Ceclor
Brand Information
| Brand name | Ceclor |
| Active ingredient | Cefaclor |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ceclor.
Summary CMI
CECLOR®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about taking this medicine, speak to your doctor or pharmacist.
1. Why am I taking CECLOR?
CECLOR contains the active ingredient cefaclor. CECLOR is used to treat infections caused by bacteria in different parts of the body. For more information, see Section 1. Why am I taking CECLOR? in the full CMI.
2. What should I know before I take CECLOR?
Do not take if you have ever had an allergic reaction to CECLOR, other cephalosporins or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I take CECLOR? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with CECLOR and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take CECLOR?
- Your doctor will tell you how much CECLOR you need to take. This will depend on the type of infection you have.
- Shake the bottle well and accurately measure the dose with a medicine measure.
More instructions can be found in Section 4. How do I take CECLOR? in the full CMI.
5. What should I know while taking CECLOR?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking CECLOR? in the full CMI.
6. Are there any side effects?
Tell your doctor or pharmacist if you notice any of the following: diarrhoea; itchy rash; oral thrush; vaginal thrush. Tell your doctor as soon as possible if you notice any of the following: nausea; vomiting; drowsiness; headache; hyperactivity, insomnia, confusion, dizziness, hallucinations; severe muscle stiffness; swelling of the joints with or without fever. Tell your doctor immediately if you notice any of the following, particularly if they occur several weeks after stopping treatment with CECLOR: severe abdominal cramps or stomach cramps; watery and/or severe diarrhoea which may also be bloody; fever, in combination with one or both of the above. Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of the following: rash, itching or hives on the skin with swelling of the face, lips, tongue or other parts of the body; seizures; confusion, memory loss, personality changes, trouble thinking clearly or focusing; involuntary muscle movements such as jerks, tremors or eye movements. This not a complete list of all possible side effects. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
CECLOR®
Active ingredient(s): cefaclor (as monohydrate)
Consumer Medicine Information (CMI)
This leaflet provides important information about taking CECLOR. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about taking CECLOR.
Where to find information in this leaflet:
1. Why am I taking CECLOR?
2. What should I know before I take CECLOR?
3. What if I am taking other medicines?
4. How do I take CECLOR?
5. What should I know while taking CECLOR?
6. Are there any side effects?
7. Product details
1. Why am I taking CECLOR?
CECLOR contains the active ingredient cefaclor monohydrate. CECLOR belongs to a group of antibiotics called cephalosporins. These antibiotics works by killing the bacteria that are causing your infection.
CECLOR is used to treat infections caused by bacteria in different parts of the body. This includes infections of the:
- ears, nose, throat and tonsils (upper respiratory tract)
- chest and lungs (lower respiratory tract)
- bladder and kidneys (lower urinary tract)
- skin
Your doctor may have prescribed it for another reason.
This medicine is not addictive.
Ask your doctor if you have any questions about why CECLOR has been prescribed for you.
2. What should I know before I take CECLOR?
Warnings
Do not take CECLOR if:
- you are allergic to any medicine containing cefaclor monohydrate, other cephalosporins, or any of the ingredients listed at the end of this leaflet. Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Always check the ingredients to make sure you can use this medicine. - you have had a serious allergic reaction to penicillin.
Do not give this medicine to a child under the age of one month.
Safety and effectiveness in children younger than one month have not been established.
Do not take this medicine after the expiry date printed on the carton and bottle label or if the packaging is torn or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Check with your doctor if you:
- are allergic to any other medicines or any foods, preservatives or dyes.
- have or have had any of the following medical conditions:
- kidney disease
- severe bowel conditions
- liver disease
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Your doctor can discuss with you the risks and benefits involved.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
If you have not told your doctor about any of the above, tell them before you start taking CECLOR.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and CECLOR may interfere with each other. These include:
- antacids, a medicine used to neutralise stomach acid to relieve heartburn, indigestion or an upset stomach
- probenecid, a medicine used to treat gout and to promote the excretion of uric acid
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking CECLOR.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect CECLOR.
4. How do I take CECLOR?
Follow all directions given to you by your doctor or pharmacist carefully.
They may differ from the information contained in this leaflet.
If you do not understand the instructions on the carton or bottle, ask your doctor or pharmacist for help.
How much to take
- Your doctor will tell you how much CECLOR you need to take.
