Belkyra
Brand Information
| Brand name | Belkyra |
| Active ingredient | Deoxycholic acid |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Belkyra.
Summary CMI
BELKYRA®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using BELKYRA® injection?
BELKYRA® injection contains the active ingredient deoxycholic acid. BELKYRA® injection is used to reduce the fat under the chin, without surgery, resulting in a more contoured neck profile and jawline.
For more information, see Section 1. Why am I using BELKYRA® injection? in the full CMI.
2. What should I know before I use BELKYRA® injection?
Check the list of ingredients at the end of the CMI. Do not use BELKYRA® injection if you have ever had an allergic reaction to any of them.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use BELKYRA® injection? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with BELKYRA® injection and affect how it works, or BELKYRA® injection may interfere with other medicines and affect how they work.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How BELKYRA® injection is used?
- BELKYRA® is injected under the skin
- The number of injections and treatment sessions you receive is calculated by your doctor based on your specific needs
- BELKYRA® can only be injected by a suitably qualified doctor.
More instructions can be found in Section 4. How BELKYRA® injection is used? in the full CMI.
5. What should I know while using BELKYRA® injection?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using BELKYRA® injection? in the full CMI.
6. Are there any side effects?
- The most common reactions include bruising, pain, numbness, swelling, redness, itching around the treatment area, tingling, formation of small areas of hardness under the skin where BELKYRA® has been injected, high blood pressure. Some serious effects can occur in other parts of the body, including shortness of breath, and difficulty swallowing.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
BELKYRA®
Active ingredient: deoxycholic acid
Consumer Medicine Information (CMI)
This leaflet provides important information about using BELKYRA® injection. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using BELKYRA® injection.
Where to find information in this leaflet:
1. Why am I using BELKYRA®injection?
2. What should I know before I use BELKYRA® injection?
3. What if I am taking other medicines?
4. How BELKYRA® injection is used?
5. What should I know while using BELKYRA® injection?
6. Are there any side effects?
7. Product details
1. Why am I using BELKYRA® injection?
BELKYRA® injection is a prescription medicine that contains the active ingredient deoxycholic acid.
BELKYRA® injection is used to reduce fat under the chin, without the need for surgery, to result in a more contoured neck profile and jawline.
2. What should I know before I use BELKYRA® injection?
Warnings
Do not use BELKYRA® injection if:
- you are allergic to deoxycholic acid, or any of the ingredients listed at the end of this leaflet.
- You are allergic to any other similar medicines. Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin
- you have an infection in your chin or neck area where BELKYRA® is to be injected.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have allergies to any other medicines, foods, preservatives or dyes
- have had or plan to have plastic surgery on your face, chin, or neck
- have had other aesthetic treatments such as liposuction or neurotoxins (e.g. BOTOX®) (drugs sometimes used in the neck for aesthetic purposes such as to reduce wrinkling or for other medical reasons) in these areas
- have or have had difficulty swallowing
- have or have had, medical conditions in, on or near the neck
- have a bleeding disorder or are taking medicines to prevent clotting of the blood
- are on a diet that restricts sodium (or a salt-reduced diet)
- are not sure whether this medicine is right for you
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed.
It is not known if BELKYRA® injection can harm your unborn baby. Use in pregnancy is not recommended.
The active ingredient in BELKYRA® injection may pass into breast milk. Use while breast feeding is not recommended.
BELKYRA® injection is not indicated for use in children or adolescents, younger than 18 years.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are being given BELKYRA® injection.
Some medicines may interfere with BELKYRA® injection and affect how it works.
There is limited information available on the use of BELKYRA® injection with other medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while being given this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these have the possibility of affecting BELKYRA® injection.
4. How BELKYRA® injection is used?
How much is used
The number of injections you receive is calculated by your doctor based on the amount of excess fat you have under your chin. You may need up to 50 injections per treatment session.
When to use BELKYRA® injection
The total number of treatment sessions needed to achieve a satisfactory response depends upon the individual. Your doctor will determine how many treatments you need.
No more than 6 treatments can be given. They should be no closer than a minimum of 4 weeks apart.
How BELKYRA® injection is given
A doctor will administer BELKYRA® injection by injecting small amounts of this medicine into several locations in your treatment area approximately every 4 weeks for up to 6 treatments or until the desired result is achieved.
