Bisalax Enema
Brand Information
| Brand name | Bisalax Enema |
| Active ingredient | Bisacodyl |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Bisalax Enema.
Full CMI
BISALAX® enema
Active ingredient(s): bisacodyl
Consumer Medicine Information (CMI)
This leaflet provides important information about using BISALAX enemas. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using BISALAX enemas.
Where to find information in this leaflet:
1. Why am I using BISALAX?
2. What should I know before I use BISALAX?
3. What if I am taking other medicines?
4. How do I use BISALAX?
5. What should I know while using BISALAX?
6. Are there any side effects?
7. Product details
1. Why am I using BISALAX?
BISALAX contains the active ingredient bisacodyl. BISALAX is a stimulant laxative. It acts on the wall of the large bowel, encouraging bowel movements by increasing muscle contractions which push the stool mass along.
BISALAX is used to provide relief from constipation or to empty the large bowel before some medical procedures, operations or X-rays.
2. What should I know before I use BISALAX?
Warnings
Do not use BISALAX if:
- you are allergic to bisacodyl, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have:
- cramps, colic or stomach pain
- nausea or vomiting
- appendicitis, a pain in the abdomen or stomach
- inflammatory bowel disease
- a condition of the intestine called ‘ileus’ (swelling of the intestines when the bowel muscles have stopped working)
- a blocked intestine
- undiagnosed stomach pain
- dehydration
- low potassium levels in the blood (hypokalemia)
- an anal tear (anal fissure)
- inflammation or swollen veins (haemorrhoids) in or around the back passage (rectum) or rectal ulcers.
Check with your doctor or pharmacist if you:
- have any other medical conditions
- have noticed a persistent change in bowel habits
- have been using laxatives for a period longer than one week
- have rectal bleeding after the use of a laxative
- have had any unusual or no reaction to laxatives
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
BISALAX is not known to be harmful in pregnancy or while breastfeeding. However, if you are pregnant or breastfeeding you should discuss this with your doctor or pharmacist prior to treatment.
Use in children
BISALAX enemas may be used in children, however should be medically supervised.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with BISALAX and affect how it works.
In particular, tell your doctor or pharmacist if you are taking any of the following:
- diuretics (water tablets)
- corticosteroids
- digoxin (a medicine to treat heart problems)
- some controlled release medicines
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect BISALAX.
4. How do I use BISALAX?
How much and when to use BISALAX
- BISALAX enemas may be used on their own, or in combination with BISALAX tablets.
- Follow the instructions provided with the medicine.
- Do not exceed the recommended dosage.
Adults
Depending on the condition being treated, the entire contents of an enema should be inserted into the rectum either in the afternoon, evening or morning according to the dosage guidelines below or as instructed by your doctor.
DOSE – Adults; for constipation-type conditions:
| Acute constipation: | Initially one enema, then 1 or 2 BISALAX tablets late in the evening and 1 BISALAX enema in the morning (after breakfast) for about 3 days. |
| Elderly patients: | One BISALAX enema in the morning (after breakfast) on days in which bowel motion is desired. |
| Chronic constipation: | As per doctor's instructions |
| Prior to childbirth: | One BISALAX enema immediately before delivery is anticipated. |
| After childbirth or surgery: | One BISALAX enema to be used immediately after breakfast for two or three days. |
| After X-ray: | To treat constipation due to barium contrast medium, use one enema. |
DOSE – Adults; to empty the bowel for medical procedures:
| Before X-ray: | The day prior to the examination: take 2 BISALAX tablets at lunchtime and 2 tablets in the evening. The day of the X-ray: One BISALAX enema in the rectum in the morning one to two hours prior to examination. Note: No food should be eaten from the time of taking BISALAX tablets until after the X-ray has been completed. |
| Surgery: | As per doctor's instructions |
Children
BISALAX tablets are not recommended for use in children under 6 years.
