Avaxim
Brand Information
| Brand name | Avaxim |
| Active ingredient | Hepatitis A vaccine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Avaxim.
Summary CMI
AVAXIM™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using AVAXIM?
AVAXIM contains the active ingredient Hepatitis A Virus (inactivated, adsorbed). AVAXIM is a vaccine used to help prevent Hepatitis A infection.
For more information, see Section 1. Why am I using AVAXIM? in the full CMI.
2. What should I know before I use AVAXIM?
Do not use if you have ever had an allergic reaction to AVAXIM or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use AVAXIM? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with AVAXIM and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use AVAXIM?
- AVAXIM is given as an injection into your upper arm muscle by a doctor or nurse.
- Your doctor will usually give you one injection, followed by another injection 6 to 36 months later.
More instructions can be found in Section 4. How do I use AVAXIM? in the full CMI.
5. What should I know while using AVAXIM?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using AVAXIM? in the full CMI.
6. Are there any side effects?
The more common side effects of AVAXIM include local reaction around the injection site, bruising, mild fever, headaches, unusual weakness, aching muscles, muscle tenderness or weakness (not caused by exercise), painful, swollen joints, nausea, diarrhoea, vomiting or abdominal pain, fainting, severe allergic reaction and seizures. Mostly these are mild and short-lived.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using AVAXIM?
AVAXIM contains the active ingredient Hepatitis A Virus (inactivated, adsorbed). AVAXIM is a vaccine used to help prevent Hepatitis A infection.
Hepatitis A is an infection caused by a virus which is usually transmitted in unclean food or drink. It may also be transmitted by sharing needles and some sexual practices.
The vaccine contains inactivated virus and is injected into the body. The body then produces its own protection by making disease-fighting substances (antibodies) to fight the virus. The vaccine cannot cause the infection. If a vaccinated person comes into contact with live virus the body is usually ready to destroy it. However, as with all vaccines, 100% protection against hepatitis A cannot be guaranteed. AVAXIM will not protect against hepatitis caused by other agents or viruses (such as hepatitis B, hepatitis C, or hepatitis E).
As with most vaccines, AVAXIM may not protect every person.
AVAXIM is recommended in adults and children aged 2 years and older.
2. What should I know before I use AVAXIM?
Warnings
Do not use AVAXIM if:
- you are allergic to AVAXIM, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have had a severe reaction to a previous injection of this vaccine.
- the child is under 2 years of age. The safety and efficacy of AVAXIM in these children has not been established.
Check with your doctor if you:
- have any other medical conditions that may cause lowered immunity as the vaccine may not work as well as it would in healthy individuals especially
- Lowered immunity due to diseases such as some blood disorders, leukaemia, malaria, kidney disease requiring dialysis, HIV/AIDS or cancer
- Lowered immunity due to treatment with medicines such as corticosteroids, cyclosporin or other medicines used to treat cancer (including radiation therapy) - take any medicines for any other condition.
- have allergies to neomycin, any other medicines or any other substances such as foods, preservatives or dyes.
- have ever had a serious allergic reaction to a vaccine.
- Have an illness with a high temperature or any acute illness. Your doctor may decide to delay vaccine until the illness has passed. A mild illness, such as a cold, is not usually a reason to delay vaccination.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant or intend to become pregnant.
AVAXIM is not recommended for use during pregnancy. If there is a need to consider AVAXIM during your pregnancy, your doctor will discuss with you the benefits and risks of having it.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the possible risks and benefits of having AVAXIM during breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Having other vaccines
As AVAXIM does not contain any live bacteria or viruses, it can generally be given at the same time as other inactivated vaccines, but at a different injection site.
AVAXIM can be given at the same time as yellow fever vaccine or polysaccharide typhoid vaccine at different injection sites.
Other medicines should be taken as usual after the vaccination.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect AVAXIM.
4. How do I use AVAXIM?
How AVAXIM is given
- AVAXIM is given as an injection into your upper arm muscle by a doctor or nurse.
- For some people with bleeding disorders, the dose may need to be given under the skin.
- AVAXIM should not be injected directly into the veins or into the buttocks.
- The dose is the same for adults and children, 0.5 mL of vaccine. The first injection is followed by a second injection 6 to 36 months later in order to give long-term protection. AVAXIM may be given as a second injection to those who have previously been vaccinated with another inactivated hepatitis A vaccine.
- It is important to return at the scheduled date for the second dose.
- Because hepatitis A infection can go undetected for a long period of time, it is possible that an individual may already be infected at the time the vaccine is given. The vaccine may not prevent hepatitis A in these individuals.
