Bemfola
Brand Information
| Brand name | Bemfola |
| Active ingredient | Follitropin alfa |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Bemfola.
Summary CMI
Bemfola®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using BEMFOLA?
Bemfola contains the active ingredient follitropin alfa. In women, Bemfola is used to bring about the development of follicles in women who are not ovulating and who have not responded to treatment with clomiphene citrate. Bemfola is also used to stimulate the development of several follicles in women undergoing assisted reproductive technologies (ART) such as in vitro fertilisation. Bemfola can be used together with another hormone called Luteinising Hormone (LH) to stimulate development of follicles in women who have been shown to produce very low levels of some of the hormones involved in the natural reproductive cycle. In men, Bemfola is used in combination with human chorionic gonadotrophin (hCG) to stimulate the production of sperm.
For more information, see Section 1. Why am I using Bemfola? in the full CMI.
2. What should I know before I use BEMFOLA?
Do not use if you have ever had an allergic reaction to Bemfola or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, are pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Bemfola? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Bemfola and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use BEMFOLA?
- Your doctor will decide the correct dose of Bemfola for you. Your dose may be adjusted depending on your individual response to treatment.
- Follow all directions given to you by your doctor, nurse or pharmacist carefully, including the Instructions for Use provided in the pack. Do not self-inject until you are sure of how to do it.
More instructions can be found in Section 4. How do I use Bemfola? in the full CMI and in the Instructions for Use leaflet inside the pack.
5. What should I know while using BEMFOLA?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Bemfola? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. Most common side effects include injection site reactions, such as redness, pain and swelling at the site of injection. In women, common side effects may include headache, dizziness, abdominal pain, discomfort, nausea, vomiting and diarrhoea. In men, common side effects may include acne, weight gain and breast development.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Bemfola®
Active ingredient: follitropin alfa (rch)
Consumer Medicine Information (CMI)
This leaflet provides important information about using Bemfola. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Bemfola.
Where to find information in this leaflet:
1. Why am I using Bemfola?
2. What should I know before I use Bemfola?
3. What if I am taking other medicines?
4. How do I use Bemfola?
5. What should I know while using Bemfola?
6. Are there any side effects?
7. Product details
1. Why am I using BEMFOLA?
Bemfola contains the active ingredient follitropin alfa (recombinant human follicle stimulating hormone). This hormone is similar to follicle stimulating hormone found naturally in humans, but it is made by means of biotechnology. Follicle stimulating hormone belongs to the family of hormones called gonadotrophins and is necessary for the growth and development of egg cells in women and sperm production in men.
Bemfola is an approved biosimilar to the reference product Gonal-f®. Comparability in safety, efficacy and quality between Bemfola and Gonal-f® have been established.
In women, Bemfola is used to bring about the development of follicles in women who are not ovulating and who have not responded to treatment with clomiphene citrate. Bemfola is also used to stimulate the development of several follicles in women undergoing assisted reproductive technologies (ART) such as in vitro fertilisation.
Bemfola can be used together with another hormone called Luteinising Hormone (LH) to stimulate development of follicles in women who have been shown to produce very low levels of some of the hormones involved in the natural reproductive cycle.
In men, Bemfola is used in combination with human chorionic gonadotrophin (hCG) to stimulate the production of sperm.
2. What should I know before I use BEMFOLA?
Warnings
Do not use Bemfola if:
- you are allergic to follitropin alfa, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have tumours of the hypothalamus or pituitary gland.
In women, do not use Bemfola if:
- you are pregnant
- you are breastfeeding
- your ovaries are enlarged
- you have unexplained ovarian cyst
- you have unexplained vaginal or uterine bleeding
- you have cancer of the ovaries, uterus or breasts
- your ovaries have failed
- you have fibroids in your uterus which would make pregnancy impossible.
In men, do not use Bemfola if:
- you have increased levels of gonadotrophins indicating failure of testes
- your infertility is due to disorders other than hypogonadotrophic hypogonadism (low levels of gonadotrophins).
Check with your doctor if you:
- have or have had any other medical conditions, such as:
- thyroid problems
- adrenal problems
- high prolactin levels in the blood
- porphyria or a family history of porphyria. - take any medicines for any other condition.
Tell your doctor if you or a family members have or have had signs of blood clots (e.g. pain, warmth, redness, numbness or tingling in the arm or leg). Treatment with Bemfola and hCG may increase the risk of blood clots forming in your blood vessels.
Your doctor will assess you and your partner's infertility. This may include tests for other medical conditions, which may interfere with your ability to become pregnant. If necessary, other medical conditions may be treated before starting infertility treatments with Bemfola.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant while using Bemfola.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Multiple pregnancies carry risks for mothers and babies. Compared to natural conception, the frequency of multiple pregnancies and births is higher in patients receiving treatments that stimulate follicle growth for ovulation induction. Your doctor will monitor your response to minimise the chance of multiple pregnancies.
Compared to natural conception, the frequency of pregnancy loss is higher in patients undergoing fertility treatments.
There may be a slightly increased risk of birth defects in women using assisted reproductive technologies. This may be due to increased maternal age, genetic factors, multiple pregnancies or the procedure.
For men
- Elevated follicle stimulating hormone blood levels are indicative of testicular failure. Your doctor may ask you for a semen analysis to assess your response to treatment.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Bemfola and affect how it works.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Bemfola.
4. How do I use BEMFOLA?
Treatment with Bemfola should be started under the supervision of a specialist doctor experienced in the treatment of infertility.
How much to use
- Always use Bemfola exactly as your doctor has told you. You should check with your doctor if you are unsure.
- Your doctor will decide the correct dose of Bemfola for you depending on your condition.
- For women who are not ovulating, the common dose usually starts at 75 IU daily. The dose may be adjusted as your treatment progresses. The maximum daily dose is usually not higher than 225 IU.
- For women undergoing assisted reproductive technologies, the common dose usually starts at 150 IU daily. The dose may be adjusted as your treatment progresses. The maximum daily dose is usually not higher than 450 IU.
- For women who are not ovulating and have been diagnosed with very low levels of FSH and LH hormones, the common dose usually starts at 75 IU to 150 IU BEMFOLA together with 75IU lutropin alfa daily.
- For men, the common dose usually starts at 150 IU three times a week.
- Your dose may be adjusted depending on your individual response to treatment.
- Follow the instructions provided by your doctor or nurse carefully.
When to use Bemfola
- Your doctor will tell you when to use Bemfola. The length of your treatment will be determined by your doctor depending on your condition.
How to use Bemfola
- Bemfola is given as a subcutaneous (under your skin) injection in the lower abdominal area or thigh.
- The injection site should be changed daily to lessen possible injection site reactions.
- Before using Bemfola, your doctor or nurse can teach you the injection technique. Your partner may be trained to give the injection at home.
- Read the Instructions for Use provided in the pack before commencing injections. Do not self-inject until you are sure of how to do it.
If you forget to use Bemfola
If you forget an injection or are not sure what to do, contact your doctor or nurse immediately for advice.
Do not inject double dose to make up for the dose you missed.
If you use too much Bemfola
If you think that you have used too much Bemfola, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using BEMFOLA?
Things you should do
See your doctor regularly. Your doctor will monitor you closely throughout your treatment.