Follow all directions given by your doctor carefully. They may differ from the information contained in this leaflet.
If you forget a dose of BELKYRA® injection
It is important that you receive BELKYRA® injection at regular intervals to ensure the best outcome.
If you forget to attend an appointment, contact your doctor for advice and/or to reschedule.
If you use too much BELKYRA® injection
If you think you or anyone else may have swallowed or accidentally injected BELKYRA®
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia or
0800 POISON or 0800 764 766 in New Zealand)), or - contact your doctor, or
- go to Accident and Emergency Department at your nearest hospital.
You should do this even if there appears to be no signs of discomfort or poisoning. You may need urgent medical attention.
5. What should I know while using BELKYRA® injection?
Things you should do
Follow all directions given to you by your doctor before, during and after you receive your BELKYRA® injection.
They may differ from the information contained in this leaflet.
Keep all your doctor's appointments so that your progress can be checked.
Call your doctor straight away if you:
- become pregnant while being treated with BELKYRA® injection
- have problems swallowing at any time following your BELKYRA® injection
Remind any doctor, dentist or pharmacist you visit that you are using BELKYRA® injection.
Things you should not do
Do not skip any appointments as this may result in a less satisfactory outcome.
Driving or using machines
BELKYRA® injection is not expected to affect your ability to drive or use machines.
Looking after your medicine
BELKYRA® is an injectable medicine which will only be provided to you by your healthcare practitioner in a clinic setting.
Your doctor or clinic staff will keep it where young children cannot reach it.
Getting rid of any unwanted medicine
Your doctor or clinic staff will dispose of any unused medicine.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Do not be alarmed by the following lists of side effects.
You may not experience any of them. Most patients experience mild or moderate pain or discomfort at the time BELKYRA® injection is injected.
Very Serious side effects
| Very Serious side effects | What to do |
Body as a whole:
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Serious side effects
| Serious side effects | What to do |
Area of treatment (injection):
| Call your doctor straight away if you notice any of these serious side effects. |
Less serious side effects
| Less serious side effects | What to do |
Area of treatment (injection):
| Speak to your doctor if you have any of these less serious side effects and they worry you. These are mild or moderate side effects of the medicine and are short-lived. Your doctor may ask you to use ice/cold packs, paracetamol, ibuprofen or other ways to reduce side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems in Australia or to nzphvc.otago.ac.nz/reporting in New Zealand. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop using any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription for injection by a suitably qualified doctor.
What BELKYRA® injection contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not use this medicine if you are allergic to any of these ingredients.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes
What BELKYRA® injection looks like
BELKYRA® injection is a clear, colourless liquid. It is supplied in 2 mL single-use glass vials. (Aust R 233201).
Who distributes BELKYRA® injection?
BELKYRA® injection is distributed in Australia by:
AbbVie Pty Ltd
241 O'Riordan Street
Mascot NSW 2020
AUSTRALIA Toll free: 1800 252 224 (AU)
BELKYRA® injection is distributed in New Zealand by:
AbbVie Limited
6th Floor, 156-158 Victoria St
Wellington 6011
NEW ZEALAND
Freephone: 0800 659 912 (NZ)
This leaflet was prepared in December 2025.
BELKYRA® and its design are trademarks of Kythera Holdings Ltd., an AbbVie company.
© 2025 AbbVie. All rights reserved.
Brand Information
| Brand name | Belkyra |
| Active ingredient | Deoxycholic acid |
| Schedule | S4 |
MIMS Revision Date: 01 April 2025
1 Name of Medicine
Deoxycholic acid.
2 Qualitative and Quantitative Composition
Belkyra solution for injection contains deoxycholic acid 10 mg/mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Belkyra (deoxycholic acid) injection is a formulation of synthetically derived deoxycholic acid in a sterile solution for subcutaneous injection. No human or animal-derived materials are used in the manufacture of synthetic deoxycholic acid.
Deoxycholic acid is a white to off-white crystalline powder and is freely soluble in alkaline solutions. The pKa and logP of deoxycholic acid have been determined at 25°C to be 6.0 and 4.1, respectively.
