Bicillin L-A
Brand Information
| Brand name | Bicillin L-A |
| Active ingredient | Benzathine benzylpenicillin tetrahydrate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Bicillin L-A.
Summary CMI
Bicillin® L-A
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being treated with Bicillin L-A?
Bicillin L-A contains the active ingredient benzathine benzylpenicillin. Bicillin L-A is used to treat mild-moderate infections of the upper respiratory tract and sexually transmitted infections such as syphilis, yaws, bejel and pinta. It is used to prevent the recurrence of rheumatic fever and chorea, and complications following rheumatic heart disease and severe kidney disease.
For more information, see Section 1. Why am I being treated with Bicillin L-A? in the full CMI.
2. What should I know before treatment with Bicillin L-A?
You should not be given Bicillin L-A if you have ever had an allergic reaction to benzathine benzylpenicillin or any of the ingredients listed at the end of the CMI including lecithin (from soya bean) and benzoates, or if you have experienced previous allergic reactions to any other penicillin medicine.
Talk to your doctor if you have asthma, allergies, problems with your kidneys, are sodium restricted, have had severe skin adverse reactions or diarrhoea following antibiotic use, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before treatment with Bicillin L-A? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Bicillin L-A and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Bicillin L-A given?
Bicillin L-A is given as an injection into deep muscle on your buttock or the front of the thigh for infants/children.
More instructions can be found in Section 4. How is Bicillin L-A given? in the full CMI.
5. What should I know during treatment with Bicillin L-A?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
For more information, see Section 5. What should I know during treatment with Bicillin L-A? in the full CMI.
6. Are there any side effects?
Side effects of this medicine may include feeling anxious/nervous/abnormally happy/experience tremors; sleepiness/tiredness; nausea/vomiting; headache; excessive/abnormal sweating; muscle/joint pain; pain/swelling along bones; worsening of arthritis; oral thrush; vaginal thrush; stomach cramps; fast/irregular heartbeat; frequent infections; swollen glands in neck/armpit/groin; bleeding/bruising; blood in stool/poo; weakness of muscles; numbness/tingling/weakness of arms/legs; injection site reaction; impotence/persistent painful erection of penis, blurred blindness; loss of bladder control; new snoring; severe blisters and bleeding; itchy; allergic reaction; severe agitation/confusion; abnormal taste/sounds in the ears; heart attack; rapid/shallow breathing; cold/clammy skin; a rapid/weak pulse; dizziness; weakness/fainting; yellowing of the whites of your eyes/skin; stroke; seizures/fits/convulsions; severe diarrhoea; kidney disease; blood in urine/pass little/no urine; notice any changes to skin colour or appearance; have blisters that may have a discharge; skin peeling; experience any loss of sensation/numbness; coma.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Bicillin® L-A
Active ingredient(s): benzathine benzylpenicillin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Bicillin L-A. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Bicillin L-A.
Where to find information in this leaflet:
1. Why am I being treated with Bicillin L-A?
2. What should I know before treatment with Bicillin L-A?
3. What if I am taking other medicines?
4. How is Bicillin L-A given?
5. What should I know during treatment with Bicillin L-A?
6. Are there any side effects?
7. Product details
1. Why am I being treated with Bicillin L-A?
Bicillin L-A suspension for injection contains the active ingredient benzathine benzylpenicillin. Benzathine benzylpenicillin is an antibiotic.
Bicillin L-A is used to treat the following infections:
- Mild-to-moderate infections of the upper respiratory tract caused by streptococci bacteria.
- Sexually transmitted infections such as syphilis, yaws, bejel and pinta.
Bicillin L-A is also used to prevent:
- The recurrence of rheumatic fever and chorea.
- Complications following rheumatic heart disease and severe kidney disease.
2. What should I know before treatment with Bicillin L-A?
Warnings
Do not use Bicillin L-A if:
- you are allergic to benzathine benzylpenicillin, or any of the ingredients listed at the end of this leaflet including lecithin (from soya bean) and benzoates.
- you have had previous allergic reactions to any penicillin medicines
If you are not sure if you are allergic to any of the above, ask your doctor.
Some of the symptoms of an allergic reaction may include rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or troubled breathing.
Check with your doctor if you:
- you have previous allergic reactions to penicillins or cephalosporins
- have or have had asthma, allergy, hay fever or skin problems
- have problems with your kidneys
- have had severe skin adverse reactions following antibiotic use
- have/had diarrhoea (or usually with blood and mucus, stomach pain, fever) following antibiotic use
- are on a sodium restricted diet as Bicillin L-A contains either 2.59 mg or 5.17 mg of sodium per injection, depending on the strength
- consider the symptoms of your infection have not improved within a few days, or if they have become worse
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
- Check with your doctor if you are pregnant or intend to become pregnant.
