Cipla Ipratropium Metered Dose Inhaler
Brand Information
| Brand name | Cipla Ipratropium Metered Dose Inhaler |
| Active ingredient | Ipratropium bromide monohydrate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Cipla Ipratropium Metered Dose Inhaler.
Summary CMI
Cipla Ipratropium Metered Dose Inhaler
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Cipla Ipratropium?
Cipla Ipratropium metered dose inhaler (Cipla Ipratropium MDI) contains the active ingredient ipratropium bromide monohydrate. Cipla Ipratropium is used to make breathing easier for people with asthma or chronic obstructive pulmonary disease (COPD).
For more information, see Section 1. Why am I using Cipla Ipratropium? in the full CMI.
2. What should I know before I use Cipla Ipratropium?
Do not use if you have ever had an allergic reaction to ipratropium, atropine or any medicines like atropine or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Cipla Ipratropium? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Cipla Ipratropium and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Cipla Ipratropium?
Inhale each puff through the mouth. The usual doses are:
- Adults: 2 to 4 puffs, three to four times daily (at regular intervals), up to a maximum of 16 puffs a day
More instructions can be found in Section 4. How do I use Cipla Ipratropium? in the full CMI.
5. What should I know while using Cipla Ipratropium?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Cipla Ipratropium? in the full CMI.
6. Are there any side effects?
Common but usually mild potential side effects include headache, dizziness, throat irritation, cough, dry mouth, nausea and digestion-related issues (e.g. constipation, diarrhoea, indigestion, reflux and wind).
Serious potential side effects that require medical attention include allergic reaction, difficulty in breathing, heartbeat changes, eye irritation or vision changes and difficulty in passing urine.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Cipla Ipratropium Metered Dose Inhaler
Active ingredient: ipratropium bromide monohydrate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Cipla Ipratropium. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Cipla Ipratropium.
Where to find information in this leaflet:
1. Why am I using Cipla Ipratropium?
2. What should I know before I use Cipla Ipratropium?
3. What if I am taking other medicines?
4. How do I use Cipla Ipratropium?
5. What should I know while using Cipla Ipratropium?
6. Are there any side effects?
7. Product details
1. Why am I using Cipla Ipratropium?
Cipla Ipratropium metered dose inhaler (Cipla Ipratropium MDI) contains the active ingredient ipratropium bromide monohydrate. Cipla Ipratropium is a type of bronchodilator, which are medicines that help to open up the airways.
Cipla Ipratropium is used to make breathing easier for people with asthma or chronic obstructive pulmonary disease (COPD).
Asthma is a disease where the lining of the lungs become inflamed (red and swollen), making it difficult to breathe.
COPD (which includes chronic bronchitis and emphysema) is a long-term, serious lung condition. You may have difficulty breathing (shortness of breath or wheezing), cough, tightness in your chest or coughing up phlegm.
Cipla Ipratropium begins to act quickly after use but may take up to 2 hours to give maximum benefit.
2. What should I know before I use Cipla Ipratropium?
Warnings
Do not use Cipla Ipratropium if:
- You are allergic to ipratropium, or any of the ingredients listed at the end of this leaflet.
- You are allergic to atropine or medicines like atropine.
Check with your doctor if you:
- Have or have had any of the following medical conditions:
- glaucoma (high pressure in the eye)
- difficulty or pain when passing urine
- constipation
- cystic fibrosis (a serious condition that affects the lungs and digestive system)
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant, intend to become pregnant, are breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved.
Alcohol content
- Cipla Ipratropium contains about 8.4 mg of alcohol in each puff. The amount in each puff is equal to less than 1 ml beer or 1 ml wine. The small amount of alcohol in Cipla Ipratropium will not have any noticeable effects.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may be affected by Cipla Ipratropium or may interfere with how Cipla Ipratropium works. These include:
- Medicines used to treat heart problems such as adrenaline.
- Medicines used to treat asthma or COPD such as theophylline, salbutamol and tiotropium.
You may need different amounts of your medicines, or you may need to take different medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Cipla Ipratropium.
4. How do I use Cipla Ipratropium?
How much to use
The recommended doses are:
- Adults: 2 to 4 puffs, three to four times daily (at regular intervals), up to a maximum of 16 puffs a day.
Your doctor may advise you to use a different dose, depending on your condition and specific needs.
Follow the instructions provided by your doctor or pharmacist and use Cipla Ipratropium until your doctor tells you to stop.