This will depend on the type of infection you have. - Follow the instructions provided when CECLOR was prescribed, including the number of days it should be taken.
How to take it
- Shake the bottle well and accurately measure the dose with a medicine measure.
Shaking the bottle and using a medicine measure will make sure that you get the correct dose. You can buy a medicine measure from your pharmacist. - It does not matter if you take this medicine with or without food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
It is important to complete the full course prescribed by your doctor, even if you begin to feel better after a few days. If you do not, the bacteria causing your infection may continue to grow and multiply so that your infection may not clear completely or your symptoms may return.
If you forget to take CECLOR
CECLOR should be used regularly at the same time each day.
If you miss your dose at the usual time and it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much CECLOR
If you think that you or anyone else have used too much CECLOR, urgent medical attention may be needed.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
Symptoms of an overdose may include feeling sick, vomiting, upset stomach or diarrhoea.
5. What should I know while taking CECLOR?
Things you should do
- Tell your doctor if the symptoms of your infection do not improve within a few days, or if they become worse.
- Tell any other doctors, dentists and pharmacists who treat you that you are taking CECLOR, especially if you are about to be started on any new medicines..
- If you are about to have any blood tests, tell your doctor that you are taking this medicine.
It may interfere with the results of some tests. - Keep all of your doctor's appointments so that your progress can be checked.
Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects. - If you are diabetic, check with your doctor or pharmacist before using urine sugar tests.
CECLOR may cause false test results with some urine sugar tests.
Call your doctor straight away if you:
- become pregnant while taking CECLOR.
Things you should not do
- Do not take CECLOR to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not stop taking your medicine or lower the dosage without checking with your doctor.
Driving or using machines
Be careful driving or operating machinery until you know how CECLOR affects you.
This medicine may cause dizziness or drowsiness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Children should be careful when riding bicycles or climbing trees.
Looking after your medicine
- Keep CECLOR suspension in your refrigerator at 2°C to 8°C where young children cannot reach it. Do not freeze.
- Keep the bottle tightly closed.
- The suspension should be shaken well before take and discarded after 14 days.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking CECLOR.
This medicine helps most people with infection, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. The list includes the more common side effects of CECLOR. They are usually mild and short-lived. |
Serious side effects
| Serious side effects | What to do |
Tell your doctor immediately if you notice any of the following, particularly if they occur several weeks after stopping treatment with CECLOR:
| Speak to your doctor as soon as possible if you have any of these serious side effects and they worry you. The list includes serious side effects which may require medical attention |
Do not take any diarrhoea medicine without first checking with your doctor.
You may have a serious condition affecting your bowel, requiring urgent medical attention.
More serious side effects
| More serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these more serious side effects. The list includes very serious side effects. You may need urgent medical attention or hospitalisation. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What CECLOR contains
| Active ingredient (main ingredient) | cefaclor 25 mg/mL or 50 mg/mL per bottle |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | sugars |
What CECLOR looks like
CECLOR 125 mg / 5 mL: A pink free-flowing dry powder. After constitution a red coloured suspension with a characteristic strawberry odour. (AUST R 347594)
Available in bottle of 100 mL.
CECLOR 250 mg / 5 mL: A pink free-flowing dry powder. After constitution a red coloured suspension with a characteristic strawberry odour. (AUST R 347593)
Available in bottle of 75 mL.
Sponsor
Viatris Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in July 2024.
CECLOR® is a Viatris company trade mark
CECLOR_cmi\Jul24/00
Brand Information
| Brand name | Ceclor |
| Active ingredient | Cefaclor |
| Schedule | S4 |
MIMS Revision Date: 01 September 2024
1 Name of Medicine
Cefaclor (as monohydrate).
2 Qualitative and Quantitative Composition
Each 5 mL of Ceclor powder for oral liquid upon reconstitution contains 125 mg or 250 mg of cefaclor (as monohydrate) as the active ingredient.
Excipients of known effect. Sugars.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Ceclor 125 mg/5 mL and 250 mg/5 mL powder for oral liquids are pink free-flowing dry powders. After constitution, a red coloured suspension with a characteristic strawberry odour is formed.
4 Clinical Particulars
4.1 Therapeutic Indications
Ceclor is indicated for the treatment of the following types of infections caused by or likely to be caused by susceptible organisms:
Lower respiratory infections, including pneumonia, bronchitis and exacerbations of chronic bronchitis.
