Azactam
Brand Information
| Brand name | Azactam |
| Active ingredient | Aztreonam |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Azactam.
Summary CMI
AZACTAM®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using AZACTAM?
AZACTAM contains the active ingredient aztreonam. AZACTAM is used to treat serious infections caused by bacteria.
For more information, see Section 1. Why am I using AZACTAM? in the full CMI.
2. What should I know before I use AZACTAM?
Do not use if you have ever had an allergic reaction to AZACTAM or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions or allergies, take any other medicines, if you have diarrhoea or usually get diarrhoea when you take antibiotics or have ever suffered problems with your bowel, or if you are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use AZACTAM? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with AZACTAM and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use AZACTAM?
- The dose of AZACTAM will be determined by your doctor.
- AZACTAM will be administered as an injection or infusion in a hospital or clinic.
More instructions can be found in Section 4. How do I use AZACTAM? in the full CMI.
5. What should I know while using AZACTAM?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using AZACTAM? in the full CMI.
6. Are there any side effects?
Serious side effects include signs of an allergic reaction; swelling of the face, lips, tongue and/or throat with difficulty in swallowing or breathing; severe and sudden onset of pinkish, itchy swelling; blistering on the skin; ulcers in the eyes or mouth; severe persistent of bloody diarrhoea; flu-like symptoms or fever. Other side effects include itchy skin; flushing; rash; changes to bowel habits including diarrhoea, constipation, pain in the gut, indigestion or upset stomach; oral or vaginal thrush; swelling or pain at the injection site; confusion; impaired consciousness; fits or seizures; movement disorders; bleeding under the skin; dermatitis; nausea; vomiting; bloating; altered taste.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
AZACTAM® (Ay-ZAC-TAM)
Active ingredient(s): Aztreonam (azz-TREE-oh-nam)
Consumer Medicine Information (CMI)
This leaflet provides important information about using AZACTAM. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using AZACTAM.
Where to find information in this leaflet:
1. Why am I using AZACTAM?
2. What should I know before I use AZACTAM?
3. What if I am taking other medicines?
4. How am I given AZACTAM?
5. What should I know while using AZACTAM?
6. Are there any side effects?
7. Product details
1. Why am I using AZACTAM?
AZACTAM contains the active ingredient aztreonam.
AZACTAM is used for the treatment of serious infections caused by bacteria. AZACTAM may be used in the treatment of infections present in various parts of the body and sometimes AZACTAM is given with other antibiotics.
Your doctor may have prescribed AZACTAM for another use. Ask your doctor if you have any questions about why AZACTAM was prescribed for you.
AZACTAM works by killing or preventing the growth of the bacteria that is causing your infection.
2. What should I know before I use AZACTAM?
Warnings
Do not use AZACTAM if:
- you have a severe allergic reaction to AZACTAM, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have allergies to:
- any other medicines, particularly antibiotics
- any of the substances, such as foods, preservatives or dyes. - have or have had any other medical conditions, especially the following:
- You have diarrhoea or usually get diarrhoea when you take antibiotics or have ever suffered problems with your bowel. If you develop severe, prolonged or bloody diarrhoea during or after using AZACTAM. This side effect may be serious and require treatment. Do not treat with over-the-counter diarrhoea treatments, please consult your doctor.
- You have or have had liver disease.
- You have renal (kidney) impairment.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
- Check with your doctor if you are pregnant or intend to become pregnant.
- Check with your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
These medicines may be affected by AZACTAM or they may affect how well AZACTAM works.
Tell your doctor if you are taking any other medicines; especially if you are taking any of the following:
- Probenecid (medicine to treat gout)
- Frusemide (medicine that reduces fluid)
Know the medicines you take. Keep a list of your medicines with you to show your doctor or pharmacist.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect AZACTAM.
If you have not told your doctor about any of the above, tell them before you take AZACTAM.
4. How am I given AZACTAM?
How AZACTAM is given
- AZACTAM will be administered as an injection or infusion in a hospital or clinic.
- The AZACTAM injection will be prepared and administered by your doctor or nurse. You will not administer AZACTAM yourself.
- AZACTAM will be injected directly into your vein over 3-5 minutes or it will be administered as an infusion over 30 minutes.
- AZACTAM can also be administered as an injection into a large muscle, such as the buttocks or thigh.
