Canesten Clotrimazole Thrush 3 Day Cream
Brand Information
| Brand name | Canesten Clotrimazole Thrush 3 Day Cream |
| Active ingredient | Clotrimazole |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Canesten Clotrimazole Thrush 3 Day Cream.
Full CMI
Canesten® Clotrimazole Thrush 1 Day Cream
Active ingredient: clotrimazole
Consumer Medicine Information (CMI)
This leaflet provides important information about using Canesten® Clotrimazole Thrush 1Day Cream. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Canesten® Clotrimazole Thrush 1 Day Cream.
Where to find information in this leaflet:
1. Why am I using Canesten® Clotrimazole Thrush 1 Day Cream?
2. What should I know before I use Canesten® Clotrimazole Thrush 1 Day Cream?
3. What if I am taking other medicines?
4. How do I use Canesten® Clotrimazole Thrush 1 Day Cream?
5. What should I know while using Canesten® Clotrimazole Thrush 1 Day Cream?
6. Are there any side effects?
7. Product details
1. Why am I using CANESTEN® CLOTRIMAZOLE THRUSH 1 DAY CREAM?
Canesten® Clotrimazole Thrush 1 Day Cream contains the active ingredient clotrimazole which belongs to a group of medicines called azole antifungals. Canesten® Clotrimazole Thrush 1 Day Cream is used to treat a fungal infection in the vagina and vulval area (thrush).
2. What should I know before I use CANESTEN® CLOTRIMAZOLE THRUSH 1 DAY CREAM?
Warnings
Do not use Canesten® Clotrimazole Thrush 1 Day Cream if:
- you are allergic to clotrimazole, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include swelling of the parts of the body to which treatment has been applied; rash, itching or hives on the skin.
Check with your doctor or pharmacist if:
- you are under the age of 18 years or over the age of 60 years
- you are unsure whether you have thrush or this is the first time you have had thrush
- you have had 3 or more infections in the last 6 months
- your symptoms include lower abdominal pain or back pain, fever or a foul smelling, unusual coloured or blood stained discharge
- you have diabetes
- you have allergies to any other topical treatments
- you take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Your doctor may also advise you to use a pessary during pregnancy, instead of a cream, as this can be inserted without an applicator.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor or pharmacist if you are taking tacrolimus, or other medicines used to prevent organ transplant rejection.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Canesten® Clotrimazole Thrush 1 Day Cream.
4. How do I use CANESTEN® CLOTRIMAZOLE THRUSH 1 DAY CREAM?
How much Canesten® Clotrimazole Thrush 1 Day Cream to use
This treatment is a full dose in one application. The 5g tube contains enough cream for the one dose.
If you are not sure what to do, ask your doctor or pharmacist.
When to use Canesten® Clotrimazole Thrush 1 Day Cream
The cream should preferably be inserted in the evening, just before bed time, with the use of an applicator. Using it before you go to sleep will have the best effect.
How to use Canesten® Clotrimazole Thrush 1 Day Cream
Wash your hands thoroughly before handling the cream or using the applicator. To avoid the possibility of re-infection, strict personal hygiene must be observed.
Loading an applicator
- Wash your hands thoroughly.
- Pull out the plunger of the disposable applicator until it stops (B).
- Open the Canesten® cream tube.
Attach the applicator (A) to the tube and hold it firmly pressed against the tube.
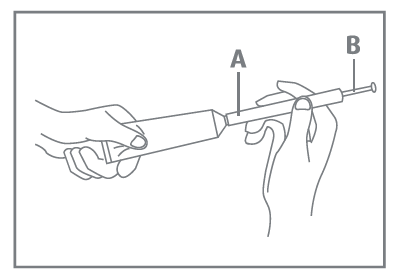
- Squeeze the cream tube until the applicator is full.
- Remove the tube of cream and replace the cap.
Inserting the applicator
- Carefully insert the end of the applicator containing the cream as gently and deeply into the vagina as possible. This is best achieved when lying on your back with your knees bent.
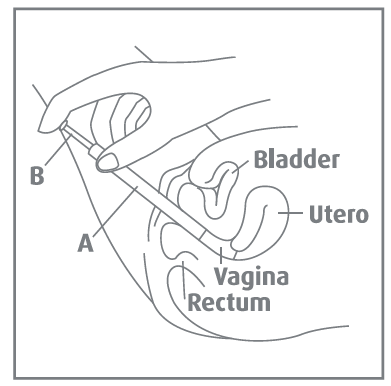
- Hold the applicator in position and slowly push the plunger with the forefinger until it stops. This places the cream in the vagina.