- Tell your doctor if you are breastfeeding or plan to breastfeed. Bicillin L-A may pass into breastmilk, your doctor can discuss the risks and benefits of treatment while breastfeeding.
Use in the elderly
The risk of toxic reactions may be increased in patients greater than 65 years of age who have kidney problems. Your doctor should monitor you carefully in this situation.
3. What if I am taking other medicines?
Tell your doctor, nurse or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Bicillin L-A, these include:
- Tetracyclines, a class of antibiotics that treat several kinds of bacterial infections, e,g, doxycycline and minocycline,
- Probenecid, a medicine used to treat chronic gout or gouty arthritis and may be used in HIV treatment
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Bicillin L-A.
4. How is Bicillin L-A given?
How much is given
- Your doctor will decide how much Bicillin L-A you should receive and the length of time for which you should receive it based on what it is being used to treat and your age.
How is Bicillin L-A given
Bicillin L-A is given as an injection into deep muscle on your buttock (bottom cheek). For infants and children, it may be given in the middle front thigh. If injections must be repeated, they should not be given in exactly the same place each time.
If you use too much Bicillin L-A
As Bicillin L-A is given under the close supervision of your doctor, it is very unlikely that you will receive too much. If you experience any side effects, tell your doctor or nurse immediately.
Tell your doctor or nurse immediately if you are concerned that you may have been given too much Bicillin L-A. Or you should immediately phone the Poisons Information Centre (by calling 13 11 26).
5. What should I know during treatment with Bicillin L-A?
Things you should do
Keep all of your doctor's appointments and attend any tests as directed, as treatment with Bicillin L-A may require your doctor to monitor your kidneys and blood.
Tell your doctor or nurse straight away if you:
- do not experience an improvement of the symptoms of your infection within a few days, or if they become worse
- have any allergic reactions
- have flu-like symptoms including fever, sore throat and fatigue with severe skin problems such as a painful rash, blisters which may have pus or peeling
- have sores in the mouth, eyes, nose and genitals
- have redness and swelling of the eyes or conjunctivitis
- have swelling of the face
- experience any fungal or candida/thrush infections (sore, creamy-yellow, raised patches in the mouth or vaginal itching, burning with a thick, white, 'cottage cheese' discharge)
- have a rapid onset of weakness, pain, and numbness in the legs and/or arms, often with a bandlike tightness around the chest or stomach, and/or any problems when going to the toilet such as incontinence (with both urine/wee and poo) or inability to wee
- notice any changes to skin colour or appearance, especially on your fingers or toes such as turning red, brown, bronze, purple, blue, or black; have blisters that may have a discharge; peeling; or experience any loss of sensation or numbness
- paleness, mottling or a blue colour of the skin, or severe swelling around or near where the injection was given
- have diarrhoea (usually with blood and mucus) or stomach pain, even if it happens several weeks after treatment with Bicillin L-A has been stopped.
Remind any doctor, nurse, dentist or pharmacist who are treating you that you are being given Bicillin L-A.
Things you should not do
Do not take any other medicines whether they require a prescription or not without first telling your doctor or consulting a pharmacist.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Bicillin L-A affects you.
Bicillin L-A may cause dizziness or visual disturbances in some people
Drinking alcohol
No information is available.
Looking after your medicine
Bicillin L-A is stored in the pharmacy, clinic or on the hospital ward under the correct conditions.
Disposal of Bicillin L-A
The hospital or clinic staff will dispose of any leftover Bicillin L-A.
6. Are there any side effects?
All medicines can have side effects. Some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
| Side effects | What to do |
| Speak to your doctor if you have any of these side effects and they worry you. |
| Serious side effects that may require medical attention | What to do |
| Speak to your doctor or nurse as soon as possible if you notice any of these side effects. |
| Very serious side effects. You may need urgent medical attention or hospitalisation You may not experience any of them. | What to do |
| Tell your doctor or nurse immediately, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Some people may get other side effects whilst receiving Bicillin L-A.
Do not take any diarrhoea medicine without first checking with your doctor. Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list
Some of these side effects (for example blood abnormalities) can only be found when your doctor does tests from time to time to check your progress.