How much to take
- The dose of AZACTAM will be determined by your doctor; your doctor will consider the severity and location of your infection, your size and your medical history.
- The dose of AZACTAM can range from 1.5g to 8g each day.
When to take AZACTAM
- AZACTAM is given over 2, 3 or 4 injections throughout a 24-hour period.
If you miss a dose of AZACTAM
- AZACTAM is administered in a hospital or clinic by a doctor or nurse. If you believe you have missed a dose inform your doctor or nurse straight away.
If you are given to much
- As your dose of AZACTAM will be determined and administered by a doctor, the chance of receiving an overdose is most unlikely. However, if an overdose should be given your doctor will give the appropriate treatment.
5. What should I know while using AZACTAM?
Things you should do
- Tell all the doctors, dentists and pharmacists who are treating you that you are taking AZACTAM.
- If you are about to be started on any new medicine, tell your doctor and pharmacist that you are taking AZACTAM.
- Tell your doctor if you notice anything that is making you feel unwell. Other side effects not listed may also occur in some patients.
Call your doctor straight away if you:
- If you notice any of the following side effects, particularly if they occur several weeks after the treatment with AZACTAM has stopped:
- severe abdominal cramps or stomach cramps
- watery and severe diarrhoea, which may also be bloody
- fever with one or both of the above symptoms
Although these side effects are rare, you may have a serious condition affecting your bowel and may need urgent medical attention. - become pregnant during AZACTAM treatment
Things you should not do
- Do not take any diarrhoea medicine without checking with your doctor first.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how AZACTAM affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
- AZACTAM will be stored in the hospital or pharmacy under the appropriate conditions.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
Allergic Reaction
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Other side effects
| Less serious side effects | What to do |
Skin
| Tell your doctor if you have any of these side effects. |
| Common side effects | What to do |
Skin
| Speak to your doctor if you have any of these side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
During treatment with AZACTAM you will require close medical supervision.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What AZACTAM contains
| Active ingredient (main ingredient) | Aztreonam |
| Other ingredients (inactive ingredients) | L-arginine |
Do not take this medicine if you are allergic to any of these ingredients.
What AZACTAM looks like
AZACTAM for is a white powder that is prepared a solution intended for injection by your doctor or nurse.
AZACTAM 1g vial (pack of 10 vials) (AUST R 14032)
Who distributes AZACTAM
Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
This leaflet was prepared in February 2026.
Brand Information
| Brand name | Azactam |
| Active ingredient | Aztreonam |
| Schedule | S4 |
MIMS Revision Date: 01 April 2026
1 Name of Medicine
Aztreonam.
2 Qualitative and Quantitative Composition
Azactam 1 g powder for injection.
Azactam for injection is sodium free and contains approximately 750 mg L-arginine per g of aztreonam. The pH of Azactam solution, depending on the type and amount of diluent used, ranges between 4.5 and 7.5.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Powder for injection. A white to off-white, free flowing sterile non-pyrogenic powder blend of aztreonam and L-arginine which upon reconstitution is intended for intravenous or intramuscular administration.
4 Clinical Particulars
4.1 Therapeutic Indications
Azactam is indicated for use in the following.
As a single agent in the treatment of infections known or strongly suspected to be due to susceptible gram negative aerobes, such as urinary tract infection, gonorrhoea, etc.
In combination with other suitable antibiotics to treat serious infections due to problem organisms known or likely to be susceptible to aztreonam*.
Meningitis caused by Haemophilus influenzae or Neisseria meningitidis. Azactam should not be used alone, but only in combination with other suitable antibiotics, in cases where meningitis is known or presumed to be due to the above organisms. Appropriate therapy should be instituted when results of sensitivity tests are known.
*Some patients with serious Pseudomonas infections may benefit from concurrent use of Azactam and an aminoglycoside because of synergistic action. However, this enhanced activity is not predictable. If such concurrent therapy is considered in patients with serious infections, susceptibility tests should be performed in vitro to determine the activity of the drugs in combination. Because of the potential nephrotoxicity and ototoxicity of aminoglycoside antibiotics, renal function should be monitored according to the aminoglycoside manufacturer's prescribing information, especially if high dosages of the aminoglycoside are to be used or if therapy is prolonged.
Before instituting treatment with Azactam, appropriate specimens should be obtained for isolation of the causative organism(s) and for determination of susceptibility to aztreonam.
