Atropt 1%
Brand Information
| Brand name | Atropt 1% |
| Active ingredient | Atropine sulfate monohydrate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Atropt 1%.
Summary CMI
ATROPT 1% eye drops
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ATROPT 1% Eye Drops?
ATROPT 1% eye drops contain the active ingredient atropine (as atropine sulfate monohydrate). ATROPT 1% eye drops are used in eye examinations as well as other circumstances when the pupil is to be widened and/or the accommodation frozen.
For more information, see Section 1. Why am I using ATROPT 1% eye drops? in the full CMI.
2. What should I know before I use ATROPT 1% Eye Drops?
Do not use if you have ever had an allergic reaction to ATROPT 1% or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ATROPT 1% eye drops? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ATROPT 1% and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ATROPT 1% Eye Drops?
- Your doctor will advise you on how much and how often you should use ATROPT 1% eye drops.
- Gently pull out the lower eyelid to form a sac. Allow one drop to fall into the sac without touching the eye with the dropper end of the bottle.
- To minimise absorption of the drug other than into the eye, apply gentle pressure to the tear duct for approximately one minute immediately after application.
More instructions can be found in Section 4. How do I use ATROPT 1% eye drops? in the full CMI.
5. What should I know while using ATROPT 1% eye drops?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ATROPT 1% Eye Drops? in the full CMI.
6. Are there any side effects?
Common side effects of ATROPT 1% Eye Drops mainly relate to the eye or eye area. These include temporarily blurred vision; burning, stinging, redness, or watering of the eyes; and conjunctivitis (or pink eye). Serious side effects may also occur, mainly if too much of the medication is absorbed into the rest of the body. Signs of this include flushing and dryness of the skin; rapid and irregular pulse; fever, mental aberrations; loss of muscular coordination; dizziness or light-headedness; fatigue or lethargy; shortness of breath; or confusion, disorientation or altered mental state.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using ATROPT 1% eye drops?
ATROPT 1% eye drops contains the active ingredient atropine (as atropine sulfate monohydrate). ATROPT 1% belongs to a group of medications called anticholinergics, and works by widening the pupil of the eye, blocking its ability to change the focus of the eye.
ATROPT 1% eye drops are used in eye examinations as well as other circumstances when the pupil is to be widened and/or the accommodation frozen.
Ask your doctor if you have any questions about why ATROPT 1% eye drops have been prescribed for you.
Your doctor may have prescribed it for another reason.
2. What should I know before I use ATROPT 1% eye drops?
Warnings
Do not use ATROPT 1% if:
- you are allergic to atropine, or any of the ingredients listed at the end of this leaflet.:
- some symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or any other parts of the body; rash, itching or hives on the skin.
- always check the ingredients to make sure you can use this medicine. - you have angle closure glaucoma or a family history of glaucoma.
- the packaging shows signs of tampering.
- the expiry date on the label has passed.
Check with your doctor if you:
- have any other medical conditions, especially glaucoma or a family history of glaucoma
- wear contact lenses
- take any medicines for any other condition
- have had any allergy to any other medicines or any other substances, such as food, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Use in children
- Do not use in children less than 2 years old unless directed to by your doctor.
- Do not use in children who have had a previous reaction to atropine.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ATROPT 1% and affect how it works. These include:
- medicines to treat glaucoma
- some medicines used to treat depression
- some antihistamines
- some medicines used to treat Parkinson's disease
- some medicines used to treat mental illness
- medicines used to treat muscle weakness
- potassium supplements.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ATROPT 1% eye drops.
4. How do I use ATROPT 1% eye drops?
How much to use
Your doctor will advise you on how much and how often you should use ATROPT 1% eye drops.
The usual dosage is one drop into the eye as required for treatment.
How to use it
Gently pull out the lower eyelid to form a sac. Allow one drop to fall into the sac without touching the eye with the dropper end of the bottle.
To minimise absorption of the drug other than into the eye, apply gentle pressure to the tear duct for approximately one minute immediately after application.
If you forget to use ATROPT 1% eye drops
Use it as soon as you remember and then return to your normal dosing time.
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much or accidently swallow ATROPT 1% eye drops
If you think that you have swallowed or used too much ATROPT 1%, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ATROPT 1% eye drops?
Things you should do
Tell your doctor if you feel that ATROPT 1% eye drops are not helping your condition.
Tell your doctor if, for any reason, you have not used your medicine as prescribed.
Otherwise your doctor may think that it was not effective and change your treatment unnecessarily.
Always discuss with your doctor any problems or difficulties during or after using this medicine.
Remind any doctor, dentist or pharmacist you visit that you are using ATROPT 1% eye drops.
Things you should not do
- Do not give ATROPT 1% to anyone else even though their symptoms seem similar to yours.
- Do not use it for other conditions unless your doctor tells you.