When to use Cipla Ipratropium
- Use Cipla Ipratropium at about the same time each day.
How to use Cipla Ipratropium
Leaflet instructions for Use (with diagrams) on how to use Cipla Ipratropium is enclosed in your carton.
- Inhale each puff properly through the mouth.
- Ask your doctor or pharmacist for help if:
- you are not sure how to use the inhaler
- you find it difficult to breathe in and use your inhaler at the same time. Your doctor or pharmacist may recommend another method, such as using a spacer with the inhaler.
Testing your inhaler
- When you use the inhaler for the first time or if you have not used the inhaler for three days or more:
- remove the mouthpiece cover
- point the mouthpiece away from you and press the canister twice (once at a time) to release two puffs into the air
Cleaning instructions
- Clean your mouthpiece at least once a week. It is important to keep the mouthpiece of your inhaler clean to ensure that medicine does not build up and block the spray.
- For cleaning, first take off the dust cap and remove the canister from the mouthpiece. Rinse warm water through the mouthpiece until no medication build-up and/or dirt is visible.
- After cleaning, shake out the mouthpiece and let it air-dry without using any heating system. Once the mouthpiece is dry, replace the canister and the dust cap.
If you forget to use Cipla Ipratropium
If it is almost time for your next dose, skip the dose you missed and have your next dose when you are meant to.
Otherwise, use it as soon as you remember, and then go back to using your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
Ask your doctor or pharmacist if you are not sure what to do.
If you use too much Cipla Ipratropium
If you think that you have used too much Cipla Ipratropium, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose may include:
- a fast or irregular heartbeat
- dry mouth
- blurred vision.
5. What should I know while using Cipla Ipratropium?
Things you should do
- Stop using Cipla Ipratropium and contact your doctor immediately if you get sudden tightness of the chest, coughing, wheezing or breathlessness immediately after using Cipla Ipratropium. These may be signs of a condition called bronchospasm.
- Contact your doctor immediately if you experience irritation or a feeling of having something in the eye, or any disturbances with your sight (blurred vision, visual halos or coloured images) together with red eyes, during or after using Cipla Ipratropium. This may mean that you have developed a serious eye condition called narrow-angle glaucoma. This can happen if the spray gets in your eyes.
- Tell your doctor immediately if you become pregnant
- If you have an Asthma or COPD Action Plan that you have agreed with your doctor, follow it closely at all times.
- If you find that the usual dose of Cipla Ipratropium is not giving as much relief as before, or you need to use it more often, contact your doctor so that your condition can be checked.
- Remind any doctor, dentist or pharmacist you visit that you are using Cipla Ipratropium.
- If you plan to have surgery, tell the surgeon or anaesthetist that you are using Cipla Ipratropium. It may affect other medicines used during surgery.
- Keep all of your doctor's appointments so that your progress can be checked.
- Keep your Cipla Ipratropium with you at all times.
Things you should not do
- Do not take any other medicines for your breathing problems without checking with your doctor.
- Do not give Cipla Ipratropium to anyone else, even if they have the same condition as you.
- Do not use Cipla Ipratropium to treat any other complaints unless your doctor or pharmacist tells you to.
- Do not stop using Cipla Ipratropium or lower the dosage without checking with your doctor or pharmacist.
- Do not allow the Cipla Ipratropium spray to enter your eyes.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Cipla Ipratropium affects you.
Cipla Ipratropium may cause dizziness and blurred vision in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Looking after your medicine
- Clean and dry your mouthpiece at least once a week. Follow the cleaning directions in the Instructions for Use leaflet (leaflet is enclosed in the carton). It is important to keep the mouthpiece of your inhaler clean and dry. It may not work as well if it gets dirty.
- Keep your inhaler in a cool dry place where the temperature stays below 25°C.
- Protect from frost. As with most inhaled medications in pressurised canisters, the therapeutic effect of this medication may decrease when the canister is cold. If the Inhaler has been exposed to low temperatures, the patient should take the metal canister out of the plastic case and warm it in their hands for a minimum of two minutes.
- Do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills. - Keep Cipla Ipratropium where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
Do not puncture the container or throw it into a fire.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Less serious side effects
| Less serious side effects | What to do |
Brain and nerves:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs of an allergic reaction:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Cipla Ipratropium contains
Each dose (puff) of Cipla Ipratropium contains:
| Active ingredient (main ingredient) | ipratropium bromide monohydrate (20.87 micrograms) |
| Other ingredients (inactive ingredients) | citric acid purified water ethanol absolute norflurane |
| Potential allergens | alcohol |
Do not take this medicine if you are allergic to any of these ingredients.