Treatment with Azactam should normally be initiated on the basis of susceptibility tests, but it may be initiated in the above situations before the results of identification and sensitivity testing of the causative organism become available.
4.2 Dose and Method of Administration
Azactam for Injection may be administered intravenously or by intra-muscular injection.
Dosage and route of administration should be determined by susceptibility of the causative organisms, severity of infection, and the condition of the patient (see Table 1).

The intravenous route is recommended for patients requiring single doses greater than 1 g or those with bacterial septicaemia, localised parenchymal abscess (e.g. intra-abdominal abscess), peritonitis or other severe systemic or life threatening infections. Because of the serious nature of infections due to Pseudomonas aeruginosa, dosage of 2 g every 6 or 8 hours is recommended, at least for initial therapy in systemic infections caused by this organism. (See Section 5.1 Pharmacodynamic Properties, Mechanism of action, Microbiology.)
A single dose of 1 g Azactam administered intramuscularly is effective in the treatment of acute uncomplicated gonorrhoea and acute uncomplicated cystitis.
Renal impairment. Since aztreonam is mostly eliminated by the kidney it is recommended that after an initial loading dose of 1 to 2 g, the dose of Azactam should be reduced in patients with estimated creatinine clearances as shown in Table 2.
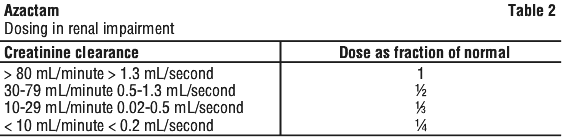
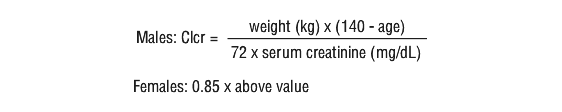
Paediatric patients. The usual dosage for patients older than one week is 30 mg/kg/dose every 8 hours. For severe infections in patients 2 years of age or older, 50 mg/kg/dose every 6 or 8 hours is recommended. Total maximum daily dose should not exceed recommended dose for adults. Dosage information is not yet available for newborns less than one week old.
Constitution and stability. Depending upon the concentration of aztreonam and diluent used, constituted Azactam for Injection yields a colourless to light straw yellow solution which may develop a slight pink tint on standing. The pH of Azactam solutions, depending on the type and amount of diluent used, ranges between 4.5 and 7.5.
Parenteral drug products should be inspected visually for particulate matter and discoloration whenever solution and container permit.
Upon the addition of the diluent the contents should be shaken immediately and vigorously. Vials of constituted Azactam for Injection are not intended for multiple dose use. Should the entire volume in the container not be used for a single dose, the unused solution must be discarded. Azactam should not be admixed with any other drugs or antibiotics. Only those diluents listed below should be used.
Intramuscular administration. Azactam for Injection should be constituted with at least 3 mL of diluent per g of aztreonam. Azactam is given by deep injection into a large muscle mass (such as the upper outer quadrant of the gluteus maximus or lateral part of the thigh). Since Azactam is well tolerated no local anaesthetic agent is required; therefore, compatibility studies have not been performed.
Azactam may be diluted with Water for Injection or Sodium Chloride Injection, or the corresponding bacteriostatic preparations containing either benzyl alcohol* or parabens as preservatives.
*Diluents containing benzyl alcohol are not suitable for use in the newborn.
Intravenous administration. For bolus injection. The selected dose should be constituted with 6 to 10 mL Water for Injection BP, and the resulting solution slowly injected directly into the vein over a period of 3 to 5 minutes.
For infusion. Each g of aztreonam supplied in 15 mL vials should be initially constituted with at least 3 mL of Water for Injection BP provides 1 g of aztreonam in a total volume of approximately 4 mL. The resulting initial solution should be diluted with an appropriate infusion solution to a final concentration not exceeding 2% w/v (at least 50 mL solution per g aztreonam).
The Azactam infusion should be administered over a 30 minute period.
With intermittent infusion of Azactam and another drug via a common delivery tube, the tube should be flushed before and after delivery of Azactam with any appropriate infusion solution compatible with both drug solutions. The drugs should not be delivered simultaneously.
A volume control administration set may be used to deliver the initial solution of Azactam for Injection into a compatible infusion solution being administered. With use of a Y-tube administration set, careful attention should be given to the calculated volume of Azactam solution required so that the entire dose will be infused.
