Benzatropine Injection
Brand Information
| Brand name | Benzatropine Injection |
| Active ingredient | Benzatropine mesilate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Benzatropine Injection.
Summary CMI
BENZATROPINE INJECTION
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why is Benzatropine Injection being given?
Benzatropine Injection contains the active ingredient benzatropine mesilate. Benzatropine Injection is used to treat some of the symptoms of parkinsonism.
For more information, see Section 1. Why is Benzatropine Injection being given? in the full CMI.
2. What should I know before Benzatropine Injection is given?
Do not use if you have ever had an allergic reaction to Benzatropine Injection or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before Benzatropine Injection is given? in the full CMI.
3. What if I am using other medicines?
Some medicines may interfere with Benzatropine Injection and affect how it works.
A list of these medicines is in Section 3. What if I am using other medicines? in the full CMI.
4. How is Benzatropine Injection given?
Benzatropine Injection must only be given by a doctor or nurse. The dose is given as an injection into a vein or a muscle.
More instructions can be found in Section 4. How is Benzatropine Injection given? in the full CMI.
5. What should I know while using Benzatropine Injection?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Benzatropine Injection? in the full CMI.
6. Are there any side effects?
The most common and serious side effects include constipation, dry mouth, nausea, loss of appetite, blurred vision, dilated pupils, increased heart rate, difficult or painful urination, an allergic reaction, for example skin rash, mood or mental changes, numb fingers, visual hallucinations, heat stroke, fever, urinary retention. Other side effects not listed can also occur. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why is Benzatropine Injection being given?
Benzatropine Injection contains the active ingredient benzatropine mesilate. Benzatropine Injection belongs to a group of medicines called anticholinergics. It comes as an injection. Benzatropine Injection works by blocking the effects of acetylcholine, a naturally occurring chemical produced by brain cells that can interfere with the regions of the brain that control muscle movement. In many patients, this reduces the symptoms of shaking and muscle stiffness.
Benzatropine Injection is used to treat some of the symptoms of parkinsonism.
The symptoms of parkinsonism include slow and unsteady movement, muscle stiffness and shaking (tremors). If untreated, parkinsonism can cause difficulty in performing normal daily activities.
Symptoms of parkinsonism can be caused by certain diseases of the brain affecting movement, such as Parkinson's disease. They can also be caused by some medicines that are used to treat certain mental conditions.
Benzatropine Injection is most helpful in improving muscle stiffness and shaking.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
2. What should I know before Benzatropine Injection is given?
Warnings
You should not be given Benzatropine Injection if:
- you are allergic to benzatropine mesilate, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not give this medicine to a child under three years old.
Also, it should be used with caution in older children. You should not be given Benzatropine Injection if the solution is discoloured, cloudy, turbid, or a precipitate is present. The solution is normally a clear, particle free solution.
The doctor or nurse will check to ensure the medicine is not past its expiry date and has not been tampered with.
If you are not sure whether you should be given this medicine, talk to your doctor.
Check with your doctor if you:
- have allergies to any other medicines, foods, preservatives or dyes.
- have or have had any of the following medical conditions:
- heart problems, such as fast heartbeat
- enlarged prostate
- mental illness
- glaucoma
- uncontrolled movements of hands, mouth or tongue.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell him/her before you are given Benzatropine Injection.
3. What if I am using other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Benzatropine Injection and affect how it works. These include:
- some medicines used to treat mental illness or psychotic disorders
- a group of medicines called tricyclic antidepressants used to treat depression
- other anticholinergic medicines, such as benzhexol, biperiden, procyclidine, orphenadrine.
These medicines may be affected by Benzatropine Injection or may affect how well it works. Being given Benzatropine Injection with these medicines may cause fever, heat intolerance, and stomach or bowel problems. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor has more information on medicines to be careful with or avoid while being given this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Benzatropine Injection.
4. How is Benzatropine Injection given?
How much is given
- Your doctor will decide what dose of Benzatropine Injection you will receive and how long you will receive it for. This depends on your medical condition and other factors, such as your weight, age and whether you are taking other medicines.
How it is given
- Benzatropine Injection must only be given by a doctor or nurse.
- The dose is given as an injection into a vein or a muscle.
If too much Benzatropine Injection is given
Symptoms of an overdose are the same as side effects but may be more severe. The symptoms of a side effect are listed under Side effects below. You may also have other effects, including decreased or increased reaction time, dizziness, poor coordination, muscle weakness, inability to perspire, hot and dry flushed skin, headache, breathing difficulties, fits and unconsciousness.
Also, symptoms of existing mental illness may worsen after taking an overdose.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Benzatropine Injection?
Things you should do
- If you are about to be started on any new medicine, remind your doctor and pharmacist that you have been given Benzatropine Injection.