Cipla Ipratropium does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
What Cipla Ipratropium looks like
Cipla Ipratropium (AUST R 410010) is supplied in a can with a mouthpiece and it contains 200 puffs.
Who distributes Cipla Ipratropium
Cipla Ipratropium is distributed in Australia by:
Cipla Australia Pty Ltd.,
Level 1 / 132-136 Albert Road,
South Melbourne Vic 3205.
drugsafety@cipla.com
Phone: 1800-569-074
This leaflet was prepared in April 2025.
Brand Information
| Brand name | Cipla Ipratropium Metered Dose Inhaler |
| Active ingredient | Ipratropium bromide monohydrate |
| Schedule | S4 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Ipratropium bromide monohydrate.
2 Qualitative and Quantitative Composition
Cipla Ipratropium contains ipratropium bromide monohydrate 20.87 micrograms [equivalent to 20 micrograms of ipratropium bromide] per actuation.
Excipients with known effect. Contains 8.4 mg ethanol absolute per actuation.
For full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Cipla Ipratropium is a pressurised inhalation solution.
Each Cipla Ipratropium metered dose aerosol is a colourless solution, filled in an aluminum container fitted with a suitable metering valve and a plastic actuator.
4 Clinical Particulars
4.1 Therapeutic Indications
Cipla Ipratropium metered aerosol is indicated as a bronchodilator for maintenance treatment of bronchospasm associated with asthma and chronic obstructive pulmonary disease (COPD).
4.2 Dose and Method of Administration
Note. One puff (metered dose) of Cipla Ipratropium contains 20.87 micrograms of ipratropium bromide monohydrate [equivalent to 20 micrograms of ipratropium bromide].
If the response to the treatment is inadequate, medical advice should be sought so that appropriate measures can be taken. It is advisable not to greatly exceed the recommended daily dose as this suggests additional therapeutic modalities may be needed.
If therapy does not produce a significant improvement or if the patient's condition gets worse, medical advice must be sought in order to determine a new plan of treatment. In the case of acute or rapidly worsening dyspnoea, a doctor should be consulted immediately.
Adults. Two puffs 3 to 4 times daily, although some patients may need up to 4 puffs at a time to obtain maximum benefit during early treatment.
Before first use of the inhaler, or if the inhaler has not been used for three days or more, prime the inhaler by releasing two puffs into the air.
Patients should be instructed to clean mouthpiece at least once a week. It is important to keep the mouthpiece of inhaler clean to ensure that medicine does not build up and block the spray.
Patients with poor inhaler technique will benefit from the consistent use of a spacer device with their metered aerosol. Use of a spacer will also decrease the amount of drug deposited in the mouth and back of the throat, and therefore, reduce the incidence of local irritation in susceptible patients.
In those people using a spacer, a change in formulation of the drug used, or a change in the make of the spacer may be associated with alterations in the amount of drug delivered to the lungs. The clinical significance of these alterations is uncertain. However, in these situations, the person should be monitored for any change in their condition.
If using a spacer, the patient should be instructed to breathe in and out after each actuation of the drug into the spacer. Any delay should be kept to a minimum.
Static on the walls of the spacer may cause variability in drug delivery. Patients should be instructed to wash the spacer in warm water and detergent and allow it to dry without rinsing or drying with a cloth. This should be performed before initial use of the spacer and at least monthly thereafter.
The Cipla Ipratropium metered aerosol can must only be used with the mouthpiece supplied with the product.
4.3 Contraindications
Known hypersensitivity to atropine or its derivatives (such as the active substance ipratropium bromide), or to any of the other ingredients of Cipla Ipratropium.
4.4 Special Warnings and Precautions for Use
Hypersensitivity. Immediate hypersensitivity reactions may occur after administration of ipratropium bromide pressurised inhalation aerosol, as demonstrated by rare cases of urticaria, angio-oedema, rash, bronchospasm, oropharyngeal oedema and anaphylaxis.
Paradoxical bronchospasm. As with other inhaled medicines ipratropium bromide may result in paradoxical bronchospasm that may be life-threatening. If paradoxical bronchospasm occurs ipratropium bromide pressurised inhalation aerosol should be discontinued immediately and substituted with an alternative therapy.