Tell your doctor immediately if you become pregnant while using Bemfola.
If you plan to have surgery, tell your doctor or dentist that you are using Bemfola.
Remind any doctor, dentist or pharmacist you visit that you are using Bemfola.
Things you should not do
- Do not stop using this medicine suddenly without telling your doctor.
- Do not change the dose unless your doctor tells you to.
- Do not give this medicine to anyone else even if they have the same condition as you.
Ovarian Hyperstimulation Syndrome (OHSS)
Treatment with Bemfola may increase your risk of developing a condition called ovarian hyperstimulation syndrome (OHSS). This is when the ovaries overreact to the hormonal treatment and become larger.
The most common symptom is lower abdominal pain. During stimulation your doctor will monitor your treatment by using ultrasound and/or blood tests to help determine if you are likely to develop OHSS. If necessary, your doctor will delay or cancel your Bemfola injection. You may also be advised to refrain from sexual intercourse until the end of the cycle if this occurs.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Bemfola affects you.
Bemfola may cause dizziness in some people.
Looking after your medicine
- Prior to use, store Bemfola in the original package at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light.
- Before opening and within its shelf-life, the medicine may be removed from the refrigerator, without being refrigerated again, for upto 3 months kept at or below 25°C.
- The product if stored at or below 25°C, must be discarded if it has not been used after 3 months.
Discard used pen and needle immediately after injection.
Follow the instructions in the carton on how to take care of your medicine properly.
Do not use Bemfola if the solution contains particle or is not clear.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Injection site related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Symptoms of allergic reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems.
In New Zealand, you can report side effects to Medsafe online at pophealth.my.site.com/carmreportnz/s/.
By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Bemfola contains
| Active ingredient (main ingredient) | Follitropin alfa (rch) |
| Other ingredients (inactive ingredients) | Sucrose Dibasic sodium phosphate dihydrate Monobasic sodium phosphate monohydrate Phosphoric acid Poloxamer Methionine Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Bemfola looks like
Bemfola contains sterile solution for injection in a cartridge, pre-assembled in a pen.
Bemfola Pen Injector is pre-filled with a clear, colourless solution of 0.125 mL (75 IU), 0.25 mL (150 IU), 0.375 mL (225 IU), 0.5 mL (300 IU) or 0.75 mL (450 IU).
Bemfola Pen Injector is available in five strengths:
- 75 IU/0.125 mL (5.5 micrograms/0.125 mL) (AUST R 231039);
- 150 IU/0.25 mL (11 micrograms/0.25 mL) (AUST R 231046);
- 225 IU/0.375 mL (16.5 micrograms/0.375 mL) (AUST R 231051);
- 300 IU/0.50 mL (22 micrograms/0.5 mL) (AUSTR 231052); and
- 450 IU/0.75 mL (33 micrograms/0.75 mL) (AUSTR 231 053).
Pack sizes of 1, 5 and 10 pre-filled pens. Not all pack sizes may be marketed. Each pack also contains the corresponding number of needles to be used with the pen for administration.
Bemfola Pens are not designed to allow the cartridge to be removed.
Who distributes Bemfola is supplied in Australia by:
Gedeon Richter Australia Pty Ltd
Suite 902, 15 Blue Street
North Sydney NSW 2060
Australia
Phone number: 1300 GEDEON (1300 433 366)
Bemfola is supplied in New Zealand by:
Pharmacy Retailing (NZ) Ltd t/a Healthcare Logistics
58 Richard Pearse Drive
Airport Oaks Mangere
AUCKLAND
Phone: 0800 GEDEON (0800 433 366)
This leaflet was prepared in June 2024.
Instructions for Use
CONTENTS
1. How to use the Bemfola pre-filled pen
2. Before you start using your pre-filled pen
3. Getting your pre-filled pen ready for injection
4. Setting the dose
5. Injecting the dose
6. After the injection
- How to use the Bemfola pre-filled pen
- Before starting to use your pre-filled pen, please read these instructions the whole way through first.
- Only use this pen for you – do not let anyone else use it.
- The numbers on the dose display are measured in International Units or IU. Your doctor will have told you how many IU to inject each day.
- Your doctor/pharmacist will tell you how many Bemfola pens you need to use for your complete treatment course.
- Give yourself the injection at the same time each day.
- Before you start using your pre-filled pen
2.1. Wash your hands
- It is important that your hands and the things you use to get your pen ready are as clean as possible.
2.2. Find a clean area
- A good place is a clean table or surface.
- Getting your pre-filled pen ready for injection
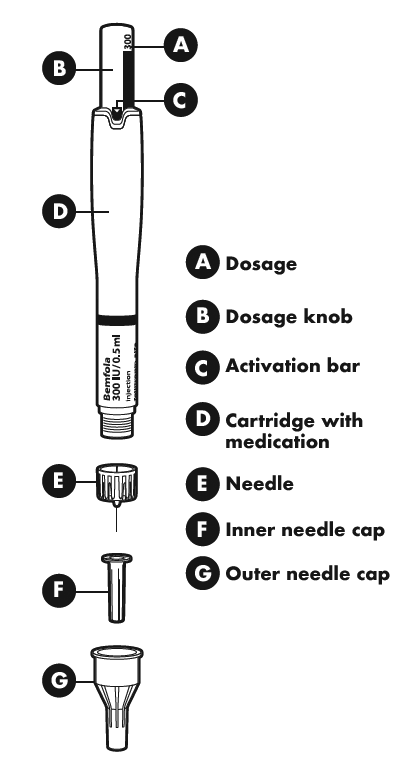
The different parts of your pre-filled pen
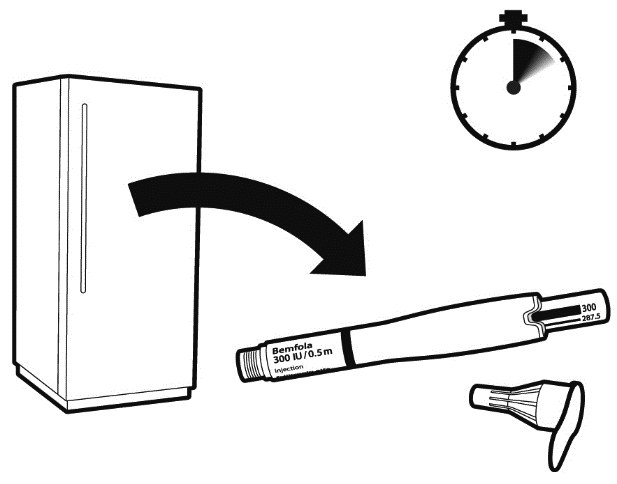
Perform the injection every day around the same time. Take the pen out of the fridge 5 to 10 minutes before using it.
Note: Please check that the medicine is not frozen.