- Remove the applicator from the vagina and dispose of the applicator.
How long to use Canesten® Clotrimazole Thrush 1 Day Cream
Use the cream once.
If you use too much Canesten® Thrush 1 Day Cream
If you think that you have used too much Canesten® Clotrimazole Thrush 1 Day Cream you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling: Australia 13 11 26 or New Zealand 0800 764 766 [0800 POISON]) for advice, or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using CANESTEN® CLOTRIMAZOLE THRUSH 1 DAY CREAM?
Things you should do
- Discontinue treatment and tell your doctor or pharmacist if you develop a local reaction to this product.
- See your doctor if the symptoms do not improve within 4 days or if the problem returns within 2 months.
Remind any doctor, dentist or pharmacist you visit that you are using Canesten® Clotrimazole Thrush 1 Day Cream
Things you should not do
- Do not use Canesten® Clotrimazole Thrush 1 Day Cream to treat any other complaints unless your doctor tells you to.
- Do not give your treatment to anyone else, even if you suspect they have the same condition as you.
- Do not use tampons, intravaginal douches, spermicides or other vaginal products while using this product.
Things to be careful of
- Preferably the treatment should be timed to avoid the menstrual period.
This will allow the treatment to be most effective. Ask your doctor if you have questions about this. - Canesten® Clotrimazole Thrush 1 Day Cream may reduce the effectiveness and safety of latex products such as condoms and diaphragms if it comes into contact with them.
Therefore you should prevent Canesten® Clotrimazole Thrush 1 Day Cream from coming into contact with these products. If Canesten® Clotrimazole Thrush 1 Day Cream does accidentally come into contact with a condom or diaphragm, replace the condom or diaphragm before using it as contraception. - Avoid vaginal intercourse while you have thrush and during use of this product because your partner could become infected.
Things that may help to resolve Thrush
Some self help measures suggested below may help your condition.
Your doctor or pharmacist can give you more information about these measures.
- Blot with toilet paper after urinating (instead of wiping back to front).
- Change underwear, night attire, towels and linen daily.
Avoid where possible:
- Perfumed toilet paper
- Fragrant soaps and bath oils
- Wearing tight or synthetic clothing
- Douches or vaginal deodorants
- Soaking in baths
Looking after your medicine
Keep your cream in the tube until it is time to use it.
If you take the cream out of the tube it may not keep well. Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Store Canesten® Clotrimazole Thrush 1 Day Cream below 25°C.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
Local reaction to the cream:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergy or reaction related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Canesten® Clotrimazole Thrush 1 Day Cream contains
| Active ingredient (main ingredient) | clotrimazole |
| Other ingredients (inactive ingredients) | Sorbitan monosterate Polysorbate 60, Cetyl palmitate, Cetostearyl alcohol, Isopropyl myristate, Benzyl alcohol, Purified water |
| Potential allergens | Benzyl alcohol |
Do not use this medicine if you are allergic to any of these ingredients.
What Canesten® Clotrimazole Thrush 1 Day Cream looks like
Canesten® Clotrimazole Thrush 1 Day cream is a white opaque cream. The package also contains 1 disposable plastic applicator. AUST R 68570
Who distributes Canesten® Clotrimazole Thrush 1 Day Cream
Bayer Australia Ltd
Sydney NSW
Toll Free: 1800 008 757
www.canesten.com.au
Bayer New Zealand Limited
Auckland New Zealand
Freecall: 0800 229 376
www.canesten.co.nz
® = Registered Trademark
This leaflet was prepared November 2023
Brand Information
| Brand name | Canesten Clotrimazole Thrush 3 Day Cream |
| Active ingredient | Clotrimazole |
| Schedule | S3 |
MIMS Revision Date: 01 April 2023
1 Name of Medicine
Clotrimazole.
2 Qualitative and Quantitative Composition
Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Contains 10 mg/g (1%) of clotrimazole.
Canesten Clotrimazole Thrush 3 Day Cream. Contains 20 mg/g (2%) of clotrimazole.
Canesten Clotrimazole Thrush 1 Day Cream. Contains 100 mg/g (10%) of clotrimazole.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Contains 100 mg of clotrimazole.