Anticholinergic effects. Like other drugs with anticholinergic activity, ipratropium bromide should be avoided or used with caution in patients where atropine-like effects may precipitate or exacerbate a pre-existing clinical condition. Patients at particular risk are those with eyes with narrow iridocorneal angles as acute angle-closure glaucoma may be precipitated, or patients with a tendency towards urinary retention or constipation.
Ocular complications. Ipratropium bromide pressurised inhalation aerosol should be used with caution in patients predisposed to narrow-angle glaucoma.
There have been isolated reports of ocular complications (mydriasis, increased intraocular pressure, acute angle glaucoma, eye pain) as a result of direct eye contact of ipratropium bromide formulations. Thus, patients must be instructed in the correct administration of ipratropium bromide pressurised inhalation aerosol and warned not to allow the aerosol to enter the eyes.
Patients who may be predisposed to glaucoma should be specifically warned to protect their eyes. Eye pain or discomfort, blurred vision, visual halos or coloured images in association with red eyes from conjunctival congestion and corneal oedema, may be signs of acute angle-closure glaucoma. Should any combination of these symptoms develop, treatment with miotic drops should be initiated and specialist advice sought immediately.
Renal and urinary effects. Ipratropium bromide pressurised inhalation aerosol should be used with caution in patients with pre-existing urinary outflow tract obstruction (e.g. prostatic hyperplasia or bladder-neck obstruction).
Gastro-intestinal motility disturbances. Patients with cystic fibrosis may be more prone to gastrointestinal motility disturbances.
Propellants. Cipla Ipratropium CFC-free metered aerosol contains the hydrofluoroalkane propellant norflurane. In animal studies, norflurane has been shown to have no significant pharmacological effects, except at very high exposure concentrations when narcosis and a relatively weak sensitisation to the arrhythmogenic effects of catecholamines were found. The potency of the cardiac sensation was less than that of trichloromethane.
Excessive inhalation of the aerosol should, however, be avoided as this carries a potential hazard, both from the propellant as well as from overdosage of the active therapeutic agent contained in the formulation. The recommended dose should not be exceeded and the patients should be so informed.
Alcohol content. Cipla Ipratropium contains about 8.4 mg of alcohol (ethanol) in each actuation. The amount in each actuation of this medicine is equivalent to less than 1 mL beer or 1 mL wine. The small amount of alcohol in this medicine will not have any noticeable effects.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The chronic co-administration of ipratropium bromide pressurised inhalation aerosol with other anticholinergic drugs has not been studied. Therefore, the chronic co-administration of ipratropium bromide pressurised inhalation aerosol with other anticholinergic drugs is not recommended.
Beta-adrenergics and xanthine preparations may intensify the bronchodilatory effect of ipratropium bromide.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Clinical data on fertility are not available for ipratropium bromide.
Use in pregnancy. (Category B1)
Care is recommended during pregnancy, particularly in the first trimester. The safety of ipratropium bromide pressurised inhalation aerosol during pregnancy has not been established. The benefits of using ipratropium bromide pressurised inhalation aerosol when pregnancy is confirmed or suspected must be weighed against possible hazards to the foetus. Studies in rats, mice and rabbits showed no embryo-toxic nor teratogenic effects.
Use in lactation. No specific studies are available to determine the excretion of ipratropium bromide in human breast milk. Although lipid-insoluble quaternary cations pass into breast milk, it is unlikely that ipratropium bromide would reach the infant to an important extent, especially when administered by inhalation. However, as many drugs are excreted into breast milk, caution should be exercised when ipratropium bromide pressurised inhalation aerosol is administered to breastfeeding mothers.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. However, patients should be advised that they may experience undesirable effects such as dizziness, accommodation disorder, mydriasis and blurred vision during treatment with ipratropium bromide pressurised inhalation aerosol. Therefore, caution should be recommended when driving a car or operating machinery. If patients experience the above mentioned side effects they should avoid potentially hazardous tasks such as driving or operating machinery.
4.8 Adverse Effects (Undesirable Effects)
Many of the listed undesirable effects can be assigned to the anticholinergic properties of ipratropium bromide pressurised inhalation aerosol. As with all inhalation therapy ipratropium bromide pressurised inhalation aerosol may show symptoms of local irritation. The most frequent side effects reported in clinical trials were headache, dizziness, throat irritation, cough, gastrointestinal disorders (including constipation, diarrhoea, gastrointestinal motility disorder, dry mouth, nausea, stomatitis, oedema mouth and vomiting).
