Biodone Forte
Brand Information
| Brand name | Biodone Forte |
| Active ingredient | Methadone hydrochloride |
| Schedule | S8 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Biodone Forte.
Summary CMI
BIODONE FORTE™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I using Biodone Forte?
Biodone Forte contains the active ingredient methadone hydrochloride. Biodone Forte is used for treating people with a dependency on opioid analgesic drugs.
For more information, see Section 1. Why am I using Biodone Forte? in the full CMI.
2. What should I know before I use Biodone Forte?
Do not use if you ever had an allergic reaction to Biodone Forte or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Biodone Forte? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Biodone Forte and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Biodone Forte?
- Biodone Forte is to be taken by mouth only.
- Each dose, number of dose/s for each day, when to take your dose/s is calculated by your doctor or pharmacist which is appropriate for you to stop you having withdrawal symptoms.
- Follow the instructions given to you by your doctor or your pharmacist.
More instructions can be found in Section 4. How do I use Biodone Forte? in the full CMI.
5. What should I know while using Biodone Forte?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Biodone Forte? in the full CMI.
6. Are there any side effects?
Common side effects include drowsiness, dizziness, vomiting and sweating. Serious side effects include symptoms of an allergic reaction such as wheezing, swelling of the lips/mouth, difficulty in breathing and fainting
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
WARNINGS
Hazardous and harmful use
Biodone Forte poses risks of abuse, misuse and addiction which can lead to overdose and death. Your doctor will monitor you regularly during your treatment.
Life threatening respiratory depression
Biodone Forte can cause life-threatening or fatal breathing problems (slow, shallow, unusual or no breathing) even when used as recommended. These problems can occur at any time during use, but the risk is higher when first starting Biodone Forte and after a dose increase, if you are older, or have an existing problem with your lungs. Your doctor will monitor you and change the dose as appropriate.
Use of other medicines while using Biodone Forte
Using Biodone Forte with other medicines that can make you feel drowsy such as sleeping tablets (e.g. benzodiazepines), other pain relievers, antihistamines, antidepressants, antipsychotics, gabapentinoids (e.g. gabapentin and pregabalin), cannabis and alcohol may result in severe drowsiness, decreased awareness, breathing problems, coma and death. Your doctor will minimise the dose and duration of use; and monitor you for signs and symptoms of breathing difficulties and sedation. You must not drink alcohol while using Biodone Forte.
BIODONE FORTE™
Active ingredient(s): methadone hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using Biodone Forte. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Biodone Forte.
Where to find information in this leaflet:
1. Why am I using Biodone Forte?
2. What should I know before I use Biodone Forte?
3. What if I am taking other medicines?
4. How do I use Biodone Forte?
5. What should I know while using Biodone Forte?
6. Are there any side effects?
7. Product details
1. Why am I using Biodone Forte?
Biodone Forte contains the active ingredient methadone hydrochloride. It belongs to a group of medicines called opioid analgesics.
Biodone Forte has been approved for use in treating people with a dependency on opioid analgesic drugs.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Biodone Forte is only available with a doctor's prescription.
2. What should I know before I use Biodone Forte?
Warnings
Addiction
You can become addicted to Biodone Forte even if you take it exactly as prescribed. Biodone Forte may become habit forming causing mental and physical dependence. If abused, it may become less able to reduce pain.
Dependence
As with all other opioid containing products, your body may become used to you taking this medicine. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Biodone Forte suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance
Tolerance to Biodone Forte may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
Biodone Forte given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
BIODONE FORTE works like other opioid analgesic drugs and so it may be used to replace heroin or other morphine-like drugs. Because BIODONE FORTE tends to stay in the body longer, it helps stop the withdrawal symptoms that are experienced by people who are dependent on opioid drugs. The withdrawal symptoms are less intense but more prolonged than those produced by morphine or diamorphine. They develop more slowly and do not usually appear until 24 to 48 hours after the last dose.
Do not use Biodone Forte if:
- you have ever had an allergic reaction to methadone hydrochloride, any other opioid drug or any of the ingredients listed toward the end of this leaflet.