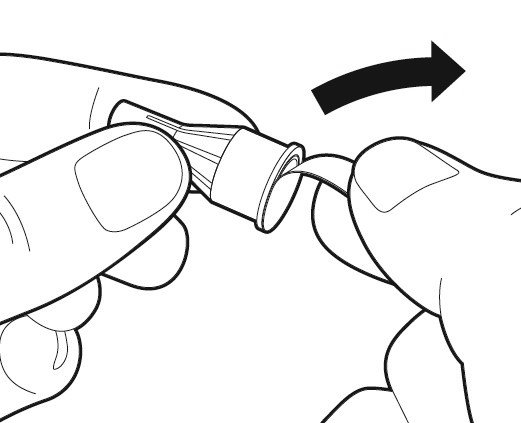
Prepare your needle for injection
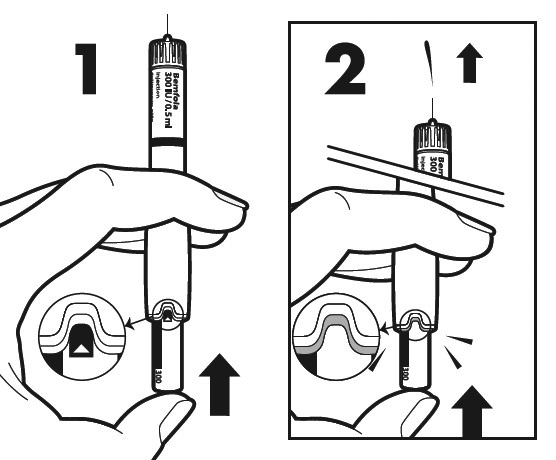
Get a new needle - only use the single-use needles supplied in the package. Hold the outer needle cap firmly.
Check in all cases that the peel-off seal on the outer needle cap is not damaged or loose.
Remove the peel-off seal from the injection needle.
Caution: If the peel-off seal is damaged or loose, do not use the needle. Throw it away in a sharps disposal container. Get a new needle.
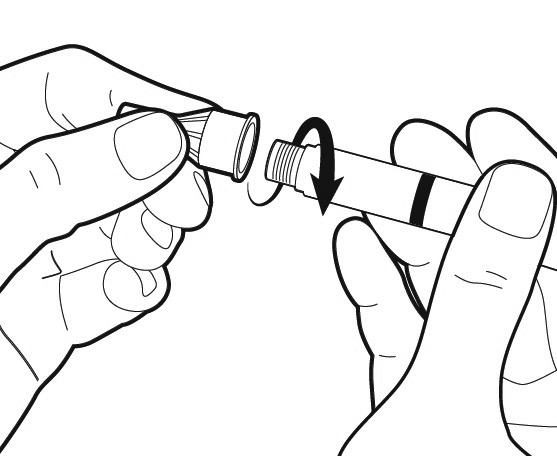
Attach the needle.
Hold the pen by its sides and keep the needle in line with it while attaching the needle. Screw the needle clockwise onto the tip of the pen all the way until firmly attached. Make sure that the needle is properly secured on the pen in a straight position.
Caution: Do not overtighten the needle. Do not push the dosage knob in while attaching the needle.
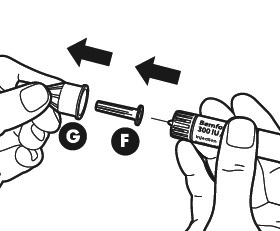
Remove the outer needle cap (G). Keep it for later. You will need it after the injection to discard the pen.
Remove the inner needle cap (F).
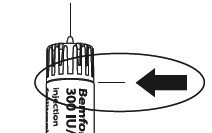
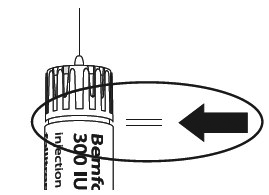
Make sure that the needle is in the correct position.
Correct position of the needle
Incorrect position of the needle
- Setting the dose prescribed by your doctor
First, hold the pen so that the needle is pointing upwards. To remove the air bubbles from the system, gently tap the side of the pen to make the any existing air bubbles rise to the top.
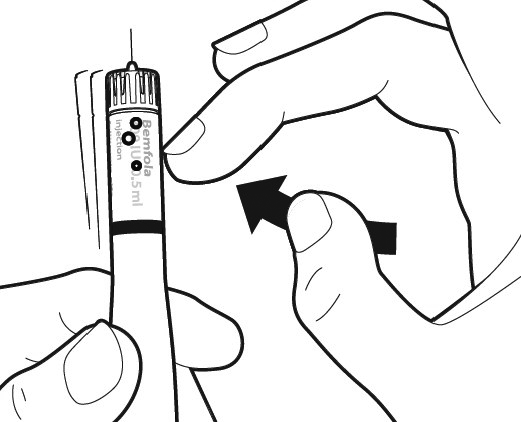
Still holding the pen upright, push the dosage knob in until the activation bar with the small arrow disappears. You should also hear a click, and some liquid will splash out (this is normal).The pen is now ready to set the dose.
Caution: If no liquid splashes out, or liquid leakage is detected at the junction of the needle and the pen, the pen should not be used. Tell your doctor or pharmacist if you notice any problems.
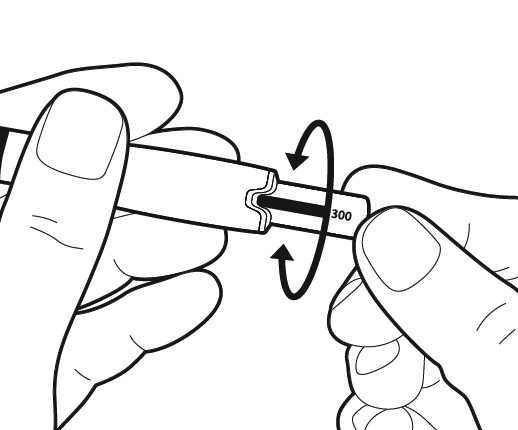
Gently turn the dosage knob until the bar of your prescribed dose is in line with the middle of the indent on the pen.
Note: For the Bemfola 75 IU/0.125 mL pre-filled pen, the dosage knob cannot go around completely, but it can still be rotated backwards.
The pen is now ready for injection.
Caution: Do not push the dosage knob in any further, at this point.
- Injecting the dose
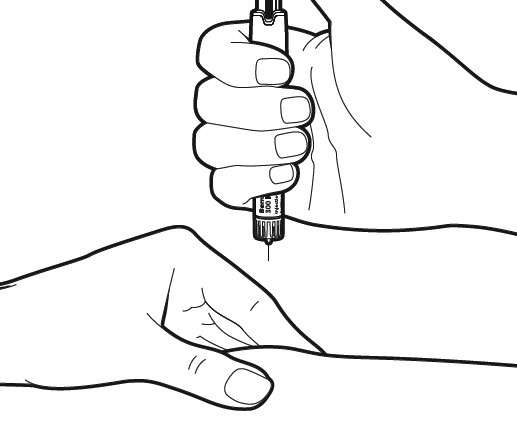
Now you are ready to immediately give yourself the injection: Your doctor or nurse will have already advised you where to inject (e.g. tummy, front of thigh). To minimise skin irritation, select a different injection site each day.
Check again that the correct dose is displayed on the pen. Lightly pinch the skin of the injection area. Hold the pen at approximately a right angle (900 angle) and insert the needle completely in one steady movement into your skin.
Caution: Do not push the dosage knob, while inserting the needle and do not change the direction of the needle as it goes in.
