Biological Therapies Methylcobalamin 10 mg in 2 mL Injection
Brand Information
| Brand name | Biological Therapies Methylcobalamin 10 mg in 2 mL Injection |
| Active ingredient | Mecobalamin (co-methylcobalamin) |
| Schedule | Unscheduled |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Biological Therapies Methylcobalamin 10 mg in 2 mL Injection
Summary CMI
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Methylcobalamin 10 mg in 2 mL Injection contains the active ingredient mecobalamin (Vitamin B12). Mecobalamin is used to help with the symptoms of pernicious anaemia, or for megaloblastic anaemia if Vitamin B12 deficiency is the diagnosed cause. Methylcobalamin Injection may help to treat peripheral and diabetic neuropathies (diseases of the nerves) and may improve your symptoms. Ask your doctor if you have any questions about why Methylcobalamin Injection has been prescribed for you. For more information, see Section 1. Why am I using Biological Therapies Methylcobalamin 10 mg in 2mL Injection? in the full CMI.
2. What should I know before I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Do not use if you have ever had an allergic reaction to mecobalamin or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Methylcobalamin 10 mg in 2 mL Injection and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
- Your doctor will tell you how much Methylcobalamin Injection you will need to be given and for how long it is to be given. This is determined by many factors including your body weight and your medical condition. Methylcobalamin Injection is used in one patient on one occasion only. The usual dose is 10 mg by slow intramuscular injection. Methylcobalamin 10 mg in 2 mL Injection will be injected intramuscularly (into the buttock muscle) by your doctor. More instructions can be found in Section 4. How do I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection? in the full CMI.
5. What should I know while using Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Biological Therapies Methylcobalamin 10 mg in 2mL Injection? in the full CMI.
6. Are there any side effects?
Methylcobalamin Injection may have unwanted side effects in a few people which are usually mild but may be serious. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection contains the active ingredient Mecobalamin. Mecobalamin belongs to a group of vitamins called cobalamins. The cobalamins are various different forms of Vitamin B12.
Mecobalamin is used to help with the symptoms of pernicious anaemia, or for megaloblastic anaemia if Vitamin B12 deficiency is the diagnosed cause. Methylcobalamin Injection may help to treat peripheral and diabetic neuropathies and may improve your symptoms. Your doctor may prescribe Methylcobalamin Injection for another reason. Ask your doctor if you have any questions about why Methylcobalamin Injection has been prescribed for you.
2. What should I know before I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Warnings
Do not use Biological Therapies Methylcobalamin 10 mg in 2 mL Injection if:
- You are allergic to B vitamins, or any of the ingredients listed at the end of this leaflet.
- You are pregnant and are diagnosed with megaloblastic anaemia.
- You are breastfeeding infants
- You do not have a confirmed diagnosis that Vitamin B12 may be useful.
- The solution in the bottle is not clear or contains particles.
- The packaging is torn or shows signs of tampering.
- Always check the ingredients to make sure you can use this medicine.
- Precaution should be taken if you have low potassium and are diagnosed with pernicious anaemia.
Check with your doctor if you:
- Have any other medical conditions such as an allergy to any of the ingredients listed at the end of this leaflet.
- Have had an allergy to any other medicines or any other substances, such as foods, preservatives or dyes.
Your doctor may test if you have an allergy to mecobalamin by injecting a small amount under your skin.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Do not have Methylcobalamin 10 mg in 2mL Injection administered if you are pregnant and are diagnosed with megaloblastic anaemia. Cobalamins can mask the effects of megaloblastic anaemia caused by folate deficiency. Folate deficiency in pregnancy is a risk for birth defects in your baby. You must have Vitamin B12 deficiency confirmed by blood tests before using Methylcobalamin Injection to treat megaloblastic anaemia.
Women receiving Methylcobalamin 10 mg in 2mL Injection should not breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Your doctor or pharmacist has more information about medicines to be careful with, use correctly or to avoid while you are being treated with Methylcobalamin Injection.
Some medicines may interfere with Biological Therapies Methylcobalamin 10 mg in 2mL Injection and affect how it works.
- Colchicine (an anti-inflammatory used to treat gout), para-aminosalicylic acid (an antibiotic used to treat tuberculosis), and heavy alcohol intake longer than 2 weeks may lower vitamin B12 absorption.
- Oral contraceptives may reduce the amount of cyanocobalamin in your blood.
- Vitamin B12 concentrations in the blood may be reduced following administration of large and continuous doses of folic acid.