- Some symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or any other parts of the body; rash, itching or hives on the skin. - you are taking or have recently taken antidepressants of the type called monoamine oxidase inhibitors (MAOIs)
- you have any other medical condition including:
- a lung disorder such as asthma, or any illness causing difficulty in breathing, especially if there is excessive phlegm or skin is bluish in colour
- recent head injury or increased pressure in the head
- a bowel condition known as ulcerative colitis
- certain liver or kidney conditions
- certain heart conditions
- alcoholism. - biliary and renal tract spasm
Check with your doctor if you:
- are under 18 years of age
- are allergic to foods, dyes, preservatives or any other medicines
- have any other medical condition including:
- hormone problems
- an underactive thyroid gland
- diabetes
- prostate disease
- phaeochromocytoma, a rare tumour of the adrenal gland. Symptoms include bouts of anxiety and headaches. There may be palpitations (banging of the heart felt in the chest), dizziness, weakness, nausea, vomiting, diarrhoea, dilated pupils, blurry vision, stomach pains and raised blood pressure. - intend to drink alcohol while you are taking Biodone Forte.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are or intend to become pregnant, or are about to give birth, or are breastfeeding.
Use in children
Biodone Forte is not recommended for use in children under 18 years of age.
Use in the elderly
The doctor may prescribe a smaller dose in elderly patients.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some of the medicines that may interfere with this medicine include:
- St John's wort
- rifampicin, an antibiotic
- medicines to treat epilepsy
- medicines to treat HIV infections
- fluconazole, an antifungal
- selective serotonin re-uptake inhibitors (SSRIs) which are medicines to treat depression or obsessive-compulsive disorders e.g. fluvoxamine
- any other medicines to treat depression
- medicines to treat mental illness
- medicines to treat heart conditions
- other pain relievers, muscle relaxants or tranquilisers
- fluid or water tablets
- laxatives
- fludrocortisone, a medicine used in certain hormone conditions.
You may need to take different amounts of your medicine or you may need to take different medicines. Your doctor or pharmacist has a more complete list of medicines to avoid while taking this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Biodone Forte.
4. How do I use Biodone Forte?
How much to take
Take this medicine as directed by your doctor or pharmacist.
- Your doctor will tell you how much to take each day.
- Follow the instructions provided and take Biodone Forte until your doctor tells you to stop.
- Your doctor will review your dosage regularly and change it as needed. Each dose is worked out specifically for you to stop you having withdrawal symptoms.
- If your dose has increased from the last one you took, you may experience some intolerance to the higher dosage for a few days.
Do not take more than the recommended dose.
It can result in low blood sugar.
How to take Biodone Forte
Biodone Forte is designed specifically to be taken by mouth only.
It is NOT SUITABLE for injection. Injecting this medicine can be extremely hazardous.
How long to take it for
Do not stop taking this medicine or change the dose without first checking with your doctor.
Your doctor may want to gradually reduce the amount you are taking before stopping completely in order to lessen the chance of possible withdrawal side effects.
If you use too much Biodone Forte
You will usually take your dose under the supervision of your pharmacist or doctor who has calculated the appropriate amount for you. Therefore, overdosage should not normally occur. Take home doses have also been calculated specifically for you and should not result in an overdosage.
If you or someone else receive too much (overdose) and experience one or more of the symptoms below, call triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used Biodone Forte that was prescribed for you.
If someone takes an overdose, they may experience one or more of the following symptoms:
- slow, unusual or difficult breathing
- drowsiness, dizziness or unconsciousness
- slow or weak heartbeat
- nausea or vomiting
- convulsions or fits
Other signs of overdose can also include problems with the 'nervous system' caused by damage to the white matter of the brain (known as toxic leukoencephalopathy).
If you think that you have used too much Biodone Forte, you may need urgent medical attention
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Biodone Forte?
Things you should do
Tell your doctor if, for any reason, you have not taken your medicine exactly as directed.
Otherwise your doctor may think that it was not working as it should and change your treatment unnecessarily.
If you forget to take a dose, take it as soon as you remember and then go on as before, but remember not to take the medicine more often than recommended by your doctor.
Seek medical help immediately if Biodone Forte is accidentally taken by a child.
Things you should not do
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Do not use it to treat any other complaints unless your doctor says to.
Things to be careful of
Particular care should be taken when starting this medicine or increasing the dose. Methadone can decrease heart and breathing rates, which if severe, may lead to death.
