Chlorhexidine Acetate with Cetrimide Antiseptic Solution
Brand Information
| Brand name | Chlorhexidine Acetate with Cetrimide Antiseptic Solution |
| Active ingredient | Chlorhexidine acetate + Cetrimide |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Chlorhexidine Acetate with Cetrimide Antiseptic Solution.
Summary CMI
Chlorhexidine Acetate with Cetrimide Antiseptic Solution
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I receiving Chlorhexidine with Cetrimide?
Chlorhexidine Acetate with Cetrimide Antiseptic Solution (Chlorhexidine with Cetrimide) contains the active ingredients chlorhexidine acetate and cetrimide. Chlorhexidine with Cetrimide is used on the skin to clean and disinfect wounds and treat burns, helping prevent infections and promote healing. For more information, see Section 1. Why am I receiving Chlorhexidine with Cetrimide? in the full CMI.
2. What should I know before I receive Chlorhexidine with Cetrimide?
Do not use if you have ever had an allergic reaction to chlorhexidine acetate, cetrimide or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I receive Chlorhexidine with Cetrimide? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Chlorhexidine with Cetrimide and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Chlorhexidine with Cetrimide given?
- Your doctor or trained nurse will apply Chlorhexidine with Cetrimide to your skin as needed to clean and disinfect the wound or burn area/s.
- Your doctor will decide how much Chlorhexidine with Cetrimide to give. The amount depends on your need and condition.
More instructions can be found in Section 4. How is Chlorhexidine with Cetrimide given? in the full CMI.
5. What should I know while receiving Chlorhexidine with Cetrimide?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while receiving Chlorhexidine with Cetrimide? in the full CMI.
6. Are there any side effects?
Serious side effects are possible, including severe allergic reactions, chemical burns (in newborns) and sudden vision changes when the product comes into contact with the eyes. For more information on what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Chlorhexidine Acetate with Cetrimide Antiseptic Solution
Active ingredients: Chlorhexidine acetate and cetrimide
Consumer Medicine Information (CMI)
This leaflet provides important information about using Chlorhexidine Acetate with Cetrimide Antiseptic Solution (Chlorhexidine with Cetrimide). You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about using Chlorhexidine with Cetrimide.
Where to find information in this leaflet:
1. Why am I receiving Chlorhexidine with Cetrimide?
2. What should I know before I receive Chlorhexidine with Cetrimide?
3. What if I am taking other medicines?
4. How is Chlorhexidine with Cetrimide given?
5. What should I know while receiving Chlorhexidine with Cetrimide?
6. Are there any side effects?
7. Product details
1. Why am I receiving Chlorhexidine with Cetrimide?
Chlorhexidine with Cetrimide contains the active ingredients chlorhexidine acetate and cetrimide. Chlorhexidine with Cetrimide is an antiseptic and disinfectant. It helps to kill microbes on the skin.
Chlorhexidine with Cetrimide is used on the skin to clean and disinfect wounds, as well as to treat burns. It helps prevent infections and promote healing.
It should never be injected or swallowed.
2. What should I know before I receive Chlorhexidine with Cetrimide?
Warnings
Do not use Chlorhexidine with Cetrimide:
- If you are allergic to chlorhexidine acetate, cetrimide or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- Around the face or head. It must not come into contact with the eyes, on the tissues around the brain, or in the ear canal (especially perforated eardrums).
- In the mouth.
Chlorhexidine with Cetrimide mouthwash has been linked to colour changes of the tongue, teeth and dentures/fillings/ crowns. Do not swallow Chlorhexidine with Cetrimide.
Check with your doctor if you:
- have any other medical conditions
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor if you use any lotions, soaps or other skin products.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Chlorhexidine with Cetrimide.
4. How is Chlorhexidine with Cetrimide given?
How it is given
- Your doctor or trained nurse will apply Chlorhexidine with Cetrimide to your skin as needed to clean and disinfect the wound or burn area/s.
- It is usually given in a hospital, clinic or nursing home.
How much is given
- Your doctor will decide how much Chlorhexidine with Cetrimide to give. The amount depends on your need and condition.
- Your doctor will decide how long you need to be treated with Chlorhexidine with Cetrimide.
