Blooms the Chemist Famciclovir Once
Brand Information
| Brand name | Blooms the Chemist Famciclovir Once |
| Active ingredient | Famciclovir |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Blooms the Chemist Famciclovir Once.
Summary CMI
BLOOMS THE CHEMIST FAMCICLOVIR ONCE
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
BLOOMS THE CHEMIST FAMCICLOVIR ONCE contains the active ingredient famciclovir. It is an antiviral medicine that is used to treat recurrent outbreaks of cold sores in adults who have a normal immune system.
For more information, see Section 1. Why am I using BLOOMS THE CHEMIST FAMCICLOVIR ONCE? in the full CMI.
2. What should I know before I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
Do not use if you have ever had an allergic reaction to BLOOMS THE CHEMIST FAMCICLOVIR ONCE or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with BLOOMS THE CHEMIST FAMCICLOVIR ONCE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
- Your doctor will give you instruction on how to take BLOOMS THE CHEMIST FAMCICLOVIR ONCE tablets.
More instructions can be found in Section 4. How do I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE? in the full CMI.
5. What should I know while using BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using BLOOMS THE CHEMIST FAMCICLOVIR ONCE? in the full CMI.
6. Are there any side effects?
- Side effects of this medicine may include headache; dizziness; abdominal pain or bloating; nausea; vomiting; diarrhoea; itching or an itchy rash (urticaria); abnormal liver function test results; extreme sleepiness; hallucinations; painful or swollen joints; aching muscles; yellowing of the skin or eyes; palpitations; a rash on other parts of your body; swelling below the surface of the skin; severe blistering of the skin; unexplained bruising; purple patches, itching, burning of the skin; seizures or fits; difficulty breathing or swallowing, wheezing or coughing.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
BLOOMS THE CHEMIST FAMCICLOVIR ONCE is an antiviral medicine that is used to treat recurrent outbreaks of cold sores in adults who have a normal immune system.
Cold sores are an infection caused by a virus called herpes simplex type 1 (HSV-1). The infection is most commonly acquired as a baby or child from contact with parents or relatives, often from kissing.
Cold sores usually begin on or around the lips, mouth, and nose as small red bumps that turn into fluid-filled blisters. Cold sores can be tender and painful. Many people who get cold sores know when one is coming by a tingling, burning, itchy or painful sensation or redness in the area. This can happen very rapidly.
After redness and swelling develop, blisters form. The blisters may weep or burst and this can be painful. Then a shallow ulcer and yellow crust form as the cold sore dries. The crust eventually falls off, exposing new pink-coloured skin. Generally, the sores heal without scarring. After the initial infection has healed, the virus becomes dormant in nerve cells.
Cold sores can be unpredictable. The virus can become active again in the body, even after many years, resulting in recurrent outbreaks.
Even after many years, some people may experience recurring cold sores due to viral reactivation.
Some common triggers to a cold sore may include:
- sun exposure
- stress
- fatigue
- menstrual periods
- fever
- illness
- dry chapped lips
- skin trauma
- a cold.
BLOOMS THE CHEMIST FAMCICLOVIR ONCE does not cure the viral infection, however it helps to relieve the symptoms and shorten the duration of an outbreak.
The best results are obtained if the medicine is started as soon as possible after the first symptoms are noticed. These include tingling, itching or burning, or the appearance of redness or swelling. This is when the virus is reproducing rapidly.
2. What should I know before I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
Warnings
Do not use BLOOMS THE CHEMIST FAMCICLOVIR ONCE if you are allergic medical conditions:
- Famciclovir, the active ingredient
- Penciclovir, a related antiviral medicine
- Any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include shortness of breath, Skin rash, itching or hives, swelling of the face, lips, tongue or other parts of the body, wheezing or troubled breathing.
Do not use BLOOMS THE CHEMIST FAMCICLOVIR ONCE if:
- the packaging is torn or shows signs of tampering
- the expiry date printed on the pack has passed.
Tell your doctor or pharmacist if you are allergic to any other medicines, foods, preservatives, or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- problems with your immune system (which helps fight off infections)
- kidney disease
- liver disease.