- If you become pregnant while being given this medicine, tell your doctor immediately.
- If you experience problems with sweating while using Benzatropine Injection, tell your doctor immediately.
Remind any doctor, dentist or pharmacist you visit that you are given Benzatropine Injection.
Things you should not do
Under certain conditions Benzatropine Injection may make you perspire or sweat less, which may cause heat stroke. This may happen during hot weather, especially if you:
- drink large quantities of alcohol
- are being treated with other anticholinergic medicines for a chronic illness
- have a nervous (neurological) disorder
- will be working in a hot environment
- have an existing problem with sweating.
Your doctor may need to reduce your dose of Benzatropine Injection.
Try to avoid becoming overheated during exercise or hot weather while you are using this medicine.
Benzatropine Injection may make you sweat less, causing your body temperature to increase. If you overheat during exercise or hot weather, you may get heat stroke.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Benzatropine Injection affects you.
This medicine may cause some people to have blurred vision or to become less alert than normal. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol
Tell your doctor if you consume large quantities of alcohol.
Looking after your medicine
Benzatropine Injection will be stored in the surgery, pharmacy or ward of a hospital. The injection is kept in a cool dry place protected from light where the temperature stays below 30°C.
Benzatropine Injection will only be opened when it is time for you to have the injection.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. These are possible side effects which are usually mild. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. Serious side effects are generally rare. |
Very serious side effects:
| You may need urgent medical attention or hospitalisation. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Benzatropine Injection contains
| Active ingredient | benzatropine mesilate 2 mg |
| Other ingredients (inactive ingredients) | sodium chloride water |
Do not take this medicine if you are allergic to any of these ingredients.
This medicine does not contain lactose, sucrose, gluten, tartrazine, alcohol, dyes or preservatives.
What Benzatropine Injection looks like
Benzatropine Injection is a clear, particle free solution in a clear glass vial sealed with a grey rubber stopper and aluminium seal with a blue plastic flip off cap.
Benzatropine Injection is supplied in a 2 mL vial and is available in cartons containing 5 or 10 vials (AUST R 276242).
Not all pack sizes may be marketed.
Who distributes Benzatropine Injection
Benzatropine Injection is made and supplied in Australia by:
Phebra Pty Ltd
17-19 Orion Road
Lane Cove West, NSW 2066
Australia
Telephone: 1800 720 020

The leaflet was prepared in December 2025.
Phebra and the Phi symbol are registered trademarks of Phebra Pty Ltd.
Brand Information
| Brand name | Benzatropine Injection |
| Active ingredient | Benzatropine mesilate |
| Schedule | S4 |
MIMS Revision Date: 01 February 2019
1 Name of Medicine
Benzatropine mesilate.
2 Qualitative and Quantitative Composition
Benzatropine Injection is available as 2 mg in 2 mL vials.
Benzatropine Injection contains benzatropine mesilate as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Benzatropine Injection, 2 mg in 2 mL, clear, colourless, particle free solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Benzatropine Injection is recommended for all forms of parkinsonism - including arteriosclerotic, post-encephalitic, idiopathic, as well as drug-induced extrapyramidal disorders (except tardive dyskinesia). It can be effective at any stage of the disease, even when a patient has become bedridden. Benzatropine Injection often is helpful in patients who have become unresponsive to other agents. Benzatropine Injection is a powerful anticholinergic agent which is mainly effective in relieving tremor and rigidity. Therapy is directed toward control of disturbing symptoms to permit the patient maximum integration of function with minimum discomfort.
In non-drug-induced parkinsonism, partial control of symptoms is usually achieved.
4.2 Dose and Method of Administration
Benzatropine Injection is available as an injection for intravenous and intramuscular use. Each mL of the injection contains: benzatropine mesilate 1.0 mg; sodium chloride 9.0 mg; water for injection q.s 1.0 mL.
Because Benzatropine Injection is cumulative in action, therapy should be initiated with a small dose which then can be increased gradually at five- or six-day intervals. Increases in dosage should be made in increments of 0.5 mg, to a maximum of 6 mg.
The injection is especially useful for psychotic patients with acute dystonic reactions or other reactions that make oral medication difficult or impossible.
There is no significant difference in the onset of effect following intravenous or intramuscular injection. Improvement is noticeable within a few minutes after injection.
In emergency situations, when the patient's condition is alarming, administration of 1 to 2 mL of Benzatropine Injection will provide quick relief. If the signs of parkinsonism begin to return, the dose can be repeated.
Some patients experience greatest relief when taking the entire dose at bedtime; others react more favourably to divided doses, two to four times a day.
The long duration of action of Benzatropine Injection makes it particularly suitable for administration at bedtime when the effects may persist throughout the night. Consequently, Benzatropine Injection enables the patient to turn in bed more easily and to rise in the morning.
