Biological Therapies B-Dose Forte 2.5 mL Injection
Brand Information
| Brand name | Biological Therapies B-Dose Forte 2.5 mL Injection |
| Active ingredient | Thiamine hydrochloride + Riboflavine sodium phosphate + Nicotinamide + Dexpanthenol + Pyridoxine hydrochloride + Cyanocobalamin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Biological Therapies B-Dose Forte 2.5 mL Injection.
Summary CMI
Biological Therapies B-Dose Forte 2.5 mL Injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given this medicine?
This medicine contains the vitamins B1, B2, B3, B5, B6 and B12 as the following active ingredients: Thiamine hydrochloride (B1), Riboflavine sodium phosphate (B2), Nicotinamide (B3), Dexpanthenol (B5), Pyridoxine hydrochloride (B6), Cyanocobalamin (B12). It is used for the prevention and treatment of specific vitamin B deficiencies. For more information, see Section 1. Why am I being given this medicine? in the full CMI.
2. What should I know before I am given this medicine?
Do not use if you have ever had an allergic reaction to this medicine or any of the ingredients listed at the end of the CMI. You should not be given this medicine if you are pregnant and are diagnosed with megaloblastic anaemia, if you are a haemophiliac or if you have mechanical intestinal obstruction (ileus).
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given this medicine? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with B-Dose Forte 2.5 mL Injection and affect how it works. You should not be given B-Dose Forte Injection if you are taking high dose vitamin B vitamins.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is this medicine given to me?
This medicine is given as an injection, intramuscularly (into the buttock muscle). This medicine must only be given by a doctor or nurse. The amount given depends on many factors including your body weight and medical condition. The usual dose given is 2.5 mL weekly for 2 to 6 weeks.
More instructions can be found in Section 4. How is this medicine given to me? in the full CMI.
5. What should I know while being given this medicine?
| Things you must do |
|
| Things you must not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given this medicine? in the full CMI.
6. Are there any side effects?
The more common side effects of this medicine are skin irritation, pain or bruising the injection site, mild nausea or vomiting, headaches, numbness in hands or feet, hives, heartburn, fatigue, or sore throat. Serious side effects include severe allergic reactions, sneezing, severe rash, temporary itchiness, blue discolouration of the skin, bleeding, prolonged stomach pain, severe dizziness or drowsiness, muscular paralysis, low blood pressure, prolonged nausea or vomiting, restlessness, scaling of the facial skin and inability to focus eyes.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Biological Therapies B-Dose Forte 2.5 mL Injection
Active ingredient(s): Thiamine hydrochloride (B1), Riboflavine sodium phosphate (B2), Nicotinamide (B3), Dexpanthenol (B5), Pyridoxine hydrochloride (B6), Cyanocobalamin (B12).
Consumer Medicine Information (CMI)
This leaflet provides important information about using Biological Therapies B-Dose Forte 2.5 mL Injection. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using B-Dose Forte 2.5 mL Injection.
Where to find information in this leaflet:
1. Why am I being given this medicine?
2. What should I know before I am given this medicine?
3. What if I am taking other medicines?
4. How is this medicine given to me?
5. What should I know while being given this medicine?
6. Are there any side effects?
7. Product details
1. Why am I being given this medicine?
B-Dose Forte 2.5 mL Injection contains the Vitamins B1, B2, B3, B5, B6 and B12 as the following active ingredients:
B1 - Thiamine hydrochloride,
B2 - Riboflavine sodium phosphate,
B3 - Nicotinamide, B5 - Dexpanthenol,
B6 - Pyridoxine hydrochloride, B12 - Cyanocobalamin.
These B vitamins are required by the body for normal metabolic processes in your cells, including the production of energy. Different metabolic processes need several of these B vitamins at the same time. Deficiencies of one, some or all of these vitamins in your cells can have serious consequences for your health.
Vitamin B deficiencies:
Vitamin B deficiencies may occur as a result of a diet deficient in B vitamins, malnutrition, or changes to the gastrointestinal tract which slow or prevent the absorption of B vitamins (e.g. abdominal surgery or some diseases of the gastrointestinal tract).
A specific deficiency of vitamin B1 (thiamine) may cause the disease beriberi.
A specific deficiency of vitamin B3 (niacin) may cause the disease pellagra.
A specific deficiency of vitamin B12 (cobalamin) may cause the disease pernicious anaemia.
Some of the B vitamins need other B vitamins to work properly. Your doctor may prescribe B-Dose Forte Injection to treat specific vitamin B deficiencies, or to raise the levels of B vitamins in your body.