Reporting side effects
After you have received medical advice for any side effects you may experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Bicillin L-A contains
| Active ingredient (main ingredient) | Benzathine benzylpenicillin tetrahydrate |
| Other ingredients (inactive ingredients) | Sodium citrate Water for injections Soya bean products (lecithin) Carmellose sodium Povidone Methyl hydroxybenzoate Propyl hydroxybenzoate |
| Potential allergens | Methyl hydroxybenzoate Propyl hydroxybenzoate Soya bean products (lecithin) |
Do not take this medicine if you are allergic to any of these ingredients.
Each Bicillin L-A contains benzathine benzylpenicillin tetrahydrate 442 mg/mL.
What Bicillin L-A looks like
Bicillin L-A is white fluid in a pre-filled glass syringe.
Aust R 147169, 293456
Who distributes Bicillin L-A
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Bicillin L-A |
| Active ingredient | Benzathine benzylpenicillin tetrahydrate |
| Schedule | S4 |
MIMS Revision Date: 01 May 2024
1 Name of Medicine
Benzathine benzylpenicillin tetrahydrate.
2 Qualitative and Quantitative Composition
Bicillin L-A contains benzathine benzylpenicillin (the benzathine salt of benzylpenicillin) in aqueous suspension.
600,000 units/1.17 mL pre-filled syringe, containing benzathine benzylpenicillin tetrahydrate 517 mg/1.17 mL.
1,200,000 units/2.3 mL pre-filled syringe, containing benzathine benzylpenicillin tetrahydrate 1016.6 mg/2.3 mL.
Excipient(s) with known effect. Methyl hydroxybenzoate, propyl hydroxybenzoate, soya bean products (lecithin).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Suspension for injection.
4 Clinical Particulars
4.1 Therapeutic Indications
Intramuscular benzathine benzylpenicillin is indicated in the treatment of infections due to penicillin-sensitive micro-organisms that are susceptible to the low and very prolonged serum levels common to this particular dosage form. Therapy should be guided by bacteriological studies (including sensitivity tests) and by clinical response.
The following infections will usually respond to adequate dosage of intramuscular benzathine benzylpenicillin:
Streptococcal infections (group A - without bacteraemia). Mild to moderate infections of the upper respiratory tract (e.g. pharyngitis).
Venereal infections. Syphilis, yaws, bejel and pinta.
Medical conditions in which benzathine benzylpenicillin therapy is indicated as prophylaxis:
Rheumatic fever and/or chorea. Prophylaxis with benzathine benzylpenicillin has proven effective in preventing recurrence of these conditions. It has also been used as follow up prophylactic therapy for rheumatic heart disease and acute glomerulonephritis.
4.2 Dose and Method of Administration
Dosage. Use a concentration of 442 mg/mL when measuring part doses. The quantity of benzathine benzylpenicillin is based on 1,200 units/mg potency.
Streptococcal (group A) upper respiratory infections (for example, pharyngitis). A single injection of 1,200,000 units for adults.
A single injection of 900,000 units for older children.
A single injection of 300,000 to 600,000 units for infants and for children under 27 kg.
Venereal infections. Syphilis - primary, secondary and latent. 2,400,000 units (1-dose). Late (tertiary including neurosyphilis): 2,400,000 units at 7-day intervals for three doses.
Congenital (with normal CSF) - under 2 years of age. 50,000 units/kg body weight; ages 2-12 years: adjust dosage based on adult dosage schedule.
Yaws, bejel and pinta. 1,200,000 units (single injection).
Prophylaxis - for rheumatic fever and glomerulonephritis. Following an acute attack, benzathine benzylpenicillin (parenteral) may be given in doses of 1,200,000 units once a month or 600,000 units every 2 weeks.
Method of administration. Because of the high concentration of suspended material in this product, the needle may be blocked if the injection is not made at a slow, steady rate.
Administer by deep, intramuscular injection in the upper, outer quadrant of the buttock. In infants and small children, the midlateral aspect of the thigh may be preferable. Administration in the anterolateral thigh is not recommended due to the adverse effects observed (see Section 4.4 Special Warnings and Precautions for Use), and vascularity of this region. When doses are repeated, vary the injection site.
Method of administration is the same as with conventional syringe. Remove needle cover by grasping it securely; twist and pull. Introduce needle into patient, aspirate by pulling back slightly on the plunger, and inject.
Discard any unused portion.
4.3 Contraindications
Previous hypersensitivity reaction to any of the penicillins.