Your doctor may want to take special care if you have any of these conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
BLOOMS THE CHEMIST FAMCICLOVIR ONCE should not be used during pregnancy unless necessary. Your doctor will discuss with you the potential risks of taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE during pregnancy, and will also advise you if you should take BLOOMS THE CHEMIST FAMCICLOVIR ONCE while breast-feeding, based on the benefits and risks of your personal situation.
Talk to your doctor or pharmacist if you are not sure whether you should start taking this medicine.
If you have not told them about any of the above, tell him/her before you start taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE against the benefits they expect it will have for you.
There is no evidence that BLOOMS THE CHEMIST FAMCICLOVIR ONCE is addictive.
BLOOMS THE CHEMIST FAMCICLOVIR ONCE is not recommended for use in children or adolescents under 18 years of age.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and BLOOMS THE CHEMIST FAMCICLOVIR ONCE may interfere with each other. These include:
- probenecid, a prescription medicine used to treat gout (a disease with painful, swollen joints caused by uric acid crystals) and to increase blood levels of penicillin-type antibiotics
- raloxifene, a medicine used to treat osteoporosis (a disease which causes bones to become less dense, gradually making them weaker, more brittle and likely to break)
- medicines that can affect your kidneys.
You may need to take different amounts of these medicines or you may need to take different medicines. Your doctor and pharmacist have more information.
Your doctor OR Pharmacist can tell you what to do if you are taking any of these medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
4. How do I use BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
The usual dose is three 500 mg tablets taken together as a single dose. Swallow the tablets whole with a full glass of water.
The tablets may be taken with or without food. It is not necessary to chew or crush the tablet.
Follow all directions given to you by your doctor or pharmacist carefully.
These directions may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask the doctor or pharmacist for help.
When to take BLOOMS THE CHEMIST FAMCICLOVIR ONCE
Take BLOOMS THE CHEMIST FAMCICLOVIR ONCE tablets as soon as possible after the first symptoms of a cold sore appear.
Symptoms include tingling, itching or burning or the appearance of redness and swelling.
Do not take the tablets if a hard crust has already formed on the cold sore.
Keep the tablets for the next episode.
How long to take it for
A single dose of BLOOMS THE CHEMIST FAMCICLOVIR ONCE is all that is necessary for treating each episode of cold sores. The dose may be repeated if cold sores recur. Each pack of BLOOMS THE CHEMIST FAMCICLOVIR ONCE for cold sores contains enough medicine for one dose.
If you use too much (Overdose)
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
You may need medical attention.
5. What should I know while using BLOOMS THE CHEMIST FAMCICLOVIR ONCE?
Things you should do
If you become pregnant, make sure you tell your doctor or pharmacist before taking any further doses of BLOOMS THE CHEMIST FAMCICLOVIR ONCE.
They can discuss with you the risks of taking it while you are pregnant.
Remind your doctor and pharmacist that you are taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE if you are about to be started on any new medicine.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE.
Things you should not do
Do not take less than the recommended dose of 3 tablets, unless advised by your pharmacist.
If you stop your tablets suddenly, your condition may worsen or you may have unwanted side effects.
Do not take BLOOMS THE CHEMIST FAMCICLOVIR ONCE to treat any other conditions unless your doctor or pharmacist tells you to.
Do not give BLOOMS THE CHEMIST FAMCICLOVIR ONCE to anyone else, even if they have the same condition as you.
Driving or using machines
Be careful driving or operating machinery until you know how BLOOMS THE CHEMIST FAMCICLOVIR ONCE affects you.
This medicine can cause dizziness, sleepiness or confusion in some people.
Things that may help your condition
Cold sores are contagious and the virus can be passed on from person to person through close physical contact or saliva, even when blisters are not present. The risk is much higher when the cold sore is visible, as the virus can be shed, making it easy to infect other people.
Take the following precautions to avoid spreading the virus:
- keep the areas affected by the virus as clean and dry as possible
- avoid touching or scratching the sore area as you may spread the virus on your fingers
- do not share any objects that have been in contact with a cold sore (e.g. drinking glasses, eating utensils, or towels)
- avoid direct skin-to-skin contact of the area with other people (e.g. kissing) until the cold sore has healed.
Looking after your medicine
Keep your tablets in the blister pack, until it is time to take them.
If you take the medicine out of the pack it may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store BLOOMS THE CHEMIST FAMCICLOVIR ONCE or any other medicines in a bathroom or near a sink.