Upper respiratory infections, including pharyngitis, tonsillitis and otitis media.
Skin and skin structure infections.
Urinary tract infections, including pyelonephritis and cystitis.
Note. 1. Penicillin is the usual drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever. Ceclor appears to be as effective as phenoxymethylpenicillin in the eradication of streptococci from the nasopharynx; however, substantial data establishing the efficacy of cefaclor in the subsequent prevention of rheumatic fever are not available at present.
2. Appropriate culture and susceptibility studies should be performed to determine susceptibility of the causative organism to cefaclor.
4.2 Dose and Method of Administration
Ceclor is administered orally.
Directions for reconstitution of Ceclor for oral suspension. 125 mg/5 mL. Add 60 mL of water in two portions to the dry mixture in the bottle. Shake well after each addition.
250 mg/5 mL. Add 45 mL of water in two portions to the dry mixture in the bottle. Shake well after each addition.
Adults. The usual adult dosage is 250 mg every 8 to 12 hours. For bronchitis and pneumonia, the dosage is 250 mg administered 3 times daily. For more severe infections or those caused by less susceptible organisms, doses may be doubled (500 mg 8 hourly).
Doses of 2 g/day should not be exceeded.
For skin and skin structure infections, the dosage is 250 mg 2-3 times a day.
Children. The usual recommended daily dosage for children with mild to moderate infections is 20 mg/kg/day in divided doses every 8 hours (maximum 1 g/day).
For streptococcal pharyngitis/ tonsillitis and impetigo, 12 hourly administration appears equally effective.
In more serious infections, otitis media, and infections caused by less susceptible organisms, the recommended dosage is 40 mg/kg/day in divided doses every 8 to 12 hours (maximum 2 g/day). For otitis media, 12 hourly administration appears equally effective.
Ceclor may be administered in the presence of impaired renal function. Under such a condition, the dosage usually is unchanged (see Section 4.4 Special Warnings and Precautions for Use).
In the treatment of beta-haemolytic streptococcal infections, a therapeutic dosage of Ceclor should be administered for at least 10 days.
4.3 Contraindications
Ceclor is contraindicated in patients with known allergy to the cephalosporin group of antibiotics or who have previously experienced a major allergy to penicillin (see Section 4.4 Special Warnings and Precautions for Use) or any of the excipients.
Ceclor is also contraindicated in infants under the age of one month as safety and efficacy of this product has not been established in prematures and infants under one month of age.
4.4 Special Warnings and Precautions for Use
As with antibiotic therapy in general, administration of Ceclor should be continued for a minimum of 48 to 72 hours after the patient becomes asymptomatic or after evidence of bacterial eradication has been obtained. A minimum of ten days of treatment is recommended in infections caused by group A beta-haemolytic streptococci in order to guard against the risk of rheumatic fever or glomerulonephritis.
Prolonged use of Ceclor may result in the overgrowth of non-susceptible organisms. Careful observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Allergic reaction (anaphylaxis). In penicillin-sensitive patients, cephalosporin antibiotics should be administered cautiously. There is clinical and laboratory evidence of partial cross-allergenicity of the penicillins and the cephalosporins and there are instances in which patients have had reactions, including anaphylaxis, to both drug classes. Serious and occasionally fatal hypersensitivity (anaphylactic/ anaphylactoid) reactions have been reported in patients on penicillin/ cephalosporin therapy. Although anaphylaxis is more frequent following parenteral therapy, it has occurred in patients on oral penicillins/ cephalosporins. These reactions are more likely to occur in individuals with a history of penicillin hypersensitivity and/or a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin/ cephalosporin hypersensitivity who have experienced severe reactions when treated with a penicillin/ cephalosporin.
Past history of a severe allergic reaction to penicillin/ cephalosporin is a contraindication to the use of Ceclor. Before initiating therapy with any cephalosporin, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, or other allergens. If an allergic reaction occurs, Ceclor should be discontinued and the appropriate therapy instituted. Serious anaphylactoid reactions require immediate emergency treatment with adrenaline (epinephrine). Oxygen, intravenous steroids and airway management, including intubation, should also be administered as indicated.