4.4 Special Warnings and Precautions for Use
Allergic reactions. Serious and occasionally fatal hypersensitivity (anaphylactoid) reactions have been reported in patients on penicillin therapy. Although anaphylaxis is more frequent following parenteral therapy, it has occurred in patients on oral penicillins. These reactions are more apt to occur in individuals with a history of sensitivity to multiple allergens. There have been well documented reports of individuals with a history of penicillin hypersensitivity reactions who have experienced severe hypersensitivity reactions when treated with a cephalosporin. Before therapy with a penicillin, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins and other allergens. If an allergic reaction occurs, the drug should be discontinued and the patient treated with the usual agents, e.g. pressor amines, antihistamines and corticosteroids. Severe anaphylactoid reactions require emergency treatment with adrenaline. Oxygen, intravenous corticosteroids and airway management, including intubation, should also be administered as indicated.
Penicillin should be used with caution in individuals with histories of significant allergies and/or asthma.
Whenever allergic reactions occur, penicillin should be withdrawn unless, in the opinion of the physician, the condition being treated is life-threatening and amenable only to penicillin therapy.
Severe cutaneous adverse reactions. Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in patients taking beta-lactam antibiotics. When SCAR is suspected, Bicillin L-A should be discontinued immediately and an alternative treatment should be considered.
Administration precautions. Do not inject intravenously or admix with other intravenous solutions. There have been reports of inadvertent intravenous administration of benzathine which has been associated with cardiorespiratory arrest and death.
Inadvertent intravascular administration, including inadvertent direct intra-arterial injection or injection immediately adjacent to arteries, of Bicillin L-A and other penicillin preparations has resulted in severe neurovascular damage, including transverse myelitis with permanent paralysis, gangrene requiring amputation of digits and more proximal portions of extremities, and necrosis and sloughing at and surrounding the injection site consistent with the diagnosis of Nicolau syndrome (also known as livedoid dermatitis or embolia cutis medicamentosa). Such severe effects have been reported following injections into the buttock, thigh and deltoid areas. Other serious complications of suspected intravascular administration which have been reported include immediate pallor, mottling or cyanosis of the extremity, both distal and proximal to the injection site, followed by bleb formation; severe oedema requiring anterior and/or posterior compartment fasciotomy in the lower extremity.
Severe effects and complications following accidental intravascular administration have most often occurred in infants and small children. Prompt consultation with an appropriate specialist is indicated if any evidence of compromise of the blood supply occurs at, proximal to, or distal to the site of injection.
Do not inject into or near a nerve. Injection into or near a nerve may result in permanent neurological damage.
Quadriceps femoris fibrosis and atrophy have been reported following repeated intramuscular injections of penicillin preparations into the anterolateral thigh. Because of these adverse effects and the vascularity of this region, administration in the anterolateral thigh is not recommended.
Antibiotic-associated pseudomembranous colitis. Antibiotic-associated pseudomembranous colitis has been reported with many antibiotics including penicillin. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of Clostridioides difficile.
C. difficile produces toxins A and B which contribute to the development of Clostridioides difficile associated diarrhoea (CDAD). Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhoea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
The severity of the colitis may range from mild to life-threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with antibiotic use (this may occur up to several weeks after cessation of antibiotic therapy). Mild cases usually respond to drug discontinuation alone. However, in moderate to severe cases, appropriate therapy with a suitable oral antibacterial agent effective against C. difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated. Drugs which delay peristalsis, e.g. opiates and diphenoxylate with atropine (Lomotil) may prolong and/or worsen the condition and should not be used.
Non-susceptible organisms and superinfections. Prolonged use of antibiotics may promote the overgrowth of non-susceptible organisms, including fungi. Should superinfection occur, appropriate measures should be taken.
Streptococcal infections. In streptococcal infections, therapy must be sufficient to eliminate the organism otherwise the sequelae of streptococcal disease may occur. Cultures should be taken following completion of treatment to determine whether Streptococci have been eradicated.
Blood and kidney function tests. In prolonged therapy with penicillin and particularly with high-dosage schedules, periodic evaluation of the renal and haematopoietic systems is recommended.
Fluids, electrolytes and protein replacement therapy should be provided when indicated.
Use in the elderly. Bicillin L-A is known to be mainly excreted by the kidney, the risk of toxic reactions to this drug may be greater in patients with impaired renal function (see Section 5.2 Pharmacokinetic Properties). It may be useful to monitor renal function in elderly patients.
Paediatric use. (See Section 4.1 Therapeutic Indications; Section 4.2 Dose and Method of Administration.)