Do not leave it in the car or on windowsills.
Heat and dampness can destroy some medicines.
Keep BLOOMS THE CHEMIST FAMCICLOVIR ONCE where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
When to discard your medicine
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
6. Are there any side effects?
Tell the doctor or pharmacist as soon as possible if you or your child do not feel well while you are taking BLOOMS THE CHEMIST FAMCICLOVIR ONCE.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects.
You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Less serious side effects
| Less Serious Side effects | What to do |
| Speak to your doctor or Pharmacist if you have any of these less side effects and they worry you. |
These side effects are usually mild.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
These are serious side effects. You may need urgent medical attention or hospitalisation. These side effects are rare.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What BLOOMS THE CHEMIST FAMCICLOVIR ONCE 500 mg tablet contains
| Active ingredient (Main ingredient) | Famciclovir |
| Other ingredients (Inactive ingredients) | poloxamer stearic acid hypromellose titanium dioxide macrogol 8000 |
This medicine does not contain gluten, lactose, sucrose, tartrazine and other azo dyes.
What BLOOMS THE CHEMIST FAMCICLOVIR ONCE looks like
White, oval, biconvex, film-coated and engraved "FAM500" on one side and "APO" on the other side.
Available in blister packs of 3 tablets. AUST R 306770
Who distributes BLOOMS THE CHEMIST FAMCICLOVIR ONCE
Arrotex Pharmaceuticals Pty Ltd
15 -17 Chapel Street; Cremorne
VIC 3121
This leaflet was prepared in March 2025.
Brand Information
| Brand name | Blooms the Chemist Famciclovir Once |
| Active ingredient | Famciclovir |
| Schedule | S3 |
MIMS Revision Date: 01 July 2023
1 Name of Medicine
Famciclovir.
2 Qualitative and Quantitative Composition
Famciclovir is a synthetic guanine derivative. It is a white to pale yellow crystalline solid with a melting point of 103°C.
Each tablet contains 500 mg famciclovir. In addition, each tablet also contains poloxamer, stearic acid, hypromellose, titanium dioxide and macrogol 8000.
3 Pharmaceutical Form
Blooms the Chemist Famciclovir Once (Famciclovir) 500 mg tablets are white, oval, biconvex, film-coated and engraved "FAM500" on one side and "APO" on the other side.
Blooms the Chemist Famciclovir Once (Famciclovir) is intended for oral administration.
4 Clinical Particulars
4.1 Therapeutic Indications
Blooms the Chemist Famciclovir Once is indicated for treatment of recurrent herpes labialis (cold sores) in immunocompetent adults aged 18 years and over.
4.2 Dose and Method of Administration
The recommended dosage is 1500 mg taken as a single dose. Initiate therapy at the earliest sign or symptom of a cold sore (e.g. tingling, itching or burning). Treatment was initiated within 1 hour of symptom onset in the recurrent herpes labialis clinical study.
Blooms the Chemist Famciclovir Once can be taken without regard to meals (see Section 5.2 Pharmacokinetic Properties, Effect of food).
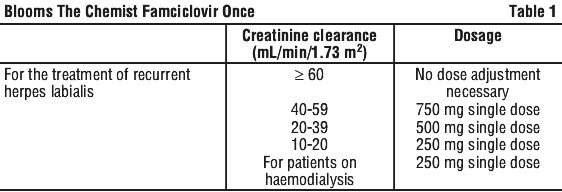
Renal impairment. As reduced clearance of penciclovir is related to reduced renal function, as measured by creatinine clearance, special attention should be given to dosages in patients with impaired renal function. The recommended modifications in dosage are tabulated in Table 1. As these recommendations are not based on repeated dose data, patients with impaired renal function should be closely monitored for adverse effects. There are insufficient data to recommend a dosage for patients with creatinine clearance less than 10 mL/min/1.73 m2.
For a patient on haemodialysis who has been prescribed famciclovir for conditions other than herpes labialis, a dosage interval of 48 hours is recommended for periods between dialysis. Since 4-hour haemodialysis results in approximately 75% reduction in plasma concentrations of penciclovir, the full adjusted dose (for patients with severe renal impairment) of famciclovir should be administered immediately following dialysis.