History of colitis or gastrointestinal disease. Broad-spectrum antibiotics should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Pseudomembranous colitis. Antibiotic associated pseudomembranous colitis has been reported with many antibiotics including cefaclor. A toxin produced with Clostridium difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with antibiotic use (this may occur up to several weeks after cessation of antibiotic therapy). Mild cases usually respond to drug discontinuation alone. However, in moderate to severe cases appropriate therapy with a suitable oral antibacterial agent effective against C. difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated. Drugs which delay peristalsis, e.g. opiates and diphenoxylate with atropine, may prolong and/or worsen the condition and should not be used.
History of bleeding disorders. All cephalosporins may cause hypoprothrombinemia and, potentially, bleeding.
Neurotoxicity. There have been reports of neurotoxicity associated with cephalosporin treatment. Symptoms of neurotoxicity include encephalopathy, seizures and/or myoclonus (see Section 4.8 Adverse Effects (Undesirable Effects)). Risk factors for developing neurotoxicity with cephalosporin treatment include being elderly, renal impairment, central nervous system disorders and intravenous administration. Withdrawal of the medicine should be considered if there are signs of neurotoxicity.
Severe cutaneous adverse reactions. Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in patients taking beta-lactam antibiotics. When SCAR is suspected, Ceclor should be discontinued immediately and an alternative treatment should be considered.
Use in hepatic impairment. Ceclor should be used with caution in patients with liver disease, as documented clinical experience in this group of patients is lacking.
Use in renal impairment. Ceclor should be administered with caution in the presence of markedly impaired renal function. Since the half-life of cefaclor in anuria is 2.3 to 2.8 hours, dosage adjustments for patients with moderate or severe renal impairment are usually not required. Clinical experience with cefaclor under such conditions is limited; therefore, careful clinical observation and laboratory studies should be made.
Use in the elderly. In elderly subjects (over age 65) with normal serum creatinine values, a higher peak plasma concentration and AUC are effects resulting from mildly diminished renal function and are not expected to have clinical significance. Therefore, dosage changes are not necessary in elderly subjects with normal renal function.
Paediatric use. Safety and effectiveness of this product for use in infants less than one month of age have not been established. Serum sickness-like reactions including arthritis and arthralgia have been reported more frequently in children than in adults.
Effects on laboratory tests. Administration of Ceclor may result in a false-positive reaction for glucose in the urine. This phenomenon has been seen in patients taking cephalosporin antibiotics when the test is performed using Benedict's and Fehling's solutions and also with Clinitest tablets but not with Tes-Tape (Glucose Enzymatic Test Strip, USP).
Positive direct Coombs tests have been reported during treatment with Ceclor. In haematologic studies or in transfusion cross-matching procedures when anti-globulin tests are performed on the minor side, or in Coombs' testing of newborns whose mothers have received cephalosporin antibiotics before parturition, it should be recognised that a positive Coombs' test may be due to the drug.
4.5 Interactions with Other Medicines and Other Forms of Interactions
As all cephalosporins can inhibit vitamin K synthesis by suppressing gut flora, prophylactic vitamin K therapy is recommended when any of these medications is used for prolonged periods in malnourished or seriously ill patients.
The extent of absorption of cefaclor is diminished if aluminium hydroxide-or magnesium-containing antacids are taken within 1 hour of administration.
Probenecid decreases renal tubular secretion of cephalosporins excreted by this mechanism, resulting in increased and prolonged cephalosporin serum concentrations, prolonged elimination half-life, and increased risk of toxicity.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Adequate and well-controlled studies in humans have not been done. However, studies in animals have not shown that cefaclor causes impaired fertility.
Use in pregnancy. (Category B1)
The oral administration of high dose cefaclor (500 mg/kg) in pregnant rats and mice has resulted in a slight increase of minor skeletal malformations. Safety of this product for use during pregnancy has not been established. Cefaclor should not be used in women of childbearing potential unless, in the judgment of the treating clinician, its use is considered essential to the welfare of the patient and the expected benefits outweigh potential risks.
Use in lactation. Small amounts of cefaclor have been detected in mother's milk following administration of single 500 mg doses of cefaclor. Average levels were 0.18, 0.20, 0.21 and 0.16 microgram/mL at 2, 3, 4 and 5 hours respectively. Trace amounts were detected at 1 hour. The effect on nursing infants is not known. Caution should be exercised when Ceclor is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Gastrointestinal. The most frequent side effect has been diarrhoea.