Belkyra injection is a clear, colourless liquid essentially free of visible particulates. Each single-use 2 mL vial contains 20 mg (10 mg/mL) of deoxycholic acid formulated in a sterile solution of sodium hydroxide, dibasic sodium phosphate, sodium chloride and water for injections. The formulation is adjusted to pH 8.3 with hydrochloric acid or sodium hydroxide and has a tonicity compatible with that of biological tissues and fluids.
4 Clinical Particulars
4.1 Therapeutic Indications
Belkyra (deoxycholic acid) injection is indicated for improvement in the appearance of moderate to severe convexity or fullness associated with submental fat in adults.
4.2 Dose and Method of Administration
Dosing considerations. Belkyra injection should be administered by a medical practitioner, who understands the relevant submental anatomy and associated neuromuscular structures and any alterations to the anatomy in a particular individual patient (e.g. due to prior surgical or aesthetic procedures) (see Section 4.4 Special Warnings and Precautions for Use).
Screen patients for other potential causes of submental convexity/fullness (e.g. thyromegaly and cervical lymphadenopathy).
Use caution in patients who have had prior surgical or aesthetic treatment of the submental area. Changes in anatomy/landmarks or the presence of scar tissue may impact the ability to safely administer Belkyra injection or to obtain the desired aesthetic result.
Recommended dose. Belkyra is injected into subcutaneous fat tissue in the submental area using an area-adjusted dose of 2 mg/cm2.
A single treatment consists of up to a maximum of 50 injections, 0.2 mL each (up to a total of 10 mL), spaced 1 cm apart.
Up to 6 single treatments may be administered at intervals no less than 1 month apart.
Administration. Belkyra injection is supplied in vials containing 2 mL of a 10 mg/mL solution and should be clear, colourless and free of particulate matter.
Visually inspect Belkyra vials for particulate matter and/or discolouration and discard the vial if the solution is discoloured and/or contains particulate matter.
Gently invert the vial several times prior to use. Do not dilute. After use, discard any remaining solution in the vial. Product is for single use in one patient only.
Injection technique. The safe and effective use of Belkyra injection depends on the use of the correct number and locations for injections, proper needle placement and administration techniques (see Section 4.4 Special Warnings and Precautions for Use).
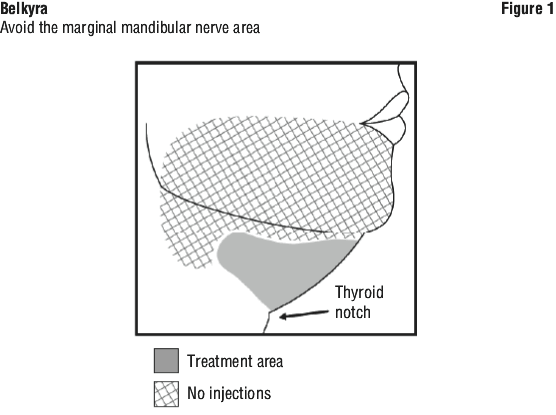
Avoid injections near the area of the marginal mandibular nerve (see Section 4.4 Special Warnings and Precautions for Use). Needle placement with respect to the mandible is very important as it reduces the potential for injury to the marginal mandibular nerve, a motor branch of the facial nerve. Injury to the nerve presents as an asymmetrical smile due to paresis of lip depressor muscles (see Section 4.4 Special Warnings and Precautions for Use).
To avoid injury to the marginal mandibular nerve:
do not inject above the inferior border of the mandible;
do not inject within a region defined by a 1-1.5 cm line below the inferior border (from the angle of the mandible to the mentum);
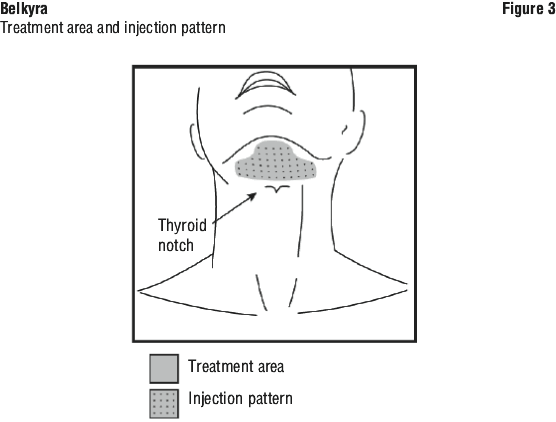
inject Belkyra only within the target submental fat treatment area (see Figures 1 and 3).
