Azarga
Brand Information
| Brand name | Azarga |
| Active ingredient | Brinzolamide + Timolol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Azarga.
Summary CMI
Azarga®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Azarga?
Azarga contains the active ingredient Brinzolamide and timolol maleate. Azarga is used to treat certain types of eye conditions such as high pressure inside your eye and open angle glaucoma (an eye condition caused by fluid buildup that damages the optic nerve).
For more information, see Section 1. Why am I using Azarga? in the full CMI.
2. What should I know before I use Azarga?
Do not use if you have ever had an allergic reaction to Azarga or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Azarga? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Azarga and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Azarga?
- Follow the instructions provided and use Azarga until your doctor tells you to stop.
- Put one drop of Azarga in the conjunctival sac of the affected eye(s) twice daily.
More instructions can be found in Section 4. How do I use Azarga? in the full CMI.
5. What should I know while using Azarga?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Azarga? in the full CMI.
6. Are there any side effects?
Common side effects include temporary blurred vision, eye pain, bitter or unusual taste in the mouth after using Azarga, dry eye, eye irritation, vision problems, eyelid swelling.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Azarga® Eye Drops
Active ingredients: Brinzolamide and timolol maleate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Azarga. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Azarga.
Where to find information in this leaflet:
1. Why am I using Azarga?
2. What should I know before I use Azarga?
3. What if I am taking other medicines?
4. How do I use Azarga?
5. What should I know while using Azarga?
6. Are there any side effects?
7. Product details
1. Why am I using Azarga?
Azarga contains the active ingredients Brinzolamide and timolol maleate. Brinzolamide belongs to class of drugs called carbonic anhydrase inhibitors and timolol maleate belongs to class of drugs called beta-blockers.
Azarga is used to treat certain types of eye conditions such as high pressure inside your eye and open angle glaucoma (an eye condition caused by fluid buildup that damages the optic nerve).
The active ingredients of Azarga eye drops help in reducing the production of fluid within the eye.
2. What should I know before I use Azarga?
Warnings
Do not use Azarga:
- If you are allergic to Brinzolamide and other sulphonamides, timolol maleate, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Symptoms of an allergic reaction may include shortness of breath, difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, hives. - In children under 18 years old.
- If you suffer from any respiratory problems such as asthma (symptoms such as wheezing, coughing, shortness of breath) or a history of asthma, or shortness of breath, persistent cough or any symptoms such as runny nose, sneezing, itchy, watery eyes.
- If you have any heart problems such as problems with your heart beat, problems with heart function, any history of heart attack or any other heart diseases.
- If you are allergic to any other drugs similar to timolol maleate (other beta blockers).
- If you suffer from symptoms such as headache, lack of energy, vomiting and feeling sick.
- If you have any kidney problems.
Check with your doctor if you:
- take any medicines for any other condition
- have any skin rash, problems with your skin
- have any problems with your blood vessels, having symptoms such as skin colour changes, affected part feels cold or numb, skin ulcers.
- have diabetes or problems with low blood sugar levels (symptoms like sweating, confusion, shakiness)
- suffer from problems with your thyroid gland
- have any problems with your blood circulation or if you have low blood pressure, symptoms include dizziness, blurred vision, fatigue
- have a history of allergic problems (symptoms such as hives, shortness of breath, difficulty in breathing)
- have any other type of Glaucoma
- have any problems with your cornea.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Your doctor will advise you regarding the possible risks and benefits of using Azarga during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will advise you regarding the possible risks and benefits of using Azarga during breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Azarga and affect how it works.
Tell your doctor if you use any of the following medicines:
- Drugs used to treat high blood pressure or heart problems, e.g. beta-blockers (Metoprolol), calcium channel blockers or digitalis glycosides or Pilocarpine (parasympathomimetics)
- Digoxin, a medicine used to treat heart failure
- Medicines used to treat an irregular heartbeat e.g. amiodarone and quinidine
- Nonsteroidal anti-inflammatory drugs (NSAIDs) eg Ibuprofen, Aspirin and salicylates or carbonic anhydrase inhibitors such as acetazolamide
- Antifungal medicines such as ketoconazole, itraconazole, clotrimazole
- Ritonavir, a medicine used to treat HIV
- Cimetidine, a medicine used to treat stomach or intestine ulcers
- Clonidine, a medicine used to treat high blood pressure and migraines
- Any other medicines or eye drops used for treatment of glaucoma
- Medicine used to treat severe allergic reaction e.g. adrenaline
- Medicines used to treat high blood sugar.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Azarga.
4. How do I use Azarga?
How much to use
- Follow the instructions provided and use Azarga until your doctor tells you to stop.
- Put one drop of Azarga in the conjunctival sac of the affected eye(s) twice daily.
- Do not exceed the dose of one drop in the affected eye(s) twice daily.
When to use Azarga
- Use Azarga eye drops at about the same time each day, unless your doctor tells you otherwise.
How to use Azarga
- In a sitting or lying down position might make the process simpler.