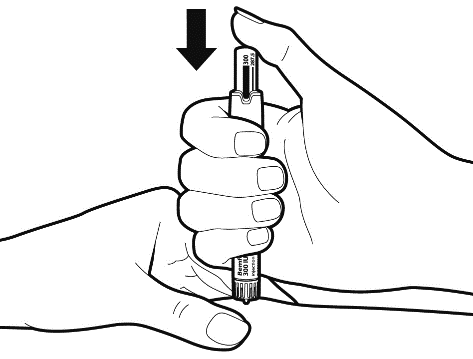
Once the needle is fully inserted in the injection site, push in the dosage knob slowly and continuously until it stops, and the bar of the set dose has disappeared.
Do not remove the needle immediately, wait at least for 5 seconds, before pulling it out to ensure you have injected the full dose.
Caution: If liquid leakage is detected at the junction of the needle and the pen during injecting, tell your doctor or pharmacist.
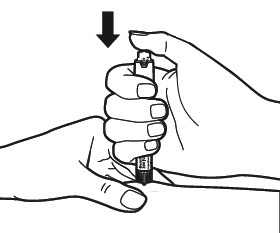
- After the injection
Replace the outer needle cap onto the needle carefully.
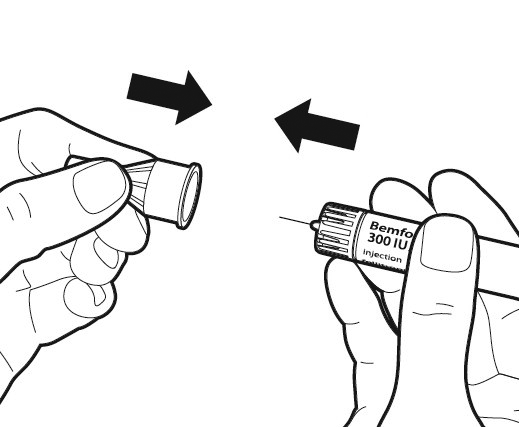
You can use the pre-filled injection pen only once and must discard the pen even if any liquid remains in the pen after injection.
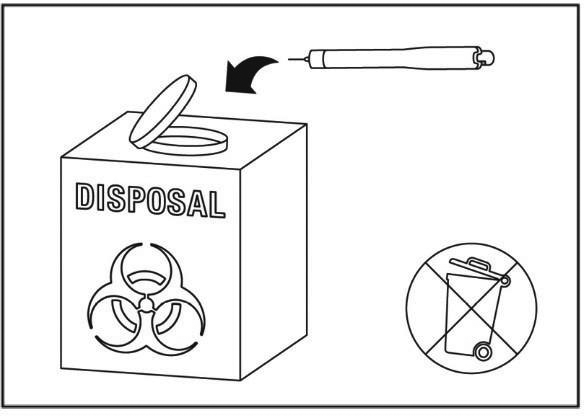
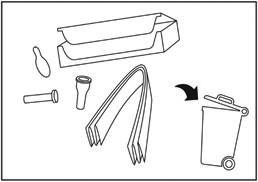
Throw away the packaging box, inner needle cap, peel tab and the Instructions for use in the normal household waste. Do not throw away any medicines via your sink, toilet or in your household waste. The used pen needs to be discarded in a sharps container and returned to the pharmacy for a correct disposal. Ask your pharmacist how to dispose of medicines you no longer use.
Brand Information
| Brand name | Bemfola |
| Active ingredient | Follitropin alfa |
| Schedule | S4 |
MIMS Revision Date: 01 December 2022
1 Name of Medicine
Follitropin alfa (rch).
2 Qualitative and Quantitative Composition
Bemfola is a biosimilar medicinal product, i.e. a medicine that has been demonstrated to be similar in quality, safety and efficacy to the reference medicinal product Gonal-f.
Bemfola contains the active ingredient follitropin alfa (rch). This is produced by a Chinese Hamster Ovary cell line transfected with the human FSH subunit genes (i.e. by recombinant DNA technology).
Clear glass cartridges containing Bemfola solution for injection are designed for subcutaneous injection pre-assembled in a disposable pen. Bemfola is available as solution for injection, containing follitropin alfa (rch) 75 IU/0.125 mL (5.5 microgram/0.125 mL), 150 IU/0.25 mL (11 microgram/0.25 mL), 225 IU/0.375 mL (16.5 microgram/0.375 mL), 300 IU/0.5 mL (22 microgram/0.5 mL) or 450 IU/0.75 mL (33 microgram/0.75 mL) in pre-filled pens.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Bemfola is a clear, colourless solution for injection for subcutaneous injection pre-assembled in a disposable pen.
4 Clinical Particulars
4.1 Therapeutic Indications
In adult women. Bemfola is indicated for the treatment of anovulatory infertility in women who have been unresponsive to clomiphene citrate or where clomiphene citrate is contraindicated.
Controlled ovarian hyperstimulation in women undergoing assisted reproductive technologies.
Bemfola in association with a luteinising hormone (LH) preparation is recommended for the stimulation of follicular development in women with severe LH and FSH deficiency. In clinical trials these patients were defined by an endogenous serum LH level < 1.2 IU/L.
In adult men. Bemfola is indicated with concomitant human chorionic gonadotrophin (hCG) therapy for the stimulation of spermatogenesis in gonadotrophin deficient men in whom hCG alone is ineffective.
4.2 Dose and Method of Administration
Treatment with Bemfola should be initiated under the supervision of a physician experienced in the treatment of fertility disorders.
Bemfola should be administered subcutaneously. The injection site should be alternated daily to prevent lipoatrophy. Self administration of Bemfola should only be performed by patients who are well motivated, adequately trained and who have access to expert advice.
The solution should not be administered if it contains particles or is not clear.
Women with anovulatory infertility (WHO group II). The objective of Bemfola therapy is to develop a single mature Graafian follicle from which the ovum will be liberated after the administration of hCG.
Bemfola may be given as a course of daily injections. In menstruating patients treatment should commence within the first 7 days of the menstrual cycle. Treatment should be tailored to the individual patient's response as assessed by measuring 1) follicle size by ultrasound and/or 2) oestrogen secretion. A commonly used regimen commences at 75-150 IU (5.5 to 11 microgram) FSH daily and is increased in increments of 37.5 IU (2.75 microgram) up to 75 IU (5.5 microgram) at 7 or 14 day intervals if necessary, to obtain an adequate, but not excessive response. If a patient fails to respond adequately after 5 weeks of treatment, that cycle should be abandoned. The Bemfola Pen provides treating physicians with capability of 12.5 IU increments to adapt patient's dose with flexibility.
When an optimal response is obtained, a single injection of 250 microgram r-hCG or 5000 IU up to 10,000 IU urinary human chorionic gonadotropin (u-hCG) should be administered 24-48 hours after the last Bemfola injection. The patient is recommended to have coitus on the day of, and the day following hCG administration.
If an excessive response is obtained, treatment should be stopped and hCG withheld (see Section 4.4 Special Warnings and Precautions for Use). Treatment should recommence in the next cycle at a dosage lower than that of the previous cycle.
Women undergoing assisted reproductive technologies. A commonly used regimen for superovulation involves the administration of 150 IU (11 microgram) to 225 IU (16.5 microgram) of Bemfola daily, commencing on days 2 or 3 of the cycle. Treatment is continued until adequate follicular development has been achieved (as assessed by monitoring of serum oestrogen concentrations and/or ultrasound examination), with the dose adjusted according to the patient's response, to usually not higher than 450 IU (33 microgram) daily.