When administration by mouth is not feasible or appropriate:
Your doctor may decide that B-Dose Forte Injection is the best way for you to take B vitamins.
Debilitated or elderly patients where the diet is inadequate:
Confinement to bed, debility or old age may lead to an inadequate diet. In these cases, vitamin B deficiencies may occur. Your doctor may prescribe B-Dose Forte to supplement B vitamins missing in your diet.
Malnutrition resulting from alcoholism:
Alcoholism can lead to a diet deficient in B vitamins. The normal metabolism of alcohol in your liver also requires B vitamins, especially thiamine. Thiamine may be prescribed to treat Wernicke's encephalopathy, a disorder caused by thiamine deficiency in alcoholism.
B-Dose Forte Injection contains a high dose of thiamine and may be prescribed by your doctor to help the effects of alcoholism.
Peripheral neuritis and Carpal tunnel syndrome:
Peripheral neuritis means nerve pain, usually in the arms or hands, legs or feet (the peripheral parts). Many of the B vitamins are important for the normal functioning of the nervous system. Long-term alcoholism, beriberi, pellagra or long-term gastrointestinal disease may lead to B vitamin deficiencies and peripheral nerve pain.
Carpal tunnel syndrome and its symptoms may be relieved by the administration of vitamin B6 (pyridoxine). Carpal tunnel syndrome symptoms include pain in the hands and wrists, weakness of the hands and loss of grip strength, and coldness of the hands and fingers.
Your doctor may prescribe B-Dose Forte Injection to treat peripheral nerve pain or carpal tunnel syndrome.
Pernicious anaemia:
Anaemia occurs when red blood cells cannot carry sufficient oxygen to meet the requirements of the body's cells. This may be due to a deficiency of oxygen, lack of ability of red blood cells to bind oxygen, or some defect of red blood cells. Vitamin B12 is needed to make red blood cells grow correctly, without it the red blood cells become large and less effective. This is a type of anaemia called megaloblastic (large cells) anaemia. Megaloblastic anaemia may also be caused by a folate (folic acid) deficiency. The “pernicious” part is because prolonged
Vitamin B12 deficit can also lead to damage to the nervous system. The combination of nervous system damage and anaemia is called Pernicious Anaemia.
Symptoms of pernicious anaemia include tiredness, breathlessness, lack of energy and different sensations of the nervous system, such as pins and needles and loss of strength.
B-Dose may be prescribed to help with the symptoms of pernicious anaemia, or for megaloblastic anaemia if Vitamin B12 deficiency is the diagnosed cause.
Your doctor may have prescribed B-Dose Forte Injection for another reason.
Ask your doctor if you have any questions about why B-Dose Forte Injection has been prescribed for you.
B-Dose Forte Injection is not addictive.
2. What should I know before I am given this medicine?
Warnings
You must not be given this medicine if:
- You are allergic to B vitamins, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- You must not be given this medicine if you are taking high dose B vitamins. This medicine should not be given to you if you have high levels of B vitamins in your body from other sources, such as high dose vitamin supplements.
- You must not be given this medicine if you a haemophiliac. Dexpanthenol, the form of vitamin B5 used in B-Dose Forte Injection should not be used if you have haemophilia.
- You must not be given this medicine if you have mechanical intestinal obstruction (ileus)
Dexpanthenol, the form of vitamin B5 used in B-Dose Forte Injection is often used to treat ileus (lack of forward movement of intestinal contents), but should not be used if you have ileus due to a mechanical intestinal obstruction (something stuck in there). - You must not be given this medicine if you are pregnant and are diagnosed with megaloblastic anaemia. Cobalamins can mask the effects of megaloblastic anaemia caused by folate deficiency. Folate deficiency in pregnancy is a risk for birth defects in your baby. You must have Vitamin B12 deficiency confirmed by blood tests before using B-Dose Forte Injection to treat megaloblastic anaemia.
- You must not be given this medicine if the solution in the vial is not clear or contains particles.
- If you are not sure whether you should be given this medicine, talk to your doctor.
Check with your doctor if you:
- Are pregnant and are diagnosed with megaloblastic anaemia.
- Have any other medical conditions.
- Take any medicines for any other condition.
- Have had an allergy to any of the ingredients listed at the end of this leaflet. Your doctor may test if you have an allergy to this medicine by injecting a small amount under your skin.