Speak to your doctor immediately if you have any concerns.
Driving or using machines
Biodone Forte may cause dizziness in some people. It is recommended that you don't drive, use machinery or undertake any activities where alertness is required.
Drinking alcohol
Do not drink alcohol while taking Biodone Forte.
Looking after your medicine
Store Biodone Forte in a cool, dry place where it stays below 25°C. Protect from light. Do not freeze.
Keep this medicine where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. With prolonged use, the dose may have to be increased to achieve the same benefit, whilst a sudden decrease in dose or interruption of therapy may give rise to possible withdrawal symptoms.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these side effects and they worry you. |
| Speak to your doctor if you have any of these side effects that may be associated with long-term use. |
Serious side effects
| Serious side effects | What to do |
Allergic reaction including:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these side effects. |
Biodone Forte can cause sleep-related breathing disorders such as sleep apnoea and sleep related hypoxemia (low oxygen level in the blood). The symptoms can include breathing pauses during sleep, night awakening due to shortness of breath, difficulties to maintain sleep or excessive drowsiness during the day.
Contact your doctor if you experience severe upper abdominal pain possibly radiating to the back, nausea, vomiting or fever as this could be symptoms associated with inflammation of the pancreas (pancreatitis) and the biliary tract system.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Biodone Forte contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
Biodone Forte is preservative free.
What Biodone Forte looks like
Biodone Forte is a clear, clean and pink coloured solution
(Aust R 64800).
Who distributes Biodone Forte
Biomed Aust Pty Ltd
Suite 5, Level 1
24 Hickson Road
Millers Point
NSW 2000
AUSTRALIA
This leaflet was revised in July 2025.
Brand Information
| Brand name | Biodone Forte |
| Active ingredient | Methadone hydrochloride |
| Schedule | S8 |
MIMS Revision Date: 01 December 2025
Life threatening respiratory depression. Serious, life‐threatening or fatal respiratory depression may occur with the use of Biodone Forte. Be aware of situations which increase the risk of respiratory depression, and monitor patients closely, especially on initiation or following a dose increase (see Section 4.4 Special Warnings and Precautions for Use).
Concomitant use of benzodiazepines and other central nervous system (CNS) depressants, including alcohol. Concomitant use of opioids with benzodiazepines, gabapentinoids, antihistamines, tricyclic antidepressants, antipsychotics, cannabis or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Patients and their caregivers should be made aware of the symptoms of respiratory depression. Patients and their caregivers should also be informed of the potential harms of consuming alcohol while taking Biodone Forte.
1 Name of Medicine
Methadone hydrochloride.
2 Qualitative and Quantitative Composition
Oral liquid containing methadone hydrochloride 5 mg/mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Biodone Forte is a clear, clean and pink coloured solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Biodone Forte is indicated for the detoxification and maintenance treatment of dependence on opioid drugs.
4.2 Dose and Method of Administration
Dosage and duration of treatment should be individualised.
Treatment of dependence on opioid drugs. A dose of 10 to 20 mg by mouth may be given initially and increased as necessary by 5 to 10 mg daily. The dose must not be increased by more than 5 to 10 mg daily and by no more than 30 mg in any seven day period. After stabilisation, which can often be achieved with a daily dose of 30 to 50 mg daily (up to a maximum of 80 mg daily), the dose of methadone is gradually decreased until total withdrawal is achieved. Some treatment schedules for opioid dependence involved prolonged maintenance therapy with methadone where the daily dose is adjusted carefully for the individual.
The dose of Biodone Forte required is to be measured accurately, using a calibrated dropper or other appropriate method.
Dilution may be required by local protocols and this dilution should be made with distilled water if the solution is for immediate consumption, or with a solution containing 0.1% sodium benzoate for take away doses, which should be used within five days of preparation. Dilution of take away doses, usually to 200 mL, is a strategy intended to reduce the likelihood of injection and of small children consuming sufficient of the drug to cause overdose. Take away solutions should be packaged in registered Quinex containers and sealed with a child-proof cap.
4.3 Contraindications
Biodone Forte is contraindicated in individuals who are hypersensitive to methadone or permicol red, which are the only components in the formulation.
Like other opioids, methadone is contraindicated in patients with respiratory depression, especially in the presence of cyanosis and excessive bronchial secretions.