Therapy with other agents in parkinsonism should not be terminated abruptly when Benzatropine Injection is started, but reduced or discontinued gradually. Many patients obtain the greatest relief with a combination of Benzatropine Injection and other drugs.
Benzatropine Injection may be used concomitantly with combinations of carbidopa/ levodopa, or with levodopa in which case periodic dosage adjustment may be required in order to maintain optimum response.
Arteriosclerotic, idiopathic and postencephalitic parkinsonism. The usual daily dose of Benzatropine Injection is 1 to 2 mg, with a range of 0.5 to 6 mg parenterally.
Dosage must be individualised. In determining the dosage, the age and weight of the patient and the type of parkinsonism must be taken into consideration. Older patients, thin patients and patients with arteriosclerotic parkinsonism generally cannot tolerate large doses. However, most patients with postencephalitic parkinsonism require and, indeed, tolerate fairly large doses. Patients with a poor mental outlook are usually poor candidates for therapy.
In arteriosclerosis and idiopathic parkinsonism, therapy may be initiated with a single daily dose of 0.5 mg to 1 mg at bedtime. This dosage will be adequate in some patients, whereas 4 mg to 6 mg a day may be required by others.
In postencephalitic parkinsonism, therapy may be initiated in most patients with 2 mg a day in one or more doses. In highly sensitive individuals, therapy may be initiated with 0.5 mg at bedtime and increased as necessary.
Drug-induced parkinsonism. When treating extrapyramidal disorders due to central nervous system drugs such as phenothiazines or reserpine, a dosage of 1 to 4 mg once or twice a day is recommended.
Dosage should be varied to suit the needs of the patient. After one or two weeks of administration, Benzatropine Injection should be withdrawn to determine the continued need for medication.
If parkinsonism recurs, therapy with Benzatropine Injection can be reinstituted.
Usually the injection of 1 to 2 mL of Benzatropine Injection quickly relieves acute dystonic reactions.
4.3 Contraindications
Because of the atropine-like side effects, Benzatropine Injection is contraindicated in children under three years of age, and should be used with caution in older children.
Benzatropine Injection is contraindicated in patients who are hypersensitive to any component of this product.
4.4 Special Warnings and Precautions for Use
Since benzatropine mesilate has cumulative action, continued supervision is advisable. Patients with a tendency to tachycardia and patients with prostatic hypertrophy, should be closely observed during treatment.
In large doses, the drug may cause complaints of weakness and inability to move particular muscle groups. For example, if the neck has been rigid and suddenly relaxes, it may feel weak, causing some concern. In this event, dosage adjustment may be required.
Mental confusion and excitement may occur with large doses, or in susceptible patients. Visual hallucinations have been reported occasionally. Furthermore, in the treatment of extrapyramidal symptoms due to central nervous system drugs, such as phenothiazines and reserpine, in patients with mental disorders, occasionally there may be intensification of mental disorders. In such cases antiparkinsonian drugs can precipitate a toxic psychosis.
Patients with mental disorders should be kept under careful observation, especially at the beginning of treatment or if dosage is increased.
Tardive dyskinesia may appear in some patients on long-term therapy with phenothiazines and related agents, or may occur after therapy when these drugs have been discontinued. Antiparkinsonian agents usually do not alleviate their symptoms of tardive dyskinesia, and in some instances may aggravate or unmask such symptoms. Benzatropine Injection is not recommended in tardive dyskinesia.
Since benzatropine mesilate contains structural features of atropine, it may produce anhydrosis. For this reason, it should be given with caution during hot weather, especially when given concomitantly with other atropine-like drugs to the chronically ill, the alcoholic, those who have central nervous system disease and those who do manual labour in a hot environment.
Anhydrosis may occur more readily when some disturbance of sweating already exists. If there is evidence of anhydrosis, the possibility of hyperthermia should be considered. Dosage should be decreased at the discretion of the physician so that the ability to maintain body heat equilibrium by perspiration is not impaired. Severe anhydrosis and fatal hyperthermia have occurred.
The physician should be aware of the possible occurrence of glaucoma. Although the drug does not appear to have any adverse effect on simple glaucoma, Benzatropine Injection probably should not be used in narrow-angle glaucoma.
Use in the elderly. No data available.
Paediatric use. See Section 4.3 Contraindications.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
When Benzatropine Injection is given concomitantly with phenothiazines, haloperidol or other drugs with anticholinergic or antidopaminergic activity, patients should be advised to report fever, heat intolerance and gastrointestinal complaints promptly. Paralytic ileus, sometimes fatal, has occurred in patients taking anticholinergic-type antiparkinsonism drugs, including Benzatropine Injection, in combination with phenothiazines and/or tricyclic antidepressants.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
It is not known whether Benzatropine Injection can cause foetal harm when administered to a pregnant woman or can affect reproductive capacity. Benzatropine Injection should be given to a pregnant woman only if clearly needed.