- Have allergies to any other medicines or any other substances, such as foods, preservatives or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
There is an increased need for some B vitamins during pregnancy. It is recommended that you and your doctor discuss your requirements during pregnancy and the possible risks and benefits of having this medicine during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
There is an increased need for some B vitamins during breastfeeding. It is recommended that you and your doctor discuss your requirements during breastfeeding and the possible risks and benefits of having this medicine during breastfeeding.
If you have not told your doctor about any of the above, tell them before you are given B-Dose Forte Injection.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with B-Dose Forte Injection and affect how it works.
These include:
- Vitamin and Mineral Supplements
- Levodopa (a medicine used to treat Parkinson's disease)
- Oestrogen containing medicines, including oral contraceptives (the pill)
- Immunosuppressants, such as azathioprine
- Some antiepileptic drugs such as phenobarbitone and phenytoin
- Hydralazine (a medicine to treat high blood pressure)
- Some drugs used to treat infections, such as isoniazid, penicillamine, cycloserine and pyrazinamide.
These medicines may be affected by B-Dose Forte Injection, or affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information about medicines to be careful with, or avoid while you are being treated with this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect B-Dose Forte 2.5 mL Injection.
4. How is this medicine given to me?
This medicine must only be given by a doctor or nurse.
B-Dose Forte Injection will be injected intramuscularly (into the buttock muscle) by your doctor.
How much is given
Your doctor will tell you how much B-Dose Forte Injection will need to be given and for how long it will need to be given. This is determined by many factors including your body weight and your medical condition. The usual dose is 2.5 mL by slow intramuscular injection.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
How long is the treatment time with this medicine
Each person will respond differently to B-Dose Forte Injection.
Treatment times will differ depending on the reason for prescribing this medicine.
Typically, treatment will be once each week for 2-6 weeks. Your doctor will let you know how long you will need to be treated with this medicine.
If you miss an appointment
If you miss an appointment, talk to your doctor and arrange another appointment as soon as possible.
If you are not sure what to do, contact your doctor or pharmacist as soon as possible.
If too much of this medicine is given
Your doctor should be the only person to inject B-Dose Forte Injection, so an overdose is unlikely to occur.
If you think that you or anyone else may have been given too much B-Dose Forte Injection you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while being given this medicine?
Things you must do
- If you about to be started on any new medicine, remind your doctor and pharmacist that you are being treated with B-Dose Forte Injection.
- Tell all doctors, dentists and pharmacists who treat you that you are being given this medicine.
- If you are about to have any urine or blood tests tell your doctor that you are being treated with this medicine. It may interfere with the results of some tests.
- Tell your doctor if you become pregnant while being treated with B-Dose Forte Injection.
- Tell your doctor if you feel that giving B-Dose Forte Injection is not helping your condition.
- Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you must not do
- Do not attempt to inject this medicine yourself.
- Do not take any other medicines, whether they require a prescription or not, without first telling your doctor or consulting a pharmacist.
- Do not use this medicine if the packaging is torn or shows signs of tampering.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how B-Dose Forte Injection affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcoholism can lead to a diet deficient in B vitamins. The normal metabolism of alcohol in your liver also requires B vitamins.
Looking after your medicine
B-Dose Forte 2.5 mL Injection is usually stored in the doctor's surgery or clinic, or at the pharmacy.
If you need to store B-Dose Forte 2.5 mL Injection keep it in the original pack until it is time for it to be given. If you take the vials out of the pack they may not keep well.
Store it in a refrigerator (store at 2°C to 8°C). Do not freeze. Protect from light.
Keep it where young children cannot reach it.
When to discard your medicine
Each vial is for SINGLE USE in one patient on one occasion only. It will be used once only and then it will be discarded. It must never be stored after it is opened or used for more than one person.
Getting rid of any unwanted medicine
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any vials that are left over.
Do not use this medicine after the expiry date. If you are given this medicine after the expiry date has passed, it may not work and it may be harmful to you. If it is expired or damaged, return it to your pharmacist for disposal.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are being given or treated with B-Dose Forte Injection.
This medicine helps most people, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor, nurse or pharmacist as soon as possible, if you have any of these less serious side effects and they worry you. |
The above list includes the mild side effects of this medicine but could be serious.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
The above list includes serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell, even if is not on this list. This is not a complete list of all possible side effects.
Others may occur in some people and there may be some side effects not yet known.