Methadone should not be given during an attack of bronchial asthma.
Methadone is contraindicated in the presence of acute alcoholism, head injury and raised intracranial pressure.
Methadone is contraindicated in individuals receiving monoamine oxidase inhibitors or within 14 days of stopping such treatment.
As with other opioids, methadone is contraindicated in patients with ulcerative colitis, since it may precipitate toxic dilation or spasm of the colon.
As with all opioid analgesics, methadone should not be administered to patients with severe hepatic impairment as it may precipitate hepatic encephalopathy (see Section 4.4 Special Warnings and Precautions for Use).
Methadone is contraindicated in biliary and renal tract spasm.
Methadone is contraindicated in individuals with existing QT prolongation, including those with congenital long QT syndrome (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Harmful and hazardous use. Biodone Forte is subject to misuse, abuse and diversion, similar to other opioids, legal or illicit. Monitor patients carefully for progression of opioid dependence and other drug use.
Respiratory depression. Serious, life‐threatening, or fatal respiratory depression has been reported with the use of methadone, even when used as recommended. It can occur at any time during the use of Biodone Forte but the risk is greatest during initiation of therapy or following an increase in dose. Patients should be monitored closely for respiratory depression at these times.
Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol. Concomitant use of methadone and benzodiazepines or other CNS depressants (including other opioids, gabapentinoids, cannabis, sedatives, hypnotics, tricyclic antidepressants, antipsychotics, antihistamines, and centrally‐active anti-emetics) increases the risk of adverse events and may result in sedation, respiratory depression, coma and death. Patients and their caregivers should be made aware of these symptoms. Patients and their caregivers should also be informed of the potential harms of consuming alcohol while taking Biodone Forte.
Cardiac repolarisation. Methadone should be administered with particular caution to patients at risk for development of prolonged QT interval.
In vivo and in vitro studies have demonstrated that methadone inhibits cardiac potassium channels and prolongs cardiac repolarisation (i.e. prolongs the QT interval). QT interval prolongation and serious arrhythmia (torsades de pointes) have been observed during treatment with methadone and appear to be more common with higher doses. Particular caution and careful monitoring is recommended in patients at risk of prolonged QT interval (e.g. cardiac hypertrophy, concomitant diuretic use, hypokalaemia, hypomagnesaemia), patients with a previous history of cardiac repolarisation prolongation, those taking medications affecting cardiac repolarisation or methadone metabolism, and in patients with an increased risk of arrhythmia (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). Patients developing QT prolongation while on methadone treatment should be evaluated for modifiable risk factors, such as concomitant medications with cardiac effects, drugs which might cause electrolyte abnormalities, and drugs which might act as inhibitors of methadone metabolism.
Dependence. In common with all opioids, prolonged use of methadone has the potential to produce dependence of the morphine type. The withdrawal symptoms are less intense but more prolonged than those produced by morphine or diamorphine. They develop more slowly and do not usually appear until 24 to 48 hours after the last dose. Discontinuation of methadone therapy should be carried out gradually in patients who may have developed physical dependence on the medicine so as to avoid precipitating withdrawal symptoms (see Section 4.8 Adverse Effects (Undesirable Effects)).
Phaeochromocytoma. Extreme caution should be exercised when administering methadone to patients with phaeochromocytoma, since aggravated hypertension has been reported in association with diamorphine.
Ambulatory patients. Methadone should be used with caution in the presence of hypothyroidism, adrenocortical insufficiency, hypopituitarism, prostatic hypertrophy, shock and diabetes mellitus.
Hypoglycaemia. Hypoglycaemia has been observed in the context of methadone overdose or dose escalation. Regular monitoring of blood sugar is recommended during dose escalation (see Section 4.8 Adverse Effects (Undesirable Effects); Section 4.9 Overdose).
Sleep-related breathing disorders. Opioids can cause sleep-related breathing disorders including central sleep apnoea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the total opioid dosage.
Adrenal insufficiency. Adrenal insufficiency has been reported with opioid use, more often following long-term use. Symptoms may include nausea, vomiting, anorexia, fatigue, weakness, dizziness, or low blood pressure. If adrenal insufficiency is suspected, appropriate laboratory testing is recommended and discontinuation of treatment with Biodone Forte should be considered.