Depending on the child's age, either half or the entire contents of an enema should be inserted into the rectum either in the afternoon, evening or morning according to the dosage guidelines below or as instructed by your doctor.
DOSE – Children; for constipation:
| Under 3 Years: | Half a BISALAX enema (Note: only half the point of the tube should be inserted). |
| 3 to 6 years: | One BISALAX enema as required. |
| 6-12 years: | One BISALAX tablet in the evening and one enema in the morning. |
DOSE – Children; to empty the bowel for stomach x-ray examination:
| Under 1 year: | Half a BISALAX enema the night before the examination and half an enema in the morning on the day of the examination. |
| 1 to 3 years: | One BISALAX enema the night before the examination and one enema in the morning of the day of the examination. |
| 3 to 6 years: | One BISALAX enema in the afternoon of the day before the examination and one BISALAX enema in the morning of the day of the examination. |
| 6 to 12 years: | One BISALAX tablet at 10am and one enema in the afternoon of the day before the examination. |
| Over 12 years: | Two BISALAX tablets at 10am and two tablets in the evening of the day before the examination and one BISALAX enema on the morning of the day of the examination. |
How to use BISALAX
- Take the enema out of the refrigerator and allow it to reach room temperature before use.
- Shake the enema well.
- Take off the plastic cap from the end of the nozzle.
- If required, lubricate the tip with a lubricant to aid insertion.
- Lie on your left side with the left leg extended and the right leg bent, in preparation for administration of the enema.
- Carefully insert the applicator nozzle tip into the rectum, then press on the end of the plastic tube to squeeze the liquid out gently.
- Remain lying down for a couple of minutes before sitting or standing up.
- Discard the container after use, even if you have only used half the contents.
- Visit a toilet as soon as the need to pass a bowel motion occurs. A bowel motion should normally occur within 5-15 minutes.
How long to use BISALAX
- Laxative products such as BISALAX should not be used for a period longer than one week, unless directed by a doctor or pharmacist. Prolonged use of laxatives is undesirable, and may lead to dependence and further reliance on the laxative to achieve a bowel motion. This can cause a continuing cycle of constipation.
- If you require longer use, or if BISALAX does not produce a bowel movement as expected, consult your doctor or pharmacist.
If you use too much BISALAX
If you think that you have used too much BISALAX, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using BISALAX?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using BISALAX.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how BISALAX affects you.
BISALAX may cause dizziness in some people.
Looking after your medicine
- Store BISALAX enemas in the original packaging at a temperature of 2°C to 8°C (Refrigerate. Do not freeze).
- Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
Stomach or bowel-related:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs and symptoms of an allergic reaction:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What BISALAX contains
| Active ingredient (main ingredient) | Each enema contains 10 mg bisacodyl/5 mL tube |
| Other ingredients (inactive ingredients) | citric acid monohydrate macrogol 3000 macrogol 400 macrogol 4000 sodium citrate |
Do not take this medicine if you are allergic to any of these ingredients.
What BISALAX looks like
BISALAX comes as a yellow tube with yellow flip off cap which contains a white to grey viscous material. Available in packs of 25 enemas (AUST R 27900).
Who distributes BISALAX
Orion Pharma (Aus) Pty Limited
Level 24, Tower 3, 300 Barangaroo Avenue,
Sydney NSW 2000, Australia
Telephone: 1800 861 913
This leaflet was prepared in June 2025.
Brand Information
| Brand name | Bisalax Enema |
| Active ingredient | Bisacodyl |
| Schedule | Unscheduled |
MIMS Revision Date: 01 January 2023
1 Name of Medicine
Bisacodyl.
2 Qualitative and Quantitative Composition
Bisalax enema contains 10 mg bisacodyl/5 mL tube.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Enema.
White to grey viscous material.