The following intravenous solutions may be used as diluents for the administration of Azactam for Injection by intravenous infusion: water for injection, sodium chloride injection, Ringer's injection, lactated Ringer's injection, glucose injection (5%), glucose injection (10%), glucose (5%) with sodium chloride (0.9%) injection, glucose (5%) with sodium chloride (0.45%) injection, glucose (5%) with sodium chloride (0.2%) injection, sodium lactate (M/6 sodium lactate), Ionosol B and 5% glucose, Isolyte E, Isolyte E with 5% glucose, Isolyte M with 5% glucose, Normosol-R, Normosol R and 5% glucose, Normosol (R) M and 5% glucose, 10% Travert injection, mannitol injection (5%), mannitol injection (10%), lactated Ringer's and 5% glucose injection, Plasma-Lyte M and 5% glucose injection, 10% Travert in Electrolyte No. 1 Injection, 10% Travert in Electrolyte No. 2 Injection, 10% Travert in Electrolyte No. 3 Injection.
4.3 Contraindications
Azactam is contraindicated in patients with known hypersensitivity to aztreonam or any other component in the formulation.
4.4 Special Warnings and Precautions for Use
As with other drugs inquiry should be made regarding a history of hypersensitivity reactions.
Antibiotics, like other drugs, should be given with caution to any patient with a history of allergic reaction to structurally related compounds. If an allergic reaction occurs, discontinue the drug and institute supportive treatment as appropriate. Serious hypersensitivity reactions may require adrenaline and other emergency measures.
Antibiotic associated pseudomembranous colitis has been reported with many antibiotics including Azactam. A toxin produced with Clostridium difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with antibiotic use (this may occur up to several weeks after cessation of antibiotic therapy). Mild cases usually respond to drug discontinuation alone. However, in moderate to severe cases appropriate therapy such as oral antibiotic agents effective against Clostridium difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated.
Drugs which delay peristalsis, e.g. opiates and diphenoxylate with atropine (Lomotil) may prolong and/or worsen the condition and should not be used.
Azactam is not indicated for the treatment of gynaecological infections or for other sites where aerobic gram negative organisms are not the common infective agents, but may be used if the infection can be shown to be due to susceptible gram negative organisms only.
Experience with patients with impaired hepatic function is limited. Appropriate monitoring of liver function in such patients is recommended during therapy.
Therapy with Azactam may result in overgrowth of nonsusceptible organisms which may require therapy.
Use of beta-lactam containing therapies, including aztreonam, can cause encephalopathy (e.g. confusion, impairment of consciousness, epilepsy, movement disorders); particularly in patients with renal impairment and in association with beta-lactam overdose.
Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in patients taking beta-lactam antibiotics. When SCAR is suspected, beta-lactam antibiotics should be discontinued immediately and an alternative treatment should be considered.
Use in hepatic impairment. A dose reduction is recommended for long-term treatment of patients with alcoholic cirrhosis, especially in cases when renal function is also impaired.
Use in renal impairment. No data available.
Use in the elderly. No data available.
Paediatric use. Data on safety and effectiveness in neonates younger than one week are limited; use in this population needs to be carefully assessed. (See Section 4.2 Dose and Method of Administration.)
Azactam contains arginine. Studies in low birth weight infants have demonstrated that arginine administered in the Azactam formulation may result in increases in serum arginine, insulin and indirect bilirubin. The consequences of exposure to this amino acid during treatment of neonates have not been fully ascertained.
Effects on laboratory tests. A positive direct or indirect Coombs test may develop during treatment with Azactam. See Section 4.8 Adverse Effects (Undesirable Effects), Haematological.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant administration of probenecid or frusemide and Azactam cause clinically insignificant increases in the serum levels of aztreonam. Single dose pharmacokinetic studies have not shown any significant interaction between aztreonam and gentamicin, nafcillin sodium, cephradine, clindamycin or metronidazole. No reports of disulfiram-like reactions with alcohol ingestion have been noted.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B1)
Aztreonam crosses the placenta and enters the foetal circulation.
Since studies in pregnant women have not been done, Azactam should be used during pregnancy only if clearly needed.
Use in lactation. Studies in lactating women have shown that aztreonam is excreted in breast milk in concentrations that are less than 1% of concentrations determined in simultaneously obtained maternal serum, consideration should be given to temporary discontinuation of nursing during treatment with Azactam.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Azactam is generally well tolerated.