Ask your doctor or pharmacist if you don't understand anything in this list.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What B-Dose Forte 2.5 mL Injection contains
| Active ingredient (main ingredient) | Thiamine hydrochloride (B1) Riboflavine sodium phosphate (B2) Nicotinamide (B3) Dexpanthenol (B5) Pyridoxine hydrochloride (B6) Cyanocobalamin (B12) |
| Other ingredients (inactive ingredients) | Disodium Edetate Benzyl alcohol Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What B-Dose Forte 2.5 mL Injection looks like
Biological Therapies B-Dose Forte Injection is a clear amber colour, contained in an amber glass vial sealed with a rubber stopper and an aluminium cap.
Australian Registration Number: AUST R 22408.
This medicine may be available in 4 pack sizes:
1 x 2.5 mL vial
3 x 2.5 mL vials
5 x 2.5 mL vials
6 x 2.5 mL vials
Who distributes B-Dose Forte 2.5 mL Injection
Biological Therapies
A Division of Orthomolecular Medisearch Laboratories Pty Ltd
5 / 20 – 30 Malcolm Road, Braeside VIC 3195
Australia
Tel: + 61 3 9587 3948
Fax: +61 3 9587 1720
Website: www.biologicaltherapies.com.au
Email: biol@biol.com.au
This leaflet was prepared in September 2025.

Brand Information
| Brand name | Biological Therapies B-Dose Forte 2.5 mL Injection |
| Active ingredient | Thiamine hydrochloride + Riboflavine sodium phosphate + Nicotinamide + Dexpanthenol + Pyridoxine hydrochloride + Cyanocobalamin |
| Schedule | S4 |
MIMS Revision Date: 01 December 2019
1 Name of Medicine
Thiamine hydrochloride, riboflavine sodium phosphate, nicotinamide, dexpanthenol, pyridoxine hydrochloride, cyanocobalamin.
2 Qualitative and Quantitative Composition
Biological Therapies B-Dose Forte 2.5 mL Injection is a B complex mixture of B1, B2, B3, B5, B6 and B12.
Each vial contains a minimum of 2.5 mL of solution.
2.5 mL of this formulation contains:
(B1) Thiamine hydrochloride 250 mg.
(B2) Riboflavine sodium phosphate 5 mg.
(B3) Nicotinamide 50 mg.
(B5) Dexpanthenol 50 mg.
(B6) Pyridoxine hydrochloride 100 mg.
(B12) Cyanocobalamin 1 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Biological Therapies B-Dose Forte 2.5 mL Injection is a clear, amber colour.
4 Clinical Particulars
4.1 Therapeutic Indications
B vitamin deficiencies may exist that are unable to be corrected by oral intake and in these cases parenteral administration may be preferable. Oral administration of B vitamins may be insufficient in: alcoholism, pernicious anaemia, malabsorption disorders, gastrectomy, gastrointestinal pathologies and debilitated and elderly patients.
Specific indications include:
1. Rapid saturation of the B group vitamins.
2. When oral administration is not feasible or appropriate.
3. For alcoholic, debilitated and elderly patients when their diet is inadequate.
4. Beriberi and Wernicke's syndrome as a result of Vitamin B1 deficiency.
5. Pellagra as a result of Vitamin B3 deficiency. Patients with pellagra may have a concurrent deficiency of B1, B2, B6 and B12.
6. Peripheral neuritis caused by various B group deficiencies.
7. Carpal tunnel syndrome.
8. Pernicious anaemia as a result of in situ B12 deficiency.
General indications include:
1. Angular stomatitis and glossitis have been shown in some instances to respond to the administration of either riboflavine or pyridoxine.
2. For general energy depletion.
4.2 Dose and Method of Administration
Biological Therapies B-Dose Forte 2.5 mL Injection is suitable for patients with acute symptoms of the indications listed (see Section 4.1).
The B vitamins tend to act synergistically in the body and are, in general, best injected as a complex.
Biological Therapies B-Dose Forte 2.5 mL Injection is administered by slow intramuscular (deep intragluteal) injection weekly for 2 to 6 weeks or as determined by the physician. In severe cases, one vial daily until the acute symptoms subside. This product is for single use only. Use once only and discard any residue. For patient comfort, warm solution to body temperature before administration. If glucose is used to treat hypoglycaemia associated with Wernicke's syndrome, it is preferable to give B-Dose Forte 2.5 mL Injection before the glucose administration to prevent further reduction of thiamine levels by the glucose. In general, high dose B complex injections are best delivered IM, however IV thiamine doses as high as 250 mg daily have been given in the successful treatment of Wernicke's encephalopathy (Martindale). If the product is to be used IV care should be taken to only slowly infuse diluted solutions such as those typically given in TPN, always being on the alert for any sign of anaphylaxis.