- Remove contact lenses if you are wearing them before using the eye drops.
- Shake the bottle well prior to use.
- Follow the steps below to use Azarga:
- Wash your hands thoroughly with soap and water.
- Before using a bottle for the first time, break the safety seal around the neck area and throw the loose plastic ring away.
- Remove the cap from the bottle.
- After the cap is removed, if tamper evident snap collar is loose, this should be removed before using the product.
- Mix the contents of the bottle by inverting 5 to 10 times.
- Hold the bottle upside down in one hand between your thumb and middle finger (refer diagram 1).
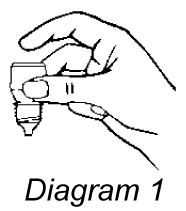
- Tilt your head back, gently pull down the lower eyelid of your eye to form a pouch/pocket.
- Place the tip of the bottle close to your eye. Do not touch the tip to your eye as this may cause injury to your eye.
- Do not touch the dropper tip to your eyelid or to surrounding areas or to any surface to avoid contamination of the dropper tip and solution.
- Release one drop into the pouch/pocket formed between your eye and eyelid by gently tapping or pressing the base of the bottle with your forefinger (refer diagrams 2 and 3).
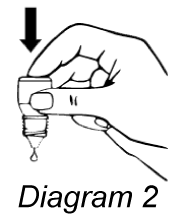
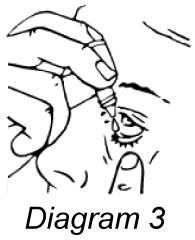
- Gently close your eye(s) for 2 minutes. Do not blink or rub your eye(s).
- When your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to prevent the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body and may result in less side effects.
- This will also help to prevent the unpleasant taste sensation that some people experience while using these eye drops.
- If necessary, repeat the above steps for the other eye.
- You may feel a slight burning sensation in the eye shortly after using Azarga. If it continues, or is uncomfortable, contact your doctor or pharmacist.
- If you want to use any other eye drops wait at least 5 minutes after using Azarga.
- It is normal for a small amount of eye drops to spill onto your cheek since your eyelids can only hold less than one drop at a time. Wipe away any spillage with a tissue.
- Replace the cap on the bottle and close it tightly.
- Always keep the bottle tightly closed when not in use.
- Wash your hands again with soap and water to remove any residue.
- Wait for 15 minutes before putting your contact lenses in.
- Discard Azarga eye drops 4 weeks after opening.
If you forget to use Azarga
Azarga should be used regularly at the same time each day. If you miss your dose at the usual time, treatment should be continued with the next dose as planned.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Azarga
If you think that you have used too much Azarga, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you accidentally put several Azarga Eye Drops in your eyes, immediately wash your eyes with warm tap water.
5. What should I know while using Azarga?
Things you should do
- Tell your doctor immediately if you are going to have any surgery.
- Use Azarga for eye use only.
- Remove soft contact lenses before putting in Azarga eye drops. Benzalkonium chloride one of the ingredients in Azarga may cause eye irritation and is known to discolour soft contact lenses.
- Put your lenses back in 15 minutes after using the eye drops.
Call your doctor straight away if you:
- develop any signs of allergic reaction.
- become pregnant, are planning to become pregnant or if you are breast feeding while using Azarga.
- suffer from any heart or breathing problems.
- develop a skin rash, hives or other skin problems while on treatment with Azarga.
Remind any doctor, dentist, pharmacist or specialist you visit that you are using Azarga.
Things you should not do
- Do not stop using this medicine suddenly.
- Do not use Azarga in children.
- Do not give Azarga to anyone else, even if they seem to have the same condition as you.
- Do not touch the dropper tip to any surface.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Azarga affects you.
Azarga may impair the ability to perform tasks requiring mental alertness and/or physical coordination. It may also cause temporary blurred vision or other visual problems in some people.
If blurred vision occurs when you use Azarga, wait until your vision clears before driving or using machinery.
Looking after your medicine
- Store below 25°C
- Do not freeze.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard Azarga 4 weeks after opening.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Eye Problems:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Eye Problems:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Azarga contains
| Active ingredient (main ingredient) | Brinzolamide and timolol maleate. |
| Other ingredients (inactive ingredients) | Mannitol, carbomer 974P, sodium chloride, tyloxapol, disodium edetate, sodium hydroxide and/or hydrochloric acid (for pH adjustment) purified water and benzalkonium chloride. |
| Potential allergens | Benzoates, sulfites and hydroxybenzoates. |
Do not take this medicine if you are allergic to any of these ingredients.
What Azarga looks like
Azarga eye drops is a white to off-white uniform suspension and is presented in an 8 mL round opaque low density polyethylene bottle dispenser containing 5 mL suspension.
Australian registration number
AUST R 156500.
Who distributes Azarga
This product is supplied in Australia by:
Novartis Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1-800-671-203
Web site: www.novartis.com.au
This product is supplied in New Zealand by:
Novartis New Zealand Limited
PO Box 99102
Newmarket
Auckland 1149
New Zealand
Free Phone: 0800 354 335.