4.3 Contraindications
Famciclovir is contraindicated in patients with known hypersensitivity to famciclovir or to any of the ingredients in this medicine.
It is also contraindicated in those patients who have shown hypersensitivity to penciclovir, the active metabolite of famciclovir.
Patients who are immunocompromised.
Children and adolescents under 18 years of age.
4.4 Special Warnings and Precautions for Use
Identified precautions. Genital herpes is a sexually transmitted disease. The risk of transmission is increased during acute episodes. Patients should be advised to use condoms between episodes to reduce the risk of transmission and to avoid sexual intercourse when symptoms are present, even if treatment with an antiviral has been initiated. Genital herpes can also be transmitted in the absence of symptoms through asymptomatic viral shedding. Therefore, in addition to therapy with famciclovir, it is recommended that patients use "safer sex" practices.
Efficacy has not been studied in ophthalmic zoster, chicken pox or zoster encephalomyelitis patients.
Use in renal impairment. Special attention should be paid to patients with impaired renal function and dosage adjustment may be necessary. Appropriate dosage adjustments for renally-impaired patients are provided (see Section 4.2 Dose and Method of Administration).
Use in hepatic impairment. Famciclovir has not been studied in patients with severe hepatic impairment. Conversion of famciclovir to the active metabolite penciclovir may be impaired in these patients resulting in lower penciclovir plasma concentrations, and thus possibly a decrease of efficacy of famciclovir (see Section 5 Pharmacological Properties).
Use in the elderly. No special precautions are required for elderly patients with normal renal function and well-compensated hepatic impairment.
Paediatric use. Safety and efficacy in children and adolescents under the age of 18 years has not been established. Therefore, use of this medicine in this age group is not recommended.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Effects of other medicines on famciclovir. No clinically significant interactions have been identified with famciclovir or penciclovir (the active metabolite of famciclovir).
Probenecid. Concurrent use of probenecid may result in increased plasma concentrations of penciclovir (the active metabolite of famciclovir, see Section 5 Pharmacological Properties).
Other drugs that affect renal physiology could affect plasma levels of penciclovir (the active metabolite of famciclovir, see Section 5 Pharmacological Properties).
Evidence from preclinical studies has shown no potential for induction of cytochrome P450.
Zidovudine. In a phase I study, no significant drug interactions were observed after co-administration of zidovudine and famciclovir.
The conversion of the inactive metabolite 6-deoxy penciclovir to penciclovir is catalyzed by aldehyde oxidase. Interactions with other drugs metabolized by this enzyme and/or inhibiting this enzyme could potentially occur. Clinical interaction studies of famciclovir with cimetidine and promethazine, in vitro inhibitors of aldehyde oxidase, did not show relevant effects on the formation of penciclovir. However, raloxifene, the most potent aldehyde oxidase inhibitor observed in vitro, could affect the formation of penciclovir and thus the efficacy of famciclovir. When raloxifene is co-administered with famciclovir, the clinical efficacy should be monitored.
Effects of famciclovir on other medicines. Although famciclovir is only a weak inhibitor of aldehyde oxidase in vitro, interactions with drugs metabolized by aldehyde oxidase could potentially occur. Evidence from preclinical studies has shown no potential for induction of cytochrome P450 enzymes or inhibition of CYP3A4.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Testicular toxicity was observed in rats, mice and dogs following repeated administration of famciclovir or penciclovir. Testicular changes included atrophy of seminiferous tubules, reduction in sperm count and/or increased incidence of sperm with abnormal morphology or reduced motility. The degree of testicular toxicity was related to dose and duration of exposure and tended to reverse after the cessation of dosing. In male rats, decreased fertility was observed after 10 weeks dosing at 500 mg/kg/day, or approximately 3 - 20 times the human systemic exposure (AUC). Testicular toxicity was also seen in mice and dogs following chronic administration at exposures to penciclovir ranging from 2 - 14 times the human systemic exposure (AUC). However, there were no clinically significant effects on sperm count, morphology and motility in male patients receiving 250 mg famciclovir b.i.d. for 18 weeks. Famciclovir had no effect on fertility in female rats at doses of up to 1000 mg/kg/day, approximately 4 - 27 times the human systemic exposure (AUC).