Canesten Clotrimazole Thrush 1 Day Pessary. Contains 500 mg of clotrimazole.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Includes:
One pessary containing 500 mg of clotrimazole;
One tube of antifungal cream containing 10 mg/g (1%) of clotrimazole.
Excipients with known effect. Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Benzyl alcohol 2% w/w.
Canesten Clotrimazole Thrush 3 Day Cream. Benzyl alcohol 2% w/w.
Canesten Clotrimazole Thrush 1 Day Cream. Benzyl alcohol 1% w/w.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Lactose monohydrate.
Canesten Clotrimazole Thrush 1 Day Pessary. Lactose monohydrate.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Lactose monohydrate, benzyl alcohol 2% w/w.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Vaginal creams. White cream for vaginal use.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Oblong white vaginal tablet with markings "AD" on one side and "Bayer" on the other.
Canesten Clotrimazole Thrush 1 Day Pessary. Oblong white vaginal tablet with markings "MU" on one side and "Bayer" on the other.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Oblong white vaginal tablet with markings "MU" on one side and "Bayer" on the other, plus white cream for vaginal use.
4 Clinical Particulars
4.1 Therapeutic Indications
Vaginal creams. Canesten clotrimazole vaginal creams are indicated for the topical treatment of vulvovaginal candidiasis.
Vaginal tablets (compressed pessaries). Canesten clotrimazole vaginal pessaries are indicated for the topical treatment of vaginal candidiasis.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten clotrimazole composite pack is indicated for the treatment of candidal infections of the vagina and vulvovagina and sexual partner.
4.2 Dose and Method of Administration
No dosage adjustment is necessary in patients with renal or hepatic impairment due to limited systemic absorption following topical or vaginal application.
Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Once daily, preferably at bedtime for six successive days, one applicator should be filled with cream (approximately 5 g) and inserted as deeply as possible into the vagina with the patient lying on her back with the legs slightly drawn up. The 35 g tube of cream for vaginal use provides for six such doses.
Canesten Clotrimazole Thrush 3 Day Cream. Once daily, preferably at bedtime for three successive days, one applicator should be filled with cream (approximately 5 g) and inserted as deeply as possible into the vagina with the patient lying on her back with the legs slightly drawn up. The 20 g tube of cream for vaginal use provides for three such doses.
Canesten Clotrimazole Thrush 1 Day Cream. The disposable applicator should be filled with the vaginal cream, ensuring the entire contents of the tube are used (approximately 5 g). The cream is then inserted as gently and deeply as possible into the vagina with the patient lying on her back with the legs slightly drawn up. The cream should be used preferably at bedtime as a single dose of treatment.
Canesten clotrimazole vaginal creams may also be used in conjunction with Canesten clotrimazole vaginal pessaries in the management of Candida vulvovaginitis or infection of the peri-anal area while application of the cream to the glans penis of the partner may help prevent re-infection of the female.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary, Canesten Clotrimazole Thrush 1 Day Pessary. A course of treatment normally consists of either a single 500 mg pessary (1 Day Pessary) or of six 100 mg pessaries (6 Day Pessary). The latter may be given either as two pessaries, inserted one after the other, daily for three days or as one pessary daily for six days. Clinical investigations have shown comparable efficacy from either dosage scheme. Where a first course proved unsuccessful, a second course produced success in 8 of 12 women treated.
Canesten clotrimazole vaginal tablets (pessaries) should be inserted as deeply as possible into the vagina once daily, preferably at bedtime. This is best achieved using the plastic applicator provided and following the directions on the patient instruction sheet. In late pregnancy digital insertion is preferable to use instead of the applicator.
The vaginal tablets need moisture in the vagina to dissolve completely, otherwise undissolved pieces of the vaginal tablet might crumble out of the vagina. To prevent this it is important to insert the pessaries as deeply as possible into the vagina preferably at bedtime. Should the vaginal tablet not dissolve completely within one night the use of a vaginal cream should be considered.
It is suggested that treatment be timed so as to avoid the menstrual period and to be complete before the onset of menstruation.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. The pessary should be inserted gently and as deeply as possible into the vagina, preferably at bedtime. This is best achieved using the plastic applicator provided and following the directions on the instruction leaflet. In late pregnancy digital insertion is preferable to use instead of the applicator. The 500 mg pessary provides therapy for three to four days as it creates high intravaginal concentrations of clotrimazole, however the symptoms may persist for these three to four days.