4 Clinical Particulars
4.1 Therapeutic Indications
Constipation due to confinement to bed, unaccustomed diet or change of environment. Constipation in cases of serious diseases, circulatory disturbances and metabolic disorders; occasional use in chronic constipation; bowel re-training; postoperatively in cases of haemorrhoids and anal fissures; colostomies; preparation for radiography; preparation for sigmoidoscopy or proctoscopy; preoperative preparation; postoperative care; antepartum and postpartum constipation; and preparation for delivery.
4.2 Dose and Method of Administration
The enemas can be used in conjunction with tablets. Taken at bedtime, Bisalax tablets are almost invariably effective by the following morning. When taken before breakfast, they usually produce an effect within 6-8 hours. The effects of the enema occur normally within 5 to 15 minutes.
Adults. Preparation for X-ray examination of abdominal organs. The day prior to the examination, 2 tablets are taken orally at lunchtime and 2 tablets in the evening. One enema is inserted into the rectum in the morning one to two hours prior to examination. No food should be given following the administration of the tablets to prevent re-accumulation of material in the caecum.
After X-ray examination. To treat constipation due to barium contrast medium, use one enema.
Acute constipation. Initially one enema, then 1 or 2 tablets late in the evening and 1 enema in the morning (after breakfast) for about three days.
Constipation in elderly patients. One enema in the morning (after breakfast) on days in which defecation is desired.
Chronic constipation and bowel retraining. By gradually increasing the interval between doses as colonic tone improves, the drug has been found to be effective in redeveloping proper bowel hygiene. There is no tendency to "rebound".
Antepartum use. One enema immediately before delivery is anticipated.
Postpartum or postoperative constipation. One enema to be used immediately after breakfast for two or three days.
Preparation for sigmoidoscopy proctoscopy. For sigmoidoscopy scheduled in advance, administration of tablets the night before and one enema rectally one hour before the examination will almost invariably result in adequate preparation.
Preparation for surgery of the colon. Two tablets at lunchtime and two tablets in the evening on the day before the operation. One enema should be inserted into the rectum on the morning of surgery.
Colostomies. Two tablets the night before inserted into the colostomy opening in the morning will frequently make irrigations unnecessary and in other cases will expedite the procedure.
Children. Preparation for X-ray examination of abdominal organs. Under 1 year. Half an enema the night before the examination and half an enema in the morning on the day of the examination.
1 to 3 years. One enema the night before the examination and one enema in the morning of the day of the examination.
3 to 6 years. One enema in the afternoon of the day before the examination and one enema in the morning of the day of the examination.
6 to 12 years. One tablet at 10 am and one enema in the afternoon of the day before the examination.
Over 12 years. Two tablets at 10 am and two tablets in the evening of the day before the examination and one enema on the morning of the day of the examination.
Constipation. Under 3 years. Half an enema. (Note. Only half the point of the tube should be inserted).
3 to 6 years. One enema as required.
6 to 12 years. One tablet in the evening, and one enema in the morning.
(Note: Bisalax tablets are not recommended for use in children under 6 years of age.)
Advice to patients. If you have noticed a persistent change in bowel habits, consult a physician before using a laxative.
Laxative products should not be used longer than one week unless directed by a physician (see Section 4.4 Special Warnings and Precautions for Use).
Rectal bleeding or failure to have a bowel movement after use of a laxative may indicate a serious condition. Discontinue use and consult a physician.
Bisalax enemas should be administered rectally when required (see Section 4.2 Dose and Method of Administration). Pull off plastic cap from end of nozzle, insert nozzle in rectum and squeeze tube to administer contents. The nozzle of the Bisalax enema may be coated with a lubricant to aid insertion.
4.3 Contraindications
Bisacodyl has the same contraindications as other laxatives.
All laxatives are contraindicated in patients with cramps, colic, nausea, vomiting or other symptoms as in appendicitis, inflammatory bowel disease, acute abdominal situations such as ileus, intestinal obstruction or any other undiagnosed abdominal pain.