Endocrine effects. Opioids, such as Biodone Forte, may influence the hypothalamic-pituitary-adrenal or -gonadal axes. Hormonal disturbances that have been observed include an increase in serum prolactin and decreases in plasma cortisol and testosterone. Clinical symptoms may manifest from these hormonal changes.
Androgen deficiency may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility.
Neonatal withdrawal syndrome. Chronic use of methadone by the mother at the end of pregnancy may result in a withdrawal syndrome (e.g. hypertonia, neonatal tremor, neonatal agitation, myoclonus, convulsions, apnoea or bradycardia) in the neonate. In many reported cases the withdrawal was serious and required treatment. The syndrome is generally delayed for several hours to several days after birth. (See Section 4.6, Use in pregnancy).
Hepatobiliary disorders. Opioids may cause dysfunction and spasm of the sphincter of Oddi, thus raising intrabiliary pressure and increasing the risk of biliary tract symptoms and pancreatitis. Therefore, Biodone Forte has to be administered with caution in patients with pancreatitis and diseases of the biliary tract.
Gastrointestinal toxicity. Reports of significant oesophageal dysfunction have been observed via high-resolution manometry in patients taking opioid medicines on a long-term basis. Discontinuation or weaning of opioids should be considered in patients presenting with oesophageal complaints including but not limited to dysphagia, regurgitation, or non-cardiac chest pain.
Use in hepatic impairment. Particular care should be taken when methadone is to be used in patients with hepatic impairment as these patients metabolise methadone more slowly than normal patients. Where not contraindicated methadone should be given at less than the normal recommended dose and the patient's response used as a guide to further dosage requirements (see Section 4.3 Contraindications).
Use in renal impairment. Methadone should be used with caution in patients with renal dysfunction.
Use in the elderly. Methadone has a long plasma half-life, which may lead to accumulation, particularly if renal function is impaired (see Use in renal impairment).
In common with other opioids, methadone may cause confusion in this age group, therefore careful monitoring is advised.
Paediatric use. Methadone is not recommended for use in children less than 18 years of age since documented clinical experience has been insufficient to establish a suitable dosage regimen, furthermore, children are particularly sensitive to the respiratory and central nervous system effects of methadone.
Effects on laboratory tests. The serum BSP retention test may be increased (hepatotoxic effect or spasm of sphincter of Oddi). Plasma cortisol may be increased in response to cold to an extent not seen in controls. An increase in the serum albumin, prolactin and immunoglobulin (IgG) levels may be seen as a response to chronic administration. A significant decrease in serum indocyanine green level has been observed in a small series of patients with normal liver function tests. PCO2 may be increased due to decreased pulmonary ventilation. False positive urine pregnancy tests have occurred, mainly with the Gravindex test. Physiological changes in thyroid hormones may be seen - decrease in serum thyroxine (T4), a decrease in free thyroxine and an increase in tri-iodothyronine (T3).
Biodone Forte is not intended for administration by injection.
Biodone Forte is for oral use only.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Methadone is metabolised by various cytochrome P450 (CYP450) enzymes. Therefore, coadministration of drugs known to interfere with the CYP450 enzymes may affect its clinical activity.
Some compounds may increase the metabolism of methadone, e.g. rifampicin, phenytoin, carbamazepine, St John's wort and antiretroviral agents used in the treatment of HIV infection (particularly nevirapine, efavirenz and some protease inhibitors). This has the potential to result in withdrawal symptoms.
Patients on methadone maintenance who are also taking enzyme inducers such as carbamazepine, may require higher than typical doses of methadone.
Some compounds may decrease the metabolism of methadone, e.g. fluconazole and some serotonin reuptake inhibitors (SSRIs), particularly fluvoxamine. This may increase the likelihood of toxicity.
In addition to compounds that may decrease the metabolism of methadone, extreme caution is necessary when any drug known to have the potential to prolong the QT interval is prescribed in conjunction with methadone (see Section 4.4 Special Warnings and Precautions for Use). Interactions may occur with methadone and potentially arrhythmogenic agents such as class I and III antiarrhythmics, some neuroleptics and tricyclic antidepressants and calcium channel blockers. Caution should also be exercised when prescribing concomitant drugs capable of inducing electrolyte disturbances that may prolong the QT interval (hypokalaemia, hypomagnesaemia). These include diuretics, laxatives and in rare cases mineralocorticoid hormones.