The antifungal cream may be used in conjunction with the pessary in the management of candidal vulvovaginitis or infection of the peri-anal area while application of the cream to the glans penis of the partner may help prevent reinfection of the female. This should be applied thinly to the affected areas (external genital organs to the anus in the woman; glans and prepuce in men) 2 - 3 times daily and rubbed in.
It is suggested that the treatment be timed so as to avoid the menstrual period and to be complete before the onset of menstruation. If the treatment does coincide with a menstrual period, use sanitary pads not tampons during therapy.
4.3 Contraindications
Known hypersensitivity to clotrimazole, cetostearyl alcohol (only for products containing cetostearyl alcohol), and/or to any other excipients (see Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
If evidence of local intolerance develops, consider withdrawal of the drug and institution of appropriate therapy.
Canesten clotrimazole vaginal creams and vaginal pessaries are for intravaginal use only and are not to be taken orally. Avoid contact with eyes.
A doctor should be consulted before use in the following instances:
if it is the first occurrence of the problem;
if there have been three or more thrush infections in the previous six months;
if pregnant;
if diabetic;
if under 18 years of age or over the age of 60 years;
if not better in four days.
If the patient has a fever (temperature of 38°C or above), lower abdominal pain, back pain, foul smelling vaginal discharge, nausea, vaginal haemorrhage, and/or associated shoulder pain the patient should consult a doctor.
Only for products containing cetostearyl alcohol (see Section 6.1 List of Excipients): cetostearyl alcohol may cause local skin reactions (e.g. contact dermatitis).
Canesten clotrimazole creams are not intended for ophthalmic use.
If symptoms persist for more than 4 days the patient may have a medical condition that requires treatment by a doctor.
The treatment can be repeated if necessary, however, recurrent infections may indicate an underlying medical cause, including diabetes or HIV infection. Patients should seek medical advice if symptoms return within 2 months.
Since the vagina and vulva are usually both affected, a combination treatment (treatment of both of these areas) should be performed.
lf the labia and adjacent areas are simultaneously infected, local treatment with an external cream should also be given. The sexual partner should also undergo local treatment if symptoms, e.g. pruritus, inflammation, etc. are present.
Treatment during the menstrual period should not be performed. The treatment should be finished before the onset of menstruation.
Do not use tampons, intravaginal douches, spermicides or other vaginal products while using these products.
Avoidance of vaginal intercourse is recommended while using this product because the infection could be transferred to the partner, and the effectiveness and safety of latex products such as condoms and diaphragms may be reduced (see Interactions with latex).
Interactions with latex. Canesten clotrimazole vaginal creams and vaginal pessaries may reduce the effectiveness and safety of latex products, such as condoms and diaphragms. This effect is temporary and occurs only during treatment.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant treatment with vaginal clotrimazole and oral tacrolimus (FK-506, an immunosuppressant) might lead to increased tacrolimus plasma levels. Patients should thus be thoroughly monitored for symptoms of tacrolimus overdosage, if necessary by determination of the respective plasma levels.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No human studies of the effects of clotrimazole on fertility have been performed. However, animal studies have not demonstrated any effects of the medicine on fertility.
Use in pregnancy. (Category A)
There is a limited amount of clinical data in pregnant women. Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity (see Section 5.3 Preclinical Safety Data).
Clotrimazole can be used during pregnancy, but only under the direction of a health care professional.
If treatment is carried out during pregnancy, Canesten clotrimazole vaginal pessaries are the preferable choice as they can be inserted without the use of an applicator. Sanitation of the birth canal should be ensured particularly during the last 4-6 weeks of pregnancy.
Use in lactation. Although systemic absorption following topical or vaginal administration is low, caution should be exercised when clotrimazole is administered to nursing mothers as there is no information on whether or not clotrimazole is excreted in breast milk.
4.7 Effects on Ability to Drive and Use Machines
No effects on ability to drive and use machines have been observed.
4.8 Adverse Effects (Undesirable Effects)
The listed adverse drug reactions are based on spontaneous reports, thus, an organisation according to CIOMS III categories of frequency is not possible.
Immune system disorders. Anaphylactic reaction, angioedema, hypersensitivity.
Vascular disorder. Syncope, hypotension.
Respiratory, thoracic and mediastinal disorders. Dyspnoea.
Skin and subcutaneous tissue disorders. Rash, urticaria.