How much is injected
- Your doctor will usually give you one injection, followed by another injection 6 to 36 months later.
If you forget to use AVAXIM
If you miss a scheduled dose, talk to your doctor and arrange another visit as soon as possible.
If you use too much AVAXIM
Overdose is unlikely as your doctor is giving you the injection.
If you have any concerns, ask your doctor.
5. What should I know while using AVAXIM?
Things you should do
- Keep an updated record of your vaccinations.
- Attend any other appointments made by your doctor or nurse.
- Report any side effects to your doctor.
Remind any doctor, nurse, dentist or pharmacist you visit that you are using AVAXIM.
Looking after your medicine
AVAXIM is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store AVAXIM
- Keep AVAXIM in the original pack until it is time for it to be given.
- Keep it in the refrigerator, between 2°C and 8°C. Do not freeze AVAXIM. Freezing destroys the vaccine.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergy-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems in Australia or in New Zealand at pophealth.my.site.com/carmreportnz/s/. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What AVAXIM contains
| Active ingredient (main ingredient) | 160 antigen units hepatitis A virus (inactivated, adsorbed) The hepatitis A virus that this vaccine contains was grown in a cell line derived from human embryonic lung in the 1960s. |
| Other ingredients (inactive ingredients) |
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product. |
Do not take this medicine if you are allergic to any of these ingredients.
What AVAXIM looks like
Each pack of AVAXIM contains one syringe filled with inactivated Hepatitis A vaccine. (Aust R 194815).
Who distributes AVAXIM
Distributed in Australia by:
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
Freecall: 1800 818 806
Email: medinfo.australia@sanofi.com
Distributed in New Zealand by:
Pharmacy Retailing (NZ) Ltd t/a Healthcare Logistics
PO Box 62027
Sylvia Park Auckland 1644
Freecall: 0800 283 684
Email: medinfo.australia@sanofi.com
This leaflet was prepared in July 2025.
avaxim-ccdsv11-cmiv7-02jul25
Brand Information
| Brand name | Avaxim |
| Active ingredient | Hepatitis A vaccine |
| Schedule | S4 |
MIMS Revision Date: 01 July 2025
1 Name of Medicine
Hepatitis A virus (inactivated, adsorbed).
2 Qualitative and Quantitative Composition
Avaxim is a sterile suspension for injection containing formaldehyde-inactivated hepatitis A virus (GBM strain) adsorbed onto aluminium hydroxide hydrate.
Each 0.5 mL dose contains:
Active ingredient. Hepatitis A virus* 160 antigen units**.
* GBM strain cultured on MRC-5 human diploid cells. MRC-5 is a cell line that was derived from human embryonic lung tissue in the 1960s.
** In the absence of an international standardised reference, the antigen content is expressed using an in-house reference.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
Contains phenylalanine and residual neomycin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Suspension for injection.
Avaxim is a cloudy, whitish suspension.
4 Clinical Particulars
4.1 Therapeutic Indications
Avaxim is indicated for active immunisation against hepatitis A infection in adults and children 2 years and over.
Vaccination against viral hepatitis A is recommended for individuals who are or will be at increased risk of infection:
travellers to areas of moderate or high endemicity for hepatitis A;
visitors to rural and remote indigenous communities;
child daycare and preschool personnel;
the intellectually disabled and their carers;
healthcare providers;
sewerage workers;
men who have sex with men;
injecting drug users;
patients with chronic liver disease;
haemophiliacs who may receive pooled plasma concentrates.
4.2 Dose and Method of Administration
The dose is 0.5 mL for each injection. The dose is the same for adults and children.
The primary vaccination is performed with one single dose of vaccine. The booster injection can be given 6 to 36 months after the primary vaccination.
Avaxim may be used as a booster in individuals previously vaccinated with another inactivated hepatitis A vaccine.
The combined purified Vi polysaccharide typhoid and inactivated hepatitis A vaccine (Vivaxim) may be given as a booster injection 6 to 36 months after primary vaccination with Avaxim, in individuals over 16 years travelling to areas where hepatitis A and typhoid are endemic.
Avaxim may be used as a booster injection 6 to 36 months after a primary vaccination performed by the combined purified Vi polysaccharide typhoid and inactivated hepatitis A vaccine (Vivaxim) to ensure long-term protection against infection with hepatitis A virus.
As the vaccine is adsorbed, it must be injected by the intramuscular route in order to minimise local reactions. The recommended injection site is the deltoid region.
Do not administer by intradermal or intravenous injection. Ensure that the needle does not enter a blood vessel.
Shake the prefilled syringe before injection to obtain a homogenous suspension.
This vaccine must not be mixed with other vaccines in the same syringe.