Use in pregnancy. (Category B1)
Famciclovir was tested for effects on embryo-foetal development in rats and rabbits at oral doses up to 1000 mg/kg/day (approximately 4 - 27 times and 2 - 12 times the human systemic exposure to penciclovir in rats and rabbits, respectively [AUC]), and intravenous doses of 360 mg/kg/day in rats (1.9 - 12 times the human dose based on body surface area [BSA] comparisons) or 120 mg/kg/day in rabbits (1.2 - 7.1 times the human dose [BSA]). No adverse effects were observed on embryo-foetal development. Similarly, no adverse effects were observed following intravenous administration of penciclovir to rats (80 mg/kg/day, 0.4 - 2.6 times the human dose [BSA]) or rabbits (60 mg/kg/day, 0.6 - 3.6 times the human dose [BSA]). Although animal studies have not shown any embryotoxic or teratogenic effects with famciclovir or penciclovir (the active metabolite of famciclovir), the safety of famciclovir in human pregnancy has not been established. Famciclovir should therefore not be used during pregnancy unless the potential benefits are considered to outweigh the potential risks associated with treatment.
Category B1 - Drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed. Studies in animals have not shown evidence of an increased occurrence of foetal damage.
Use in lactation. Famciclovir should not be used by nursing mothers unless the potential benefits are considered to outweigh the potential risks associated with treatment. Following oral administration of famciclovir to lactating rats, penciclovir was excreted in milk at concentrations higher than those seen in plasma. There is no information on excretion in human milk.
4.7 Effects on Ability to Drive and Use Machines
Patients who experience dizziness, somnolence, confusion or other central nervous system disturbances while taking famciclovir should refrain from driving or operating machinery.
4.8 Adverse Effects (Undesirable Effects)
Famciclovir has been well tolerated in human studies. Headache, fatigue and nausea have been reported in clinical trials. These were generally mild or moderate and occurred at a similar incidence in patients receiving placebo treatment. Confusion, predominantly in the elderly, has been reported rarely. See Tables 2-4.
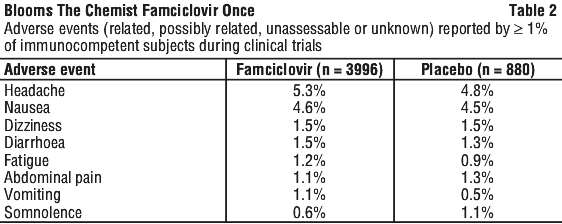
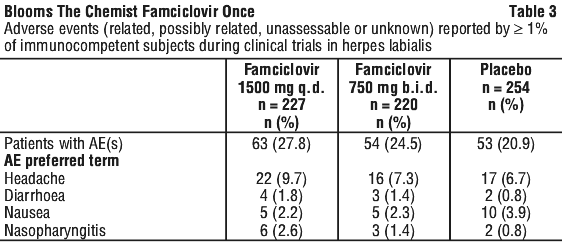
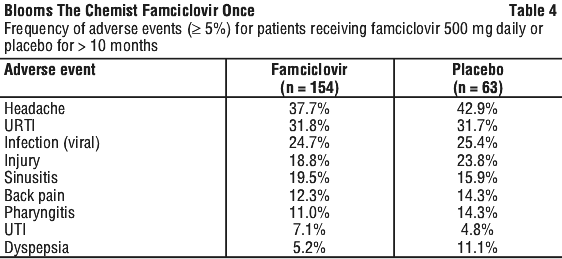
Post-marketing data. In addition to the adverse events reported in the clinical trials, the following adverse reactions have been reported rarely in post-marketing surveillance.
Adverse reactions are ranked under headings of frequency, using the following convention: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 10,000, < 1/1,000); very rare (< 1/10,000), including isolated reports.
Blood and lymphatic system disorders. Very rare: Thrombocytopenia.
Psychiatric disorders. Uncommon: Confusion (predominantly in the elderly).
Rare: Hallucinations.
Nervous system disorders. Very common: Headache.
Common: Dizziness.
Uncommon: Somnolence (predominantly in the elderly).
Not known: Seizure.
Cardiac disorders. Rare: Palpitations.
Gastrointestinal disorders. Common: Nausea, vomiting, abdominal pain, diarrhoea.