In clinical studies, adverse effects were infrequent with less than 2% of patients having therapy discontinued. Effects considered related or of uncertain relationship to Azactam therapy are:
Hypersensitivity. Anaphylaxis, angioedema, bronchospasm.
Dermatological. Rash, pruritus, petechiae, purpura, diaphoresis, flushing, urticaria, erythema multiforme, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), acute generalised exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS) and exfoliative dermatitis.
Haematological. Eosinophilia, increases in prothrombin and partial thromboplastin time, thrombocytosis, thrombocytopenia, leukocytosis, neutropenia, anaemia, pancytopenia, bleeding and positive Coombs Test have occurred rarely.
Hepatobiliary. Elevations of hepatic transaminases and alkaline phosphatase levels usually reversing during therapy and usually without overt signs or symptoms of hepatobiliary dysfunction. Clinical diagnosis of jaundice and hepatitis were reported rarely.
Gastrointestinal. Diarrhoea, nausea and/or vomiting, abdominal cramps, mouth ulcer and altered taste. Abdominal distension has been noted in children.
Rare cases of C. difficile associated diarrhoea, including pseudomembranous colitis, or gastro-intestinal bleeding have occurred.
Renal. Aztreonam was not associated with changes in renal function in healthy subjects. Renal function was monitored using standard tests (serum creatinine, creatinine clearance, BUN, urinalysis and total urinary protein excretion) as well as special tests (excretion of N-acetyl-B-glucosaminidase, alanine aminopeptidase and B2-microglobulin).
Local reactions. Discomfort at the IV injection site and phlebitis/ thrombophlebitis; mild discomfort was noted at IM injection site.
Nervous system disorders. Encephalopathy (e.g. confusion, impairment of consciousness, epilepsy, movement disorders). (See Section 4.4 Special Warnings and Precautions for Use.)
Miscellaneous. Rare instances of the following reactions have been reported. Vaginitis, vaginal candidiasis, hypotension, seizure, diplopia, weakness, paraesthesia, confusion, dizziness, vertigo, insomnia, ECG changes, tinnitus, headache, breast tenderness, halitosis, altered taste, muscle aches, fever, malaise, sneezing and nasal congestion, wheezing, dyspnoea and chest pain. Transient increase in serum creatinine were uncommon.
Reporting suspected adverse events. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
If necessary, aztreonam may be cleared from the serum by haemodialysis and/or peritoneal dialysis. Aztreonam has been shown to be cleared from the serum by continuous arteriovenous hemofiltration.
Use of beta-lactam containing therapies, including aztreonam, can cause encephalopathy (e.g. confusion, impairment of consciousness, epilepsy, movement disorders); particularly in patients with renal impairment and in association with beta-lactam overdose.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Microbiology. Aztreonam exhibits bactericidal activity against a number of gram-negative aerobes. The bactericidal action of aztreonam results from the inhibition of bacterial cell wall synthesis due to its binding to Penicillin Binding Protein 3 (PBP3). Aztreonam is resistant to hydrolysis by many beta-lactamases (i.e. penicillinases and cephalosporinases) produced by gram-negative and gram-positive pathogens. In vitro resistance to aztreonam can be induced by repeated passage through antibiotic containing media in the same manner as with other beta-lactam antibiotics.
Aztreonam, unlike the majority of beta-lactam antibiotics, is usually not an inducer of beta-lactamase activity. Aztreonam is active in vitro against most strains of the following susceptible organisms:
Escherichia coli; Enterobacter species; Klebsiella species, including K. pneumoniae and K. oxytoca (except those producing K-1 type beta-lactamase); Proteus mirabilis; Proteus vulgaris; Morganella morganii (formerly Proteus morganii); Providencia species, including P. stuartii and P. rettgeri (formerly Proteus rettgeri); Serratia marcescens; Neisseria gonorrhoeae (including penicillinase producing strains); Haemophilus influenzae (including ampicillin resistant and other penicillinase producing strains); Citrobacter species; Pseudomonas aeruginosa*.
*Pseudomonas aeruginosa strains usually are either sensitive or have intermediate sensitivity (see Susceptibility testing) to aztreonam. Other Pseudomonas species are usually resistant.
Aztreonam and aminoglycosides are synergistic in vitro against many of the strains of P. aeruginosa. However, such synergy is not always predictable.
