Bactroban Nasal Ointment
Brand Information
| Brand name | Bactroban Nasal Ointment |
| Active ingredient | Mupirocin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Bactroban Nasal Ointment.
Summary CMI
BACTROBAN NASAL OINTMENT
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using BACTROBAN nasal ointment?
BACTROBAN nasal ointment contains the active ingredient mupirocin calcium. BACTROBAN nasal ointment is used to remove bacteria, including the bacteria called 'Staphylococcus aureus' (golden staph.) from inside your nose.
For more information, see Section 1. Why am I using BACTROBAN nasal ointment? in the full CMI.
2. What should I know before I use BACTROBAN nasal ointment?
Do not use if you have ever had an allergic reaction to mupirocin calcium or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, are pregnant or plan to become pregnant, or are breastfeeding.
For more information, see Section 2. What should I know before I use BACTROBAN nasal ointment? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with BACTROBAN nasal ointment and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use BACTROBAN nasal ointment?
- The usual recommendation is for a small amount of BACTROBAN nasal ointment to be applied two or three times a day. Your doctor or pharmacist will tell you how many times to apply the ointment. Only use this ointment in your nose.
More instructions can be found in Section 4. How do I use BACTROBAN nasal ointment? in the full CMI.
5. What should I know while using BACTROBAN nasal ointment?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using BACTROBAN nasal ointment? in the full CMI.
6. Are there any side effects?
Less serious side effects which have been reported include irritation, itching, tingling, burning, stinging, soreness, facial pain, post nasal drip, sinus pain, runny or blocked nose, or conjunctivitis. Serious side effects include allergic reaction or inflammation of the colon. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
BACTROBAN NASAL OINTMENT
Active ingredient(s): Mupirocin calcium (2% w/w)
Consumer Medicine Information (CMI)
This leaflet provides important information about using BACTROBAN nasal ointment. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using BACTROBAN nasal ointment.
Where to find information in this leaflet:
1. Why am I using BACTROBAN nasal ointment?
2. What should I know before I use BACTROBAN nasal ointment?
3. What if I am taking other medicines?
4. How do I use BACTROBAN nasal ointment?
5. What should I know while using BACTROBAN nasal ointment?
6. Are there any side effects?
7. Product details
1. Why am I using BACTROBAN nasal ointment?
BACTROBAN nasal ointment contains the active ingredient mupirocin calcium (2% w/w). Mupirocin calcium belongs to a group of medicines called antibiotics. Antibiotics work by killing bacteria which can cause infection.
BACTROBAN nasal ointment is used to remove bacteria, including the bacteria called 'Staphylococcus aureus' (golden staph.) from inside your nose.
BACTROBAN nasal ointment is for use in your nose only.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed BACTROBAN nasal ointment for another reason.
There is no evidence that BACTROBAN nasal ointment is addictive.
2. What should I know before I use BACTROBAN nasal ointment?
Warnings
Do not use BACTROBAN nasal ointment if:
- you are allergic to mupirocin calcium, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- the expiry date (EXP) printed on the pack has passed, or if the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- are not sure whether you should start using this medicine.
- have any allergies to foods, dyes, preservatives or any other medicines.
- have any other medical conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the risks and benefits with you.
Children
The use of BACTROBAN nasal ointment in children under two years of age is not recommended.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with BACTROBAN nasal ointment and affect how it works.
Do not mix BACTROBAN nasal ointment with any other preparation. This may prevent BACTROBAN nasal ointment from working.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect BACTROBAN nasal ointment.
4. How do I use BACTROBAN nasal ointment?
Follow your doctor's instructions about how and when to use BACTROBAN nasal ointment.
The instructions from your doctor may differ from the information contained in this leaflet.
If you have any questions about BACTROBAN nasal ointment and how to use it, please ask your doctor or pharmacist.
How much to use
- Use BACTROBAN nasal ointment as directed by your doctor or pharmacist.
When to use BACTROBAN nasal ointment
- Use BACTROBAN nasal ointment for as long as your doctor or pharmacist tells you to.
- BACTROBAN nasal ointment is usually applied two or three times a day for between 5 and 7 days. Your doctor will tell you how many times to apply the ointment. Do not use it for more than 10 days.
- It is important that you take the full course of BACTROBAN nasal ointment. Do not stop early as the amount of bacteria in your nose may not be reduced.
How to apply BACTROBAN nasal ointment
- Always wash your hands before and after applying BACTROBAN nasal ointment.