A single injection of 250 microgram r-hCG or 5000 IU up to 10,000 IU u-hCG is administered 24-48 hours after the last Bemfola injection to induce final follicular maturation. In clinical trials, final follicular maturation was judged to be when at least two follicles were ≥ 16 mm mean diameter and when E2 levels were within the physician's acceptable range for the number of follicles present.
Downregulation with either a GnRH agonist or antagonist is now commonly used in order to suppress the endogenous LH surge and to control tonic levels of LH. Dosage regimes should be customised in order to achieve the desired result. In a commonly used protocol Bemfola is started approximately 2 weeks after the start of agonist treatment, both being continued until adequate follicular development is achieved. For example, following two weeks treatment with an agonist, 225 IU (16.5 microgram) Bemfola is administered (subcutaneously) for the first 7 days. The dose is then adjusted according to the ovarian response.
Women with anovulation resulting from severe LH and FSH deficiency. In LH and FSH deficient women (hypogonadotrophic hypogonadism), the objective of Bemfola therapy in association with lutropin alfa is to develop a single mature Graafian follicle from which the oocyte will be liberated after the administration of human chorionic gonadotropin (hCG). Follitropin alfa should be given as a course of daily injections simultaneously with lutropin alfa. Since these patients are amenorrhoeic and have low endogenous oestrogen secretion, treatment can commence at any time.
A recommended regimen commences at 75 IU of lutropin alfa daily with 75-150 IU FSH. Treatment should be tailored to the individual patient's response as assessed by measuring follicle size by ultrasound and oestrogen response.
If an FSH dose increase is deemed appropriate, dose adaptation should preferably be after 7-14 day intervals and preferably by 37.5-75 IU increments. It may be acceptable to extend the duration of stimulation in any one cycle to up to 5 weeks.
When an optimal response is obtained, a single injection of 250 microgram r-hCG or 5,000 IU up to 10,000 IU hCG should be administered 24-48 hours after the last follitropin alfa and lutropin alfa injections. The patient is recommended to have coitus on the day of, and on the day following, hCG administration.
Alternatively, IUI may be performed.
Luteal phase support may be considered since lack of substances with luteotrophic activity (LH/hCG) after ovulation may lead to premature failure of the corpus luteum.
If an excessive response is obtained, treatment should be stopped and hCG withheld. Treatment should recommence in the next cycle at a dose of FSH lower than that of the previous cycle.
Men with hypogonadotrophic hypogonadism. Prior to combined therapy with Bemfola and hCG, pretreatment should begin with hCG alone at the appropriate dosage to achieve masculinisation and serum testosterone level within the eugonadal range (> 9-10 nanomol/L). This starting dose should be increased to the necessary dosage in order to obtain normal testosterone values. If after an inadequate trial of hCG alone (usually 6 months) at effective doses, Bemfola should be given concomitantly at the dosage of 150 IU (11 microgram) three times a week. This regimen should be continued for a minimum of 4 months. If after this period, the patient has not responded, the combination treatment (hCG plus Bemfola 150 IU (11 microgram) 3 times a week) may be continued. Current clinical experience indicates that prolonged treatment for up to 18-24 months may be necessary to achieve spermatogenesis or fertility.
4.3 Contraindications
Bemfola is contraindicated for safety reasons in:
cases of prior hypersensitivity to follitropin alfa, or to any excipients of Bemfola;
tumours of the hypothalamus or pituitary gland.
FSH therapy is contraindicated for safety reasons where the following exist:
In women. Pregnancy and lactation;
ovarian enlargement or ovarian cyst of unknown aetiology;
gynaecological haemorrhages of unknown aetiology;
ovarian, uterine or breast carcinoma.
FSH is contraindicated when an effective response cannot be obtained, such as:
In women. Primary ovarian failure as indicated by high levels of FSH (ovarian dysgenesis, premature menopause);
malformations of sexual organs incompatible with pregnancy;
fibroid tumours of the uterus incompatible with pregnancy.
In men. Elevated gonadotrophin levels that indicate primary testicular failure;
infertility disorders other than hypogonadotrophic hypogonadism.
Use in elderly and children. Bemfola should not be used in the elderly or children.
4.4 Special Warnings and Precautions for Use
Bemfola is a potent gonadotrophic substance capable of causing mild to severe adverse reactions, and should only be used by physicians who are thoroughly familiar with infertility problems and their management.
Gonadotrophin therapy requires a certain time commitment by physicians and supportive health professionals, as well as the availability of appropriate monitoring facilities. In women, safe and effective use of Bemfola calls for monitoring of ovarian response with ultrasound, alone or preferably in combination with measurement of serum oestradiol levels, on a regular basis. There may be a degree of interpatient variability in response to FSH administration, with a poor response to FSH in some patients and exaggerated response in others. The lowest effective dose in relation to the treatment objective should be used in both men and women.
Before starting treatment, the couple's infertility should be assessed as appropriate and putative contraindications for pregnancy evaluated. In particular, patients should be evaluated for hypothyroidism, adrenocortical deficiency, hyperprolactinaemia, and appropriate specific treatment given.
Bemfola should be used with caution in patients with known hypersensitivity to gonadotrophin presentations that do not contain FSH, due to the risk of cross sensitivity. The first injection of Bemfola in such patients must be performed under direct medical supervision.
Self administration of Bemfola should only be performed by patients who are well motivated, adequately trained and with access to expert advice. During training of the patient for self administration, special attention should be given to specific instructions for the use of the prefilled pen.
Treatment in women. Patients should be selected carefully according to the following guidelines: a thorough gynaecological and endocrinological evaluation must be performed; presence of early pregnancy should be ruled out; aetiology of any abnormal vaginal bleeding should be established before starting Bemfola therapy; evaluation of semen quality of the partner should be performed; or other appropriate investigations should be performed as required.
Ovarian hyperstimulation syndrome (OHSS). Mild to moderate uncomplicated ovarian enlargement, which may be accompanied by abdominal distension and/or abdominal pain, occurs in approximately 20% of those treated with follitropin and hCG, and generally regresses without treatment within two or three weeks. In the presence of marked ovarian enlargement, treatment should be discontinued.
Patients undergoing superovulation are at an increased risk of developing ovarian hyperstimulation syndrome (OHSS) in view of the excessive oestrogen response and multiple follicular development. Distinct from uncomplicated ovarian enlargement, OHSS is a condition that can manifest itself with increasing degrees of severity. It comprises marked ovarian enlargement, high serum sex steroids and an increase in vascular permeability which can result in an accumulation of fluid in the peritoneal, pleural and, rarely, in the pericardial cavities.
OHSS can become a serious complication of human gonadotrophin therapy and sometimes leads to fatal complications if not adequately treated.
Mild manifestations of OHSS include abdominal pain, abdominal discomfort and distension, and enlarged ovaries. Moderate OHSS may additionally present with nausea, vomiting, ultrasound evidence of ascites and marked ovarian enlargement. Severe OHSS further includes symptoms such as severe ovarian enlargement, weight gain, dyspnoea or oliguria. Clinical evaluation may reveal hypovolaemia, haemoconcentration, electrolyte imbalances, ascites, pleural effusions or acute pulmonary distress. Rarely, severe OHSS may be complicated by ovarian torsion or thromboembolic events, such as pulmonary embolism, ischaemic stroke or myocardial infarction.