The prefilled syringe is for single use only and any residue must be discarded.
4.3 Contraindications
Avaxim should not be administered to anyone with a history of severe allergic reaction to any component of the vaccine (see Section 2 Qualitative and Quantitative Composition) or after previous administration of the vaccine or vaccine containing the same components or constituents.
Vaccination must be postponed in case of febrile or acute disease.
4.4 Special Warnings and Precautions for Use
As each dose contains formaldehyde, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to this product.
As the vaccine may contain undetectable traces of neomycin, which is used during vaccine production, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to this antibiotic (and other antibiotics of the same class).
This vaccine contains polysorbate which may cause local skin reactions.
As with other injectable vaccines, appropriate medical treatment and supervision should always be available in case of anaphylactic reactions. Adrenaline should always be readily available whenever the injection is given.
Immunogenicity of the vaccine could be impaired by immunosuppressive treatment or in immunodeficiency. In such cases, it is recommended to postpone the vaccination until the end of the treatment and/or the resolution of the disease. Nevertheless, vaccination of individuals with chronic immunodeficiency such as HIV infection is recommended even if the antibody response might be limited.
Because of the incubation period of the disease, infection may be present but not clinically apparent at the time of vaccination. In this case, the vaccination may have no effect on the development of hepatitis A.
The vaccine does not provide protection against infection caused by hepatitis B virus, hepatitis C virus, hepatitis E virus or by other liver pathogens.
As with any vaccine, vaccination with Avaxim may not protect 100% of susceptible individuals.
Do not administer by intravascular injection: ensure that the needle does not penetrate a blood vessel.
As with all injectable vaccines, the vaccine must be administered with caution to individuals with thrombocytopenia or a bleeding disorder since bleeding may occur following an intramuscular administration to these individuals.
In exceptional circumstances, the vaccine may be administered by the subcutaneous route in individuals suffering from thrombocytopenia or in individuals at risk of haemorrhage.
Syncope (fainting) can occur following, or even before, any vaccination as a psychogenic response to the needle injection. Procedures should be in place to prevent falling injury and manage syncopal reactions.
Use in the elderly. Immunogenicity and clinical experience with Avaxim in the elderly is limited.
Paediatric use. Safety and effectiveness of Avaxim below the age of 2 years have not been established.
Effects on laboratory tests. Interference of Avaxim with laboratory tests has not been studied.
4.5 Interactions with Other Medicines and Other Forms of Interactions
For individuals requiring immediate and longer-term protection, such as travellers departing on short notice to endemic areas or contacts of infected individuals requiring longer term postexposure prophylaxis, Avaxim may be administered concomitantly with immunoglobulin. The vaccine may be administered concurrently with immunoglobulin providing different injection sites are used. Seroconversion rates are unaffected although antibody titres may be lower than those obtained with vaccine alone.
Information on the concomitant use of Avaxim and other vaccines is limited. There is evidence for concurrent administration of Typhim Vi (typhoid Vi polysaccharide vaccine) or live yellow fever vaccine without any interference with the immune response.
Separate injection sites and separate syringes must be used in case of concomitant administration with other medicinal products.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Avaxim has not been evaluated for the effects on fertility.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with this vaccine. Data on the use of this vaccine in pregnant women are limited. Therefore, the administration of the vaccine during pregnancy is not recommended. Avaxim should be given to pregnant women only if clearly needed, and following an assessment of the risks and benefit.
Use in lactation. It is not known whether this vaccine is excreted in human milk. Caution must be exercised when Avaxim is administered to a nursing mother.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
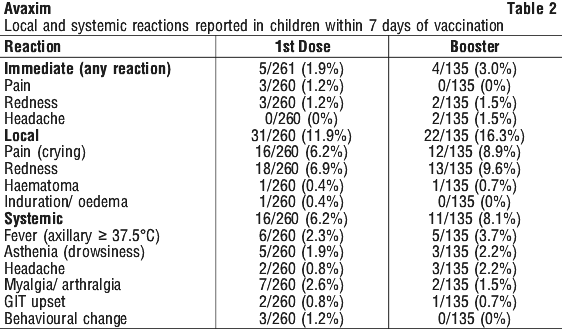
Clinical trial experience. The adverse reactions reported during clinical trials were generally mild, short-term and resolved without treatment.
Local reactions at the injection site. This was most frequently a local pain sometimes associated with erythema. The appearance of a nodule at the injection site was observed in very rare cases.
Systemic reactions. Mild fever, asthenia, headache, myalgia or arthralgia and gastrointestinal disorder were most frequently reported.
Mild reversible elevation of serum transaminase has been observed on rare occasions.