- Squeeze a small amount (about the size of a matchstick head) onto the tip of your little finger and apply to the inside surface at the front of each nostril.
- Gently press your nostrils together to help spread the ointment throughout your nose.
- A swab, such as a cotton bud, may be used for application to infants or patients who are very ill.
If you forget to use BACTROBAN nasal ointment
If you forget an application, apply some BACTROBAN nasal ointment as soon as you remember, then continue as before.
Do not use a double dose to make up for the dose you missed.
If you use too much BACTROBAN nasal ointment (overdose)
If you think that you have used too much BACTROBAN nasal ointment, or have swallowed it accidentally, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using BACTROBAN nasal ointment?
Things you should do
- Stop using BACTROBAN nasal ointment and tell your doctor if you develop an allergic reaction (such as a rash, local pain or swelling).
- Keep the ointment away from your eyes. If BACTROBAN nasal ointment is accidentally applied to the eyes, wash the eyes thoroughly with water.
- Remind any doctor or pharmacist you visit that you are using BACTROBAN nasal ointment.
Call your doctor straight away if you:
- have not used BACTROBAN nasal ointment exactly as directed. Otherwise, your doctor may think it was not working as it should and change your treatment unnecessarily.
- become pregnant while using BACTROBAN nasal ointment.
Things you should not do
- Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
- Do not stop using BACTROBAN nasal ointment, or change the dose, without first checking with your doctor.
- Do not use BACTROBAN nasal ointment to treat any other complaints unless your doctor says to.
- Do not use BACTROBAN nasal ointment to treat infections in or near your eyes.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in the original pack in a cool dry place away from moisture, heat or sunlight where the temperature stays below 25°C.
Do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine, it has expired or is damaged, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date or if the packaging is torn or shows signs of tampering.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin and subcutaneous tissue disorders:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Immune disorders and allergic reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What BACTROBAN nasal ointment contains
| Active ingredient | Mupirocin calcium |
| Other ingredients | Bis-diglyceryl polyacyladipate-2 White soft paraffin |
Do not take this medicine if you are allergic to any of these ingredients.
What BACTROBAN nasal ointment looks like
BACTROBAN nasal ointment is a white ointment supplied in 3 g tubes (AUST R 11199).
Who distributes BACTROBAN nasal ointment
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street,
Abbotsford, Victoria, 3067
Phone: 1800 033 109
www.gsk.com.au
This leaflet was prepared on 22 July 2025.
The information provided applies only to BACTROBAN nasal ointment.
Trademarks are owned by or licensed to the GSK group of companies.
© 2025 GlaxoSmithKline group of companies or its licensor.
Version 5.0
Brand Information
| Brand name | Bactroban Nasal Ointment |
| Active ingredient | Mupirocin |
| Schedule | S4 |
MIMS Revision Date: 01 April 2021
1 Name of Medicine
Mupirocin calcium.
2 Qualitative and Quantitative Composition
Bactroban nasal ointment contains mupirocin calcium 2% w/w as the active ingredient.
Each gram of Bactroban nasal ointment 2% contains 20 mg mupirocin as the calcium salt.
List of excipients with known effect. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Nasal ointment.
4 Clinical Particulars
4.1 Therapeutic Indications
Bactroban (mupirocin) nasal ointment is indicated for the elimination of nasal carriage of Staphylococci including methicillin resistant Staphylococcus aureus (MRSA).
4.2 Dose and Method of Administration
Adults and children. Bactroban nasal ointment should be applied to the anterior nares two to three times a day as follows.
A small amount of the ointment, about the size of a match head, is placed on the little finger and applied to the inside of each nostril. The nostrils are closed by pressing the side of the nose together; this will spread the ointment throughout the nares. A swab may be used for application to infants or patients who are very ill.
Nasal carriage should normally clear within 5-7 days of commencing treatment. Treatment should not continue for more than 10 days.
4.3 Contraindications
Bactroban nasal ointment is contraindicated in patients who have demonstrated hypersensitivity to mupirocin calcium or any components of the formulation.
4.4 Special Warnings and Precautions for Use
If a reaction suggesting sensitivity or chemical irritation should occur with the use of Bactroban nasal ointment, treatment should be discontinued, the product should be wiped off and appropriate alternative therapy for the infection instituted.
Bactroban nasal ointment formulation is not suitable for ophthalmic use.