Reproductive system and breast disorders. Vaginal exfoliation, vaginal discharge, vaginal haemorrhage, vulvovaginal erythema, vulvovaginal pruritus, vulvovaginal discomfort, vulvovaginal burning sensation, vulvovaginal pain.
Gastrointestinal disorders. Abdominal pain, nausea.
General disorders and administration site conditions. Application site irritation, oedema, pain.
Eleven (1.6%) of 689 patients treated with Canesten clotrimazole vaginal pessaries complained of possibly drug-related effects. Mild burning occurred in four patients while other complaints such as skin rash and lower abdominal cramps were mentioned. Slight urinary frequency and burning or irritation in the sexual partner occurred rarely. In no case was it necessary to discontinue Canesten clotrimazole vaginal pessaries.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There have been no reports of overdosage with clotrimazole.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group (ATC code): G01A F02.
Clotrimazole is an imidazole derivative with a broad spectrum antimycotic activity.
Mechanism of action. Clotrimazole acts against fungi by inhibiting ergosterol synthesis. Inhibition of ergosterol synthesis leads to structural and functional impairment of the cytoplasmic membrane.
Clotrimazole has a broad antimycotic spectrum of action in vitro and in vivo, which includes dermatophytes, yeasts, moulds, etc.
Under appropriate test conditions, the minimum inhibitory concentration (MIC) values for these types of fungi are in the region of less than 0.062-8.0 microgram/mL substrate. The mode of action of clotrimazole is primarily fungistatic or fungicidal depending on the concentration of clotrimazole at the site of infection. In vitro activity is limited to proliferating fungal elements; fungal spores are only slightly sensitive.
In addition to its antimycotic action, clotrimazole also acts on Trichomonas vaginalis, gram-positive microorganisms (Streptococci/Staphylococci), and gram-negative microorganisms (Bacteroides/Gardnerella vaginalis).
In vitro clotrimazole inhibits the multiplication of Corynebacteria and gram-positive cocci - with the exception of Enterococci - in concentrations of 0.5-10 microgram/mL substrate and exerts a trichomonacidal action at 100 microgram/mL.
Primarily resistant variants of sensitive fungal species are very rare; the development of secondary resistance by sensitive fungi has so far only been observed in very isolated cases under therapeutic conditions.
Pharmacodynamic effects. A single course of intravaginal Canesten clotrimazole cream has produced mycological cure of vaginal candidiasis as follows:
one 100 mg pessary daily for six days - 80%;
two 100 mg pessary daily for three days - 75%;
one 500 mg pessary - 84%;
5 g of 1% cream daily for six days - approx 75%.
When a first course proved unsuccessful, a second course produced success in 8 of 12 women treated.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption and distribution. Studies using 14C-labelled clotrimazole have shown absorption of about 3% of the administered dose from normal or inflamed human vaginal mucosa (peak serum level of 0.03 microgram/mL) 24 hours after insertion of a 100 mg tablet.
Studies in normal volunteers after vaginal insertion of one 500 mg pessary showed that plasma levels of clotrimazole up to 10 nanogram/mL were reached during the period of assay (up to 72 hours after insertion). Significant concentrations of clotrimazole were present in the vaginal secretion for up to 48 hours after insertion.
Canesten clotrimazole cream containing 14C-labelled clotrimazole was administered intravaginally to five women (four without gynaecological disorder and one with Candida vaginitis). A maximum serum level of 0.01 microgram/mL was found between 10 and 30 hours after administration.
Six hours after application of labeled topical Canesten clotrimazole cream the concentrations of clotrimazole ranged from 100 microgram/cm3 in the stratum corneum to 0.05-1.0 microgram/cm3 in the stratum reticulare and 0.1 microgram/cm3 in the subcutis.
No measurable radioactivity was found in the serum within 48 hours after application of 0.8 g of the cream.
Pharmacokinetic investigations after dermal application have shown that only a small amount of clotrimazole (< 2% of the dose) is absorbed. The resulting peak plasma concentrations of the active ingredient are < 10 nanogram/mL (i.e. below the detection limit) and do not lead to measurable side effects.
Metabolism and excretion. Studies of urinary excretion have shown that less than 0.5% of dermally applied Canesten clotrimazole cream appears in the urine over a five-day period of observation. Faecal excretion, the route by which most of the absorbed drug is likely to be eliminated, has not been studied in man.