If the solution is cloudy or contains visually detectable particles, please contact the Quality Control Officer at Biological Therapies.
Note. Practitioners are advised to consider the Biological Therapies range of other B-Dose products viz B-Dose AE 2 mL Injection, B-Dose 2 mL Injection, B-Dose Plus 2 mL Injection, or IV B-Dose 2 mL Injection before selecting the most appropriate B Complex injection.
4.3 Contraindications
Known hypersensitivity to any of the ingredients, or pre-existing B hyper-vitaminosis. Dexpanthenol is contraindicated in haemophiliacs and in patients with ileus due to mechanical obstruction.
Cyanocobalamin should not be administered for megaloblastic anaemia caused by folate deficiency. Do not use Biological Therapies B-Dose Forte 2.5 mL Injection with neonates due to the benzyl alcohol content.
4.4 Special Warnings and Precautions for Use
This product contains pyridoxine hydrochloride which may be dangerous when used in large amounts or for a long time. Rare cases of anaphylactic shock have been known to occur after parenteral administration of thiamine. A few unconfirmed allergic reactions have been noted from dexpanthenol and a rare number of allergic hypersensitivity reactions from administration of cyanocobalamin.
Note. 1. Should Anaphylactic Shock occur, immediately stop administration of the product and administer the appropriate dose of adrenalin 1:1000 (1 mg/1 mL) intramuscularly. This may be repeated at 5 minute intervals, if clinically indicated. Additionally, intravenous administration of corticosteroids and antihistamines may be considered.
2. Hypersensitivity to the B vitamins may be checked by intradermal injection. Patients with a documented allergic disposition, for whom parenteral thiamine therapy is considered essential, should receive a carefully supervised intradermal test dose of B-Dose Forte 2.5 mL Injection prior to administration of this vitamin preparation.
Use in the elderly. No data available.
Paediatric use. Do not use Biological Therapies B-Dose Forte 2.5 mL Injection with neonates due to the benzyl alcohol content.
Effects on laboratory tests. Large doses of riboflavine result in a bright yellow discolouration of urine which may interfere with certain laboratory tests.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No data available.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. Australian pregnancy classification. Exempt from classification.
Use in lactation. No data available.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Thiamine hydrochloride. Adverse effects are rare but can occur after parenteral administration. Anaphylactic shock has been known to occur after parenteral administration. The most severe reactions have usually followed intravenous administration.
Nicotinamide. Chronic administration of nicotinamide at 3 grams/day for 3-36 months has had various side effects such as heartburn, nausea, headaches, hives, fatigue, sore throat and inability to focus eyes.
Pyridoxine hydrochloride. Long term administration of large doses of pyridoxine is associated with the development of severe peripheral neuropathies. However, risk appears to occur at doses above 1000 mg per day for long periods of time. Pyridoxine reduces the effects of levodopa but this does not occur if a dopa decarboxylase inhibitor is also given.
Riboflavine sodium phosphate. Large doses of riboflavine result in a bright yellow discolouration of urine which may interfere with certain laboratory tests.
Dexpanthenol. A few unconfirmed allergic reactions have been noted.
Cyanocobalamin. Allergic hypersensitivity reactions have occurred rarely following the administration of cyanocobalamin and hydroxocobalamin. Administration of doses greater than 10 micrograms daily may produce a haematological response in patients with folate deficiency, indiscriminate use of cyanocobalamin may mask the precise diagnosis.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
The B vitamins are critically tied to the metabolism of carbohydrates, amino acids and fats and are essential for the production of energy in cells, as well as carrying out many other functions. Deficiencies of one or more B vitamins may occur in debilitated states, with inadequate diet, with gastrointestinal pathologies, in alcoholism and the elderly. Deficiencies of several of the B vitamins are associated with severe neurological and skin disorders. The same disease condition in the body may result from the decreased level of just one of the B vitamins, or indeed from a simultaneous decrease in several of the B vitamins.
Except in rare cases of metabolic disorders affecting a single vitamin, it is unusual for a person to have a deficiency of one B vitamin without a deficiency of others. The B vitamins are synergistic in metabolism and as such it is recommended that they are administered together.