Methadone can also affect the metabolism of other drugs. Plasma concentrations of some drugs may be increased, e.g. nelfinavir, zidovudine, fluconazole and desipramine, whereas concentrations of other drugs may be decreased, e.g. abacavir and amprenavir.
Monoamine oxidase inhibitors (MAOIs) may prolong and enhance the respiratory depressant effects of methadone. Opioids and MAOIs used together may cause fatal hypotension and coma.
The general depressant effects of methadone may be enhanced by other centrally-acting agents such as alcohol, antihistamines, antipsychotics, barbiturates, benzodiazepines, cannabis, centrally-active anti-emetics, gabapentinoids, hypnotics, neuromuscular blocking agents, other opioids, phenothiazines, sedatives, tricyclic antidepressants and tranquillisers (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol).
Some psychotropic drugs, may potentiate the analgesic effects of methadone.
The intestinal effects of methadone may delay the absorption of mexiletine.
Propranolol has been reported to enhance the lethality of toxic doses of opioids in animals. Although the significance of this finding is not known for man, caution should be exercised when such drugs are coadministered.
Opioid analgesics may antagonise the effects of agents that stimulate gastrointestinal motility (metoclopramide, domperidone, cisapride).
Anticholinergics increase the risk of constipation, urinary retention and so on. Antihypertensives may aggravate the hypotensive effects of opioid analgesics.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Methadone does not appear to impair human female fertility.
Studies in men on methadone maintenance programs have shown that methadone reduces serum testosterone and markedly depresses the ejaculate volume and sperm motility. The sperm counts of methadone subjects were twice that of controls but this reflected the lack of dilution from seminal secretions. A reduction in libido has been reported as well as impotence, delayed and/or failed ejaculation.
Use in pregnancy. (Category C)
There is insufficient evidence on which to determine the safety profile of methadone in pregnancy, therefore, it should only be used if the potential benefit outweighs the potential risk.
Opioid analgesics may cause respiratory depression in the newborn infant. During the last 2-3 hours before expected delivery, opioid analgesics should therefore only be used after weighing the needs of the mother against the risk to the foetus. Methadone is not recommended for use during labour because its prolonged duration of action increases the risk of respiratory depression in the neonate.
Like other opioids, methadone crosses the placenta during pregnancy, and most neonates born to mothers on methadone maintenance will suffer from withdrawal if left untreated.
Withdrawal symptoms pertaining to the central nervous system, gastrointestinal system and respiratory system may be observed in infants born to mothers receiving methadone maintenance. Neonatal abstinence syndrome may not occur until some days after birth. Therefore, in addition to initial monitoring of respiratory depression, neonates should undergo prolonged monitoring for signs and symptoms of methadone withdrawal.
Infants born to mothers on methadone maintenance have been reported to have smaller birthweights when compared to infants of nondrug exposed mothers. The infants born to mothers on methadone were not small for gestational age and by six months of age, these infants did not exhibit any general development sequelae.
Use in lactation. Caution should be exercised when methadone is administered to a nursing woman due to the risks of sedation and respiratory depression in the infant. Serious harm, including death, has been reported in infants following exposure to methadone through breast feeding.
Breastfeeding is permissible in mothers receiving methadone for maintenance therapy but specialist care from obstetric and paediatric staff with experience in such management is required. The baby should be monitored to avoid sedation. Therefore, breast feeding mothers should be counselled on how to identify respiratory depression and sedation in their babies and when it may be necessary to seek immediate medical care. The dose of methadone should be as low as possible.
Methadone is distributed into breast milk with a mean ratio of milk to plasma concentration of 0.44. However, doses of methadone to the infant by breast milk are low, estimated at 3% of maternal dose, on average, and insufficient to prevent neonatal abstinence syndrome in infants born to mothers on methadone maintenance.
4.7 Effects on Ability to Drive and Use Machines
In common with other opioids methadone may produce orthostatic hypotension and drowsiness in ambulatory patients. They should be cautioned, therefore, against driving vehicles, operating machinery or other activities requiring vigilance.