® Registered trademark
This leaflet was prepared in August 2025.
Internal document code
aza271123c_V2 based on PI aza271123i
Brand Information
| Brand name | Azarga |
| Active ingredient | Brinzolamide + Timolol |
| Schedule | S4 |
MIMS Revision Date: 01 January 2024
1 Name of Medicine
Brinzolamide and timolol maleate.
2 Qualitative and Quantitative Composition
The eye drops suspension contains 10 mg/1 mL (1%) brinzolamide and 5 mg/1 mL (0.5%) timolol (as timolol maleate) and also 0.1 mg/1 mL benzalkonium chloride as a preservative. The pH of the eye drops suspension is approximately 7.2.
Excipient with known effect. Benzoates, sulfites and hydroxybenzoates.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Eye drops.
Azarga is a white to off-white uniform suspension.
4 Clinical Particulars
4.1 Therapeutic Indications
Decrease of intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension for whom monotherapy with either component provides insufficient IOP reduction.
4.2 Dose and Method of Administration
The recommended dosage is one drop of Azarga in the conjunctival sac of the affected eye(s) twice daily. Patient should be instructed to shake the bottle well before use.
Nasolacrimal occlusion and closing the eyelid for two minutes, after instillation are recommended. This may reduce the systemic absorption of eye drops, decrease in systemic adverse reactions and an increase in local activity.
If more than one topical ophthalmic medicine is being used, the medicines must be administered at least 5 minutes apart.
If a dose is missed, treatment should be continued with the next dose as planned. The dose should not exceed one drop in the affected eye(s) twice daily.
When substituting another ophthalmic antiglaucoma agent with Azarga, the other agent should be discontinued and Azarga should be started the following day.
To avoid contamination, the dropper tip should not touch any surface. The dropper tip should also not come into contact with the eye as this may cause injury to the eye. Patient should be instructed to keep the bottle tightly closed when not in use.
Patients must be instructed to remove soft contact lenses prior to application of Azarga and to wait fifteen minutes after instillation of the dose before reinsertion.
After cap is removed, if tamper evident snap collar is loose, this should be removed before using the product.
4.3 Contraindications
A history of hypersensitivity to brinzolamide and other sulphonamides, timolol, or any other component of the medication.
The following conditions may also contraindicate the use of Azarga:
reactive airway disease including bronchial asthma, a history of bronchial asthma, or severe chronic obstructive pulmonary disease;
hypersensitivity to other beta-blockers;
sinus bradycardia, second or third degree atrioventricular block, overt cardiac failure, cardiogenic shock, sino-atrial block, sick sinus syndrome;
severe allergic rhinitis and bronchial hyperreactivity;
hyperchloraemic acidosis;
severe renal impairment (see Section 4.4 Special Warnings and Precautions for Use, Use in hepatic impairment, Use in renal impairment).
Azarga is not recommended for use in children below 18 years due to a lack of data on safety and efficacy.
4.4 Special Warnings and Precautions for Use
For topical use only - not for injection or oral ingestion.
Azarga should be discontinued at the first appearance of a skin rash or any other sign of hypersensitivity.
Systemic effects. Like other topically applied ophthalmic agents, brinzolamide and timolol are absorbed systemically. Systemic absorption can be minimised by nasolacrimal occlusion or closing the eyelids for two minutes (see Section 4.2 Dose and Method of Administration). This may result in a decrease in systemic side effects and an increase in local activity.
Brinzolamide. Azarga contains brinzolamide, a sulphonamide. The same types of undesirable effects that are attributable to sulphonamides may occur with topical administration. Hypersensitivity reactions reported with sulphonamide derivatives, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), can occur in patients receiving Azarga as it is absorbed systemically. At the time of prescription, patients should be advised of the signs and symptoms and monitored closely for skin reactions. If signs of serious reactions or hypersensitivity occur, discontinue use of this medicine immediately.
Acid-base disturbances have been reported with oral carbonic anhydrase inhibitors. Azarga should be used with caution in patients with risk of renal impairment because of the possible risk of metabolic acidosis.
There is potential for an additive effect on the known systemic effects of carbonic anhydrase inhibition in patients receiving an oral carbonic anhydrase inhibitor and Azarga. The concomitant administration of Azarga and oral carbonic anhydrase inhibitors has not been studied and is not recommended.
Timolol. Cardiovascular safety. Due to the beta-adrenergic component, timolol, the same types of cardiovascular and pulmonary adverse reactions as seen with systemic beta-adrenergic blocking agents may occur. Cardiac failure should be adequately controlled before beginning therapy with timolol. Patients with a history of severe cardiac disease should be watched for signs of cardiac failure and have their pulse rates checked.
In patients with cardiovascular diseases (e.g. coronary heart disease, Prinzmetal's angina and cardiac failure) and hypotension, therapy with beta-blockers should be critically assessed and the therapy with other active substances should be considered. Patients with cardiovascular diseases should be watched for signs of deterioration of these diseases and of adverse reactions.