Hepatobiliary disorders. Common: Abnormal liver function tests.
Rare: Cholestatic jaundice.
Immune system disorders. Not known: Anaphylactic shock, anaphylactic reaction.
Skin and subcutaneous tissue disorders. Common: Rash, pruritus.
Uncommon: Angioedema (e.g. face oedema, eyelid oedema, periorbital oedema, pharyngeal oedema), urticaria.
Very rare: Serious skin reactions (e.g. erythema multiforme, Stevens-Johnson syndrome and toxic epidermal necrolysis).
Not known: Hypersensitivity vasculitis.
Musculoskeletal disorders. Very rare: Arthralgia, myalgia.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems or contact Arrotex Medical Information enquiries/Adverse Drug Reaction Reporting on 1800 195 055.
4.9 Overdose
Symptomatic and supportive therapy should be given as appropriate. Acute renal failure has been reported rarely in patients with underlying renal disease. The famciclovir dosage in these patients had not been appropriately reduced for the level of renal function.
Penciclovir, the active metabolite of famciclovir, is dialysable; plasma concentrations are reduced by approximately 75% following 4-hour haemodialysis.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Virology. Famciclovir is the oral form of penciclovir. Famciclovir is rapidly converted in vivo into penciclovir, which has demonstrable in vitro activity against herpes simplex viruses (HSV types 1 and 2) and varicella zoster virus (VZV). The antiviral effect of orally administered famciclovir has been demonstrated in several animal models: this effect is due to in vivo conversion to penciclovir.
Penciclovir targets virus-infected cells where it is rapidly converted into penciclovir-triphosphate (mediated via virus-induced thymidine kinase). The triphosphate inhibits viral DNA polymerase by competition with deoxyguanosine triphosphate and is incorporated into the extending DNA chain, preventing significant chain elongation. Consequently, viral DNA synthesis and, therefore, viral replication are inhibited.
This triphosphate persists in infected cells in excess of 12 hours. The long intracellular half-life of penciclovir triphosphate ensures prolonged antiviral activity, as demonstrated in cell cultures with HSV-1 and HSV-2 and in animal studies.
Penciclovir is only readily phosphorylated in virus-infected cells. In uninfected cells treated with penciclovir, concentrations of penciclovir-triphosphate are only barely detectable. Accordingly, uninfected cells are unlikely to be affected by therapeutic concentrations of penciclovir.
The most common form of resistance encountered with aciclovir among HSV strains is a deficiency in the production of the thymidine kinase (TK) enzyme. Such TK deficient strains would be expected to be cross-resistant to both penciclovir and aciclovir. However, penciclovir has been shown to be active against a clinically isolated aciclovir-resistant herpes simplex type 1 strain with an altered DNA polymerase.
The results from penciclovir and famciclovir patient studies, including studies of up to four months treatment with famciclovir, showed that no resistance occurred as a result of treatment with either famciclovir or penciclovir. Penciclovir-resistant isolates were found at the start of treatment or in the placebo groups in 0.25% of the 1976 total isolates from HSV and VZV (5/1976) and in 0.19% of the 533 virus isolates from immunocompromised patients (1/533).
Clinical trials. Herpes labialis (cold sores). Placebo-controlled trial. In one large placebo-controlled trial, 701 immunocompetent adults with recurrent herpes labialis were treated with famciclovir 1500 mg once (n=227), famciclovir 750 mg b.i.d. (n=220) or placebo (n=254) for 1 day. As well, patients also had to be in good general health, aged at least 18 years, have normal renal and hepatic function, had prior pregnancy tests if they were females of reproductive age and have experienced 3 or more episodes of cold sores in the preceding 12 months. Patients were required to have a history of prodromal symptoms preceding at least 50% of the recurrent episodes and at least 50% of these episodes had to have progressed to the vesicular lesion stage. Women of childbearing potential had to agree to use reliable birth control measures during the study. Pregnant or breast-feeding women were excluded. Patients were excluded if they had received an investigational drug in the 4 weeks prior to the study, had been previously vaccinated against herpes or were using a topical immunosuppressive agent on or near the face or a systemic immunosuppressive agent within 1 month of screening. Patients were also excluded if they were immunosuppressed due to underlying disease or concomitant treatment, had a recent history of drug or alcohol abuse, were suffering from inflammatory skin diseases (e.g. eczema or dermatitis) that would interfere with the assessment of lesions or were allergic or hypersensitive to products containing aciclovir, penciclovir, famciclovir or other nucleoside analogues.