4.8 Adverse Effects (Undesirable Effects)
Respiratory disorders. The major side effect of methadone is respiratory depression.
Frequency not known: central sleep apnoea syndrome.
Gastrointestinal disorders. Reported events include nausea*, vomiting*, dry mouth*, loss of appetite and constipation.
Less common reactions include stomach cramps or pain. Methadone, in common with other opioids may cause spasm of the biliary tract (see Section 4.3 Contraindications).
Frequency not known: pancreatitis.
Hepatobiliary disorders. Frequency not known: spasm of sphincter of Oddi.
Neurological disorders. Reported events include dizziness*, drowsiness*, lightheadedness*, sweating* and confusion*, weakness, fatigue and low blood pressure. Less common reactions include blurred vision. Euphoria has been reported at higher doses in tolerant patients.
Cardiovascular disorders. Hypotension, collapse, and generalised oedema have occasionally been reported. Less common reactions include bradycardia, tachycardia and palpitations. ECG changes including QT prolongation and torsades de pointes have occurred very rarely, usually in patients with risk factors or receiving high doses of methadone (see Section 4.4 Special Warnings and Precautions for Use).
Renal disorders. Methadone, in common with other opioids may cause spasm of the biliary and renal tracts (see Section 4.3 Contraindications). It also possesses antidiuretic properties, and urinary retention or hesitancy has been reported.
Endocrine disorders. Prolonged use of methadone in men has been reported to be associated with the development of gynaecomastia. Long term use may also cause decreased libido, impotence and amenorrhea.
Frequency not known: adrenal insufficiency and androgen deficiency.
Withdrawal (abstinence) syndrome. Chronic use of opioid analgesics may be associated with the development of physical dependence. A withdrawal (abstinence) syndrome may be precipitated when opioid administration is suddenly discontinued or opioid antagonists administered. Withdrawal symptoms that may be observed after discontinuation of opioid use include body aches, diarrhoea, piloerection, anorexia, nervousness or restlessness, rhinorrhoea, sneezing, tremors or shivering, abdominal colic, nausea, sleep disturbance, unusual increase in sweating or yawning, weakness, tachycardia and unexplained fever. With appropriate dose adjustment and gradual withdrawal these symptoms are usually mild.
* These effects appear to be more common in ambulatory patients and in those receiving oral therapy.
Metabolism and nutrition disorders. Frequency (not known): hypoglycaemia.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms and signs. The symptoms and signs of overdosage with methadone parallel these for other opioids, namely profound respiratory depression, pinpoint pupils, hypotension, circulatory failure and pulmonary oedema, coma and death. Hypoglycaemia has been reported.
Mydriasis may replace miosis as asphyxia intervenes. Drowsiness, floppiness, pinpoint pupils and apnoea have been reported in children.
Toxic leukoencephalopathy has been observed with opioid overdose.
Treatment. General supportive measures, including ECG monitoring, should be employed as required. The specific opioid antagonist naloxone can be used for the reversal of coma and the restoration of spontaneous respiration. Intravenous infusion is the preferred route of administration in the management of methadone overdose because of the short half-life of naloxone relative to the longer half-life of methadone. Continuous infusion reduces the possibility of prolonged respiratory depression and the risk of relapse, which can occur suddenly. It should be noted that QT prolongation will not be reversed by naloxone.
In opioid dependent patients the administration of the usual dose of an opioid antagonist will precipitate an acute withdrawal syndrome. The severity of this syndrome will depend on the degree of physical dependence and the dose of the antagonist administered. The use of an opioid antagonist should be avoided if possible. If it must be used to treat respiratory depression in the physically dependent person, the antagonist should be administered with extreme care and by titration with smaller than usual doses of the antagonist.
Patients should be monitored closely for at least 48 hours after apparent recovery in case of relapse, since the duration of action of the antagonist may be substantially shorter than that of methadone.
The use of the respiratory or central stimulants is not recommended.
Acidification of the urine will enhance urinary excretion of methadone.
Methadone is not dialysable by either peritoneal dialysis or haemodialysis.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The pharmacological actions of methadone are qualitatively similar to those of morphine. Significant quantitative differences are its effective analgesic activity after administration by the oral route and its tendency to show persistent effects with repeated administration.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Methadone hydrochloride is readily absorbed after administration by mouth and has high oral bioavailability. Peak plasma concentrations have been reported 1 to 5 hours after oral administration of a single dose in tablet form.