If the substance enters the eyes by inappropriate handling, mild and reversible disturbance of accommodation may occur. Other ocular complications have also been reported (see Section 4.4 Special Warnings and Precautions for Use). However, acute angle-closure glaucoma has been reported following direct eye contact.
Allergic-type reactions such as angio-oedema of the tongue, lips and face may also occur.
The following adverse reactions have been reported during use of ipratropium bromide pressurised inhalation aerosol in clinical trials and during the post-marketing experience at the following frequency: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000); very rare (< 1/10,000). See Table 1.
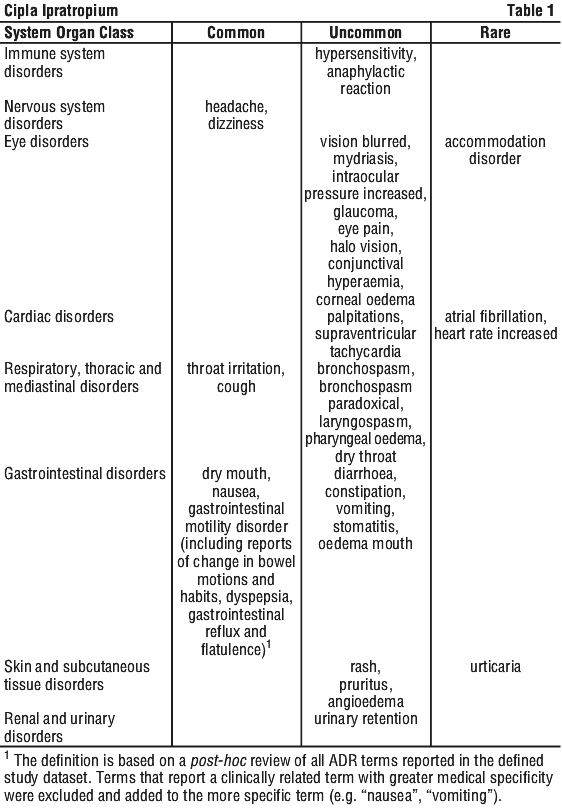
4.9 Overdose
No symptoms specific to over-dosage have been encountered. In view of the wide therapeutic range and topical administration of ipratropium bromide pressurised inhalation aerosol, no serious anticholinergic symptoms are to be expected. Minor systemic manifestations of anticholinergic action, including dry mouth, visual accommodation disorder and tachycardia may occur.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
Pharmacotherapeutic group: Anticholinergics. ATC code: R03BB01.
5.1 Pharmacodynamic Properties
Mechanism of action. Ipratropium bromide pressurised inhalation aerosol is an anticholinergic bronchodilator. It appears to inhibit vagally mediated reflexes by antagonising the action of acetylcholine, the transmitter agent released from the vagal nerve. Anticholinergics prevent the increase in intracellular calcium concentration caused by interaction of acetylcholine with the muscarinic receptor on bronchial smooth muscle. Bronchodilation following inhalation of ipratropium bromide pressurised inhalation aerosol is a local, site specific effect at the bronchial smooth muscle. Ipratropium bromide pressurised inhalation aerosol has no deleterious effect on airway mucous secretion or mucociliary clearance.
The time course of action of ipratropium bromide pressurised inhalation aerosol also differs from the β2 agonists in that although the onset of bronchodilator response is seen within 3-5 minutes of administration, peak response is not reached until 1.5-2 hours after inhalation. The duration of significant bronchodilator action is up to 6 hours.
Ipratropium bromide pressurised inhalation aerosol may be used in combination with β2 agonists. There is evidence that in patients who respond to ipratropium bromide pressurised inhalation aerosol, the concurrent administration of ipratropium bromide pressurised inhalation aerosol and β2 agonists produces a greater relief of bronchospasm than either drug given alone.
Clinical trials. The use of ipratropium bromide metered aerosol, delivered using CFC-containing propellants, is well established in clinical practice. A clinical programme has been conducted to demonstrate the therapeutic equivalence of ipratropium bromide pressurised inhalation aerosol CFC-free and CFC-containing metered aerosols. The safety and efficacy of ipratropium bromide pressurised inhalation aerosol CFC-free metered aerosol, in chronic obstructive pulmonary disease (COPD) and asthma, was established from the results of one 12-month and two 12-week safety and efficacy trials conducted in COPD, a 12-week study in asthmatic adults.