Patients were instructed to take the first dose of study medication within 1 hour of symptom onset. However, some patients commenced treatment after 1 hour of onset of symptoms. Both famciclovir regimens significantly reduced time to healing of primary vesicular herpes labialis lesions (the primary efficacy variable) in the modified ITT population compared with placebo. The median time to healing in famciclovir 1500 mg single-dose treated patients was 4.4 days compared to 4.0 days in famciclovir 750 mg bid and 6.2 days in placebo-treated patients. This translates to treatment effects of 1.8 (CI 95% 0.9, 2.7) and 2.2 (CI 95% 1.3, 3.1) days, respectively. A single 1500 mg dose of famciclovir reduced the time to resolution of pain and tenderness (median time 1.7 days versus 2.9 days) compared with placebo and was marginally more effective than famciclovir 750 mg b.i.d. (median time 2.1 days).
5.2 Pharmacokinetic Properties
Absorption. Famciclovir is the oral prodrug of penciclovir. Following oral administration, famciclovir is rapidly and extensively absorbed and converted to the antivirally active compound penciclovir. Bioavailability of penciclovir after oral famciclovir is 77%. Mean peak plasma concentrations (Cmax) of penciclovir occurred at a median time of 45 minutes following administration of single oral doses of famciclovir (as shown in Table 5). No data is available on the pharmacokinetics of 1500 mg famciclovir as a single dose. Plasma concentration-time curves of penciclovir are similar following single and repeat (b.i.d. and t.i.d.) dosing and there is no accumulation of penciclovir on repeated dosing.

Metabolism. No data available.
Excretion. Famciclovir is eliminated principally as penciclovir and its 6-deoxy precursor, which are excreted in urine. No unchanged famciclovir has been detected in urine. Tubular secretion and glomerular filtration contribute to renal elimination of the compound. The terminal plasma half-life of penciclovir after both single and repeat dosing with famciclovir is approximately 2.0 hours. There is no accumulation of penciclovir on repeated dosing with famciclovir.
Effect of food. Penciclovir Cmax was decreased by approximately 50% and Tmax was delayed by 1.5 h when a capsule formulation of famciclovir was administered 30 minutes after food. When famciclovir tablets were administered 30 minutes after food, penciclovir Cmax was reduced by approximately 20% and Tmax was delayed by 0.75 h. The systemic availability (AUC) of penciclovir following either preparation was unaffected. The clinical consequences of these effects on plasma concentration are unknown.
Characteristics in special populations. Patients with herpes zoster. Uncomplicated Herpes virus does not significantly alter the pharmacokinetics of penciclovir measured after the oral administration of famciclovir.
Renal impairment. Plasma clearance, renal clearance and plasma elimination rate constant decreased linearly with reductions in renal function. A dosage interval adjustment is recommended for patients with renal insufficiency (see Section 4.2 Dose and Method of Administration).
Hepatic impairment. Well-compensated chronic liver disease (chronic hepatitis [n=6], chronic ethanol abuse [n=8] or biliary cirrhosis [n=1]) has no effect on the extent of availability (AUC) of penciclovir following a single dose of 500 mg famciclovir. No dose adjustment is recommended for patients with well-compensated hepatic impairment (see Section 4.2 Dose and Method of Administration, Hepatic impairment; Section 4.4 Special Warnings and Precautions for Use). However, there was a 43% decrease in penciclovir mean maximum plasma concentration and the time to maximum plasma concentration was increased by a median of 0.75 h in patients with hepatic insufficiency compared to normal volunteers. Pharmacokinetics has not been evaluated in patients with severe uncompensated hepatic impairment.
Elderly patients. Based on cross-study comparisons of single dose studies, the mean penciclovir AUC was approximately 30% larger, half-life 23% longer and penciclovir body weight adjusted renal clearance reduced by 19% in healthy elderly male volunteers (n=18, aged 65 to 79 years) compared to younger volunteers. Some of this difference may be due to differences in renal function between the two groups. No dose adjustment based on age is recommended unless renal function is impaired (see Section 4.2 Dose and Method of Administration).