- Do not give this medicine to small children.
Driving or using machines
The pupil dilation may last for up to 12 days. This can make driving or using machinery difficult and possibly hazardous. While the pupil is dilated you may experience distorted vision, lack of tolerance to bright light or sunlight, and possible distortion of your balance. Special care is needed at these times.
Looking after your medicine
Keep ATROPT 1% Eye Drops in a cool dry place, protected from light where the temperature stays below 25°C.
For example, do not store it:
- In the bathroom or near a sink, or
- In the car on a hot day
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
When to discard your medicine
Discard 4 weeks after opening.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ATROPT 1% Eye Drops contains
| Active ingredient (main ingredient) | atropine sulfate monohydrate: 10 mg/mL (1%) |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What ATROPT 1% eye drops look like
ATROPT 1% eye drops are a clear or almost clear, slightly viscous, colourless sterile liquid which foams on shaking. It is odourless.
Available in a 15 mL plastic dropper bottle with a tamper proof cap.
Australian Registration number: AUST R 19648
Who distributes ATROPT 1% eye drops
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was prepared in January 2025.
Brand Information
| Brand name | Atropt 1% |
| Active ingredient | Atropine sulfate monohydrate |
| Schedule | S4 |
MIMS Revision Date: 01 September 2025
1 Name of Medicine
Atropine sulfate monohydrate.
2 Qualitative and Quantitative Composition
Atropt 1% eye drops contain atropine sulfate monohydrate (1%) in a sterile ophthalmic solution.
List of excipients with known effect. Benzalkonium chloride.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Atropt 1% eye drops are a clear or almost clear, odourless, slightly viscous, colourless liquid which foams on shaking.
4 Clinical Particulars
4.1 Therapeutic Indications
Atropt 1% eye drops are indicated where it is necessary to dilate the pupil and paralyse accommodation.
4.2 Dose and Method of Administration
Instil one drop into the eye as required for treatment. To minimise the risk of systemic absorption, gentle pressure should be applied to the tear duct for one minute after application.
4.3 Contraindications
Atropt 1% eye drops are contraindicated in the presence of angle closure glaucoma or where angle closure glaucoma is suspected. If used in glaucoma susceptible patients, an estimation of the depth of the angle of the anterior chamber should be performed prior to the initiation of therapy.
Hypersensitivity to any of the ingredients of Atropt 1% eye drops.
4.4 Special Warnings and Precautions for Use
Patients treated prior to ophthalmic examination should be escorted to and from the surgery.
Risk benefit should be considered when the following medical problems exist: keratoconus (atropine may produce fixed dilated pupil), synechiae between the iris and lens.
Use in the elderly. Geriatric patients are more susceptible to the effects of atropine, thus increasing the potential for systemic side effects.
Paediatric use. Atropt 1% must not be used in children less than 2 years old as it contains boron which may impair fertility in the future.
Atropine sulfate monohydrate should not be used in children who have previously had severe systemic reaction to atropine. An increased susceptibility to atropine has been reported in infants and young children and in children with blonde hair, blue eyes, Down's syndrome, spastic paralysis, or brain damage; therefore atropine should be used with great caution in these patients.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Anticholinergics. If significant systemic absorption of ophthalmic atropine occurs, concurrent use of other anticholinergics or medications with anticholinergic activity may result in potentiated anticholinergic effects.
Antiglaucoma agents. (Cholinergic, long acting, ophthalmic). Concurrent use with atropine may antagonise the antiglaucoma and miotic actions of ophthalmic long acting cholinergic antiglaucoma agents such as echothiophate. Concurrent use with atropine may also antagonise the anti-accommodative convergence effects of these medications when they are used for the treatment of strabismus.
Antimyasthenics, potassium citrate, potassium supplements. If significant systemic absorption of ophthalmic atropine occurs, concurrent use may increase the chance of toxicity and/or side effects of these systemic medications because of the anticholinergic induced slowing of gastrointestinal motility.
Carbachol, physostigmine or pilocarpine. Concurrent use with atropine may interfere with the antiglaucoma action of carbachol, physostigmine or pilocarpine. Also, concurrent use may counteract the mydriatic effect of atropine; this counteraction may be used to therapeutic advantage.
CNS depression producing medications. If significant absorption of systemic atropine occurs, concurrent use of medications having CNS effects, such as antiemetic agents, phenothiazines, or barbiturates, may result in opisthotonos, convulsions, coma, and extrapyramidal symptoms.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available. Excipients containing boron such as boric acid or borate compounds have been shown to cause reduced fertility and effects on embryofoetal development in animal studies and this appears to be dose related. The relevance of this to humans is uncertain.
Use in pregnancy. (Category A)
Atropine sulfate monohydrate may be systemically absorbed after ocular administration, however significant effects on the foetus have not been reported.