Effects on laboratory tests. Penicillins can interfere with the copper sulfate reagent method of testing for glycosuria, resulting in falsely elevated or falsely decreased readings. Such interference does not occur with the glucose oxidase method.
Other. Bicillin L-A contains approximately 0.11 mEq of sodium per 600,000 units of benzylpenicillin (approximately 2.59 mg of sodium per 600,000 units of benzylpenicillin) and approximately 0.22 mEq of sodium per 1,200,000 units of benzylpenicillin (approximately 5.17 mg of sodium per 1,200,000 units of benzylpenicillin).
4.5 Interactions with Other Medicines and Other Forms of Interactions
Tetracyclines may antagonise the bactericidal effect of penicillin and concurrent use of these drugs should be avoided.
The rate of excretion of the penicillins is decreased by concomitant administration of probenecid which prolongs, as well as increases, blood levels of the penicillins.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Category A - Drugs which have been taken by a large number of pregnant women and women of child-bearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
Although generally considered to be safe, Bicillin L-A should be used during pregnancy only if clearly needed.
Use in lactation. Soluble benzylpenicillin (the hydrolysate of benzathine benzylpenicillin) is excreted in breast milk. The effect on the infant, if any, is not known. Caution should be used when Bicillin L-A is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
As with other penicillins, untoward reactions of the sensitivity phenomena are likely to occur, particularly in individuals who have previously demonstrated hypersensitivity to penicillins or in those with a history of allergy, asthma, hay fever, or urticaria.
The following adverse reactions have been reported with Bicillin L-A during post-marketing experience:
Skin and other subcutaneous tissue disorders. Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) (see Section 4.4 Special Warnings and Precautions for Use).
Immune system disorders. Acute myocardial ischaemia with or without myocardial infarction may occur as part of an allergic reaction (Kounis syndrome).
The following adverse reactions have been reported with parenteral benzylpenicillin (the active moiety in Bicillin L-A):
General. Hypersensitivity reactions including the following: skin eruptions (maculopapular to exfoliative dermatitis), urticaria, laryngeal oedema, fever, eosinophilia; other serum sickness-like reactions (including chills, fever, oedema, arthralgia and prostration), and anaphylactic/ anaphylactoid reaction (including shock and death); severe cutaneous adverse reactions (SCAR), such as toxic epidermal necrolysis (TEN) and acute generalised exanthematous pustulosis (AGEP) (see Section 4.4 Special Warnings and Precautions for Use).
Fever and eosinophilia may frequently be the only reaction observed.
Gastrointestinal. Pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment (see Section 4.4 Special Warnings and Precautions for Use).
Haematologic. Haemolytic anaemia, leucopenia, thrombocytopenia.
Neurologic. Neuropathy.
Urogenital. Nephropathy, acute interstitial nephritis.
As with other treatments for syphilis, the Jarisch-Herxheimer reaction has been reported.
The following adverse events have been temporally associated with parenteral administration of benzathine benzylpenicillin (the active moiety of Bicillin L-A):
Body as a whole. Hypersensitivity reactions (including allergic vasculitis, pruritus, fatigue, asthenia, and pain), aggravation of existing disorder, headache, Nicolau syndrome (also known as livedoid dermatitis or embolia cutis medicamentosa) (see Section 4.4 Special Warnings and Precautions for Use, Administration precautions).
Cardiovascular. Cardiac arrest, hypotension, tachycardia, palpitations, pulmonary hypertension, pulmonary embolism, vasodilation, vasovagal reaction, cerebrovascular accident, syncope.
Gastrointestinal. Nausea, vomiting, blood in stool, intestinal necrosis.
Haematological. Lymphadenopathy.
Injection site. Injection site reactions (including pain, inflammation, lump, abscess, necrosis, oedema, haemorrhage, cellulitis, hypersensitivity, atrophy, ecchymosis, and skin ulcer), neurovascular reactions (including warmth, vasospasm, pallor, mottling, gangrene, numbness of the extremities, cyanosis of the extremities, and neurovascular damage).
Metabolic. Elevated BUN, creatinine, and SGOT.
Musculoskeletal. Joint disorder, periostitis, exacerbation of arthritis, myoglobinuria, rhabdomyolysis.
Nervous system. Nervousness, tremors, dizziness, somnolence, confusion, anxiety, euphoria, transverse myelitis, seizures, coma.