Distribution. It undergoes considerable tissue distribution and protein binding is reported to be 60 to 90% with α1-acid glycoprotein being the main binding protein in the plasma.
Metabolism. Metabolism to the major metabolite 2-ethylidine-1,5-dimethyl, 3,3-diphenylpyrrolidine and the minor metabolite 2-ethyl-3,3-diphenyl-5-methylpyrrolidine, both of them inactive, occurs in the liver. These metabolites are excreted in the faeces and urine together with unchanged methadone. Other metabolites, including methadol and normethadol (reported to be pharmacologically active), have also been described but account for a small proportion of the dose. The liver may also serve as a major storage site of unchanged methadone which is taken up, bound nonspecifically by the liver and released again mainly unchanged.
Excretion. Marked interindividual variations in kinetics have been observed with methadone. Elimination half-lives vary considerably (a range of 15 to 60 hours has been reported) and careful adjustment of dosage is necessary with repeated administration, after which there is a gradual accumulation in the tissues.
Plasma concentrations have been found to vary widely during methadone maintenance therapy with large differences between patients and wide fluctuations in individual patients. Declining concentrations have been reported during methadone maintenance suggesting that tolerance occurs, possibly as a result of autoinduction of hepatic microsomal enzymes.
5.3 Preclinical Safety Data
Genotoxicity. Methadone did not exhibit demonstrable mutagenic activity in a wide range of standard in vitro and in vivo mutagenicity assays. However, in a dominant lethal assay in mice treatment with methadone at doses between 1 and 6 mg/kg was associated with increased preimplantation deaths and chromosomal aberrations of sperm cells when compared with controls.
Carcinogenicity. Long-term carcinogenicity tests in rodents did not reveal any evidence of methadone related neoplasia.
Teratogenic potential. No teratogenic effects have been observed in standard teratogenicity studies in rats and rabbits given methadone at doses from ten to fifty times the average daily human maintenance dose. Developmental abnormalities of the central nervous system have been reported in hamsters and mice given high doses in early pregnancy.
6 Pharmaceutical Particulars
6.1 List of Excipients
Permicol red, water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Storage: Store below 25°C. Protect from light. Do not freeze.
6.5 Nature and Contents of Container
Pack sizes: 200 mL and 1000 mL in glass bottles with a tamper-evident cap or plastic bottles with a heat induction seal under the cap.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Methadone hydrochloride: (6-dimethylamino-4,4-diphenyl-3-heptanone hydrochloride).
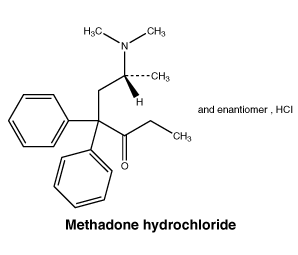
CAS number. 1095-90-5.
Methadone is a racemic mixture of two enantiomers. The l-enantiomer is more potent with respect to analgesic activity, respiratory depression and addiction liability.
7 Medicine Schedule (Poisons Standard)
S8 - Controlled Drug.
Date of First Approval
02 March 2000
Date of Revision
24 October 2025
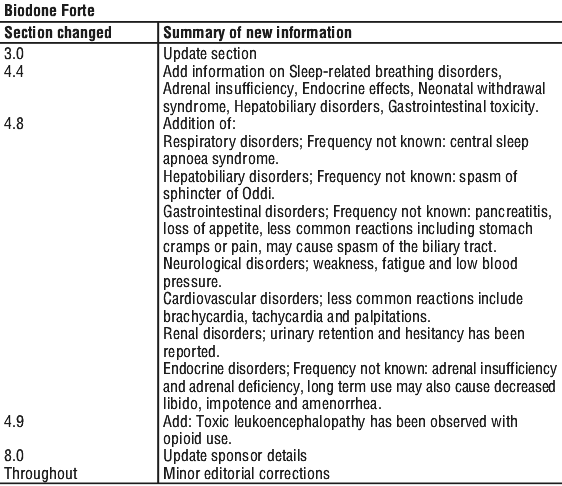
Summary Table of Changes
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.