Vascular disorders. Patients with severe peripheral circulatory disturbance/ disorders (i.e. severe forms of Raynaud's disease or Raynaud's syndrome) should be treated with caution.
Respiratory reactions. Respiratory reactions and cardiac reactions, including death due to bronchospasm in patients with asthma and, rarely, death in association with cardiac failure, have been reported following administration of timolol maleate.
Diabetes mellitus. Beta-adrenergic blocking agents should be administered with caution in patients subject to spontaneous hypoglycaemia or to patients with labile insulin-dependent diabetes, as beta-adrenergic blocking agents may mask the signs and symptoms of acute hypoglycaemia.
Thyrotoxicosis. Beta-adrenergic blocking agents may mask the signs of hyperthyroidism.
Muscle weakness. Beta-adrenergic blocking agents have been reported to potentiate muscle weakness consistent with certain myasthenic symptoms (e.g. diplopia, ptosis and generalized weakness).
Beta-adrenergic blocking agents may also cause worsening of Prinzmetal angina, severe peripheral and central circulatory disorders and hypotension.
Anaphylactic reactions. While taking beta-adrenergic blocking agents, patients with a history of atopy or a history of severe anaphylactic reaction to a variety of allergens may be more reactive to repeated challenge with such allergens and unresponsive to the usual doses of adrenaline (epinephrine) used to treat anaphylactic reactions.
Choroidal detachment. Choroidal detachment has been reported with administration of aqueous suppressant therapy (e.g. timolol, acetazolamide) after filtration procedures.
Surgical anaesthesia. Beta-blocking ophthalmological preparations may block systemic beta-agonist effects, e.g. of adrenaline (epinephrine). The anaesthesiologist should be informed when the patient is receiving timolol.
Ocular effects. Azarga has not been studied in patients with narrow-angle glaucoma and its use is not recommended in these patients.
There is limited experience with Azarga in the treatment of patients with pseudoexfoliative glaucoma or pigmentary glaucoma. Caution should be utilized in treating these patients and close monitoring of IOP is recommended.
The possible role of brinzolamide on corneal endothelial function has not been investigated in patients with compromised corneas (particularly in patients with low endothelial cell count). Specifically, patients wearing contact lenses have not been studied and careful monitoring of these patients when using brinzolamide is recommended, since carbonic anhydrase inhibitors may affect corneal hydration and wearing contact lenses might increase the risk for the cornea. The use of carbonic anhydrase inhibitors can also lead to corneal decompensation and oedema. Careful monitoring of patients with compromised corneas, such as patients with diabetes mellitus or corneal dystrophies, is recommended.
Use with contact lenses. Azarga contains benzalkonium chloride which may cause eye irritation and is known to discolour soft contact lenses. Contact with soft contact lenses is to be avoided. Patients must be instructed to remove contact lenses prior to the application of Azarga and wait 15 minutes after instillation of the dose before reinsertion.
Use in hepatic impairment. No studies have been conducted with Azarga in patients with hepatic impairment. No dosage adjustment is necessary in patients with hepatic impairment.
Use in renal impairment. No studies have been conducted with Azarga in patients with renal impairment. No dosage adjustment is necessary in patients with mild to moderate renal impairment.
Azarga has not been studied in patients with severe renal impairment (creatinine clearance < 30 mL/min) or in patients with hyperchloraemic acidosis. Since brinzolamide and its main metabolite are excreted predominantly by the kidney, Azarga is therefore contraindicated in patients with severe renal impairment.
Use in the elderly. There are no modifications to the recommended dosing regimen for elderly patients.
Paediatric use. Azarga is not recommended for use in children below 18 years due to a lack of data on safety and efficacy.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No drug interaction studies have been performed with Azarga.
Brinzolamide. Azarga contains brinzolamide, a carbonic anhydrase inhibitor and, although administered topically, it's absorbed systemically. Acid-base disturbances have been reported with oral carbonic anhydrase inhibitors. The potential for interactions (e.g. nonsteroidal anti-inflammatory drugs (NSAIDs) and salicylates) must be considered in patients receiving Azarga.
There is a potential for an additive effect on the known systemic effects of carbonic anhydrase inhibition in patients treated with an oral carbonic anhydrase inhibitor and brinzolamide eye drops. The concomitant administration of eye drops containing brinzolamide and oral carbonic anhydrase inhibitors is not recommended.
The cytochrome P450 isozymes responsible for metabolism of brinzolamide include CYP3A4 (main), CYP2A6, CYP2B6, CYP2C8 and CYP2C9. It is expected that inhibitors of CYP3A4 such as ketoconazole, itraconazole, clotrimazole and ritonavir will inhibit the metabolism of brinzolamide by CYP3A4. Caution is advised if CYP3A4 inhibitors are given concomitantly. However, accumulation of brinzolamide is unlikely as renal elimination is the major route. Brinzolamide is not an inhibitor of cytochrome P450 isozymes.
