Biological Therapies Folic Acid 15 mg in 1 mL Injection
Brand Information
| Brand name | Biological Therapies Folic Acid 15 mg in 1 mL Injection |
| Active ingredient | Folic acid |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Biological Therapies Folic Acid 15 mg in 1 mL Injection.
Summary CMI
Biological Therapies Folic Acid 15 mg in 1 mL Injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given this medicine?
This medicine contains the active ingredient folic acid. It is used for the prevention and treatment of folate deficiency. For more information, see Section 1. Why am I being given this medicine? in the full CMI.
2. What should I know before I am given this medicine?
Do not use if you have ever had an allergic reaction to this medicine or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given this medicine? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Folic Acid Injection and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is this medicine given to me?
Folic Acid Injection will be injected intramuscularly (into a muscle), intravenously (into a vein) or subcutaneously (under the skin) by your doctor. Your doctor will decide what to do. This medicine must only be given by a doctor or nurse. The amount given depends on many factors including your medical condition.
More instructions can be found in Section 4. How is this medicine given to me? in the full CMI.
5. What should I know while being given this medicine?
| Things you must do |
|
| Things you must not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given this medicine? in the full CMI.
6. Are there any side effects?
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Biological Therapies Folic Acid 15 mg in 1 mL Injection
Active ingredient(s): Folic Acid
Consumer Medicine Information (CMI)
This leaflet provides important information about using Biological Therapies Folic Acid Injection. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Folic Acid Injection.
Where to find information in this leaflet:
1. Why am I being given this medicine?
2. What should I know before I am given this medicine?
3. What if I am taking other medicines?
4. How is this medicine given to me?
5. What should I know while being given this medicine?
6. Are there any side effects?
7. Product details
1. Why am I being given this medicine?
Folic Acid Injection contains the active ingredient folic acid; (2S)-2-[[4-[[(2-amino-4-oxo-1,4-dihydropteridin-6-yl)methyl]amino]benzoyl]amino] pentanedioic acid (a member of the Vitamin B group). Once administered, folic acid is converted to folate in the body.
Folate Deficiency:
Folic Acid Injection may be prescribed if you have a diagnosed folate deficiency and are unable to absorb sufficient folate through oral intake. Oral intake may be insufficient in; malabsorption disorders, alcoholism, gastrointestinal diseases, pregnancy and lactation, haemolytic anaemia, exfoliative dermatitis and chronic infection.
For use in Pregnancy and Lactation:
Without folate, cells cannot divide normally. This is especially important for all rapidly dividing cells in the body, such as the cells in a developing baby (foetus). Your doctor may prescribe Folic Acid Injection to prevent folate deficiency in pregnancy or lactation, particularly if you have a problem that decreases your folate levels.
Megaloblastic Anaemia:
Anaemia occurs when red blood cells cannot carry enough oxygen to meet the needs of the body's cells. Folate is needed to make red blood cells grow properly, without it the red blood cells become large and less effective. This is a type of anaemia called megaloblastic anaemia. Megaloblastic anaemia may also be caused by a vitamin B12 deficiency, since vitamin B12 is required to use folate properly in the body.
Symptoms of anaemia include tiredness, breathlessness, lack of energy, fast heartbeat and pale colour of the skin.
Folic Acid Injection may be prescribed to help with the symptoms of megaloblastic anaemia if folate deficiency is the diagnosed cause.
Your doctor may have prescribed Folic Acid Injection for another reason.
Ask your doctor if you have any questions about why Folic Acid Injection has been prescribed for you.
Folic Acid Injection is not addictive.
2. What should I know before I am given this medicine?
Warnings
You must not be given this medicine if:
- You have a known allergy to folic acid or its derivatives. If you have had an allergic reaction to folic acid before, you may be allergic to Folic Acid Injection
- You have megaloblastic anaemia but have not had tests to prove folate deficiency is the cause. Vitamin B12 deficiency will also cause megaloblastic anaemia. B12 deficiency can also cause damage to the nervous system. Folic Acid Injection might correct the anaemia caused by B12 deficiency but will not stop the nerve damage.
- You must not be given this medicine if the solution in the vial is not clear or contains particles.
- If you are not sure whether you should be given this medicine, talk to your doctor.
Check with your doctor if you:
- Have any other medical conditions.
- Take any medicines for any other condition.
- Have had an allergy to any of the ingredients listed at the end of this leaflet. Your doctor may test if you have an allergy to this medicine by injecting a small amount under your skin.