If you are given too much Chlorhexidine with Cetrimide
The doctor or nurse giving you Chlorhexidine with Cetrimide is trained and qualified, so it is unlikely that you will be given an overdose.
If you think that you have been given too much, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Call the Poisons Information Centre if you swallow this product.
5. What should I know while receiving Chlorhexidine with Cetrimide?
Things you should do
Remind any doctor, nurse, dentist or pharmacist you visit that you have received Chlorhexidine with Cetrimide.
Tell your doctor immediately if you develop a rash, hives, red or tingling skin after being given Chlorhexidine with Cetrimide.
If these symptoms worsen, Chlorhexidine with Cetrimide may need to be stopped.
Things you should not do
- Do not swallow or inject this product.
- Do not use this product around the eyes, on the tissues around the brain, or in the ear canal (especially perforated eardrums).
- Do not use this product in the mouth.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Chlorhexidine with Cetrimide affects you.
This medicine is not expected to affect your ability to drive a car or operate machinery.
Drinking alcohol
Tell your doctor if you drink alcohol.
It is not known how drinking alcohol affects this medicine.
Looking after your medicine
Chlorhexidine with Cetrimide will be stored in the pharmacy or in the ward, kept below 30°C and away from light.
If for any reason, you need to keep this product at home, it should be stored in a locked cupboard.
- Store below 30°C.
- Do not freeze.
- Protect from light.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool, dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
Your doctor or nurse will discard any unused medicine. They will not save unused portions for later, either for you or anyone else.
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic-reaction related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Chlorhexidine with Cetrimide contains
| Active ingredient (main ingredient) | Chlorhexidine acetate and Cetrimide |
| Other ingredients (inactive ingredients) | Tartrazine Glacial acetic acid Water for Injections |
| Potential allergens | Chlorhexidine acetate and Cetrimide |
Do not take this medicine if you are allergic to any of these ingredients.
What Chlorhexidine with Cetrimide looks like
Baxter Chlorhexidine Acetate with Cetrimide antiseptic solutions are yellow sterile solutions.
Chlorhexidine with Cetrimide is available in the following strengths and bottle sizes:
- BAXTER 0.015% Chlorhexidine & 0.15% Cetrimide antiseptic solution bottle: 100mL, 500mL and 1000mL – Aust R 19458
- BAXTER Chlorhexidine 0.05% Cetrimide 0.5% antiseptic solution bottle: 100mL, 500mL and 1000mL – Aust R 19460
- BAXTER Chlorhexidine 0.1% Cetrimide 1% aqueous solution bottle: 100mL, 500mL and 1000mL – Aust R 19462
Not all products may be marketed.
Who distributes Chlorhexidine with Cetrimide
Baxter Healthcare Pty Ltd
1 Baxter Drive
Old Toongabbie
NSW 2146
Australia
This leaflet was prepared in November 2025.
Brand Information
| Brand name | Chlorhexidine Acetate with Cetrimide Antiseptic Solution |
| Active ingredient | Chlorhexidine acetate + Cetrimide |
| Schedule | Unscheduled |
MIMS Revision Date: 01 November 2024
1 Name of Medicine
Chlorhexidine acetate and cetrimide.
2 Qualitative and Quantitative Composition
Baxter Chlorhexidine Acetate with Cetrimide antiseptic solutions are yellow sterile solutions.
Baxter Chlorhexidine Acetate with Cetrimide antiseptic solutions are hypotonic and haemolytic. They are supplied in the following strengths:
Chlorhexidine Acetate 0.015% with Cetrimide 0.15%.
Chlorhexidine Acetate 0.05% with Cetrimide 0.5%.
Chlorhexidine Acetate 0.10% with Cetrimide 1.0%.
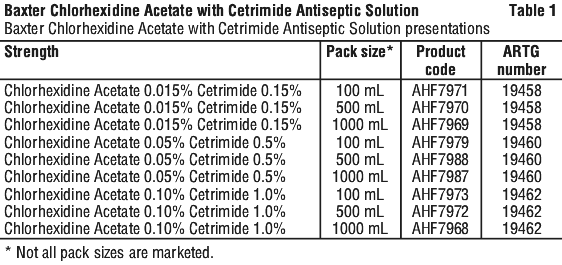
Each strength is supplied in 3 pack sizes (see Section 6.5 Nature and Contents of Container, Table 1).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Antiseptic solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution is used as a general antiseptic.