Use in lactation. Systemically absorbed atropine sulfate monohydrate is distributed into breast milk in very small amounts. It may cause adverse effects, such as rapid pulse, fever, or dry skin, in nursing infants of mothers using ophthalmic atropine.
4.7 Effects on Ability to Drive and Use Machines
The pupil dilation will last for up to 12 days. This can make driving or using machinery difficult and possibly hazardous. Patients should be advised not to drive or operate hazardous machinery until vision is clear.
4.8 Adverse Effects (Undesirable Effects)
The following adverse reactions have been reported.
Ophthalmic. Blurred vision, local irritation, follicular conjunctivitis, vascular congestion, oedema, exudate, contact dermatitis, eczematous dermatitis.
Systemic. Systemic atropine toxicity may be manifest as flushing and dryness of the skin, blurred vision, rapid and irregular pulse, fever, abdominal distension in infants, mental aberration and loss of neuromuscular coordination. Severe systemic reactions to atropine are characterised by hypotension with progressive respiratory depression. Ophthalmic atropine has been associated with cardiac arrhythmias (e.g. atrial fibrillation) in a few patients.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Signs of overdosage are similar to those described as systemic effects (see Section 4.8 Adverse Effects (Undesirable Effects)). Treatment is symptomatic and supportive.
For systemic effects, 0.2 to 1 mg (0.2 mg in children) physostigmine should be administered intravenously, as a dilution containing 1 mg in 5 mL of normal saline. The solution should be injected over a period of not less than 2 minutes. Dosage may be repeated every 5 minutes up to a total dose of 2 mg in children and 6 mg in adults in each 30 minute period. Physostigmine is contraindicated in hypertensive reactions. ECG monitoring is recommended during physostigmine administration.
Excitement may be controlled by diazepam or a short acting barbiturate.
It is recommended that 1 mg of atropine be available for immediate injection if the physostigmine causes bradycardia, convulsion, or bronchoconstriction.
Supportive therapy may require oxygen and assisted respiration; cool water baths for fever, especially in children; and catheterisation for urinary retention. In infants and small children, the body surface should be kept moist.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Atropine is a belladonna alkaloid. Atropine sulfate monohydrate acts in the eye to block the action of acetylcholine, relaxing the cholinergically innervated sphincter muscle of the iris. This results in dilation of the pupil (mydriasis). The cholinergic stimulation of the accommodative ciliary muscle of the lens is also blocked. This results in paralysis of accommodation (cycloplegia).
Atropine sulfate monohydrate has a slower onset and more prolonged effects than most other anticholinergics. Maximum mydriatic effect occurs in around 30 to 40 minutes. Maximum cycloplegia takes several hours. Mydriasis usually lasts 7 to 12 days and cycloplegia persists for 14 days or longer. Onset of effects and duration may be prolonged in heavily pigmented eyes.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Atropine is readily absorbed from the gastrointestinal tract; it is also readily absorbed from mucous membranes, the eye, and to some extent through intact skin.
Distribution. It is rapidly cleared from the blood and is distributed throughout the body. It crosses the blood brain barrier.
Metabolism. It is incompletely metabolised in the liver.
Excretion. It is excreted in the urine as unchanged drug and metabolites. A half-life of 4 hours has been reported.
5.3 Preclinical Safety Data
Genotoxicity. Studies have not been performed in either animals or humans to evaluate the potential mutagenic effects of atropine.
Carcinogenicity. Studies have not been performed in either animals or humans to evaluate the potential carcinogenic effects of atropine.
6 Pharmaceutical Particulars
6.1 List of Excipients
Disodium edetate, benzalkonium chloride, hypromellose, boric acid and water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
Discard 4 weeks after opening.
6.5 Nature and Contents of Container
15 mL PE-dropper bottles fitted with a PP tamper evident eye drop cap.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Atropine sulfate monohydrate exists as odourless, colourless crystals or white crystalline powder. It effloresces in dry air. It is soluble in water (1 in 0.5), in boiling water (1 in 2.5), in alcohol (1 in 5), in glycerol (1 in 2.5), and is practically insoluble in chloroform and ether. A 2% solution in water has a pH of 4.5 to 6.2. Solutions may be sterilised by autoclave. Atropine sulfate monohydrate should be protected from light.
Chemical structure. Atropine sulfate monohydrate is both a mydriatic and cycloplegic has the following chemical structure:
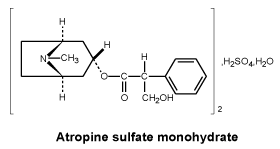
CAS number. CAS - 55-48-1 (anhydrous); CAS - 5908-99-6 (monohydrate).
7 Medicine Schedule (Poisons Standard)
Prescription only medicine: S4.
Date of First Approval
14 October 1991
Date of Revision
29 July 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.