HIV patients. Extrapolated data from a study (n=8) where famciclovir was given as a single dose resulted in a mean AUC of 24 microgram.h/mL, which is similar to that obtained in healthy subjects. However, immunocompromised patients are contraindicated.
Race. A retrospective evaluation was performed to compare the pharmacokinetic parameters obtained in Black and Caucasian subjects after single and repeat once-daily, twice-daily or three times-daily administration of famciclovir 500 mg. Data from a study in healthy volunteers (single dose), a study in subjects with varying degrees of renal impairment (single and repeat dose) and a study in subjects with hepatic impairment (single dose) did not indicate any relevant differences in the pharmacokinetics of penciclovir between Black and Caucasian subjects.
Transplant patients. Immunocompromised patients are a contraindication to using this medicine. In a study of allogenic bone marrow transplant or peripheral blood stem cell transplant or allogenic renal transplant patients (n=21), intravenous penciclovir for one month was followed by oral use of famciclovir. The doses of penciclovir and famciclovir were adjusted according to creatinine clearance. During repeat dosing with famciclovir, the AUC of penciclovir was found to be 66 microgram.h/mL in subjects with creatinine clearance greater than 50 mL/min. No safety concerns were identified despite the higher than normal AUCs reported - additional dosage adjustment in renal transplant patients is not recommended, because immunocompromised patients are contraindicated.
5.3 Preclinical Safety Data
Genotoxicity. Famciclovir and penciclovir (the active metabolite of famciclovir) were tested for genotoxic potential in a series of in vitro and in vivo assays. Famciclovir showed no genotoxic potential in a series of assays for gene mutations, chromosomal damage and DNA damage. Penciclovir was positive in the L5178Y mouse lymphoma assay for gene mutations/chromosomal damage, caused chromosomal aberrations in human lymphocytes in vitro and was positive in a mouse micronucleus assay in vivo when administered intravenously at doses toxic to bone marrow.
Carcinogenicity. Data presented below include reference to area under the plasma concentration curve (24-hour AUC) for penciclovir in humans following the lowest and highest recommended doses for famciclovir (i.e. penciclovir AUC of 4.5 microgram.h/mL at 125 mg b.i.d. for acute recurrent genital herpes, and a penciclovir AUC of 27 microgram.h/mL at 500 mg t.i.d. for herpes infections in immunocompromised patients). This is based on the assumption that the pharmacokinetics in immunocompetent subjects is similar to the pharmacokinetics in immunocompromised subjects, as shown in the study on HIV patients (see Section 5.2 Pharmacokinetic Properties). If the higher values of AUC obtained in the renal transplant patients were used as a basis for comparison, the multiples specified here would be decreased. Exposures in animal studies are expressed as multiples of human exposures at the highest and lowest dosing schedules based on penciclovir AUC or body surface area.
The carcinogenic potential of famciclovir was evaluated in 2-year dietary studies in rats and mice. A significant increase in the incidence of mammary adenocarcinoma was seen in female rats receiving 600 mg/kg/day. No increases in tumour incidences were reported for male rats treated at doses of up to 240 mg/kg/day or in mice of either sex at doses of up to 600 mg/kg/day. At the no effect levels of 240 and 200 mg/kg/day in male and female rats, the daily exposures to penciclovir based on AUC were about 40 and 29 microgram.h/mL respectively, or approximately 1 - 8 times the human systemic exposures at 500 mg t.i.d or 125 mg b.i.d. Systemic exposures at the no effect dose in male and female mice were 65 and 46 microgram.h/mL respectively, or approximately 2 - 12 times the human systemic exposure (AUC).
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
They are available in blister packs containing 3 tablets. AUST R 306770.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
Chemical Name: 9-(4-acetoxy-3-acetoxymethylbut-1-yl)-2-aminopurine.
Structural Formula:
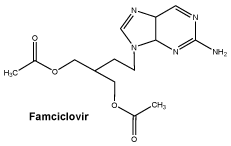
Molecular Weight: 321.332.
CAS number. CAS Registry Number: 104227-87-4.
7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist Only Medicine.
Date of First Approval
05 July 2018
Date of Revision
26 May 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.