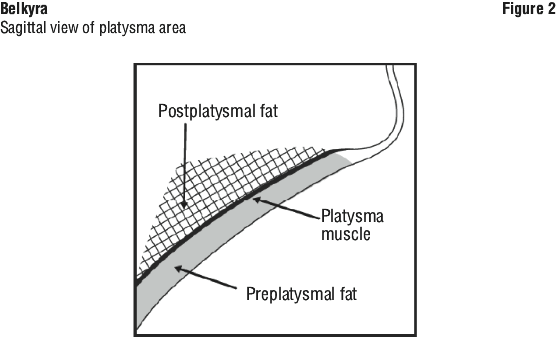
Outline the planned treatment area with a surgical pen and apply a 1 cm injection grid to mark the injection sites (see Figures 2 and 3).
Have the patient tense the platysma. Pinch the submental fat and using a 30 gauge (or smaller) 0.5-inch needle, inject 0.2 mL of Belkyra into the pre-platysmal fat (see Figure 2) next to each of the marked injection sites by advancing the needle perpendicular to the skin.
Injections that are too superficial (into the dermis) may result in skin ulceration and necrosis. Do not withdraw the needle from the subcutaneous fat during injection as this could increase the risk of intradermal exposure and potential skin ulceration and necrosis.
Avoid injecting into the post-platysmal fat by injecting Belkyra into fat tissue at the depth of approximately mid-way into the subcutaneous fat layer (see Figure 2).
If at any time resistance is met as the needle is inserted, indicating the possibility of contact with fascial or non-fat tissue, the needle must be withdrawn to an appropriate depth before the injection is administered.
Avoid injecting into other tissues such as the muscle, salivary glands (including salivary ducts), lymph nodes, arteries or veins.
Upon needle withdrawal, pressure may be applied to each injection site as necessary to minimise bleeding; an adhesive dressing may be applied.
4.3 Contraindications
Belkyra injection is contraindicated in patients who are hypersensitive to this drug or to any ingredient in the formulation or component of the container. For a complete listing, see Section 6.1 List of Excipients.
Belkyra injection is contraindicated in the presence of infection in the treatment area.
4.4 Special Warnings and Precautions for Use
General. To be administered only by subcutaneous route.
Do not inject within 1 cm of vulnerable anatomic structures. Efficacy and safety of Belkyra injection has not been established outside the submental fat area. Do not inject in any other area.
Efficacy and safety of Belkyra injection in patients with mild or extreme submental fat has not been established and is not recommended.
To achieve the efficacy and safety reported from the pre-market clinical trials, medical practitioners administering Belkyra injection must understand the relevant submental anatomy and associated neuromuscular structures; and any alterations to the anatomy in a particular individual patient (e.g. due to prior surgical or aesthetic procedures).
Marginal mandibular nerve injury. Belkyra should not be injected into or in close proximity to the marginal mandibular branch of the facial nerve to avoid the potential for motor neuropraxia, which manifests as an asymmetric smile or facial muscle weakness. The median time to resolution of marginal mandibular nerve injuries, which occurred at an incidence rate of 2.0% in the pre-market clinical trials, was 44 days; range: 1 to 298 days.
Dysphagia. Difficulty swallowing (dysphagia) occurred in the pre-market clinical trials in the setting of administration site reactions; for example, pain, swelling, and induration of the submental area. The median time to resolution of dysphagia, which occurred at an incidence rate of 1.1% in the pre-market clinical trials, was 3 days; range: 1 to 81 days.
Patients with current or prior history of dysphagia were excluded from clinical trials; avoid the use of Belkyra injection in such patients as treatment may exacerbate the condition.
Injection site haematoma/bruising. Belkyra injection should be used with caution in patients with bleeding abnormalities or who are currently being treated with anti-platelet or anti-coagulant therapy as excessive bleeding or bruising in the treatment area may occur. In pre-market clinical trials, 61% of people treated with Belkyra injection experienced injection site bleeding/haematoma.