5.3 Preclinical Safety Data
Toxicological studies in rats, dogs and primates given clotrimazole cream or tablet intravaginally showed good vaginal tolerance, and skin applications of clotrimazole cream/lotion were well tolerated in rabbits. Although limited liver toxicity was observed in rats given clotrimazole orally for 18 months; overall, preclinical data reveal no special hazard for humans based on the totality of findings from single- and repeat-dose, genotoxicity, carcinogenicity and reproductive toxicity studies.
Genotoxicity. No mutagenicity has been observed in animal studies.
Carcinogenicity. No carcinogenicity has been observed in animal studies.
6 Pharmaceutical Particulars
6.1 List of Excipients
Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Sorbitan monostearate, polysorbate 60, cetyl palmitate, cetostearyl alcohol, octyldodecanol, benzyl alcohol and purified water.
Canesten Clotrimazole Thrush 3 Day Cream. Sorbitan monostearate, polysorbate 60, cetyl palmitate, cetostearyl alcohol, octyldodecanol, benzyl alcohol and purified water.
Canesten Clotrimazole Thrush 1 Day Cream. Sorbitan monostearate, polysorbate 60, cetyl palmitate, cetostearyl alcohol, isopropyl myristate, benzyl alcohol and purified water.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Lactose monohydrate, maize starch, magnesium stearate, colloidal anhydrous silica, calcium lactate pentahydrate, crospovidone, lactic acid, hypromellose and microcrystalline cellulose.
Canesten Clotrimazole Thrush 1 Day Pessary. Lactose monohydrate, maize starch, magnesium stearate, colloidal anhydrous silica, calcium lactate pentahydrate, crospovidone, lactic acid, hypromellose and microcrystalline cellulose.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Pessary: lactose monohydrate, maize starch, magnesium stearate, colloidal anhydrous silica, calcium lactate pentahydrate, crospovidone, lactic acid, hypromellose and microcrystalline cellulose.
Cream: sorbitan monostearate, polysorbate 60, cetyl palmitate, cetostearyl alcohol, octyldodecanol, benzyl alcohol and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Store below 25°C.
Canesten Clotrimazole Thrush 3 Day Cream. Store below 25°C.
Canesten Clotrimazole Thrush 1 Day Cream. Store below 25°C.
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Store below 30°C.
Canesten Clotrimazole Thrush 1 Day Pessary. Store below 25°C.
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Store below 25°C.
6.5 Nature and Contents of Container
Vaginal creams. Canesten Clotrimazole Thrush 6 Day Cream. Presented in a 35 g tube and packed with six single-use disposable applicators and an instruction leaflet (Consumer Medicine Information).
Canesten Clotrimazole Thrush 3 Day Cream. Presented in a 20 g tube and packed with three single-use disposable applicators and an instruction leaflet (Consumer Medicine Information).
Canesten Clotrimazole Thrush 1 Day Cream. Presented in a 5 g tube and packed with one single-use disposable applicator and an instruction leaflet (Consumer Medicine Information).
Vaginal tablets (compressed pessaries). Canesten Clotrimazole Thrush 6 Day Pessary. Presented in a blister pack of six compressed pessaries, and packed with a plastic applicator and an instruction leaflet (Consumer Medicine Information).
Canesten Clotrimazole Thrush 1 Day Pessary. Presented in a blister pack containing one compressed pessary, and packed with a plastic applicator and an instruction leaflet (Consumer Medicine Information).
Composite pack: '1 day' compressed pessary plus antifungal cream. Canesten Clotrimazole Thrush 1 Day Pessary + Cream. Presented in a carton that includes a blister pack of one Canesten compressed pessary and packed with a plastic applicator. Plus, it includes one 10 g tube of Canesten antifungal cream. An instruction leaflet (Consumer Medicine Information) is also included.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Clotrimazole is a colourless, crystalline, weakly alkaline substance, melting point 141°-145°C, soluble in acetone, chloroform and ethanol and practically insoluble in water. It forms stable salts with both organic and inorganic acids. It is not photosensitive but is slightly hygroscopic, and may be hydrolysed in acid media.
Chemical structure. Chemical name: 1-(o-chloro-α,α-diphenylbenzyl) imidazole.
Structural formula:
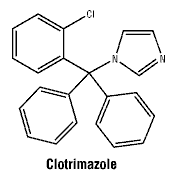
Molecular weight: 344.84.
CAS number. 23593-75-1.
7 Medicine Schedule (Poisons Standard)
Pharmacist Only Medicine (S3).
Date of Revision
08 September 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.