Mechanism of action. Thiamine is essential for the conversion of pyruvate (from glycolysis) to Acetyl Coenzyme A. Thiamine deficiency leads to elevated pyruvate concentration as well as severe depletion of aerobic respiration. Severe clinical consequences include Dry Beriberi (polyneuropathy), Cerebral Beriberi (Wernicke-Korsakoff syndrome) and Wet Beriberi (Cardiovascular Beriberi or Shosin beriberi). It is postulated that the neuropathy and encephalopathy in Wernicke's syndrome are due to pyruvate accumulation in blood. The clinical symptoms of Wernicke's syndrome are generally attenuated with immediate high dose thiamine treatment.
Riboflavine is an electron carrier essential to aerobic metabolism and other pathways. The active phosphorylated forms flavine mononucleotide (FMN) and flavine adenine dinucleotide (FAD) are involved as coenzymes in oxidative/reductive metabolic reactions. Riboflavine is necessary for the functioning of pyridoxine (B6) and nicotinic acid (B3). Riboflavine deficiency leads to the syndrome ariboflavinosis, characterized by cheilosis, angular stomatitis, glossitis, keratitis and seborrheic dermatitis.
Nicotinamide (Niacinamide) is another electron carrier essential to aerobic metabolism and other pathways. The active forms, nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP), are involved as coenzymes in several oxidative/reductive metabolic reactions. Deficiency of Vitamin B3 leads to pellagra. Symptoms include diarrhoea, dermatitis, abdominal pain, glossitis, stomatitis, loss of appetite, headache, lethargy and mental and neurological disturbances. In vivo formation of Vitamin B3 from tryptophan requires adequate B6 and B2 levels.
Vitamin B5 (as pantothenate) is a component of Coenzyme A. Without this there is a total failure of aerobic respiration due to lack of substrates for the tricarboxylic acid (TCA) cycle.
Pyridoxine is essential for amino acid metabolism and amino acid interconversion, for the metabolism of carbohydrates and fats, the production of haem and the metabolism of homocysteine. Deficiency leads to peripheral neuritis. Idiopathic carpal tunnel syndrome, with swelling of the synovia and compression of the median nerve by the transverse carpal ligament, has been attributed to a deficiency of pyridoxine. The syndrome appears to be reversible upon treatment with pyridoxine. Vitamin B6 deficiency has been reported to cause impairment in B12 absorption and reduced B12 stores in the liver.
The fraction of cyanocobalamin retained in blood and other tissues after injection is rapidly converted to the active coenzyme forms of B12; mecobalamin (co-methylcobalamin) and adenosylcobalamin. Deficiency of adenosylcobalamin leads to metabolic acidosis and an inability to use isoleucine, leucine and valine for energy production in the TCA cycle. Deficiency of mecobalamin (co-methylcobalamin) leads to the neurological consequences of pernicious anaemia. Cobalamin deficiency leads to impaired folate recycling and megaloblastic anaemia. Hydroxocobalamin is better retained than cyanocobalamin; however the administration of hydroxocobalamin has resulted in the formation of antibodies to the transcobalamin-B12 complex in some patients.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
The pharmacokinetics of combinations of B vitamins administered parenterally have not been studied, however it is expected that each individual vitamin would behave as if administered separately.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
2.5 mL of this formation contains: disodium edetate 2 mg, benzyl alcohol 52.3 mg, water for injections to 2.5 mL.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Protect from Light.
Keep out of reach of children.
6.5 Nature and Contents of Container
Container type: Amber glass vial with rubber stopper and aluminium seal.
Pack sizes: 1, 3, 5 or 6 vials per carton.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Thiamine hydrochloride. C12H17ClN4OS.HCl.
Chemical structure.
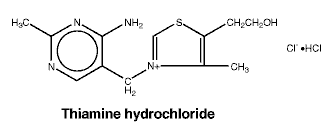
Riboflavine sodium phosphate. C17H20N4NaO9P.2H2O.
Chemical structure.
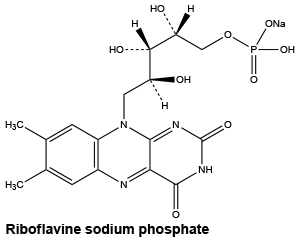
Nicotinamide. C6H6N2O.
Chemical structure.
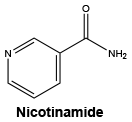
Dexpanthenol. C9H19NO4.
Chemical structure.
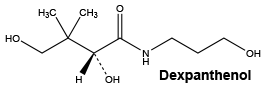
Pyridoxine hydrochloride. C8H11NO3.HCl.
Chemical structure.
Cyanocobalamin. C63H88CoN14O14P.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
08 October 1991
Date of Revision
14 October 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.