Risk of injecting in proximity to vulnerable anatomic structures. Care should be taken to avoid inadvertent intradermal or intramuscular injection. Belkyra should be injected mid-way into the pre-platysmal subcutaneous fat tissue in the submental area. Injections that are too superficial (into the dermis) may result in skin ulceration and necrosis. Do not withdraw the needle from the subcutaneous fat during injection, as this could increase the risk of intradermal exposure and potential skin ulceration and necrosis. Cases of injection site infection have been reported, some of which included cellulitis and abscess requiring additional medical treatment. Consider withholding subsequent treatment with Belkyra injection until resolution of injection site ulceration, necrosis or infection.
Avoid injection into salivary glands (including salivary ducts), the thyroid gland, lymph nodes and muscles. Care should be taken to avoid inadvertent injection directly into an artery or vein as it can result in vascular injury.
Belkyra injection should not be administered into the periorbital area.
Pre-existing conditions/treatments at or near the treatment area. Patients should be screened for other potential causes of submental convexity/fullness (e.g. thyromegaly and cervical lymphadenopathy) prior to use of Belkyra injection.
Caution should be used when Belkyra injection is administered in the presence of inflammation or induration at the proposed injection site(s).
Caution should be used when Belkyra injection is administered in patients who have had prior surgical or aesthetic treatment of the submental area. Changes in anatomy/landmarks or the presence of scar tissue may affect the ability to safely administer Belkyra injection or to obtain the desired aesthetic result.
Caution should be used in patients with excessive skin laxity, prominent platysmal bands or other conditions for which reduction of submental fat may result in an aesthetically undesirable outcome.
Controlled sodium diet. Belkyra injection contains 184 micromol (or 4.23 mg) sodium per mL. This should be taken into consideration in patients on a controlled sodium diet.
Use in the elderly (> 65 years of age). The clinical trials with Belkyra injection did not include sufficient numbers of subjects over age 65 to determine whether they respond differently than younger subjects; therefore, caution should be exercised with these patients.
Paediatric use (< 18 years of age). Safety and effectiveness in patients below the age of 18 years have not been established and Belkyra injection is not intended for use in children or adolescents.
Effects on laboratory tests. Interactions with laboratory tests have not been established.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No clinical drug interaction studies have been conducted with Belkyra injection. The use of Belkyra injection concomitantly with botulinum toxin has not been evaluated.
Results from in vitro studies on inhibition and induction of cytochrome P450 (CYP) enzymes and inhibition of uptake and efflux transporters, including those involved in the enterohepatic circulation of bile acids, did not predict a potential for drug-drug interactions with Belkyra injection at clinically relevant doses.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no clinical data on fertility.
Deoxycholic acid did not affect general reproductive performance or fertility in male or female rats at subcutaneous doses up to 50 mg/kg once weekly, yielding 5 and 3 times the plasma AUC in subjects at the maximum recommended human dose in the respective sexes.
Use in pregnancy. (Category B1)
Embryofoetal development studies have been performed in rats at doses up to 50 mg/kg and in rabbits at doses up to times 30 mg/kg, administered subcutaneously on alternate days during the period of organogenesis. These doses yielded approximately 2 and 17 times the plasma AUC in subjects at the maximum recommended human dose in the respective species. They have revealed no evidence of harm to the foetus. No adequate and well-controlled studies in pregnant women have been performed. However, because animal reproduction studies are not always predictive of human response, Belkyra injection is not recommended for use during pregnancy.
Use in lactation. Endogenous deoxycholic acid has not been observed in human milk. Because studies in nursing mothers have not been conducted, Belkyra injection is not recommended for use by nursing women.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. It is presumed to be safe to drive and use machines while using Belkyra injection.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial experience. The data described in Table 1 reflects undesirable effects reported for Belkyra treated patients (N = 1118) who were evaluated in the clinical studies that assessed the use of Belkyra injection for the treatment of submental fat.
The following side effects have been evaluated in clinical studies with the following frequencies:
Very common (≥ 1/10); Common (≥ 1/100 to < 1/10); Uncommon (≥ 1/1,000 to < 1/100); Rare (≥ 1/10,000 to < 1/1,000); Very rare (< 1/10,000); Not known (cannot be estimated from the available data).