Bisalax is also contraindicated in dehydration, hypokalaemia, hypersensitivity to bisacodyl or to any of the excipients.
Bisalax enemas are contraindicated in patients with anal fissure, ulcerative proctitis with mucosal damage and ulcerated haemorrhoids.
4.4 Special Warnings and Precautions for Use
Prolonged use of laxatives may lead to dependence, chronic constipation and loss of bowel function. "Cathartic colon" with atony, dilatation and resemblance of ulcerative colitis or proctitis, is also possible.
In addition, continued use of irritant laxatives may produce serious loss of electrolytes, especially potassium and result in disturbances of cardiac function, diarrhoea, dehydration, malabsorption and protein losing enteropathy.
Syncope has been reported during the use of Bisalax. The event could be related to defecation syncope or to a vasovagal response to abdominal pain.
Use in the elderly. No data available.
Paediatric use. Children should not make frequent use of Bisalax unless medically supervised.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant use of diuretics or systemic corticosteroids may increase the risk of electrolyte imbalance. This interaction is significant only if excessive doses of Bisalax are taken.
By increasing the motility of the small intestine and speeding the passage, Bisalax can alter the bioavailability of some drugs absorbed mainly from the small intestine such as digoxin and controlled release formulations.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Category A of Australian Categorisation of Risk of Drug Use in Pregnancy.
Use in lactation. There is no contraindication to use of Bisalax by nursing mothers, however as bisacodyl is excreted in the breast milk in low quantities, the drug should only be used during lactation when clearly indicated.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Bisacodyl is well tolerated and side effects are rare. However, side effects in the form of diarrhoea, local rectal irritation, proctitis, slight indisposition, epigastralgia, tenesmus, nausea, abdominal pain and cramping have been reported. A possible colic should be treated with atropine derivatives.
Rare cases of vomiting, mild transient skin reactions and hypersensitivity reactions including angioedema and anaphylactoid reactions have also been described.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The precise mode of action is unknown. Bisacodyl is a contact laxative. Its action is restricted mainly to the colon; the motility of the small intestine is not appreciably influenced. Bisacodyl acts locally in the gastrointestinal tract, stimulating sensory nerve endings in the colonic mucosa to produce parasympathetic reflexes resulting in increased peristaltic contractions of the colon. Local axon reflexes as well as segmental reflexes are initiated in the region of contact and contribute to the widespread peristaltic activity producing evacuation. For this reason, bisacodyl may be employed satisfactorily in patients with ganglionic blockage or spinal cord damage (paraplegia, poliomyelitis etc).
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Although previously thought not to be absorbed from the digestive tract, recent studies have shown that up to 5% of an oral dose of bisacodyl may be absorbed in man. Bisacodyl is excreted mainly in faeces, and partly in urine as the glucuronide. When taken on an empty stomach, bisacodyl tablets will exert their effect in 6 to 8 hours. When taken with food, the effect of the tablets will occur in 10 to 12 hours.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Citric acid monohydrate, macrogol 3000, macrogol 400, macrogol 4000 and sodium citrate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store Bisalax enemas in the original packaging at a temperature at 2°C to 8°C. (Refrigerate. Do not freeze). Before use, allow the enema to reach room temperature.
6.5 Nature and Contents of Container
Yellow LDPE plastic tube with yellow flip off cap. Available in packs of 25 enemas.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
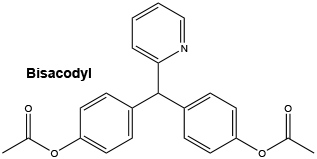
Bisacodyl is a white or almost white crystalline powder; odourless, or almost odourless; and tasteless. Chemically, bisacodyl is 4,4'-(2-pyridylmethylene)di(phenylacetate). Its molecular weight is 361.38.
7 Medicine Schedule (Poisons Standard)
Unscheduled.
Date of First Approval
10 August 1999
Date of Revision
07 November 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.