- The therapeutic response to vitamin B12 may be impaired by drugs with bone marrow suppressing effects such as chloramphenicol.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Biological Therapies Methylcobalamin 10 mg in 2mL Injection.
4. How do I use Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
How much to take / use
- Your doctor will tell you how much Methylcobalamin Injection will need to be given and for how long it is to be given. This is determined by many factors including your body weight and your medical condition. The usual dose is 10 mg by slow intramuscular Injection. Methylcobalamin injection is used in one patient on one occasion only.
- Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
When to take / use Biological Therapies Methylcobalamin 10 mg in 2mL Injection
- Methylcobalamin Injection is only available from a medical practitioner. Your doctor will determine when Methylcobalamin 10 mg in 2mL Injection is suitable.
If you forget an appointment or need to change an appointment:
You will need to make another appointment as soon as possible.
If you use too much Biological Therapies Methylcobalamin 10 mg in 2mL Injection
Your doctor should be the only person to prescribe Methylcobalamin 10 mg in 2mL Injection, so an overdose is not likely to occur.
If you think that you have used too much Methylcobalamin 10 mg in 2mL Injection, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Things you should do
Call your doctor straight away if you:
- are about to be started on any new medicine, remind your doctor and pharmacist that you are being treated with Methylcobalamin Injection.
- need to have any urine or blood tests, tell your doctor that you are being given Methylcobalamin 10mg in 2mL Injection. Methylcobalamin 10 mg in 2mL Injection may affect the results of some of these tests.
- become pregnant while being treated with Methylcobalamin Injection.
- feel that giving Methylcobalamin Injection is not helping your condition.
Remind any doctor, dentist, surgeon, anaesthetist or pharmacist you visit that you are using Methylcobalamin 10 mg in 2mL Injection.
Be sure to keep all appointments with your doctor so that your progress can be checked.
Things you should not do
- Do not attempt to inject Methylcobalamin 10 mg in 2mL Injection yourself.
- Do not take any other medicines, whether they require a prescription or not, without first telling your doctor or consulting a pharmacist.
Driving or using machines
This medicine is not expected to affect your ability to drive a car or operate machinery.
Drinking alcohol
Tell your doctor if you drink alcohol.
Heavy alcohol intake longer than 2 weeks may lower vitamin B12 absorption.
Looking after your medicine
Methylcobalamin 10 mg in 2mL Injection is usually stored in the doctor's surgery or clinic, or at the pharmacy (the following information applies to the healthcare professional).
Unopened vials of Methylcobalamin Injection are to be stored at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light.
Seal box immediately after removal of each vial and store in a dark place.
Keep it where young children cannot reach it.
When to discard your medicine
Each vial is for SINGLE USE in one patient on one occasion only. It will be used once only and then it will be discarded. It must never be stored after it is opened or used for more than one person.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Sensitivity/injection site reaction:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Severe allergic reaction:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Administration of Methylcobalamin Injection may cause anaphylactic shock. This is a serious side effect and required immediate attention by your doctor. This side effect is rare and only occurs during injection. Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side affects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Biological Therapies Methylcobalamin 10 mg in 2mL Injection contains
| Active ingredient (main ingredient) | Mecobalamin 10 mg |
| Other ingredients (inactive ingredients) | Sodium chloride Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Biological Therapies Methylcobalamin 10 mg in 2mL Injection looks like
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection is a clear bright red coloured solution. It is contained in an amber glass vial sealed with a rubber stopper and an aluminium cap.
This medicine may be available in 4 pack sizes:
6 x 2 mL vials
5 x 2 mL vials
3 x 2 mL vials
1 x 2 mL vial
Australian Registration Number: AUST R 22435
Who distributes Biological Therapies Methylcobalamin 10 mg in 2mL Injection?
Biological Therapies
A Division of Orthomolecular Medisearch
Laboratories Pty Ltd
5 / 20 – 30 Malcolm Road, Braeside VIC 3195
Australia
Tel: + 61 3 9587 3948
Fax: +61 3 9587 1720
Website: www.biologicaltherapies.com.au
Email: info@biol.com.au
This leaflet was prepared in October 2025.

Brand Information
| Brand name | Biological Therapies Methylcobalamin 10 mg in 2 mL Injection |
| Active ingredient | Mecobalamin (co-methylcobalamin) |
| Schedule | Unscheduled |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Mecobalamin.