Independent risk factors for developing OHSS have been reported to include young age, lean body mass, polycystic ovarian syndrome (PCOS), higher doses of exogenous gonadotrophins, high absolute or rapidly rising serum oestradiol levels and previous episodes of OHSS, large number of developing ovarian follicles and large number of oocytes retrieved in ART cycles.
Careful monitoring of ovarian response with ultrasound alone or preferably in combination with measurement of oestradiol levels is recommended prior to and during stimulation therapy, especially in patients with PCOS.
OHSS may progress rapidly (within 24 hours) or over several days to become a serious medical event. It most often occurs after treatment with follitropin or hCG has been discontinued, reaching its maximum at about seven to ten days following treatment. Therefore, patients should be followed for at least two weeks after follitropin or hCG administration.
If there are any symptoms or signs of OHSS, the patient must be evaluated, investigated, and monitored. Adherence to recommended Bemfola dosage and regimen of administration can minimise the risk of OHSS. Monitoring of stimulation cycles by ultrasound scans as well as oestradiol measurements are recommended to identify risk factors early.
Excessive oestrogenic response seldom gives rise to significant hyperstimulation unless hCG is administered to induce ovulation. Cases of OHSS are more common, more severe and more protracted if pregnancy occurs. Therefore, if signs of OHSS occur, it is recommended that hCG be withheld and the physician should advise the patient to refrain from intercourse for at least 4 days. Intercourse should be prohibited in those patients in whom significant ovarian enlargement occurs after ovulation because of the danger of haemoperitoneum resulting from ruptured ovarian cysts.
Mild or moderate OHSS requires careful monitoring and may resolve spontaneously. Worsening of symptoms suggests progression of OHSS and requires prompt clinical reassessment. If necessary, the physician should recommend cessation of treatment or withholding hCG injection, and closely monitor the ovarian response. Severe OHSS requires admission to hospital and commencement of appropriate therapy in addition to cessation of gonadotrophins treatment. Treatment of OHSS is primarily symptomatic, consisting of bed rest, fluid and electrolyte management, and analgesics if needed.
The phenomenon of haemoconcentration associated with fluid loss into the peritoneal cavity, pleural cavity and pericardial cavity has been seen to occur and should be thoroughly assessed in the following manner: 1) fluid intake and output, 2) weight, 3) haematocrit, 4) serum and urinary electrolytes, 5) urine specific gravity, 6) BUN and creatinine, and 7) abdominal girth. These determinations are to be performed daily or more often if the need arises. Appropriate imaging examination, especially ultrasound, should also be used for identifying, localising and quantifying fluid loss.
There is an increased risk of injury to the ovary with OHSS. The ascitic, pleural and pericardial fluids should not be removed unless absolutely necessary to relieve symptoms such as pulmonary distress or cardiac tamponade. Pelvic examination may cause rupture of an ovarian cyst, which may result in haemoperitoneum and should therefore be avoided. If this does occur, and if bleeding becomes such that surgery is required, the surgical treatment should be designed to control bleeding and to retain as much ovarian tissue as possible.
Thromboembolic events. Thromboembolic events, including thrombophlebitis, pulmonary embolism, stroke and arterial occlusion both in association with, and separate from OHSS, have been reported following gonadotrophin therapy. In rare cases, thromboembolic events have resulted in death.
In women with recent or ongoing thromboembolic disease or women with generally recognised risk factors for thromboembolic events, such as personal or family history, treatment with gonadotrophins may further increase the risk for aggravation or occurrence of such events. In these women, the benefits of gonadotrophin administration need to be weighed against the risks. It should be noted, however, that pregnancy itself, as well as OHSS, also carries an increased risk of thromboembolic events.
Multiple pregnancies. In patients undergoing induction of ovulation, the incidence of multiple pregnancy is increased compared with natural conception. The majority of multiple conceptions are twins. Multiple pregnancies, especially higher order, carry an increased risk of adverse maternal and perinatal outcomes. The patient should be advised of the potential risk of multiple births before starting treatment.
To minimise the risk of twins or higher order multiple pregnancy, careful monitoring of ovarian response is recommended. Appropriate management, such as cycle cancellation, should be considered in line with current clinical practice. The incidence of multiple pregnancy can be minimised by using the recommended dose and schedule of administration (see Section 4.2 Dose and Method of Administration).
In patients undergoing ART procedures, the risk of multiple pregnancy is related mainly to the number of embryos replaced, their quality and the patient's age. Single embryo transfer in good prognosis cycles substantially reduces the risk of multiple pregnancy with little effect on live birth rates.
Pregnancy loss. The incidence of pregnancy loss by miscarriage or abortion is higher in patients undergoing stimulation of follicular growth for ovulation induction or ART than following natural conception, but comparable with the rates found in women with other fertility problems.
Congenital anomalies. The prevalence of congenital anomalies after the use of ART may be slightly higher than after spontaneous conceptions. Possible contributing factors include aspects inherent in the couple's infertility, ovulation induction agents, other medicines used in treatment and the ART procedures. While there is no specific evidence from clinical trials or postmarketing data implicating gonadotrophin use in adverse effects on pregnancy, embryonal or fetal development, parturition or postnatal development, ovulation induction agents cannot be excluded as a contributing factor.
Treatment in men. Elevated endogenous FSH levels are indicative of primary testicular failure. Such patients are unresponsive to Bemfola/hCG therapy. Semen analysis is recommended in assessing the response to treatment.
Paediatric use. Bemfola should not be used in the paediatric population (see Section 4.3 Contraindications).
Use in the elderly. Bemfola should not be used in the elderly population (see Section 4.3 Contraindications).
Porphyria. In patients with porphyria or a family history of porphyria, Bemfola may increase the risk of an acute attack. Deterioration or a first appearance of this condition may require cessation of treatment.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No clinically significant drug interactions have been reported during Bemfola therapy. Concomitant use of Bemfola with other agents used to stimulate ovulation may potentiate the follicular response, whereas concurrent use of a GnRH agonist or antagonist to induce pituitary desensitisation may increase the dosage of Bemfola needed to elicit an adequate ovarian response.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Bemfola is indicated for use in infertility (see Section 4.1 Therapeutic Indications).
Use in pregnancy. (Category D)
Follitropin alfa is not intended for use during pregnancy (see Section 4.3 Contraindications). In rats and rabbits, follitropin alfa caused dystocia and marked postimplantation loss at subcutaneous doses of greater than 5 IU/kg/day, indicating that it is embryotoxic and fetotoxic. Follitropin alfa was not teratogenic at subcutaneous doses up to 320 IU/kg/day in rats or 5 IU/kg/day in rabbits.
Use in lactation. It is not known whether follitropin alfa is excreted in human milk. In lactating rats, follitropin alfa at doses up to 40 IU/kg did not influence lactation or have any effects on the postnatal growth and development of the offspring. Follitropin alfa was measured in the milk in early lactation.
Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in the nursing infant from Bemfola, a decision should be made whether to discontinue breastfeeding or to discontinue the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. However, adverse effect of Bemfola included dizziness which could affect the ability to drive or use machines (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.8 Adverse Effects (Undesirable Effects)
The reactions reported below are classified according to frequency of occurrence as follows:
Very common: ≥ 1/10; common: ≥ 1/100 to < 1/10; uncommon: ≥ 1/1,000 to < 1/100; rare: ≥ 1/10,000 to < 1/1,000; very rare: < 1/10,000.
Treatment in general. Immune system disorders. Very rare: mild to severe hypersensitivity reactions including anaphylactic reactions and shock.
Respiratory, thoracic and mediastinal disorders. Very rare: exacerbation or aggravation of asthma.
General disorders and administration site conditions. Very common: injection site reactions (e.g. pain, erythema, haematoma, swelling and/or irritation at the site of injection).
Treatment in women. The following adverse events have been reported during gonadotrophin therapy:
Reproductive system and breast disorders. Very common: ovarian cyst, mild to moderate ovarian enlargement. Common: mild or moderate OHSS (including symptomatology), intermenstrual bleeding. Uncommon: severe OHSS (including symptomatology). Rare: complications of severe OHSS, ectopic pregnancy, adnexal torsion associated with ovarian enlargement.
Gastrointestinal disorders. Common: abdominal pain, abdominal distension, abdominal discomfort, diarrhoea, nausea, vomiting.
Nervous system disorders. Very common: headache, dizziness.
Vascular disorders. Very rare: thromboembolism.
See Section 4.4 Special Warnings and Precautions for Use for information on symptoms and management of OHSS.
Treatment in men. Reproductive system and breast disorders. Common: gynaecomastia.
Skin and subcutaneous tissue disorders. Common: acne.
Investigations. Common: weight gain.
Reporting suspected adverse reactions. Reporting suspected adverse events after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
The effects of an overdose of Bemfola are unknown, nevertheless, there is the possibility that OHSS may occur (see Section 4.4 Special Warnings and Precautions for Use).
Advise your patients to immediately contact their doctor or the Poisons Information Centre (in Australia telephone 131 126, in New Zealand telephone 0800 764 766) if they are concerned that they have given themselves too much Bemfola.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
In females, the most important effect resulting from parenteral administration of FSH is the development of mature Graafian follicles. To complete follicular maturation and to stimulate ovulation in the absence of an endogenous luteinising hormone (LH) surge, human chorionic gonadotrophin (hCG) is given once monitoring of the patient indicates that sufficient follicular development has occurred. There may be a degree of interpatient variability in response to FSH administration, with lack of response to FSH in some patients. In males, FSH stimulates spermatogenesis without significant effect on the androgen secreting interstitial cells.
Clinical trials. WHO group II anovulatory infertile women. In a controlled study involving 222 randomised patients, cumulative ovulation rate was not significantly different between follitropin alfa and urofollitropin or urinary derived hFSH (u-hFSH) groups whether analysed on an intention to treat or evaluable patient basis. The ovulation rate in each cycle was also not different between the two medicines.
Superovulation in assisted reproduction techniques (ART). Study 21884. The safety and efficacy of follitropin alfa (r-hFSH; filled by mass) versus u-hFSH and its equivalence as compared to follitropin alfa old formulation (filled by bioactivity), all administered subcutaneously, were assessed in a multicentre, randomised, single blind, phase III study in infertile women undergoing in vitro fertilisation (IVF) and embryo transfer. All patients underwent pituitary desensitisation (downregulation) with a gonadotrophin releasing hormone (GnRH) agonist prior to and during stimulation of multiple follicular development with one of the three study treatments. Randomisation occurred when pituitary downregulation was confirmed by an E2 level of ≤ 50 picogram/mL.
The primary efficacy parameter in this study was the number of fertilised oocytes retrieved per patient. 837 patients entered the study, of whom 713 were randomised. Of these, 711 received at least one dose of FSH: 237 patients received follitropin alfa (r-hFSH; filled by mass), 237 patients received u-hFSH, and 237 patients received follitropin alfa old formulation (filled by bioactivity). The number of oocytes retrieved was similar in all treatment groups. The efficacy of follitropin alfa (r-hFSH; filled-by-mass) although not superior, led to statistically higher response rates in the number of fertilised oocytes as compared to u-hFSH. The efficacy results are summarised in Tables 1 and 2.

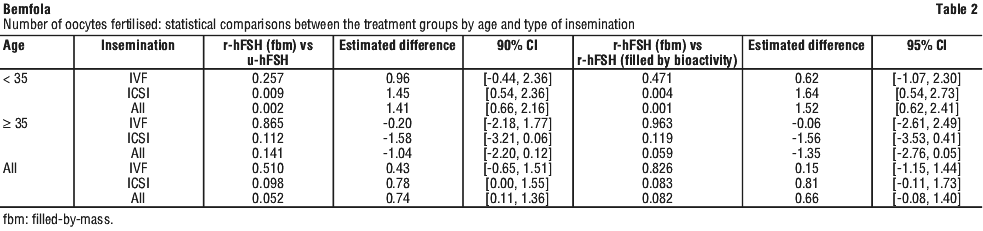
Men with hypogonadotrophic hypogonadism. Male hypogonadotrophic hypogonadism (HH) is a rare condition therefore study sizes are limited. Two phase III (open and noncomparative) studies were conducted to assess the efficacy and safety of follitropin alfa in combination with hCG in inducing spermatogenesis in men with HH. The primary efficacy endpoint was the achievement of a mature sperm density of ≥ 1.5 x 106/mL. Follitropin alfa was administered subcutaneously at a dosage of 150 IU three times a week in combination with hCG (≥ 2000 IU twice weekly) for up to 18 months.
The first study was conducted in university clinical centres in France, Germany and UK. A total of 32 patients with complete, primary isolated HH were recruited into this study. They were azoospermic before entering the study, remained so after the pretreatment phase and none had prior treatment with FSH or GnRH. In the pretreatment phase, the patients were treated with hCG alone (2000 IU twice weekly for 3-6 months) to first normalise serum testosterone levels before initiating the treatment with follitropin alfa. Of 26 patients who received follitropin alfa, 19 patients were found to be eligible for efficacy evaluation.
The primary endpoint of a sperm density of ≥ 1.5 x 106/mL was achieved in 12/19 (63%) patients. Overall 15/19 (79%) patients achieved some spermatogenesis. The median time to initiate spermatogenesis was 9 months.
The second study was conducted in 2 university clinical centres in Australia. A total of 10 patients with severe HH entered the study, but only 8 patients completed the follitropin alfa treatment phase. Similar results to the first study were obtained.
The primary endpoint of a sperm density of ≥ 1.5 x 106/mL were achieved in 5/8 (63%) patients. Overall 7/8 (88%) patients achieved some spermatogenesis. The median time to initiate spermatogenesis was 6 months. The studies also demonstrated that follitropin alfa has a good safety profile and is well tolerated over the treatment period of up to 18 months.
Comparability of Bemfola with Gonal-f. Therapeutic equivalence of Bemfola and Gonal-f was demonstrated in a multinational, multicentre, randomised, controlled, assessor blind, parallel group study in women undergoing assisted reproductive technologies.