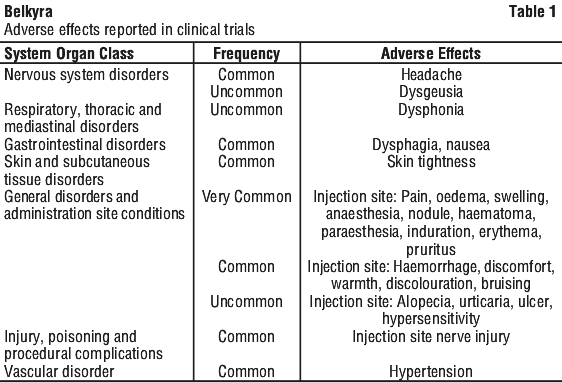
General disorders and administration site conditions. Injection site conditions such as alopecia in males, hypoaesthesia, ulceration and necrosis, injection site scar (secondary to skin ulceration or necrosis and post-injection scar tissue), injection site infection, injection site mass.
Immune system disorders. Hypersensitivity.
Nervous system disorders. Hypoaesthesia oral and paraesthesia oral.
Injury, poisoning and procedural complications. Vascular injury due to inadvertent intravascular injection.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions in Australia at www.tga.gov.au/reporting-problems and in New Zealand at pophealth.my.site.com/carmreportnz/s/.
4.9 Overdose
No overdosing with Belkyra injection in humans has been reported. Injection of increased volume or decreasing the spacing between injections of Belkyra may be expected to increase risk of local adverse effects. Non-treatment area or systemic adverse reactions were infrequent during clinical studies of doses up to 200 mg.
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia) or the National Poisons Centre on 0800 POISON (0800 764766 New Zealand).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Other dermatologicals; ATC code: D11AX.
Mechanism of action. Deoxycholic acid is a cytolytic drug which when injected into localised subcutaneous fat physically disrupts the cell membrane of adipocytes and causes cell lysis. The destruction of adipocytes elicits an expected tissue response in which macrophages are attracted to the area to eliminate cellular debris and lipids, which are then cleared through natural processes. This is followed by the appearance of fibroblasts and observed thickening of fibrous septa, likely due to an increase in total collagen (i.e. neocollagenesis).
Pharmacodynamics. In clinical dose ranging trials of submental fat reduction, higher efficacy was observed with Belkyra injection dosed at 2 mg/cm2 (based on clinician and patient rating scales, patient-reported outcomes and MRI measurements) compared with Belkyra injection dosed at 1 mg/cm2, with little or no difference in safety profile.
In clinical trials, no clinically significant effect of Belkyra injection was observed on circulating lipid levels or any other laboratory parameter.
A QT/QTc study was conducted and neither 100 mg Belkyra injection nor the supratherapeutic dose of 200 mg Belkyra injection demonstrated a propensity to prolong the ECG QT duration in healthy volunteers.
Clinical trials. Study demographics and trial design. Two identical Phase 3, randomised, multi-centre, double-blind, placebo-controlled trials of Belkyra injection for use in the improvement in the appearance of convexity or fullness associated with submental fat (SMF) were conducted in Canada and the United States as pivotal studies. Two supportive, identical Phase 3 studies of Belkyra injection for treatment of convexity or fullness associated with SMF were conducted in the European Union. The pivotal trials enrolled healthy adults (ages 19 to 65, BMI ≤ 40) with moderate or severe SMF (i.e. grade 2 or 3 on 5-point validated grading scales, where 0 = none, 4 = extreme), as judged by both clinician and patient ratings.
In the pivotal studies, subjects received up to six treatments with Belkyra injection (N = 515, combined trials) or placebo (N = 504, combined trials) at approximately 4-week intervals. Use of ice/cold packs, topical and/or injectable local anaesthesia was allowed during the clinical trials. Injection volume was 0.2 mL per injection site, spaced 1 cm apart into the submental fat tissue, which is expressed as dose per area as 2 mg/cm2. For each treatment session a maximum of 100 mg (10 mL or up to 50 injections) was permitted over the entire treatment area. The primary efficacy assessments were based on the composite clinician-reported (CR) and patient-reported (PR) ratings of SMF (concurrent improvement reported by both physician and patient) at 12 weeks after final treatment, relative to baseline. The trials employed co-primary endpoints: at least a 1-grade composite improvement, and at least a 2-grade composite improvement. As a secondary endpoint, visual and emotional impacts of SMF were evaluated using a 6-question survey (happy, bothered, self conscious, embarrassed, looking older or overweight in relation to the appearance of their SMF), with each question rated from 0 (not at all) to 10 (extremely/very much). In addition, magnetic resonance imaging (MRI) was performed in a subset of subjects (N = 449, combined trials) to confirm reduction of submental fat.