Timolol. There is a potential for additive effects resulting in hypotension and/or marked bradycardia when eye drops with timolol are administered concomitantly with oral calcium channel blockers or beta-adrenergic blocking agents, antiarrhythmics (including amiodarone), digitalis glycosides or parasympathomimetics. The use of two local beta-adrenergic blocking agents or two local carbonic anhydrase inhibitors is not recommended.
The hypertensive reaction to sudden withdrawal of clonidine can be potentiated when taking beta-adrenergic blocking agents.
Potential systemic beta-blockade (e.g. decreased heart rate) has been reported during combined treatment with CYP2D6 inhibitors (e.g. quinidine, cimetidine) and timolol.
Beta-adrenergic blocking agents may increase the hypoglycaemic effect of antidiabetic agents. Beta-adrenergic blocking agents can mask the signs and symptoms of hypoglycaemia.
Beta blockers can decrease the response to adrenaline (epinephrine) used to treat anaphylactic reactions. Special caution should be exercised in patients with a history of atopy or anaphylaxis (see Section 4.4 Special Warnings and Precautions for Use, Anaphylactic reactions).
Mydriasis resulting from concomitant use of ophthalmic beta-blockers and adrenaline (epinephrine) has been reported occasionally.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no human data on the effects of Azarga on male or female fertility. Studies in rats, in which animals were treated orally with brinzolamide up to 18 mg/kg/day or with timolol up to 100 mg/kg/day, showed no adverse effects on male or female fertility.
Use in pregnancy. (Category C)
No studies have been conducted with Azarga in pregnant women, and no animal studies have been conducted with the combined components to evaluate effects on reproduction. Studies in animals with brinzolamide have shown reproductive toxicity following systemic administration. Azarga should only be used during pregnancy if the potential benefit justifies the potential risk to the foetus. However, if Azarga is administered until delivery, the neonate should be carefully monitored during the first days of life.
Brinzolamide. Developmental toxicity studies with brinzolamide in rabbits at oral doses up to 6 mg/kg/day produced maternal toxicity at 6 mg/kg/day and a significant increase in the number of foetal variations, e.g. accessory skull bones; at 1 and 6 mg/kg/day, the incidence was only slightly higher than seen historically. In rats, statistically significant decreased bodyweights of foetuses from dams receiving oral doses of 18 mg/kg/day during gestation were proportional to the reduced maternal weight gain, with no statistically significant effects on organ or tissue development. No treatment related malformations were seen. Exposure levels are much lower following topical administration of brinzolamide. There are no adequate data from the use of brinzolamide in pregnant women. The potential risk for humans is unknown.
Timolol. Timolol maleate was not teratogenic in mice, rats and rabbits. Embryo-foetal development studies with timolol maleate in mice and rabbits showed no evidence of embryo-foetal toxicity at oral doses up to 50 mg/kg/day. At higher doses, increases in resorptions and foetal variations (14 ribs and hypoplastic sternebrae) in mice (1000 mg/kg/day) and increased resorption in rabbits (≥ 90 mg/kg/day) were observed. In rats, delayed ossification was seen at oral doses ≥ 50 mg/kg/day, and decreased number of caudal vertebral bodies and arches, and an increase in hypoplastic sternebrae were noted at 500 mg/kg/day. In humans, well-controlled epidemiological studies with systemic use of beta-adrenergic blocking agents did not indicate malformative effects, but show a risk of intrauterine growth retardation. In addition, signs and symptoms of beta-blockade (e.g. bradycardia, hypotension, respiratory distress and hypoglycaemia) have been observed in foetuses or neonates when beta-blockers have been administered until delivery. Data on a limited number of exposed pregnancies indicate no adverse effects of timolol in eye drops on pregnancy or on the health of the foetus/ newborn child but bradycardia and arrhythmia have been reported in one case in the foetus of a woman treated with timolol eye drops. To date, no other relevant epidemiological data are available.
Use in lactation. It is not known whether brinzolamide is transferred into human milk following topical ocular administration. Timolol is detectable in human milk following topical ocular administration.
Studies in animals have shown that following oral administration brinzolamide is excreted in breast milk. Following oral administration of 14C-brinzolamide to lactating rats, radioactivity was found in milk at concentrations below those in the blood and plasma. Decreases in pup bodyweights were observed at 15 mg/kg/day in a prenatal and postnatal study in which rats were given brinzolamide by oral gavage at doses up to 15 mg/kg/day.
Because of the potential for serious adverse reactions in breastfed infants from brinzolamide and timolol, a decision should be made whether to discontinue breastfeeding or to discontinue Azarga, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
However, as with any eye drops, temporary blurred vision or other visual disturbances may affect the ability to drive or use machines. If blurred vision occurs at instillation, the patient must wait until the vision clears before driving or using machinery. Carbonic anhydrase inhibitors may impair the ability to perform tasks requiring mental alertness and/or physical coordination.