- Have allergies to any other medicines or any other substances, such as foods, preservatives or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Folic Acid Injection is used in the prevention of folic acid deficiency in pregnancy and lactation.
Check with your doctor if you are pregnant or intend to become pregnant. Talk to your doctor if you are breastfeeding or intend to breastfeed.
If you have not told your doctor about any of the above, tell them before you are given Folic Acid Injection.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Your doctor and pharmacist have more information about medicines to be careful with, or avoid while you are being treated with this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Folic Acid Injection.
4. How is this medicine given to me?
This medicine must only be given by a doctor or nurse.
Folic Acid Injection will be injected intramuscularly (into a muscle), intravenously (into a vein) or subcutaneously (under the skin) by your doctor. Your doctor will decide what to do.
How much is given
Your doctor will tell you how much Folic Acid Injection will need to be given. This is determined by many factors including your medical condition. The usual amount given is 1 to 5 mg per day.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
How long is the treatment time with this medicine
Each person will respond differently to Folic Acid Injection.
Your doctor will advise you about the length of treatment with Folic Acid Injection.
If you miss an appointment or need to change an appointment
If you miss an appointment, talk to your doctor and arrange another appointment as soon as possible.
If you are not sure what to do, contact your doctor or pharmacist as soon as possible.
If too much of this medicine is given
Your doctor should be the only person to inject Folic Acid Injection, so an overdose is unlikely to occur.
If you think that you or anyone else may have been given too much Folic Acid Injection you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while being given this medicine?
Things you must do
- If you about to be started on any new medicine, remind your doctor and pharmacist that you are being treated with Folic Acid Injection.
- Tell all doctors, dentists and pharmacists who treat you that you are being given this medicine.
- Tell your doctor if you become pregnant while using Folic Acid Injection.
- Tell your doctor if you feel that giving Folic Acid Injection is not helping your condition.
- Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you must not do
- Do not attempt to inject this medicine yourself.
- Do not take any other medicines, whether they require a prescription or not, without first telling your doctor or consulting a pharmacist.
- Do not use this medicine if the packaging is torn or shows signs of tampering.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Folic Acid Injection affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
Folic Acid Injection is usually stored in the doctor's surgery or clinic, or at the pharmacy.
If you need to store Folic Acid Injection keep it in the original pack until it is time for it to be given. Unopened vials of Folic Acid Injection are to be stored at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light.
Seal box immediately after removal of each vial and store in a dark place.
Keep it where young children cannot reach it.
When to discard your medicine
Each vial is for SINGLE USE in one patient on one occasion only. It will be used once only and then it will be discarded. It must never be stored after it is opened or used for more than one person.
Getting rid of any unwanted medicine
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any vials that are left over.
Do not use this medicine after the expiry date. It may not work and it may be harmful to you.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are being given or treated with Folic Acid Injection.
This medicine helps most people, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
The above list includes rare allergic side effects and you may need urgent medical attention.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell, even if is not on this list. This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known.
Ask your doctor or pharmacist if you don't understand anything in this list.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Folic Acid 15 mg in 1 mL Injection contains
| Active ingredient (main ingredient) | Folic acid |
| Other ingredients (inactive ingredients) | Disodium edetate Sodium hydroxide Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
Folic Acid Injection does not contain gluten, lactose, sucrose, tartrazine or any other azo dyes.
What Folic Acid 15 mg in 1 mL Injection looks like
Folic Acid Injection is a clear, yellow coloured solution contained in a 2 mL amber glass vial sealed with a rubber stopper and an aluminium seal.
Australian Registration Number: AUST R 106971
This medicine may be available in 3 pack sizes:
10 x 1 mL vials per carton
6 x 1 mL vials per carton
1 x 1 mL vial per carton
Who distributes Folic Acid Injection
Biological Therapies
A Division of Orthomolecular Medisearch Laboratories Pty Ltd
5 / 20 – 30 Malcolm Road, Braeside VIC 3195
Australia
Tel: + 61 3 9587 3948
Fax: +61 3 9587 1720
Website: www.biologicaltherapies.com.au
Email: biol@biol.com.au
This leaflet was prepared in September 2025.

Brand Information
| Brand name | Biological Therapies Folic Acid 15 mg in 1 mL Injection |
| Active ingredient | Folic acid |
| Schedule | S4 |
MIMS Revision Date: 01 June 2019
1 Name of Medicine
Folic acid.
2 Qualitative and Quantitative Composition
Biological Therapies Folic Acid Injection contains folic acid adjusted to pH with sodium hydroxide in water for injections. Disodium edetate 2 mg/mL is used as a stabiliser. Each 1 mL solution contains 15 mg of folic acid as an active ingredient.