In these trials, the mean age was 49 years and the mean BMI was 29 kg/m2. Most of the subjects were women (85%) and Caucasian (87%). At baseline, 51% of the subjects had a clinician rated SMF severity of moderate and 49% had a SMF rating of severe.
In the pre-market clinical trials, Belkyra injection was administered by plastic surgeons, dermatologists and a small number of other medical practitioners with extensive experience in administration of aesthetic treatments.
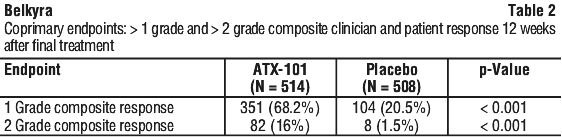
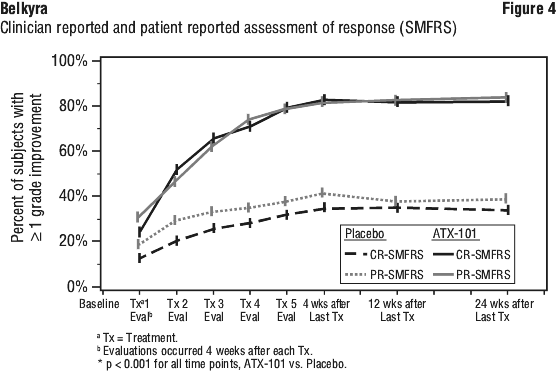
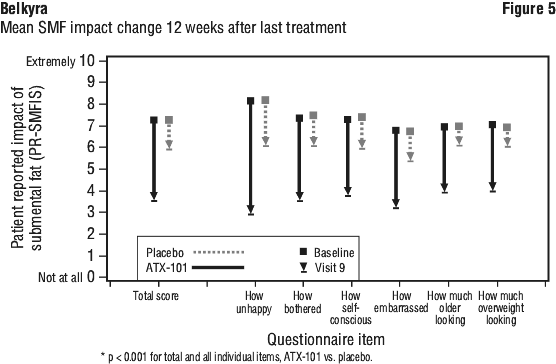
Pivotal trial results. Both 1-grade and 2-grade reductions in SMF were observed more frequently in the Belkyra injection group compared to the placebo group as measured by the composite clinician and patient ratings; 68.2% of Belkyra-treated subjects had at least a 1-grade composite Submental Fat Rating Scale (SMFRS) response compared to 20.5% of placebo-treated subjects. Sixteen percent (16.0%) of Belkyra treated subjects had at least a 2-grade composite SMFRS response compared to 1.5% of placebo-treated subjects (see Table 2). The individual clinician and patient assessments of response from which the composite response is derived are provided in Figure 4. The impact of SMF on emotional and self-perceived visual attributes showed a significant improvement in the Belkyra injection group compared to the placebo group as shown in Figure 5. Belkyra-treated patients dropped 48.6% from a baseline of 7.27 to 3.74, or -3.53, at 12 weeks after the last treatment measured on a scale of 0 (no impact at all) to 10 (extreme impact). Placebo-treated patients dropped 17.3% from a baseline of 7.28 to 6.02, or -1.26, at 12 weeks after the last treatment. Statistically significant improvements in subject satisfaction were observed more frequently in the Belkyra injection group compared to the placebo group as measured by the Subject Self Rating of Satisfaction scale. MRI measurements taken to confirm the reduction of submental fat indicate that a greater proportion of Belkyra-treated subjects had at least a 10% reduction in submental fat volume as compared to placebo-treated subjects (43% vs 5%, respectively).