2 Qualitative and Quantitative Composition
Each vial contains a minimum of 2 mL of solution and has mecobalamin 10 mg as an active ingredient.
3 Pharmaceutical Form
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection is a clear bright red coloured solution for injection supplied in amber glass vials. The pH of the solution is 6.0-8.0.
For the full list of excipients, see Section 6.1 List of Excipients.
4 Clinical Particulars
4.1 Therapeutic Indications
Cobalamin deficiency may exist that is unable to be corrected by oral intake and in these cases parenteral administration may be preferable. Oral administration of vitamin B12 may be insufficient in pernicious anaemia, malabsorption disorders, gastrectomy and gastrointestinal pathologies.
Specific indications may include:
1. Pernicious anaemia as a result of in situ B12 deficiency. In pernicious anaemia mecobalamin is disproportionately reduced in relation to other cobalamins.
2. As an adjunct to the treatment of peripheral neuropathies and diabetic polyneuropathy.
4.2 Dose and Method of Administration
Warnings. A sensitivity history should be obtained from the patient prior to administration of vitamin B12. An intradermal test dose is recommended before vitamin B12 is administered to patients who may be sensitive to cobalamins.
Anaphylactic shock and death have been reported after parenteral vitamin B12 administration. Should anaphylactic shock occur, immediately stop administration of the product and administer the appropriate dose of adrenaline 1:1000 (1 mg/1 mL) intramuscularly. This may be repeated at five minute intervals, if clinically indicated. Additionally, intravenous administration of corticosteroids and antihistamines may be considered.
Diagnosis of vitamin B12 deficiency should be confirmed by laboratory investigation before institution of vitamin B12 therapy. Do not use vitamin B12 until diagnosis is fully established, as it may mask symptoms of subacute degeneration of the spinal cord, or of the true diagnosis of pernicious anaemia.
Hematocrit, reticulocyte count, vitamin B12, folate and iron levels should be obtained prior to treatment.
If folate levels are low, folic acid should also be administered.
Patients with pernicious anemia should be informed that they will require monthly injections of vitamin B12 for the remainder of their lives. Failure to do so will result in return of the anemia and in development of incapacitating and irreversible damage to the nerves of the spinal cord. Also, patients should be warned about the danger of taking folic acid in place of vitamin B12, because the former may prevent anemia but allow progression of subacute combined degeneration.
If the solution is cloudy or contains visually detectable particles, please contact the quality assurance manager at Biological Therapies.
Single use only.
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection contains no antimicrobial agent. Use in one patient on one occasion only and discard any residue.
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection should be administered as soon as possible after removal from the box. It is to be administered by slow intramuscular (deep intragluteal) injection. Frequency as determined by the physician.
Patient monitoring. During the initial treatment of patients with pernicious anemia, serum potassium must be observed closely during the first 48 hours and potassium replaced if necessary.
Hematocrit and reticulocyte counts should be repeated daily from the fifth to seventh days of therapy and then frequently until the hematocrit is normal.
If reticulocytes have not increased after treatment or if reticulocyte counts do not continue at least twice normal while the hematocrit is less than 35%, diagnosis or treatment should be re-evaluated. Repeat determinations of iron and folic acid may reveal a complicating illness that might inhibit the response of the marrow.
Regular blood tests to determine vitamin B12 levels are advisable during treatment.
4.3 Contraindications
Known hypersensitivity to B12, or pre-existing hypervitaminosis. Known sensitivity to mecobalamin or any other ingredient in the preparation. Known sensitivity to cobalt.
Mecobalamin should not, if possible, be given to patients or used to treat megaloblastic anaemia of pregnancy without first confirming the diagnosis (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
4.4 Special Warnings and Precautions for Use
The therapeutic response to vitamin B12 may be impaired by concurrent infection, uraemia, folic acid or iron deficiency or by drugs with bone marrow suppressing effects such as chloramphenicol.
Allergy. A sensitivity history should be obtained from the patient prior to administration of vitamin B12. An intradermal test dose is recommended before vitamin B12 is administered to patients who may be sensitive to cobalamins.
Anaphylactic shock and death have been reported after parenteral vitamin B12 administration. Should anaphylactic shock occur, immediately stop administration of the product and administer the appropriate dose of adrenaline 1:1000 (1 mg/1 mL) intramuscularly. This may be repeated at five minute intervals, if clinically indicated. Additionally, intravenous administration of corticosteroids and antihistamines may be considered.
Hypokalaemia. Hypokalaemia and cardiac arrest have been reported when megaloblastic anaemia is treated intensively.