The total sodium content in undiluted Biological Therapies Folic Acid Injection 15 mg in 1 mL is approximately 2.4 mg/mL, the pH range 8 to 11.
Folic acid, (2S)-2-[[4-[[(2-amino-4-oxo-1,4-dihydropteridin-6-yl) methyl]amino] benzoyl]amino] pentanedioic acid, is prepared synthetically. Folates are complex organic compounds present in liver, yeast and natural sources.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Biological Therapies Folic Acid Injection is a yellow colour and is a clear solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Folate deficiency may exist that is unable to be corrected by oral intake and in these cases parenteral administration may be preferable. Oral intake may be insufficient in: malabsorption disorders, gastrointestinal pathologies, pregnancy and lactation, alcoholism, haemolytic anaemia, exfoliative dermatitis and chronic infection.
Specific indications may include:
1. Megaloblastic anaemia where the anaemia has been proven to be due to folate deficiency only.
2. Prevention of folic acid deficiency in pregnancy and lactation.
4.2 Dose and Method of Administration
Biological Therapies Folic Acid Injection is used for parenteral folic acid therapy, and may be administered, usually by intramuscular, or less commonly by intravenous or subcutaneous injection. Although Biological Therapies Folic Acid Injection can be given undiluted for intramuscular or subcutaneous use, it must be diluted before intravenous use. Biological Therapies Folic Acid Injection contains no antimicrobial agent. It is for single use in one patient only. Discard any residue.
A dilute solution of Biological Therapies Folic Acid Injection 15 mg in 1 mL for intravenous injection, containing 100 micrograms/mL, may be prepared by adding 1 mL (15 mg) of the injection to 149 mL of normal saline or 149 mL of 5% glucose.
For treatment of megaloblastic anaemia in adults, 1-5 mg per day is recommended. Adjust the dosage according to the severity of the anaemia and to the presence or absence of malabsorption syndromes. For the treatment of megaloblastic anaemia in pre-term infants less than 2 kg body weight, 100-200 micrograms daily, orally or intramuscularly, should be used. Treating megaloblastic anaemia in infants 5-12 months of age, satisfactory response has been obtained using 50 micrograms daily, orally or intramuscularly.
The usual therapeutic dose in paediatric practice for the treatment of megaloblastic anaemia due to folic acid deficiency is 1-5 mg daily. This supra-physiological dosage can produce a partial haematological response in primary B12 deficiency and therefore must not be used until B12 deficiency has been excluded.
For prophylaxis in pregnancy and lactation, 500 micrograms of folic acid/day prevented signs of folic acid deficiency in well-nourished pregnant women.
4.3 Contraindications
This drug is contra-indicated in patients who may be hypersensitive to it. Although rare, an anaphylactic reaction has been reported.
Folic acid should not be prescribed for megaloblastic anaemia due to vitamin B12 deficiency.
4.4 Special Warnings and Precautions for Use
Folic acid treatment may correct the haematological features of vitamin B12 deficiency without correcting the progressive neurological damage due to vitamin B12 deficiency. Therefore vitamin B12 deficiency needs to be excluded before treatment with folic acid alone.
Patients receiving diphenylhydantoin treatment should be monitored for possible loss of seizure control following large doses of folic acid. Folic acid does not correct folate deficiency due to dihydrofolate reductase inhibitors, such as methotrexate. Folinic acid should be used for this purpose. Caution is advised in patients who may have folate-dependent tumours.
Vitamin B12 concentrations in blood may be reduced by large and continuous doses of folic acid.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following drug interactions and/or related problems have been selected on the basis of their potential clinical significance (possible mechanism in parentheses where appropriate) - not necessarily inclusive (≥ major clinical significance).
Note. Combinations containing any of the following medications, depending on the amount present, may also interact with this medication.
Analgesics, long-term use or anticonvulsants, hydantoin or carbamazepine or estrogens or oral contraceptives or phenobarbital or primidone. Requirements for folic acid may be increased in patients receiving these medications. Concurrent use with folic acid may decrease the effects of hydantoin anticonvulsants by antagonism of their central nervous system (CNS) effects; an increase in hydantoin dosage may be necessary for patients who receive folic acid supplementation.
Antacids, aluminum- or magnesium-containing (with orally administered folic acid). Prolonged use of aluminum- and/or magnesium containing antacids may decrease folic acid absorption by lowering the pH of the small intestine; patients should be advised to take antacids at least 2 hours after folic acid.