It is used for the cleaning and disinfecting of wounds as an antiseptic treatment for burns.
4.2 Dose and Method of Administration
Dosage. As required to disinfect wound area. See Directions for use. Dosage and duration of administration are to be individualized and depend upon the indication for use, the patient's ages, weight, clinical condition, concomitant treatment and on patient's clinical response to treatment.
Not for intravenous or oral route of administration.
Product should be inspected visually for particulate matter and discolouration prior to administration whenever solution and container permit. Do not use unless the solution is clear and the seal is intact.
Directions for use. The area where Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution is to be used should be rinsed thoroughly with water. Apply the minimum amount necessary to cover the wound area and wash gently. Leave the area to dry by air for 3 minutes.
Use undiluted. Do not mix with detergents or other chemicals. Discard within 24 hours of opening.
The solution is used for cleaning and disinfecting wounds and as an antiseptic treatment for burns.
To open. Hold Steripour bottle and twist lid to open, breaking the tamper proof seal.
4.3 Contraindications
Known hypersensitivity to chlorhexidine or cetrimide.
Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution should not be used in the eye, intravenously, orally, in the auditory canal (especially perforated eardrums) or near meninges, brain or spinal cord.
4.4 Special Warnings and Precautions for Use
General. Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution is used as a topical solution; it must not be administered intravenously or taken orally.
This product should not be used in body cavities or as an enema. It should not be used for the disinfection of soft contact lenses.
Seek urgent medical attention if Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution is swallowed. If ingested, cetrimide may cause nausea and vomiting. Swallowing this solution may cause oesophageal damage or necrosis. Demulcents may be given, but emesis and lavage should be avoided.
Accidental intrauterine or intravenous administration may cause haemolysis.
It should not be used if you have a history of allergy to any of the ingredients of Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution.
It should not be used if the expiry date printed on the label is overdue. Do not use unless the solution is clear, free of particles and the tamperproof seal is intact.
Hypersensitivity reactions. Hypersensitivity reactions including anaphylactic/ anaphylactoid reactions have been reported with chlorhexidine. Fatal anaphylactic reactions have been reported with other products containing chlorhexidine.
If any signs or symptoms of a suspected hypersensitivity reaction develop, immediately stop use the product. Appropriate therapeutic countermeasures must be instituted as clinically indicated.
Some patients become hypersensitive to cetrimide after repeated applications. The use of chlorhexidine as a mouthwash has been associated with reversible discolouration of the tongue, teeth and silicate or composite dental restorations.
Chemical burns in neonates. The use of chlorhexidine solutions, both alcohol based and aqueous, for skin antisepsis prior to invasive procedures has been associated with skin reactions such as chemical burns in neonates. Based on available case reports in the published literature, this risk appears to be higher in preterm infants, especially those born before 32 weeks of gestation and within the first 2 weeks of life.
Remove any soaked materials, drapes or gowns before proceeding with the intervention. Do not use excessive quantities and do not allow the solution to pool in skin folds or under the patient or drip on sheets or other material in direct contact with the patient. Where occlusive dressings are to be applied to areas previously exposed to chlorhexidine, care must be taken to ensure no excess product is present prior to application of the dressing.
Preoperative skin preparation. Chlorhexidine should not be used in preoperative skin preparations for the face and head.
Chlorhexidine must not come into contact with the eye. Serious cases of persistent corneal injury, potentially requiring corneal transplant, were reported following accidental ocular exposure to chlorhexidine containing medicinal products despite taking eye protective measures due to migration of solution beyond the intended surgical preparation area. Extreme care must be taken during application to ensure that chlorhexidine does not migrate beyond its intended application site into the eyes. Particular care should be taken in anesthetized patients, who are unable to immediately report ocular exposure. If chlorhexidine comes into contact with the eyes, wash out promptly and thoroughly with water. An ophthalmologist's advice should be sought.
Use in the elderly. No data available.
Paediatric use. This product is safe for use on children.
The use of chlorhexidine solutions has been associated with skin reactions such as chemical burns in neonates.
Effects on laboratory tests. The effect of this medicine on laboratory tests has not been established.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The action of chlorhexidine is reduced by an alkaline pH, the presence of organic matter, anionic detergents and tannins.
See Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
The "Prescribing Medicines in Pregnancy" booklet categorises chlorhexidine as a Category A medicine.
Use in lactation. This product is safe for use in lactation.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Anaphylactic/ anaphylactoid reactions to chlorhexidine have been reported. Manifestations of such reactions have included cardiac arrest, circulatory collapse, hypotension, bronchospasm, rash, erythema, tachycardia, urticaria and shock. Fatal anaphylactic reaction has been reported.
Some patients may experience skin irritation or an allergic reaction/ hypersensitivity reactions on contact with this product. If this occurs, the use of this product should be stopped immediately.
Skin sensitivity to chlorhexidine has occasionally been reported.
Very occasionally the following reactions have been noted when chlorhexidine containing irrigating solutions have been used intravesically, intravaginally or topically on traumatised skin: hypotension, paraesthesia, dyspnoea, tachycardia cold sweat, generalized erythema, urticaria and loss of consciousness.
Strong solutions may cause irritation of the conjunctiva and other sensitive tissues. Transient taste disturbances and a burning sensation of the tongue may occur on initial use.
Oral desquamation and occasional parotid gland swelling have been reported with the mouthwash. If desquamation occurs, a 50% dilution of the mouthwash with water and less vigorous rinsing may allow continued use.
The adverse events reported and/or observed with other chlorhexidine products include:
Fatal anaphylactic reactions;
Chemical burns in neonates (see Section 4.4 Special Warnings and Precautions for Use);
Eye disorder. Frequency not known: corneal erosion, corneal epithelium defect/ injury corneal, visual impairment*.
*Cases of severe corneal erosion and permanent significant visual impairment due to inadvertent ocular exposure have been reported post-marketing, leading to some patients requiring corneal transplant (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
If taken by mouth, cetrimide and other quaternary ammonium compounds cause nausea and vomiting. If ingested, advice concerning treatment should be sought immediately from a doctor or contact the Poison Information Centre on 13 11 26 (Australia).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Cetrimide is a quaternary ammonium antiseptic with actions and uses typical of cationic surfactants. These surfactants dissociate in aqueous solution into a relatively large and complex cation, which is responsible for the surface activity, and a smaller inactive anion. In addition to emulsifying and detergent properties, quaternary ammonium compounds have bactericidal activity against Gram-positive and, at the higher concentrations, against some Gram-negative bacteria. Some Pseudomonas spp. are particularly resistant as are strains of Mycobacterium tuberculosis. They are ineffective against bacterial spores, have variable antifungal activity, and are effective against some viruses.
Quaternary ammonium compounds are most effective in neutral or slightly alkaline solutions and their bactericidal activity is appreciably reduced in acid media; alcohols enhance their activity.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Tartrazine, glacial acetic acid and water for injections.
6.2 Incompatibilities
Prolonged immersion of rubber appliances in these solutions should be avoided. Chlorhexidine is incompatible with soaps, other anionic materials and with potassium iodide.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Baxter Chlorhexidine Acetate with Cetrimide Antiseptic Solution should be stored below 30°C.
Protect from light.
6.5 Nature and Contents of Container
Baxter Chlorhexidine Acetate with Cetrimide antiseptic solutions are supplied in 3 strengths. Each strength has 3 pack sizes (see Table 1). Do not heat bottle in excess of 80°C.
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Chlorhexidine acetate.
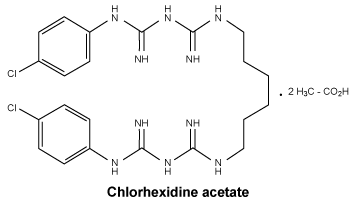
Molecular weight: 626.
Appearance: white or almost white, microcrystalline powder.
Solubility: sparingly soluble in water, soluble in ethanol (96 per cent), slightly soluble in glycerol and in propylene glycol.
Cetrimide.
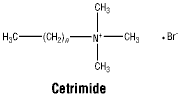
Molecular weight: 336.4.
Appearance: white or almost white, voluminous, free-flowing powder.
Solubility: freely soluble in water and in alcohol.
CAS number. Chlorhexidine acetate. CAS No.: 56-95-1.
Cetrimide. CAS No.: 505-86-2.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
30 September 1991
Date of Revision
05 September 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.