A syndrome manifested by a variety of CNS symptoms such as severe agitation with confusion, visual and auditory hallucinations, and a fear of impending death (Hoigne's syndrome), has been reported after administration of benzylpenicillin procaine and, less commonly, after injection of the combination of benzylpenicillin benzathine and benzylpenicillin procaine. Other symptoms associated with this syndrome, such as psychosis, seizures, dizziness, tinnitus, cyanosis, palpitations, tachycardia, and/or abnormal perception in taste, also may occur.
Respiratory. Hypoxia, apnoea, dyspnoea.
Skin and other subcutaneous tissue disorders. Diaphoresis.
Special senses. Blurred vision, blindness.
Urogenital. Neurogenic bladder, haematuria, proteinuria, renal failure, impotence, priapism.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There have been no reported overdoses with Bicillin L-A. Penicillin in overdosage has the potential to cause neuromuscular hyperirritability and convulsive seizures. This is particularly so if the penicillin is given intravenously or to patients with renal failure.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Benzylpenicillin exerts a bactericidal action against penicillin-sensitive microorganisms during the stage of active multiplication. It acts through the inhibition of biosynthesis of cell-wall peptidoglycan, rendering the cell wall osmotically unstable. It is not active against the penicillinase-producing bacteria or against organisms resistant to beta-lactams because of alterations in the penicillin-binding proteins.
The following in vitro data are available but the clinical significance is unknown. Benzylpenicillin exerts high in vitro activity against Staphylococci (except penicillinase producing strains), Streptococci (groups A, C, G, H, L and M) and Pneumococci. Other organisms sensitive to benzylpenicillin are: Neisseria gonorrhoea, Corynebacterium diphtheriae, Bacillus anthracis, Clostridia spp, Actinomyces bovis, Streptobacillus moniliformis, Listeria monocytogenes and Leptospira spp. Treponema pallidum is extremely sensitive to the bactericidal action of benzylpenicillin.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Intramuscular benzathine benzylpenicillin is absorbed very slowly into the bloodstream from the intramuscular site and converted by hydrolysis to benzylpenicillin. This combination of hydrolysis and slow absorption results in blood serum levels much lower but much more prolonged than other parenteral penicillins.
Intramuscular administration of 225 mg of benzathine benzylpenicillin in adults results in blood levels of 22.5 to 37.5 nanogram per mL, which are maintained for 4 to 5 days. Similar blood levels may persist for 10 days following administration of 450 mg and for 14 days following administration of 900 mg. Blood concentrations of 2.25 nanogram per mL may still be detectable 4 weeks following administration of 900 mg.
Distribution. Approximately 60% of benzylpenicillin is bound to serum protein. The drug is distributed throughout the body tissues in widely varying amounts. Highest levels are found in the kidneys with lesser amounts in the liver, skin and intestines. Benzylpenicillin penetrates into all other tissues and the spinal fluid to a lesser degree.
Excretion. With normal kidney function, the drug is excreted rapidly by tubular excretion.
In neonates and young infants and in individuals with impaired kidney function, excretion is considerably delayed.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium citrate, water for injections, soya bean products (lecithin), carmellose sodium, povidone, methyl hydroxybenzoate, propyl hydroxybenzoate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2 to 8°C. Refrigerate, do not freeze.
Bicillin L-A may be stored below 30°C, for a single period of up to 2 months, prior to expiry. The date the product is placed outside of refrigerated storage and stored below 30°C should be written in the space provided on the carton. After storage outside of refrigeration, the product should be discarded and cannot be returned to refrigerated storage.
6.5 Nature and Contents of Container
Bicillin L-A is supplied as follows:
1.17 mL pre-filled glass syringe, containing 600,000 units benzathine benzylpenicillin tetrahydrate, equivalent to 517 mg; *packs of 5 and 10 syringes.
2.3 mL pre-filled glass syringe, containing 1,200,000 units benzathine benzylpenicillin tetrahydrate, equivalent to 1016.6 mg; *packs of 5 and 10 syringes.
*Not all pack sizes available.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Benzathine benzylpenicillin occurs as a white or almost white powder. It is very slightly soluble in water, freely soluble in dimethylformamide and in formamide, slightly soluble in ethanol (96 per cent).
Chemical structure.
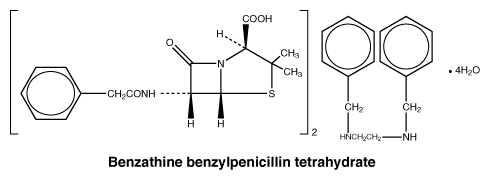
Chemical formula: (C16H18N2O4S)2.C16H20N2.4H2O.
Molecular weight: 981.18.
CAS number. 41372-02-5.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of Revision
22 March 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.