Antibiotics. May interfere with the microbiologic method of assay for serum and erythrocyte folic acid concentrations and cause falsely low results.
Cholestyramine (with orally administered folic acid). Concurrent use with folic acid may interfere with absorption of folic acid; folic acid supplementation taken at least 1 hour before or 4 to 6 hours after cholestyramine is recommended in patients receiving cholestyramine for prolonged periods.
Methotrexate or pyrimethamine or triamterene or trimethoprim. Act as folate antagonists by inhibiting dihydrofolate reductase; most significant with high doses and/or prolonged use; leucovorin calcium must be used instead of folic acid in patients receiving these medications.
Sulfonamides, including sulfasalazine (with orally administered folic acid). Inhibit absorption of folate; folic acid requirements may be increased in patients receiving sulfasalazine.
Zinc supplements (with orally administered folic acid). Some studies have found that folate may decrease the absorption of zinc, but not in the presence of excessive zinc; other studies have found no inhibition.
Absorption of folic acid is decreased in chronic alcoholics. This effect can be partially reversed by abstinence from alcohol.
Folic acid is incompatible with oxidising and reducing agents and with ions of heavy metals.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A*)
Therapeutic indications include use as a prophylactic in pregnancy and supplementation of lactating women when they are folic acid deficient. Folic acid is excreted in breast milk.
*Australian categorization definition of Category A: "Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed."
Use in lactation. Folic acid is excreted in breast milk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Note. No side effects other than an allergic reaction have been reported with folic acid administration, even at doses of up to 10 times the recommended dietary allowances (RDA) for 1 month.
The following side/adverse effects have been selected on the basis of their potential clinical significance (possible signs and symptoms in parentheses where appropriate) - not necessarily inclusive:
Those indicating need for medical attention - incidence rare. Allergic reaction, specifically bronchospasm (shortness of breath; troubled breathing; tightness of chest; wheezing), erythema (reddened skin), fever, general malaise (general weakness or discomfort), skin rash or itching.
Note. Side effects including abdominal bloating and gas, anorexia, confusion, depression, difficulty in concentrating, excitement, irritability, impaired judgment, nausea, trouble in sleeping, and unpleasant taste were reported in a study of patients taking 15 mg daily.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
In a double blind randomised trial 15 mg of folic acid per day for one month produced no signs of toxicity in healthy volunteers.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Folic acid is reduced to a number of compounds including tetrahydrofolic acid in the body. In the reduced form it is a coenzyme acting as an acceptor of one-carbon units. It is required for the synthesis of purine and pyrimidine bases, for the metabolism of amino acids such as serine-glycine interconversion, methionine methyl group biosynthesis and the degradation of histidine. It is involved in the maturation of all rapidly proliferating tissues particularly those of bone marrow and the gastrointestinal tract. Folate deficiency leads to megaloblastic anaemia.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Distribution. Protein binding is extensive (to plasma proteins). The principal storage site is the liver. Folic acid is distributed into breast milk.
Metabolism. Hepatic. Folic acid is converted (in the presence of ascorbic acid) in the liver and plasma to its metabolically active form (tetrahydrofolic acid) by dihydrofolate reductase.
Excretion. There is an enterohepatic circulation for folate; about 4 to 5 micrograms is excreted in the urine daily. Administration of larger doses of folic acid leads to proportionately more of the vitamin being excreted in the urine.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each 1 mL solution contains the following excipient ingredients:
Disodium edetate: 2 mg;
Sodium hydroxide: qs to adjust pH;
Water for injections: qs to 1 mL.
6.2 Incompatibilities
Folic acid is incompatible with oxidising and reducing agents and with ions of heavy metals (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light. Keep out of reach of children.
6.5 Nature and Contents of Container
Container type. Amber glass vial sealed with a rubber stopper and an aluminium seal with a flip-top insert.
Pack sizes. Biological Therapies Folic Acid 15 mg in 1 mL Injection is contained in a 2 mL amber glass vial and has three pack sizes:
10 x 1 mL vials per carton;
6 x 1 mL vials per carton;
1 x 1 mL vial per carton.
AUST R 106971.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Molecular formula: C19H19N7O6, Molecular weight: 441.4.
Chemical structure.
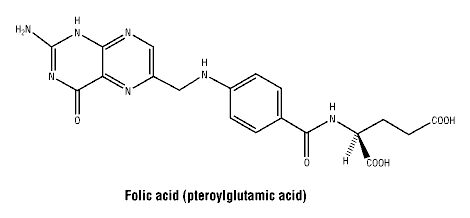
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
19 January 2006
Date of Revision
01 April 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.