Avoid contact with the eyes. If contaminated, the eyes should be thoroughly irrigated with water until the ointment residues have been removed.
As with other antibacterial products, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. Appropriate measures should be taken if this occurs.
The occurrence of resistance to topical mupirocin has occasionally been reported. The possibility of the development of resistance following intranasal use should therefore be borne in mind, particularly in treatment courses lasting longer than 5-7 days. Long-term, continuous use of Bactroban nasal ointment should be avoided to minimise this possibility, particularly in the hospital environment.
Pseudomembranous colitis has been reported with the use of antibiotics and may range in severity from mild to life threatening. Therefore, it is important to consider its diagnosis in patients who develop diarrhoea during or after antibiotic use. Although this is less likely to occur with topically applied mupirocin, if prolonged or significant diarrhoea occurs or the patient experiences abdominal cramps, treatment should be discontinued immediately and the patient investigated further.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Bactroban nasal ointment should not be mixed with other preparations as there is a risk of dilution, resulting in a reduction in the antibacterial activity and potential loss of stability of the mupirocin in the ointment.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B1)
Reproduction studies have been performed in rats and rabbits at systemic doses up to 160 mg/kg and have revealed no evidence of impaired fertility or harm to the foetus due to mupirocin.
Adequate human data on use during pregnancy are not available. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Use in lactation. Adequate human data on use during lactation are not available. Caution should be exercised when Bactroban nasal ointment is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
The following local adverse reactions with an overall incidence of approximately 2%, have been reported in connection with the use of this product: irritation, itching, tingling, burning, stinging, soreness, facial pain over maxillae, postnasal drip, sinusitis, rhinitis and conjunctivitis. However, less than 0.2% of patients withdrew due to adverse experiences.
Systemic allergic reactions including anaphylaxis, generalised rash, urticaria and angioedema have been reported very rarely.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is currently limited experience with overdosage of Bactroban.
There is no specific treatment for an overdose of Bactroban. In the event of overdose, the patient should be treated supportively with appropriate monitoring as necessary.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Mupirocin inhibits bacterial protein synthesis by reversibly and specifically binding to bacterial isoleucyl transfer RNA synthetase. It shows no cross resistance with other commonly used and clinically important antibiotics.
In vitro mupirocin is active mainly against Gram positive aerobes including Staphylococcus aureus (including MRSA positive strains), Staphylococcus saprophyticus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus viridans, Streptococcus agalactiae, and Streptococcus pneumoniae.
Group D streptococci (including S. faecalis and S. faecium), are much less sensitive to mupirocin. Most Gram negative organisms (except for H. influenzae, Neisseria and Branhamella) and anaerobes (including Propionibacterium acnes) are not sensitive to mupirocin.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
This formulation has been designed as appropriate for use in the interior nares. Limited data are available on the absorption of mupirocin following intranasal application in adults. Adverse effects from continued absorption from the nose cannot be ruled out.
Mupirocin is absorbed in neonates and premature infants following intranasal administration of mupirocin ointment. In clinical studies of neonates, intranasal administration of mupirocin for up to 5 days was well tolerated. The safety of courses lasting longer than 5 days in neonates and infants has not been studied.
If absorption occurs, mupirocin will be quickly hydrolysed to the antimicrobially inactive metabolite monic acid, which is rapidly cleared from the body.
No evidence of contact sensitisation has been demonstrated with the white soft paraffin ointment formulation of mupirocin (Bactroban nasal ointment).
Whilst mupirocin successfully eradicates S. aureus colonisation of the nasal mucosa there are currently insufficient data to determine the frequency of, and time to, recolonisation.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Bis-disglyceryl polyacyladipate-2.
White soft paraffin.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Bactroban (mupirocin) nasal ointment 2% is supplied in 3 g tubes.
6.6 Special Precautions for Disposal
Any product remaining at the end of treatment should be discarded.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
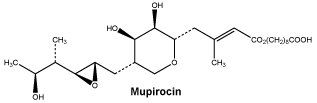
Mupirocin is a naturally occurring antibiotic, produced by fermentation of the organism Pseudomonas fluorescens.
The chemical name is 9-4-{5S-(2S,3S-epoxy-5S-hydroxy- 4S-methylhexyl)- 3R,4R-dihydroxytetrahydropyran-2S-y1}- 3-methylbut-2-(E)-enoyloxy-nonanoic acid.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
29 March 1996
Date of Revision
18 January 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.