4.8 Adverse Effects (Undesirable Effects)
In two clinical trials of 6 and 12 months duration involving 394 patients treated with Azarga, the most frequently reported adverse reaction was transient blurred vision upon instillation (3.6%), lasting from a few seconds to a few minutes.
The following adverse reactions were assessed to be treatment-related. Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
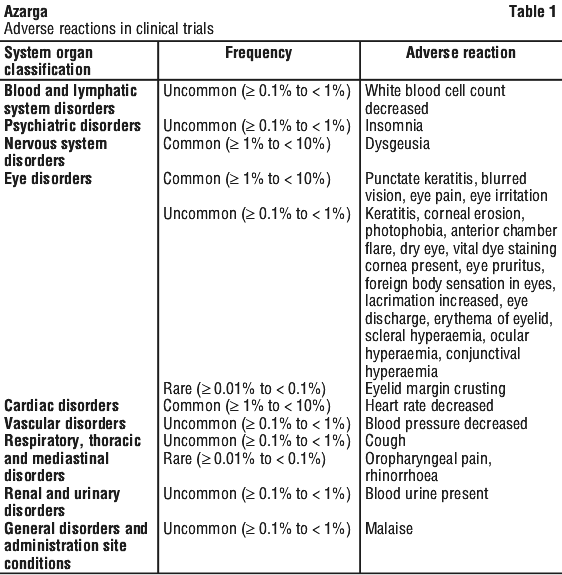
Psychiatric disorders. Uncommon (≥ 0.1% to < 1%): insomnia.
Nervous system disorders. Common (≥ 1% to < 10%): dysgeusia.
Dysgeusia (bitter or unusual taste in the mouth following instillation) was a frequently reported systemic undesirable effect associated with the use of Azarga during clinical studies. It is probably caused by passage of the eye drops in the nasopharynx via the nasolacrimal canal. Nasolacrimal occlusion and gently closing the eyelid after instillation may help reduce the incidence of this effect.
Eye disorders. Common (≥ 1% to < 10%): blurred vision, eye pain, eye irritation, foreign body sensation in eyes.
Uncommon (≥ 0.1% to < 1%): corneal erosion, punctate keratitis, dry eye, eye discharge, eye pruritus, ocular hyperaemia, blepharitis, allergic conjunctivitis, corneal disorder, anterior chamber flare, conjunctival hyperaemia, eyelid margin crusting, asthenopia, abnormal sensation in eye, eyelids pruritus, allergic blepharitis, erythema of eyelid, photophobia, lacrimation increased, sclera hyperaemia.
Vascular disorders. Uncommon (≥ 0.1% to < 1%): decreased blood pressure.
Respiratory, thoracic and mediastinal disorders. Uncommon (≥ 0.1% to < 1%): chronic obstructive pulmonary disease, pharyngolaryngeal pain, rhinorrhoea, cough.
Skin and subcutaneous tissue disorders. Uncommon (≥ 0.1% to < 1%): hair disorder, lichen planus.
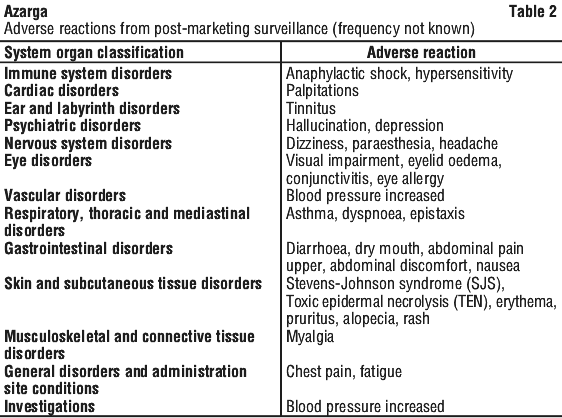
Postmarketing experience. The following adverse reactions are classified according to the following convention: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness. These adverse reactions were obtained from clinical trials. See Table 1.
Frequencies cannot be estimated from the available data. See Table 2.
4.9 Overdose
No case of overdose has been reported. A topical overdose of Azarga may be flushed from the eye(s) with warm tap water. If an overdose with Azarga occurs, treatment should be symptomatic and supportive. In case of accidental ingestion, symptoms of overdose from beta blockade may include bradycardia, hypotension, cardiac failure and bronchospasm.
Due to brinzolamide, electrolyte imbalance, development of an acidotic state, and possibly central nervous system effects may occur. Serum electrolyte levels (particularly potassium) and blood pH levels should be monitored. Studies have shown that timolol does not dialyse readily.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Azarga contains two active substances: brinzolamide and timolol maleate. These two components decrease elevated intraocular pressure (IOP) primarily by reducing aqueous humour secretion, but do so by different mechanisms of action. Clinical data demonstrates that the combined effect of these two active substances results in additional IOP reduction compared to either compound alone.