Reactions were less frequently reported after the booster injection than after the first dose.
In individuals seropositive against hepatitis A virus, this vaccine was as well tolerated as in seronegative individuals.
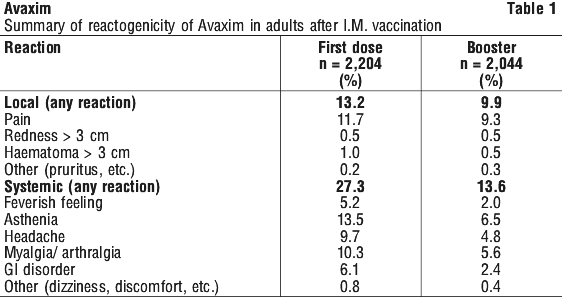
Adults. The reactogenicity of Avaxim was assessed using the same methodology in all the clinical development trials undertaken, making it possible to consolidate the results. A total of 2,204 adults received at least one dose of the final formulation of Avaxim by the intramuscular route.
Local reactions were observed in 13.2% of vaccine recipients after the first dose and 9.9% after the booster dose. General symptoms were reported in 27.3% of vaccine recipients after the first dose and 13.6% after the booster dose.
Two comparative studies compared the reactogenicity of Avaxim with a commercially available hepatitis A vaccine. The first study showed no statistical difference in the reactogenicity for seronegative patients. In the second study there was a significantly lower incidence of local reactions with Avaxim than with the comparator, both after the first and booster doses (p < 0.5). This study was randomised but not blinded. See Table 1.
Blood and lymphatic system disorder. Lymphadenopathy.
Immune system disorders. Anaphylactic reaction.
Nervous system disorders. Headache, vasovagal syncope. Convulsions with or without fever.
Gastrointestinal disorders. Nausea, diarrhoea, vomiting, abdominal pain.
Skin and subcutaneous tissue disorder. Urticaria, rashes associated or not with pruritus.
Musculoskeletal and connective tissue disorders. Arthralgia, myalgia.
General disorders and administration site condition. Injection site pain, injection site rash, injection site nodule, pyrexia, asthenia.
Investigation. Transaminases increased (mild and reversible).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Cases of administration of more than the recommended dose (overdose) have been reported with Avaxim. The adverse events reported in these cases did not differ in nature to those described in Section 4.8 Adverse Effects (Undesirable Effects).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
ATC code: J: Anti-infectives for systemic use; J07 (vaccines) B (Viral vaccines) C (Hepatitis vaccines) 02 (Hepatitis A, inactivated, whole virus).
5.1 Pharmacodynamic Properties
Mechanism of action. Hepatitis A is generally transmitted by the faecal-oral route through contaminated water or food. Blood and sexual (oral-anal relations) transmission has been demonstrated.
Avaxim confers immunity against hepatitis A virus by inducing antibodies. The measured antibody titres are greater than those obtained after passive immunisation with immunoglobulin. They are comparable to antibody titres obtained after vaccination with other inactivated hepatitis A vaccines with proven protective efficacy. The efficacy of this vaccine has not been demonstrated in field studies.
14 days after vaccination more than 90% of immunocompetent individuals are protected. One month after the first injection 100% of individuals are protected. Immunity persists for at least 36 months and is reinforced after a booster injection.
Primary immunisation should be given at least two weeks prior to anticipated exposure to the hepatitis A virus.
Data relative to long-term persistence of hepatitis A virus antibodies (anti-HAV) following vaccination with Avaxim are not currently available. Based on antibody kinetic modelling, it is predicted that anti-HAV antibody would persist for at least 10 years after the completion of the two dose vaccination schedule.
Clinical trials. The efficacy of Avaxim has been determined by the comparison of the antibody titres produced by Avaxim with that of a control hepatitis A vaccine that had previously been demonstrated to confer protective efficacy in a controlled trial of healthy children in Thailand. Seroprotection was defined as anti-HAV > 20 mIU/mL. The protective effect of Avaxim against infection with hepatitis A has not been assessed. In total 4,220 subjects received Avaxim in 9 studies in adults and 3 studies in children during the course of the clinical development program.
The pivotal studies were two multicentre, open, randomised controlled studies. In the first, 840 adults were enrolled; 420 who received Avaxim and 420 who received the control hepatitis A vaccine. In the second study 423 adults were enrolled; 212 who were inoculated with Avaxim and 211 who received the control vaccine.