5.2 Pharmacokinetic Properties
Endogenous deoxycholic acid plasma levels are highly variable within and between individuals; most of this natural bile component is sequestered in the enterohepatic circulation loop. Pharmacokinetics of exogenous deoxycholic acid administered via treatment with Belkyra injection was compared against this endogenous background.
Absorption. Deoxycholic acid from Belkyra injection is rapidly absorbed following subcutaneous injection. After dosing with the maximum recommended single treatment with Belkyra injection (100 mg), maximum plasma concentrations (mean Cmax) were observed within 30 minutes (median Tmax) after injection and mean Cmax values were 2- to 3-fold higher than average Cmax values observed during a 24-hour baseline endogenous period in the absence of Belkyra injection. After maximum recommended single treatment dose (100 mg), average deoxycholic acid exposure (AUC0-24) was less than 2-fold higher over endogenous exposure. Plasma AUC0-24 increased in a dose proportional manner up to 100 mg. Post-treatment deoxycholic acid plasma levels returned to the endogenous range within 24 hours. No accumulation is expected with the proposed treatment frequency.
Distribution. The volume of distribution of deoxycholic acid was estimated to be 193 L and is independent of the dose up to 100 mg. Deoxycholic acid is extensively bound to proteins in plasma (98%).
Metabolism and excretion. Endogenous deoxycholic acid is a product of cholesterol metabolism and is excreted intact in faeces. Deoxycholic acid from Belkyra injection joins the endogenous bile acid pool and is excreted along with the endogenous deoxycholic acid. Deoxycholic acid is eliminated via hepatic transport proteins from the blood to the bile without any significant contribution of metabolism.
Deoxycholic acid is not an in vitro inhibitor of the enzymes CYP1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6 and 3A4. Deoxycholic acid did not induce CYP1A, 2B6 and 3A at a clinical level.
Deoxycholic acid is not an in vitro inhibitor of the transporters BSEP, MRP2, MRP4, MDR1, BCRP, OATP1B1, OATP1B3, OAT1, OAT3, OCT1, OCT2, OATP2B1 and ASBT.
Deoxycholic acid inhibited NTCP with an IC50 of 2.14 microM in vitro.
Special populations and conditions. Hepatic impairment. Belkyra injection has not been studied in patients with hepatic impairment. Considering the intermittent dose frequency, the small dose administered that represents approximately 3% of the total bile acid pool, and the highly variable endogenous deoxycholic acid levels, the pharmacokinetics of deoxycholic acid following Belkyra injection is unlikely to be influenced by hepatic impairment.
Renal impairment. Belkyra injection has not been studied in patients with renal impairment. Bile acids including endogenous deoxycholic acid are excreted in the urine in negligible amounts; renal impairment is unlikely to influence deoxycholic acid pharmacokinetics.
5.3 Preclinical Safety Data
Genotoxicity. Deoxycholic acid was not genotoxic in a comprehensive battery of tests performed in vitro (microbial reverse mutation assay and chromosomal aberration test) and in in vivo (micronucleus test) genotoxicity assays.
Carcinogenicity. The carcinogenic potential of deoxycholic acid by the subcutaneous route has not been examined in long-term animal studies.
6 Pharmaceutical Particulars
6.1 List of Excipients
Dibasic sodium phosphate heptahydrate; sodium hydroxide; sodium chloride; hydrochloric acid and water for injections.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
30 months.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Belkyra deoxycholic acid 10 mg/mL solution for injection is supplied in single-use clear, colourless glass vials with a chlorobutyl rubber stopper coated with FluroTec (ETFE) on the plug surface and an aluminium seal with a polypropylene flip-top lid.
Pack size. One carton with four vials.
Each vial contains 2 mL solution for injection.
AUST R 233201.
6.6 Special Precautions for Disposal
Belkyra injection is for single use in one patient. Any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Belkyra injection contains deoxycholic acid as the active ingredient.
Chemical structure.
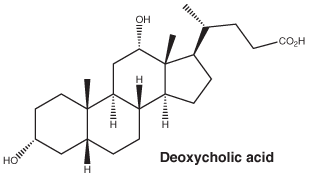
Molecular formula. C24H40O4.
Molecular mass. 392.57 g/mol.
CAS number. 83-44-3.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
21 July 2016
Date of Revision
12 February 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.