Brinzolamide exhibits a high affinity for and is a potent inhibitor of human carbonic anhydrase II (CA-II). Carbonic anhydrase exists as a number of isoenzymes and is found in many tissues in the body; CA-II is the predominant iso-enzyme in the eye. Inhibition of CA-II in the ciliary processes of the eye decreases aqueous humour secretion, presumably by slowing the formation of bicarbonate ions with subsequent reduction in sodium and fluid transport. Brinzolamide exhibited minimal cardiovascular effects, including no inducement of QTc prolongation and no or minimal effect on blood pressure and heart rate, and had no significant local anaesthetic (membrane stabilising) activity on the cornea.
Timolol is a non-selective beta-adrenergic receptor blocking agent that has no significant intrinsic sympathomimetic, direct myocardial depressant or local anaesthetic (membrane stabilising) activity. The precise mechanism of action of timolol in lowering IOP is not clearly established at this time, although tonography and fluorophotometry studies suggest that its predominant action is related to reduced aqueous humour formation; a slight increase in outflow facility was also observed in some studies.
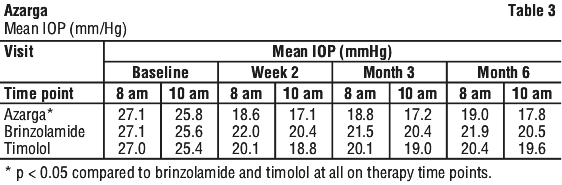
Clinical trials. In a twelve-month, double-masked, randomised clinical trial in patients (n = 437) with open angle glaucoma or ocular hypertension who, in the investigator's opinion, could benefit from combination therapy and who had baseline mean IOP of 25 to 27 mmHg, the mean IOP-lowering effect of Azarga was 7 to 9 mmHg and for dorzolamide 20 mg/mL + timolol 5 mg/mL it was 7 to 9 mmHg, when dosed twice daily. The non-inferiority of Azarga as compared to dorzolamide 20 mg/mL + timolol 5 mg/mL in the mean IOP reduction was demonstrated across all on-therapy time-points at all visits. When evaluated at each visit, up to 60% of patients in the Azarga group and up to 59% of patients in the dorzolamide 20 mg/mL group had IOP of less than 18 mmHg.
In a six-month, double-masked, randomised clinical study in patients (n = 523) with open-angle glaucoma or ocular hypertension and baseline mean IOP of 25 to 27 mmHg, the mean IOP-lowering effect of Azarga dosed twice daily was 8 to 9 mmHg, and was up to 3 mmHg greater than that of brinzolamide 10 mg/mL dosed twice daily and up to 2 mmHg greater than that of timolol 5 mg/mL dosed twice daily. A statistically superior reduction (p < 0.05) in mean IOP was observed compared to both brinzolamide and timolol at all on-therapy time-points and visits throughout the study. IOP measurements conducted at 8 am, 10 am, 12 pm, 4 pm and 8 pm confirm that diurnal IOP control is superior (p < 0.05) and clinically relevant for Azarga compared to either brinzolamide 10 mg/mL or timolol 5 mg/mL. The primary efficacy endpoint of mean IOP at 8 am and 10 am post dose for this study can be seen in Table 3.
5.2 Pharmacokinetic Properties
Absorption. Following topical ocular administration, brinzolamide and timolol are absorbed through the cornea and into the systemic circulation. In a pharmacokinetic study, healthy subjects received oral brinzolamide (1 mg) twice daily for 2 weeks to shorten the time to reach steady-state prior to starting Azarga administration. Following twice daily dosing of Azarga for 13 weeks, the concentrations of brinzolamide in erythrocytes at weeks 4, 10 and 15, averaged 18.8 ± 3.29 microM, 18.1 ± 2.68 microM and 18.4 ± 3.01 microM respectively. Steady-state concentrations of brinzolamide in erythrocytes may not bear a simple relationship to plasma concentrations of the drug, which have been observed to be low and generally below assay quantitation limits (< 10 nanogram/mL) following topical ocular administration in other studies.
At steady state, following administration of Azarga, the mean plasma Cmax and AUC0-12h of timolol were 27% and 28% lower (Cmax: 0.824 ± 0.453 nanogram/mL; AUC0-12h: 4.71 ± 4.29 nanogram.h/mL), respectively in comparison to the administration of timolol 5 mg/mL (Cmax: 1.13 ± 0.494 nanogram/mL; AUC0-12h: 6.58 ± 3.18 nanogram.h/mL). The lower systemic exposure to timolol following Azarga administration is not clinically relevant. Following administration of Azarga, mean Cmax of timolol was reached at 0.79 ± 0.45 hours.
Distribution. Plasma protein binding of brinzolamide is moderate (about 60%). Brinzolamide is sequestered in red blood cells (RBCs) due to its high affinity binding to CA-II and to a lesser extent to CA-I. Its active N-desethyl metabolite also accumulates in RBCs where it binds primarily to CA-I. The affinity of brinzolamide and metabolite to RBCs and tissue carbonic anhydrase results in low plasma concentrations.
Ocular tissue distribution data in rabbits showed that timolol can be measured in aqueous humour up to 48 hours after administration of Azarga. At steady-state, timolol is detected in human plasma for up to 12 hours after administration of Azarga.