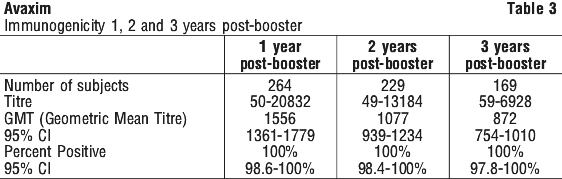
In the first pivotal study Avaxim immunogenicity was assessed at 8 weeks after the primary injection (in the subjects that were HAV seronegative (< 20 mIU/mL) at inclusion) with 99.3% of subjects achieving seropositive titres (95% CI 97.5-99.9%). The Geometric Mean Titre (GMT) was 138.4 mIU/mL (95% CI 124.5-153.9). The seroconversion rate 4 weeks post the last dose (week 28) was 100% (95% CI 99.0-100) with GMT of 4,189.6 mIU/mL (95% CI 3,792.3-4,628.6 mIU/mL). Additional analyses were made one, two and three years postbooster. All subjects tested were still seropositive. See Table 3.
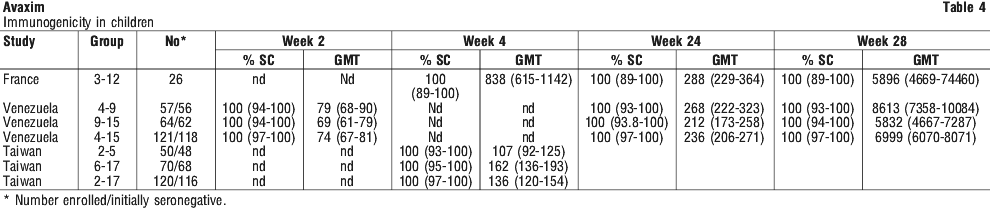
Three studies have determined the immunogenicity of Avaxim in children aged between 2 and 17 years. Study 1 was conducted in haemophiliac males in France, with the other two studies being conducted in school children in Venezuela and Taiwan. Seroprotection was defined as > 20 mIU/mL. See Table 4.
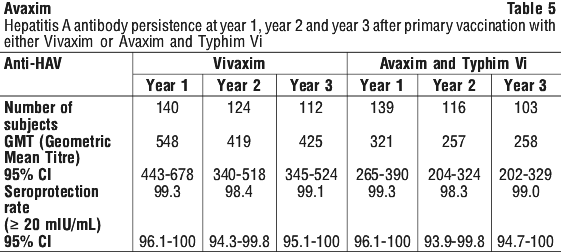
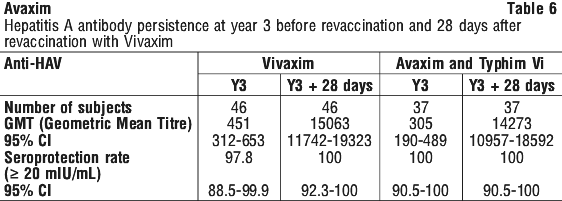
Noninferior immunogenicity of Avaxim when used as a booster following the primary dose of Vivaxim was demonstrated in a study designed to demonstrate that either of the two hepatitis A vaccines (Avaxim and Vaqta) could be used as a booster following primary vaccination with Vivaxim. This study was an open label, randomised trial which included 120 adult subjects. The results showed that one month after the booster injection, the immunogenicity elicited by vaccination with Vivaxim and booster Avaxim was noninferior to that of Avaxim and booster Avaxim in terms of GMT of anti-HAV antibody; and similarly that the immunogenicity elicited by vaccination with Vivaxim and booster Vaqta was noninferior to that of Avaxim and booster Avaxim (see Table 7).

5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. Avaxim has not been evaluated for the genotoxic potential.
Carcinogenicity. Avaxim has not been evaluated for the carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
Aluminium hydroxide hydrate (quantity expressed as aluminium) 0.3 mg, phenoxyethanol (preservative) 2.5 microL, ethanol absolute (preservative) 2.5 microL, formaldehyde (preservative) 12.5 microgram, Medium 199 (Hanks)*** up to 0.5 mL.
*** Medium 199 Hanks (without phenol red) is a complex mixture of amino acids (including phenylalanine), mineral salts, vitamins and other substances, diluted in water for injection, supplemented with polysorbate 80 and with a pH adjusted with hydrochloric acid or sodium hydroxide.
Neomycin (≤ 2.5 microgram) and bovine serum albumin (< 50 nanogram) may be present as residual traces.
6.2 Incompatibilities
This vaccine must not be mixed with other vaccines or medicinal products.
6.3 Shelf Life
36 months.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze.)
Store protected from light.
6.5 Nature and Contents of Container
Single dose prefilled syringe, 0.5 mL.
6.6 Special Precautions for Disposal
Any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Not applicable to vaccines.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of Revision
19 May 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.