for SCC. A clear cumulative dose response relationship was observed for both BCC and SCC. Another study showed a possible association between lip cancer (SCC) and exposure to HCTZ: 633 cases of lip-cancer were matched with 63,067 population controls, using a risk-set sampling strategy. A cumulative dose-response relationship was demonstrated with an adjusted OR 2.1 (95% CI: 1.7-2.6) increasing to OR 3.9 (3.0-4.9) for high use (~25,000 mg) and OR 7.7 (5.7-10.5) for the highest cumulative dose (~100,000 mg) (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
5.2 Pharmacokinetic Properties
The individual pharmacokinetic profiles of candesartan and hydrochlorothiazide were not clinically significantly affected when given in combination.
Absorption and distribution. Candesartan cilexetil. Following oral administration, candesartan cilexetil is converted to the active drug candesartan. The absolute bioavailability of candesartan is approximately 40% after an oral solution of candesartan cilexetil. The relative bioavailability of the tablet formulation compared with the same oral solution is approximately 34%, with little variability. The absolute bioavailability of candesartan following administration of the tablet is approximately 14%. The mean peak plasma concentration (Cmax) is reached 3-4 hours after taking a tablet. The candesartan plasma concentrations increase linearly with increasing doses in the therapeutic dose range.
The area under the plasma concentration versus time curve (AUC) of candesartan is not significantly affected by food. The peak concentration (Cmax) is increased by 26% and the rate of absorption is increased when taken with food. These changes are unlikely to result in clinically significant effects.
Candesartan is highly bound to plasma protein (more than 99%). The apparent volume of distribution (Vss) of candesartan is 0.1 L/kg.
Hydrochlorothiazide. Hydrochlorothiazide is rapidly absorbed from the gastrointestinal tract with an absolute bioavailability of approximately 70%. Concomitant intake of food increases the absorption by approximately 15%. The bioavailability may decrease in patients with cardiac failure and pronounced oedema.
The plasma protein binding of hydrochlorothiazide is approximately 60%. The apparent volume of distribution is approximately 0.8 L/kg.
Metabolism and excretion. Candesartan cilexetil. Candesartan cilexetil is mainly eliminated unchanged via urine and bile and is eliminated by hepatic metabolism only to a minor extent (CYP2C9). The terminal half-life of candesartan is approximately 9 hours. There is no accumulation following multiple doses.
The half-life of candesartan remains unchanged (approximately 9 h) after administration of candesartan cilexetil in combination with hydrochlorothiazide. No accumulation of candesartan occurs after repeated doses of the combination compared to monotherapy.
Total plasma clearance of candesartan is about 0.37 mL/min/kg, with a renal clearance of about 0.19 mL/min/kg. The renal elimination of candesartan is both by glomerular filtration and active tubular secretion. Following an oral dose of 14C labelled candesartan cilexetil about 30% and 70% of the total radioactivity is recovered in the urine and faeces, respectively.
Hydrochlorothiazide. Hydrochlorothiazide is not metabolised and is excreted almost entirely as unchanged drug by glomerular filtration and active tubular secretion. The terminal t1/2 of hydrochlorothiazide is approximately 8 hours. Approximately 70% of an oral dose is eliminated in the urine within 48 hours. The half-life of hydrochlorothiazide remains unchanged (approximately 8 h) after administration of hydrochlorothiazide in combination with candesartan cilexetil. No accumulation of hydrochlorothiazide occurs after repeated doses of the combination compared to monotherapy.
Pharmacokinetics in special populations. Candesartan cilexetil. In the elderly (over 65 years) both Cmax and AUC of candesartan are increased by approximately 50% and 80%, respectively in comparison to young subjects. However, the blood pressure response and the incidence of adverse events are similar after a given dose of Atacand Plus in young and elderly patients.