Due to the induction of beta-lactamases, certain antibiotics (e.g. cefoxitin, imipenem) have been found to cause antagonism with many beta-lactams, including aztreonam, for certain gram negative aerobes, such as Enterobacter species and Pseudomonas species.
Alterations of normal flora in the body by antibiotics permit overgrowth of potential pathogens, e.g. Candida and Clostridium species. Unlike broad spectrum antibiotics, aztreonam produces no effects on the normal intestinal anaerobic microflora. Clostridium difficile and its cytotoxin were not found in animal models following administration of aztreonam.
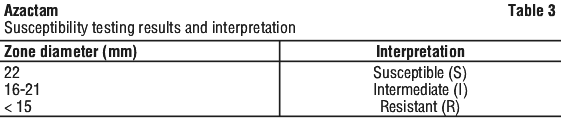
Susceptibility testing. Diffusion technique. Quantitative procedures that require measurement of zone diameters give a precise estimate of antibiotic susceptibility. One such method, recommended for use with the aztreonam 30 microgram disc, is the National Committee of Clinical Laboratory Standards (NCCLS) approved procedure.
Results in Table 3 of laboratory tests using 30 microgram aztreonam discs should be interpreted using the following criteria.
Dilution technique. Broth or agar dilution methods may be used to determine the minimal inhibitory concentration (MIC) of aztreonam. See Table 4.

Clinical trials. No data available.
5.2 Pharmacokinetic Properties
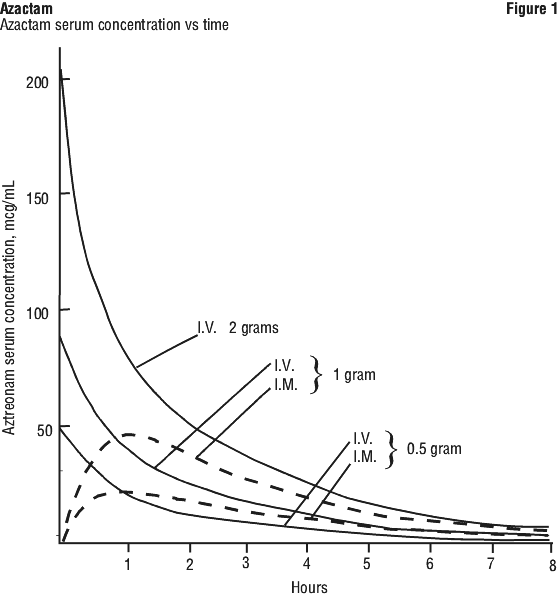
Absorption. Azactam is not intended for oral administration as it is not absorbed from the gastrointestinal tract. Single 30 minute intravenous infusions of 0.5, 1 and 2 g doses of Azactam in healthy volunteers produced serum levels of 54, 90 and 204 microgram/mL, respectively, immediately after administration (see Figure 1). Single 3-minute intravenous injections of the same doses resulted in peak serum levels of 58, 125 and 242 microgram/mL. Serum levels of aztreonam 8 hours after 3 or 30 minute infusions were 1, 3 and 6 microgram/mL.
Distribution. Aztreonam achieves measurable concentrations in the following body fluids and tissues (see Table 5).
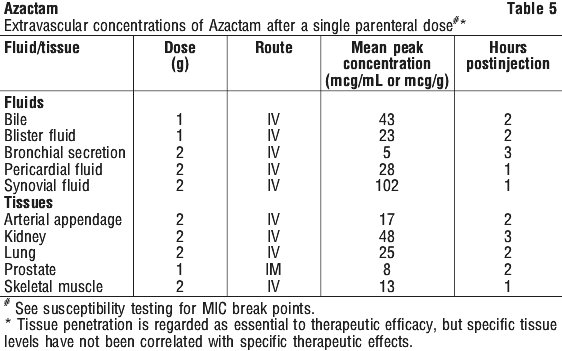
Metabolism. Serum protein binding averaged 56% and was independent of dose. An average of about 6% of a 1 g intramuscular dose was excreted as a microbiologically inactive open beta-lactam ring hydrolysis product of aztreonam in the zero to 8 hour urine collection on the last day of multiple dosing.