Use in lactation. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Benzatropine Injection is administered to a nursing mother.
4.7 Effects on Ability to Drive and Use Machines
Benzatropine mesilate may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle.
4.8 Adverse Effects (Undesirable Effects)
Adverse reactions, most of which are anticholinergic or antihistaminic in nature are listed below by body system in order of decreasing severity.
Cardiovascular. Tachycardia.
Digestive. Constipation, dry mouth, nausea, vomiting.
If dry mouth is so severe that there is difficulty in swallowing or speaking, or loss of appetite and weight occur, reduce dosage, or discontinue the drug temporarily.
Slight reduction in dosage may control nausea and still give sufficient relief of symptoms. Vomiting may be controlled by temporary discontinuation, followed by resumption at a lower dosage.
Nervous system. Toxic psychosis, including confusion, disorientation, memory impairment, visual hallucinations, exacerbation of pre-existing psychotic symptoms, nervousness, depression, listless-ness, numbness of fingers.
Special senses. Blurred vision, dilated pupils.
Urogenital. Urinary retention, dysuria.
Metabolic/immune and skin. Occasionally, an allergic reaction e.g. skin rash, develops. If this cannot be controlled by dosage reduction, the medication should be discontinued.
Other. Heatstroke, hyperthermia, fever.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. May be any of those seen in atropine poisoning or antihistamine overdosage: CNS depression, preceded or followed by stimulation; confusion; nervousness; listlessness; intensification of mental symptoms or toxic psychosis in patients with mental illness being treated with phenothiazine derivatives or reserpine; hallucinations (especially visual); dizziness; muscle weakness; ataxia; dry mouth; mydriasis; blurred vision; palpitations; tachycardia; nausea; vomiting; dysuria; numbness of fingers; dysphagia; allergic reactions, e.g. skin rash; headache; hot, dry, flushed skin; delirium; coma; shock; convulsions; respiratory arrest; anhydrosis; hyperthermia; glaucoma; constipation.
The oral LD50 in the mouse is 94 mg/kg. The intravenous LD50 in the mouse is 24 mg/kg.
Treatment. For all overdoses, the mainstay of treatment is supportive and symptomatic care. Physostigmine salicylate, 1 to 2 mg s.c. or i.v., will reverse symptoms of anticholinergic intoxication. A second injection may be given after two hours if required. Otherwise treatment is symptomatic and supportive. Maintain respiration. A short-acting barbiturate may be used for CNS excitement, but with caution to avoid subsequent depression; supportive care for depression (avoid convulsant stimulants such as picrotoxin, pentylenetetrazole or bemegride); artificial respiration for severe respiratory depression; a local miotic for mydriasis and cycloplegia; ice bags or other cold applications and alcohol sponges for hyperpyrexia, a vasopressor and fluids for circulatory collapse. Darken room for photophobia.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. If the formula for benzatropine mesilate is compared with that of atropine and that of diphenhydramine it can be seen that benzatropine contains the tropine portion of the atropine molecule and the benzohydryl portion of diphenhydramine.
Benzatropine possesses both anticholinergic and antihistaminic effects, although only the former have been established as therapeutically significant in the management of parkinsonism.
In laboratory animals the antihistaminic activity and duration of action approach those of pyrilamine maleate.
In the isolated guinea pig ileum, the anticholinergic activity of this drug is about equal to that of atropine; however, when administered orally to unanaesthetised cats, benzatropine is only about half as active as atropine.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. No data available.
Distribution. No data available.
Metabolism. No data available.
Excretion. No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each Benzatropine Injection contains: sodium chloride 18.0 mg, water for injections q.s 2.0 mL.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG)1. The expiry date can be found on the packaging.
1 AUST R 276242.
6.4 Special Precautions for Storage
Protect from light. Do not freeze. Store below 30°C.
6.5 Nature and Contents of Container
Benzatropine Injection is supplied as 2 mg/2 mL glass vials.
Phebra product code: INJ197 2 mL vial supplied in a carton containing 5 vials;
INJ187 2 mL vial supplied in a carton containing 10 vials.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
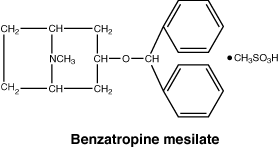
Benzatropine mesilate is a synthetic compound resulting from the combination of the active portions of atropine and diphenhydramine.
Benzatropine mesilate is a crystalline white powder and is very soluble in water.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
18 July 2017
Date of Revision
06 December 2018
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.