The primary efficacy endpoint was the number of oocytes retrieved. Equivalence of Bemfola and Gonal-f was considered to be shown if the two sided 95% CI for the difference in the number of oocytes retrieved was within the equivalence range [-2.9 oocytes, +2.9 oocytes].
The secondary endpoints 'total r-hFSH dose', 'number of days of r-hFSH stimulation', 'mean number of follicles on stimulation day 8 and day of hCG administration', 'median estradiol levels on stimulation day 8', and 'number of patients with cycle cancellation' were comparable between both treatment groups.
Differences were noted in the first treatment cycle in the 'proportion of patients requiring dose reductions due to risk of imminent OHSS' and 'median estradiol levels on the day of hCG administration'. However, these differences are considered small, i.e. 3.3% difference between Bemfola and Gonal-f in proportion of patients with dose reductions. Further, the difference in median estradiol levels is also considered small considering the range in estradiol concentrations.
5.2 Pharmacokinetic Properties
Absorption/ distribution/ metabolism/ excretion. Following intravenous administration, follitropin alfa is distributed to the extracellular fluid space with an initial half-life of around 2 hours and eliminated from the body with a terminal half-life of about 1 day. The steady-state volume of distribution and total clearance are 10 L (0.17 L/kg) and 0.6 L/h (0.01 L/h/kg), respectively. One-eighth of the follitropin alfa dose is excreted in the urine.
Following subcutaneous administration, the absolute bioavailability is about 70%. Following repeated administration, follitropin alfa accumulates 3-fold at steady state within 3-4 days. In women whose endogenous gonadotrophin secretion is suppressed, follitropin alfa has nevertheless been shown to effectively stimulate follicular development and steroidogenesis, despite unmeasurable LH levels.
A phase I (study IMP 23572), open, randomised, 2 way crossover study to assess the relative bioavailability and the tolerability of r-hFSH of a reference follitropin alfa as a monodose freeze dried formulation and a new multidose liquid formulation, administered subcutaneously in male and premenopausal female volunteers with pituitary gonadotrope cell downregulation was conducted. The pharmacokinetic parameters from this study can be seen in Table 3.
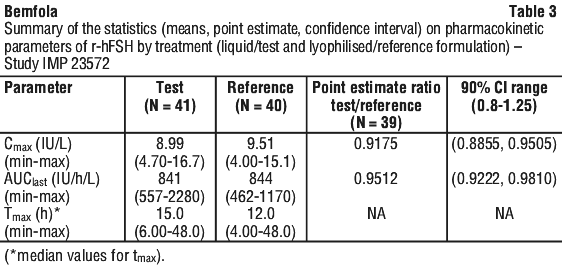
Comparability of Bemfola with Gonal-f. Equivalent pharmacokinetic (PK) profiles of Bemfola and Gonal-f have been demonstrated in a randomised, open label, two period, two treatment crossover study in 23 healthy female volunteers, with a duration of 11 weeks. The pharmacokinetic parameters are summarized in Table 4.

5.3 Preclinical Safety Data
Genotoxicity. Follitropin alfa showed no genotoxic activity in a series of assays performed to evaluate its potential to cause gene mutations (Salmonella typhimurium, Escherichia coli and Chinese hamster lung cells) and chromosomal damage (human lymphocytes and mouse micronucleus test).
Carcinogenicity. Long-term studies in animals have not been performed to evaluate the carcinogenic potential of follitropin alfa.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sucrose, methionine, phosphoric acid, dibasic sodium phosphate dihydrate, monobasic sodium phosphate dihydrate, polaxamer and water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). In New Zealand, information on the shelf-life can be found on the pubic Medsafe Product Detail information. The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Storage. Store in a refrigerator (2°C - 8°C). Do not freeze.
Before opening and within its shelf life, the medicinal product may be removed from the refrigerator, without being refrigerated again, for up to 3 months at or below 25°C. The product must be discarded if it has not been used after 3 months.
Store in the original package in order to protect from light.
Each Bemfola Pen is for individual patient use only. Discard used pen and needle immediately after injection.
6.5 Nature and Contents of Container
Solution for injection in 1.5 mL cartridge (clear type I glass), with a plunger stopper (halobutyl rubber) and an aluminium crimp cap with a rubber inlay.
Bemfola is available in packs of 1, 5 and 10 pre-filled pens. Not all pack sizes may be marketed. Each pack also contains the corresponding number of needles to be used with the pen for administration.
Bemfola Pens are not designed to allow the cartridge to be removed.
6.6 Special Precautions for Disposal
Any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Human follicle stimulating hormone (FSH) is a glycoprotein (MW about 30,000) and is characterised by two amino acid chains known as α and β. The β-chain confers biological activity. The α-chain is common to all gonadotrophins with specificity residing in the β-chain.
Chemical structure. The 92-amino-acid FSH alpha subunit in humans has the following sequence:
NH2-Ala-Pro-Asp-Val-Gln-Asp-Cys-Pro-Glu-Cys-Thr-Leu-Gln-Glu-Asn-Pro-Phe-Phe-Ser-Gln-Pro-Gly-Ala-Pro-Ile-Leu-Gln-Cys-Met-Gly-Cys-Cys-Phe-Ser-Arg-Ala-Tyr-Pro-Thr-Pro-Leu-Arg-Ser-Lys-Lys-Thr-Met-Leu-Val-Gln-Lys-Asn-Val-Thr-Ser-Glu-Ser-Thr-Cys-Cys-Val-Ala-Lys-Ser-Tyr-Asn-Arg-Val-Thr-Val-Met-Gly-Gly-Phe-Lys-Val-Glu-Asn-His-Thr-Ala-Cys-His-Cys-Ser-Thr-Cys-Tyr-Tyr-His-Lys-Ser-OH.
Note: The carbohydrate moiety is linked to the asparagine at positions 52 and 78.
The 111-amino-acid FSH beta subunit in humans has the following sequence:
NH2-Asn-Ser-Cys-Glu-Leu-Thr-Asn-Ile-Thr-Ile-Ala-Ile-Glu-Lys-Glu-Glu-Cys-Arg-Phe-Cys-Ile-Ser-Ile-Asn-Thr-Thr-Trp-Cys-Ala-Gly-Tyr-Cys-Tyr-Thr-Arg-Asp-Leu-Val-Tyr-Lys-Asp-Pro-Ala-Arg-Pro-Lys-Ile-Gln-Lys-Thr-Cys-Thr-Phe-Lys-Glu-Leu-Val-Tyr-Glu-Thr-Val-Arg-Val-Pro-Gly-Cys-Ala-His-His-Ala-Asp-Ser-Leu-Tyr-Thr-Tyr-Pro-Val-Ala-Thr-Gln-Cys-His-Cys-Gly-Lys-Cys-Asp-Ser-Asp-Ser-Thr-Asp-Cys-Thr-Val-Arg-Gly-Leu-Gly-Pro-Ser-Tyr-Cys-Ser-Phe-Gly-Glu-Met-Lys-Glu-OH.
Note: The carbohydrate moiety is linked to the asparagine at positions 7 and 24.
CAS number. 146479-72-3.
7 Medicine Schedule (Poisons Standard)
Schedule 4 Prescription Medicine.
Date of First Approval
27 November 2015
Date of Revision
21 October 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.