Excretion. After single 0.5, 1 and 2 g intravenous doses of Azactam (30 minute infusion) average urine concentrations of aztreonam were approximately 1100, 3500 and 6600 microgram/mL, respectively, within the first two hours. After intramuscular injection of a single 0.5 or 1 g dose of Azactam urinary levels were approximately 500 and 1200 microgram/mL respectively within the first two hours, declining to 180 and 470 microgram/mL in the 6 to 8 hour specimens.
In healthy subjects approximately 60 to 70% of the intravenous or intramuscular dose administered was recovered in the urine by 8 hours. Urinary excretion of a single parenteral dose was essentially complete by 12 hours after injection. About 12% of a single radio-labelled dose was recovered in the faeces; both unchanged aztreonam and the inactive beta-lactam ring hydrolysis product of aztreonam were present.
In patients with impaired renal function the serum half-life of aztreonam is prolonged. Aztreonam is cleared from the serum by haemodialysis, approx., 38% being removed in 4 hours.
Intravenous or intramuscular administration of a single 0.5 or 1 g dose of Azactam every 8 hours for 7 days to healthy subjects produced no apparent accumulation of aztreonam or modification of its disposition characteristics. The range of average concentrations for aztreonam in the 8 to 12 hour urine specimens in the above studies was 25 to 120 microgram/mL. The serum half-life of aztreonam averaged 1.7 hours (1.5 to 2.0) in subjects with normal renal function, independent of the dose and route of administration. In subjects over 65 years of age and a mean creatinine clearance of 40 mL/minute the serum half-life was 8.5 hours. The half-life of the metabolite of aztreonam is approximately 15-20 hours. In healthy subjects, based on a 70 kg person, the serum clearance was 91 mL/minute and renal clearance was 56 mL/minute; the apparent mean volume of distribution at steady-state averaged 12.6 litres, approximately equivalent to extra-cellular fluid volume.
Since the liver is a minor pathway of excretion, the serum half-life of aztreonam is only slightly prolonged in patients with hepatic impairment.
Pharmacokinetics (paediatrics). The pharmacokinetics of Azactam in paediatric patients are dependent on age and body weight. Data obtained after single doses for various patient subgroups are listed in Table 6.
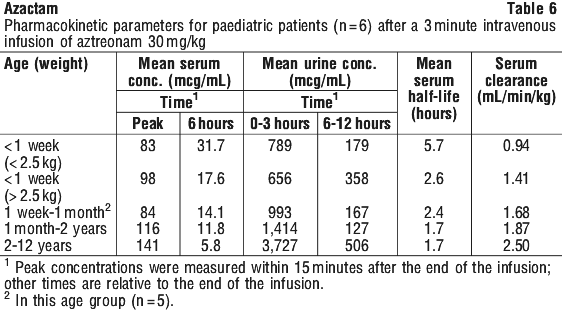
Studies in vitro demonstrated that aztreonam, at concentrations up to 660 microgram/mL, did not displace bilirubin from albumin, either in a purified bilirubin-albumin solution or in hyperbilirubinemic neonatal serum.
5.3 Preclinical Safety Data
Genotoxicity. Aztreonam produced no mutagenic changes in several standard laboratory models.
Studies in pregnant rats and rabbits disclosed no clear evidence of embryo toxicity, foetotoxicity, or teratogenicity. In rats given aztreonam during late gestation and lactation no drug-induced changes were seen in any of the maternal, foetal, or neonatal parameters that were monitored.
Carcinogenicity. Carcinogenicity studies in animals have not been performed.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
With intermittent infusion of Azactam and another drug via a common delivery tube, the tube should be flushed before and after delivery of Azactam with any appropriate infusion solution compatible with both drug solutions. The drugs should not be delivered simultaneously.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Powder for injection. Store below 30°C.
Solutions prepared for intramuscular injection must be used within 48 hours if kept below 25°C.
Solutions prepared for IV use should be used immediately.
6.5 Nature and Contents of Container
Glass vials containing 1 g in packs of 10.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Azactam (aztreonam) is a totally synthetic monocyclic beta lactam belonging to a new class of antibiotics, the monobactams. Chemically, aztreonam is designated as: (Z)-2-((((2-amino-4-thiazolyl)-(((2S,3S)-2-methyl-4-oxo-1-sulfo-3-azetidinyl) carbamoyl)methylene)-amino)oxy)-2-methylpropionic acid.
CAS number. 78110-38-0.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
05 September 1991
Date of Revision
11 February 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.