Serum potassium is to be carefully monitored during the initial phase of treatment in pernicious anaemia.
Degenerative lesions of the spinal cord. Vitamin B12 deficiency that is allowed to progress for longer than 3 months may produce permanent degenerative lesions of the spinal cord.
Do not use vitamin B12 until diagnosis is fully established, as it may mask symptoms of subacute degeneration of the spinal cord, or of the true diagnosis of pernicious anaemia.
Folic acid. Folic acid may potentiate the neurological complications of vitamin B12 deficiency, so should not be administered to patients with pernicious anaemia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Doses of folic acid greater than 0.1 mg per day may result in hematologic remission in patients with vitamin B12 deficiency. Neurologic manifestations will not be prevented with folic acid, and if not treated with vitamin B12, irreversible damage will result.
Folate deficiency. Doses in excess of 10 micrograms daily may produce a haematological response in patients with folate deficiency; indiscriminate administration may mask the true diagnosis of folate deficiency.
Diet. A vegetarian diet which contains no animal products (including milk products or eggs) does not supply any vitamin B12. Patients following such a diet, should be advised to take oral vitamin B12 regularly.
Other warnings. Treatment with vitamin B12 may unmask polycythaemia vera, because vitamin B12 deficiency may suppress the symptoms of this condition.
Early Leber's disease (hereditary optic nerve neuropathy involving impaired cyanide detoxification) is not relevant for mecobalamin as it contains no cyanide. Unlike hydroxocobalamin, mecobalamin is not recommended for treatment of cyanide poisoning.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. Most antibiotics, methotrexate and pyrimethamine invalidate folic acid and vitamin B12 blood assays.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Colchicine, para-aminosalicylic acid and heavy alcohol intake for longer than two weeks may produce malabsorption of vitamin B12.
Serum concentrations of cyanocobalamin may be lowered by oral contraceptives.
Vitamin B12 concentrations in the blood may be reduced following administration of large and continuous doses of folic acid. Folic acid administration may impair the therapeutic response to cyanocobalamin.
The therapeutic response to vitamin B12 may be impaired by drugs with bone marrow suppressing effects such as chloramphenicol.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. Exempt from classification in the Australian categorisation system for prescribing medicines in pregnancy.
The need for vitamin B12 is increased by pregnancy and lactation.
Problems in humans have not been documented with intake of normal daily amounts. Vitamin B12 crosses the placental barrier. There are no studies establishing the safety of this drug during pregnancy. It is not recommended for pregnancy unless the expected benefits outweigh any potential risk to the infant.
Megaloblastic anaemia occurring during pregnancy is usually due to folic acid deficiency rather than vitamin B12 deficiency. Mecobalamin should not be used for the treatment of megaloblastic anaemia of pregnancy caused by folic acid deficiency.
Use in lactation. Mecobalamin is distributed into breast milk. Therefore it is not recommended for breastfeeding mothers unless the expected benefits to the mother outweigh any potential risk to the infant.
Deficiency has been recognized in infants of vegetarian mothers who were breast fed, even though the mothers had no symptoms of deficiency at the time.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Generalized. Anaphylactic shock and death have been reported with administration of parenteral vitamin B12 (see Section 4.4 Special Warnings and Precautions for Use). Allergic hypersensitivity reactions have rarely occurred.
Cardiovascular. Pulmonary oedema and congestive heart failure early in treatment; peripheral vascular thrombosis, cardiac arrest, low blood pressure.
Nervous system. Severe dizziness, drowsiness, muscular paralysis, vision problems, loss of consciousness.
Hematological. Polycythemia vera, hypokalaemia.
Gastrointestinal. Mild transient diarrhea, prolonged abdominal pain, prolonged nausea or vomiting.
Dermatological. Itching; transitory exanthema.
Miscellaneous. Feeling of swelling of entire body bleeding.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
No overdosage has been reported with this medicine.
For information on the management of overdose please contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Several chemically related forms of vitamin B12, differing in slight modification of a side chain attached to the cobalamin nucleus have been isolated. Such variants of vitamin B12 include cyanocobalamin, hydroxocobalamin, adenosylcobalamin and mecobalamin.
Vitamin B12 is essential for normal growth, haematopoiesis, production of all epithelial cells and maintenance of myelin throughout the nervous system. Whenever nucleic acid synthesis occurs and therefore whenever cell reproduction occurs, vitamin B12 is required.