Nausea and vomiting have been reported rarely. Colitis, including rare instances of pseudomembranous colitis, has been reported in conjunction with therapy with Ceclor (see Section 4.4 Special Warnings and Precautions for Use).
Hepatobiliary disorder. Transient hepatitis and cholestatic jaundice have been reported rarely.
Slight elevations in AST, ALT, or alkaline phosphatase values have also been reported.
Immune system disorders. Allergic reactions, such as urticaria and morbilliform eruptions, have been observed, as have pruritus and positive Coombs' tests. These reactions usually subsided upon discontinuation of the drug. Angioedema and fever have been reported rarely.
Cases of serum sickness-like reactions have been reported with the use of cefaclor. These have been reported more frequently in children than in adults, with an overall occurrence ranging from 0.5% (1 in 200) in one trial, to 0.024% (2 in 8,346) in overall clinical trials (with an incidence in children in clinical trials of 0.055%). The worldwide reporting rate for serum sickness-like reactions in adults is very rare (< 0.01%). Serum sickness-like reactions are characterised by findings of erythema multiforme, rashes, and other skin manifestations accompanied by arthritis/ arthralgia, with or without fever, and differ from classic serum sickness in that there is infrequently associated lymphadenopathy and proteinuria, no circulating immune complexes, and no evidence to date of sequelae of the reaction. While further investigation is ongoing, serum sickness-like reactions appear to be due to hypersensitivity and more often occur during or following a second (or subsequent) course of therapy with cefaclor. Such reactions have been reported more frequently in children than in adults. Signs and symptoms usually occur a few days after initiation of therapy and subside within a few days after cessation of therapy; occasionally these reactions have resulted in hospitalisation, usually of short duration (median hospitalisation: 2 to 3 days, based on postmarketing surveillance studies). In those requiring hospitalisation, the symptoms have ranged from mild to severe at the time of admission with more of the severe reactions occurring in children. Antihistamines and glucocorticoids appear to enhance resolution of the signs and symptoms. No serious sequelae have been reported. More severe hypersensitivity reactions, including Stevens-Johnson syndrome, and toxic epidermal necrolysis have been reported rarely. Anaphylaxis may be more common in patients with a history of penicillin allergy. The worldwide reporting rate for anaphylaxis in the total population is very rare (< 0.01%).
Blood and lymphatic system disorders. Eosinophilia, transient lymphocytosis, leukopenia and rarely, thrombocytopenia, thrombocytosis, haemolytic anaemia, aplastic anaemia, agranulocytosis and reversible neutropenia of possible clinical significance.
There have been rare reports of increased prothrombin time with or without clinical bleeding in patients receiving cefaclor and warfarin concomitantly.
There have also been reports of transient fluctuations in leukocyte count, predominantly lymphocytosis in infants and young children.
Renal and urinary disorders. Reversible interstitial nephritis, slight elevations in serum urea or serum creatinine or abnormalities of urinalysis (haematuria; pyuria).
Infections and infestations. Genital pruritus, moniliasis or vaginitis.
Nervous system disorders. Reversible hyperactivity, nervousness, insomnia, confusion, hypertonia, dizziness, hallucinations, headache or somnolence have been rarely reported.
Transitory abnormalities in clinical laboratory test results have been reported, but their clinical significance is uncertain.
Severe and other subcutaneous tissue disorders. Severe cutaneous adverse reactions, including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in beta-lactam antibiotics.
Post-marketing experience. Nervous system disorders. Frequency not known: seizures, encephalopathy and/or myoclonus.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Signs and symptoms. The toxic symptoms following an overdose of Ceclor may include nausea, vomiting, epigastric distress and diarrhoea. The severity of the epigastric distress and the diarrhoea are dose related. If other symptoms are present, it is probable that they are secondary to an underlying disease state, an allergic reaction, or the effects of other intoxication.
Treatment. In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs and unusual drug kinetics in the patient.
Protect the patient's airway and support ventilation and perfusion. Meticulously monitor and maintain, within acceptable limits, the patient's vital signs, blood gases, serum electrolytes, etc. Absorption of drugs from the gastrointestinal tract may be decreased by giving activated charcoal, consider charcoal instead of or in addition to gastric emptying. Repeated doses of charcoal over time may hasten elimination of some drugs that have been absorbed. Safeguard the patient's airway when employing charcoal.