Metabolism. The metabolic pathways for brinzolamide involve N-dealkylation, O-dealkylations and oxidation of its N-propyl side chain. N-desethyl brinzolamide is a major metabolite of brinzolamide formed in humans, which also binds to CA-I in the presence of brinzolamide and accumulates in RBCs. In vitro studies show that the metabolism of brinzolamide mainly involves CYP3A4 as well as at least four other isozymes (CYP2A6, CYP2B6, CYP2C8 and CYP2C9).
Timolol is metabolised by two pathways. One route yields an ethanolamine side chain on the thiadiazole ring and the other gives an ethanolic side chain on the morpholine nitrogen and a second similar side chain with a carbonyl group adjacent to the nitrogen. Timolol metabolism is mediated primarily by CYP2D6.
Excretion. Brinzolamide is eliminated primarily by renal excretion (approximately 60%). About 20% of the dose has been accounted for in urine as metabolite. Brinzolamide and N-desethyl-brinzolamide are the predominant components found in the urine along with trace levels (< 1%) of the N-desmethoxypropyl and O-desmethyl metabolites.
Timolol and its metabolites are primarily excreted by the kidneys. Approximately 20% of a timolol dose is excreted in the urine unchanged and the remainder excreted in urine as metabolites. The plasma t½ of timolol is 4.8 hours after administration of Azarga.
5.3 Preclinical Safety Data
Genotoxicity. Brinzolamide did not display mutagenic potential in bacteria (Ames test) or produce chromosomal damage in vivo (mouse micronucleus test). Brinzolamide did induce forward mutations in the mouse lymphoma assay in vitro in the presence, but not in the absence, of metabolic activation. Brinzolamide was negative in a sister chromatid exchange assay in mice. Timolol was not genotoxic in assays for mutagenicity (Ames test) and clastogenicity (mouse micronucleus and cytogenic assays).
Carcinogenicity. No carcinogenicity studies have been conducted with the combined components of Azarga.
Brinzolamide. A 2-year bioassay, in with rats were treated with brinzolamide by oral gavage at doses up to 8 mg/kg/day, revealed no evidence of a carcinogenic effect. A similar study conducted in mice, involving oral dosing at 0, 1, 3 or 10 mg/kg/day for 2 years, revealed a statistically significant increase in urinary bladder tumours in females at 10 mg/kg/day, and dose related proliferative changes in the urinary bladder in females at all dose levels and among males at 10 mg/kg/day. The elevated bladder tumour incidence was primarily due to the increased incidence of a tumour and was considered to be unique to mice.
Timolol. No evidence of carcinogenicity was observed with timolol maleate at oral doses up to 100 mg/kg/day in rats and up to 50 mg/kg/day in mice. However, there was a statistically significant increase in the incidence of adrenal pheochromocytomas in male rats administered 300 mg/kg/day. In female mice, statistically significant increases in the incidence of benign and malignant pulmonary tumours, benign uterine polyps and mammary adenocarcinomas were found at 500 mg/kg/day. The increased occurrence of mammary adenocarcinomas was associated with elevations in serum prolactin, which occurred in female mice administered oral timolol at 500 mg/kg/day, but not at doses of 50 mg/kg/day. An increased incidence of mammary adenocarcinomas in rodents has been associated with administration of several other therapeutic agents that elevate serum prolactin.
6 Pharmaceutical Particulars
6.1 List of Excipients
Mannitol, carbomer 974P, sodium chloride, tyloxapol, disodium edetate, sodium hydroxide and/or hydrochloric acid (for pH adjustment) purified water and benzalkonium chloride.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
Discard container 4 weeks after opening.
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Refrigerate. Do not freeze.
6.5 Nature and Contents of Container
Azarga is presented in an 8 mL round opaque low density polyethylene bottle dispenser containing 5 mL suspension.
Consumer Medicine Information is supplied with this product.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Brinzolamide is a white to off-white, crystalline powder which is very slightly soluble in water at neutral pH.
Timolol maleate is a white to off-white, crystalline powder which is soluble in water, alcohol and practically insoluble in ether.
Chemical structure. The chemical structure of each active ingredient is represented below:
Brinzolamide.
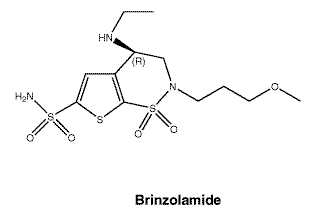
Empirical formula: C12H21N3O5S3.
Molecular weight: 383.51.
Timolol maleate.
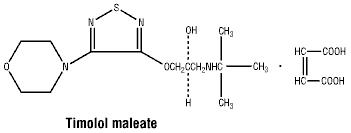
Empirical formula: C13H24N4O3S.C4H4O4.
Molecular weight: 432.50.
CAS number. Brinzolamide: 138890-62-7.
Timolol maleate: 26921-17-5.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine.
Date of First Approval
05 February 2010
Date of Revision
27 November 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.