The amounts of vitamin B12 needed to maintain normal blood forming functions are small and low doses are sufficient to correct the usual symptoms of vitamin B12 deficiency.
Vitamin B12 acts as an enzyme or coenzyme in a number of metabolic processes and is transformed in the body to at least two compounds which possess enzymatic properties.
In humans there are two active forms of vitamin B12:
i) coenzyme B12 (adenosylcobalamin) is required for conversion of propionate to succinate, thus involving vitamin B12 in both fat and carbohydrate metabolism;
ii) mecobalamin acts in a transmethylation process converting homocysteine to methionine, thus involving vitamin B12 in fat and protein metabolism.
Mecobalamin constitutes 60-80% of the total plasma cobalamin. In pernicious anaemia, mecobalamin is disproportionately reduced in relation to the other cobalamins.
Mecobalamin deficiency leads to neurological symptoms of pernicious anaemia. In some cases of vitamin B12 deficiency, severe neurological symptoms develop, as vitamin B12 is necessary for the formation of protein structures required for the integrity of the nerve cell and myelin sheath.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Absorbed vitamin B12 is transported via specific B12 binding proteins, transcobalamin I and II to the various tissues. The liver is the main organ for vitamin B12 storage.
Gastrointestinal absorption of vitamin B12 depends on the presence of sufficient intrinsic factor and calcium ions. Intrinsic factor deficiency causes pernicious anemia, which may be associated with subacute combined degeneration of the spinal cord. Prompt parenteral administration of vitamin B12 prevents progression of neurologic damage.
The average diet supplies about 5 to 15 micrograms/day of vitamin B12 in a protein bound form that is available for absorption after normal digestion. Vitamin B12 is not present in foods of plant origin, but is abundant in foods of animal origin. In people with normal absorption, deficiencies have been reported only in strict vegetarians who consume no products of animal origin (including no milk products or eggs).
Vitamin B12 is bound to intrinsic factor during transit through the stomach; separation occurs in the terminal ileum in the presence of calcium, and vitamin B12 enters the mucosal cell for absorption. It is then transported by the transcobalamin binding proteins. A small amount (approximately 1% of the total amount ingested) is absorbed by simple diffusion, but this mechanism is adequate only with very large doses. Oral absorption is considered too undependable to rely on in patients with pernicious anemia or other conditions resulting in malabsorption of vitamin B12.
Excretion. Within 48 hours after injection of 100 or 1000 micrograms of mecobalamin, 50 to 98% of the injected dose may appear in the urine. The major portion is excreted within the first eight hours. Intravenous administration results in even more rapid excretion with little opportunity for liver storage.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. Long-term studies in animals to evaluate carcinogenic potential have not been done. There is no evidence from long-term use in patients with pernicious anemia that mecobalamin is carcinogenic.
Pernicious anemia is associated with an increased incidence of carcinoma of the stomach, but this is believed to be related to the underlying pathology and not to treatment with mecobalamin.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each 2 mL solution contains the following excipient ingredients:
sodium chloride 18 mg;
water for injections qs to 2 mL.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light.
Biological Therapies Methylcobalamin 10 mg in 2 mL Injection is sensitive to light. Seal box immediately after removal of each vial and store in fridge.
6.5 Nature and Contents of Container
Container type. Amber glass vial sealed with a rubber stopper and an aluminium seal.
Pack sizes. 6 x 2 mL vials, 3 x 2 mL vials, 5 x 2 mL vials, 1 x 2 mL vial.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
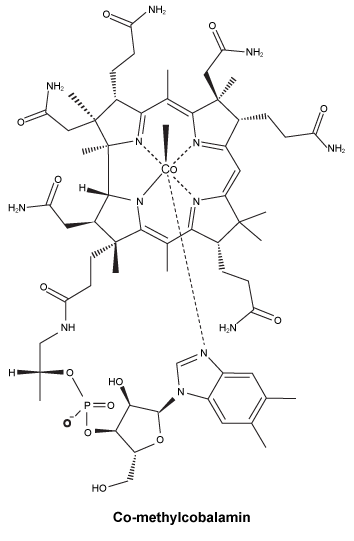
CAS number. 13422-55-4.
Mecobalamin appears as dark red crystals or crystalline powder. It is sparingly soluble in water, slightly soluble in ethanol and practically insoluble in acetonitrile.
7 Medicine Schedule (Poisons Standard)
This medicine is not scheduled.
Date of First Approval
08 October 1991
Date of Revision
21 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.