Forced diuresis, peritoneal dialysis, haemodialysis, or charcoal haemoperfusion have not been established as beneficial for an overdose of cefaclor.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Ceclor (cefaclor monohydrate) is a semi-synthetic cephalosporin antibiotic, which exercises its activity by inhibiting cell wall synthesis.
Microbiology. In vitro tests demonstrate that the bactericidal action of cephalosporins results from inhibition of cell wall synthesis. Cefaclor is stable in the presence of bacterial β-lactamases; consequently, β-lactamase producing organisms resistant to penicillins and some cephalosporins may be susceptible to cefaclor. Cefaclor has been shown to be active against most strains of the following organisms both in vitro and in clinical infections.
Staphylococci, including coagulase-positive and penicillinase-producing strains (but not methicillin resistant strains of Staph. aureus); Streptococcus pyogenes (group A beta-haemolytic streptococci); Streptococcus (Diplococcus) pneumoniae; Escherichia coli; Proteus mirabilis; Klebsiella sp; Haemophilus influenzae; Neisseria gonorrhoeae (penicillinase-producing and non-penicillinase producing strains); Moraxella (Branhamella) catarrhalis.
Note. Pseudomonas species, Acinetobacter calcoaceticus, Enterococci, Enterobacter, indole-positive Proteus, and Serratia species are resistant to cefaclor. Methicillin resistant strains are also resistant to cefaclor.
Susceptibility testing. Dilution or diffusion techniques, either quantitative (MIC) or breakpoint, should be used following a regularly updated, recognised and standardised method (e.g. NCCLS). Standardised susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures.
A report of "susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of "intermediate" indicates that the result should be considered equivocal, and if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone, which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
Note. The prevalence of resistance may vary geographically for selected species and local information on resistance is desirable, particularly when treating severe infections.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Ceclor is well absorbed after oral administration, whether taken with food or while fasting; however, when it is taken with food, the peak concentration achieved is 50% to 75% of that observed when the drug is administered to fasting subjects and generally appears from 45 minutes to 1 hour later. The presence of food in the gastrointestinal tract does not alter the total amount of cefaclor absorbed. Following administration of 250 mg, 500 mg, and 1 g doses to fasting subjects, average peak plasma levels of antibacterial activity (expressed as microgram/mL of cefaclor) of 7, 13 and 23 microgram/mL, respectively, were obtained at 30 to 60 minutes. The reduced peak serum levels resulting from the administration of cefaclor with food should be considered with reference to the sensitivity of the infecting organism, severity of illness, the dose being administered and the variability in the peak plasma levels which occur with cefaclor.
Metabolism. There is no evidence of metabolism of cefaclor in humans.
Excretion. The plasma half-life in healthy subjects is independent of dosage form and averages 40-60 minutes. See Section 4.4 Special Warnings and Precautions for Use, Use in the elderly.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
The powder for oral liquid also contains the following inactive ingredients: dimeticone 350, erythrosine, methylcellulose, sodium lauryl sulfate, Strawberry Flavour 52312 AP0551 (ARTG PI No.: 274), sucrose, pregelatinised starch and xanthan gum.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Dry powder: store below 25°C.
Reconstituted suspension: store at 2°C to 8°C (Refrigerate. Do not freeze). Use within 14 days and discard remaining portion thereafter.
6.5 Nature and Contents of Container
Container type: bottle (HDPE).
Pack sizes: 75 mL (250 mg/5 mL), 100 mL (125 mg/5 mL).
Some strengths, pack sizes and/or pack types may not be marketed.
Australian register of therapeutic goods (ARTG). AUST R 347594 - Ceclor cefaclor 125 mg/5 mL (as monohydrate) powder for oral liquid bottle.
AUST R 347593 - Ceclor cefaclor 250 mg/5 mL (as monohydrate) powder for oral liquid bottle.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Ceclor (cefaclor monohydrate) is a semisynthetic cephalosporin antibiotic for oral administration.
Cefaclor monohydrate is a white to off white crystalline powder, slightly soluble in water, but is insoluble in alcohol and chloroform.
Chemical structure.
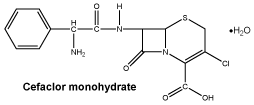
Molecular formula: C15H16ClN3O5S.
Molecular weight: 385.8.
CAS number. 70356-03-5.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
16 May 1996
Date of Revision
10 July 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.