In patients with mild to moderate renal impairment, Cmax and AUC of candesartan increased during repeated dosing by approximately 50% and 70% respectively, but t1/2 was not altered compared to patients with normal renal function. The corresponding changes in patients with severe renal impairment were approximately 50% and 110% respectively. The terminal t1/2 of candesartan was approximately doubled in patients with severe renal impairment. The pharmacokinetics in patients undergoing haemodialysis were similar to those in patients with severe renal impairment.
In patients with mild to moderate hepatic impairment, there was a 23% increase in the AUC of candesartan. No initial dosage adjustment is necessary in these patients.
Hydrochlorothiazide. The terminal t1/2 of hydrochlorothiazide is prolonged in patients with renal impairment.
5.3 Preclinical Safety Data
Genotoxicity. Candesartan cilexetil alone or in combination with hydrochlorothiazide showed no evidence of genotoxic potential in a series of assays for gene mutations (Salmonella typhimurium and Eschericia coli), chromosomal aberrations (mouse micronucleus assay) and DNA damage (unscheduled DNA synthesis in rat liver). In addition, candesartan cilexetil alone showed no evidence of genotoxic potential in further assays for gene mutations (mouse L5178Y cells and Chinese hamster ovary cells). The active metabolite, candesartan, caused an increase in chromosomal aberrations in vitro (Chinese hamster lung cells) but not in vivo (mouse micronucleus test and chromosomal aberrations in rat bone marrow). However, hydrochlorothiazide had mutagenic activity in a mammalian cell assay (mouse L5178Y cells) and caused an increase in chromosomal aberrations in vitro (Chinese hamster lung cells). Candesartan at subclastogenic concentration did not modify these effects of hydrochlorothiazide. Hydrochlorothiazide also had a genotoxic activity in the sister chromatid exchange assay in Chinese hamster ovary cells and a nondisjunction assay in Aspergillus nidulans.
Carcinogenicity. The carcinogenic potential of candesartan cilexetil in combination with hydrochlorothiazide has not been evaluated in animal studies.
Candesartan cilexetil alone was not carcinogenic when administered orally to mice and rats for 2 years at doses of up to 100 and 1000 mg/kg/day, corresponding to ca. 7 times and 260 times the clinical exposure at the maximum recommended daily human dose of 32 mg (based on AUC, respectively).
Hydrochlorothiazide alone was not carcinogenic in female mice in doses ca. 600 mg/kg/day, or in male and female rats at doses up to ca. 100 mg/kg/day in two year feeding studies. These doses correspond to ca. 110 times (female mice) or 40 times (male and female rats) the clinical exposure at the maximum recommended daily human dose of 25 mg (based on BSA). However, there was equivocal evidence for hepatocarcinogenicity in male mice that received ca. 600 mg/kg/day.
6 Pharmaceutical Particulars
6.1 List of Excipients
In addition to candesartan cilexetil and hydrochlorothiazide, Atacand Plus 16/12.5, 32/12.5 and 32/25 also contain carmellose calcium, hyprolose, iron oxide red, iron oxide yellow, lactose monohydrate, magnesium stearate, maize starch and macrogol 8000.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
All presentations are packed in blister packs of 7 and 30 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Candesartan cilexetil is a white to off white powder and is practically insoluble in water.
Hydrochlorothiazide is a sulfonamide derived drug. It is a white, or almost white crystalline powder and is very slightly soluble in water.
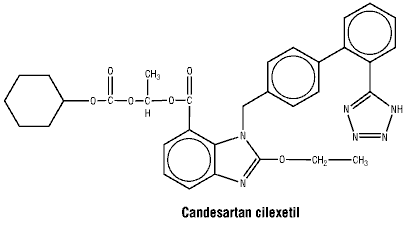
The molecular weight is 610.67.
The molecular formula is C33H34N6O6.
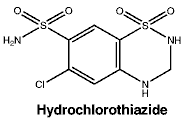
The molecular weight is 297.75.
The molecular formula is C7H8N3S2O4Cl.
CAS number. The CAS number for candesartan cilexetil is 145040-37-5.
The CAS number for hydrochlorothiazide is 58-93-5.
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
07 November 2000
Date of Revision
09 July 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.