A randomised, open-label, parallel design 12-month study in COPD patients, compared the safety and efficacy of ipratropium bromide metered aerosols containing either CFCs (n = 151) or norflurane (n = 305). Time profiles of forced expiratory volume in one second (FEV1) mean changes from baseline on all test days demonstrated the efficacy and overall comparability of the ipratropium bromide pressurised inhalation aerosol CFC-free and CFC-containing aerosols. The two formulations were generally comparable throughout the trial with respect FEV1 area under the curve for 0 to 6 hours (AUC0-6). The overall safety profile indicated that both treatments were well tolerated.
A 12-week, double-blind, randomised trial in COPD patients, comparing safety and efficacy of ipratropium bromide pressurised inhalation aerosol CFC-free and CFC-containing metered aerosols (n = 118 and 56 respectively), concluded there were no differences in respect of changes in morning and evening peak expiratory flow rates. In addition, a randomised, double-blind, placebo and active controlled study of 12-weeks duration in COPD patients, concluded that ipratropium bromide pressurised inhalation aerosol CFC-free 42 microgram (n = 125) and 84 microgram (n = 127) were significantly more effective than placebo in terms of adjusted mean FEV1 AUC0-6 and peak response. For each trial, the safety profiles of both formulations were found to be comparable.
A multi-dose comparison of ipratropium bromide pressurised inhalation aerosol CFC-free with CFC-containing metered aerosol in a 12-week, double-blind, parallel group study of adult patients with bronchial asthma, demonstrated that patients who switched to the CFC-free aerosol (n = 159) from the CFC-containing aerosol (n = 75), had no change in daily peak expiratory flow rate values and usage of rescue medication. In adult study, the adverse event profile of both formulations was very similar for all events reported.
In summary, the data shows that ipratropium bromide pressurised inhalation aerosol CFC-free metered aerosol is comparable in terms of efficacy and safety to the CFC-containing formulation.
5.2 Pharmacokinetic Properties
Absorption. Following inhalation, 10 to 30% of the dose (depending on the formulation and inhalation technique) is generally deposited in the lungs. The major part of the dose is swallowed and passes into the gastrointestinal tract. Due to the low gastrointestinal absorption of ipratropium bromide, the bioavailability of the portion of the dose swallowed, accounts for approximately 2% of the dose. This fraction of the dose does not make a relevant contribution to the plasma concentrations of the active ingredient. The portion of the dose deposited in the lungs reaches the circulation rapidly (within minutes) and has nearly complete systemic availability.
From renal excretion data (0-24 hours), the total systemic bioavailability (pulmonary and gastrointestinal portions) of inhaled doses of ipratropium bromide was estimated to be in the range 7 to 28%. This is also a valid range for inhalation from ipratropium bromide pressurised inhalation aerosol CFC-free because the kinetic results (renal excretion, AUC and Cmax) from the CFC-free and the CFC-containing formulations are approximately comparable.
Distribution. Kinetic parameters describing the disposition of ipratropium bromide were calculated from plasma concentrations after intravenous administration. A rapid biphasic decline in plasma concentrations is observed. The volume of distribution (Vz) is 338 L (approximately 4.6 L/kg). The half-life of the terminal elimination phase is about 1.6 hours. The drug is less than 20% bound to plasma proteins. The ipratropium ion does not cross the blood-brain barrier, consistent with the ammonium structure of the molecule. The main urinary metabolites bind poorly to the muscarinic receptor and have no activity.
Metabolism. The mean total clearance of the drug is 2.3 L/min. The major portion, approximately 60% of the systemically available dose, is eliminated by metabolic degradation, probably in the liver.
Excretion. Approximately 40% of the systemically available dose is cleared via urinary excretion, corresponding to an experimental renal clearance of 0.9 L/min. After oral dosing less than 1% of the dose is renally excreted, indicating an insignificant absorption of ipratropium bromide from the gastrointestinal tract.
In excretion balance studies, after intravenous administration of a radioactive dose, less than 10% of the drug-related radioactivity (including parent compound and all metabolites), are excreted via the biliary-faecal route. The dominant excretion of drug-related radioactivity occurs via the kidneys.
5.3 Preclinical Safety Data
Genotoxicity. Results of various mutagenicity studies (Ames test, mouse dominant lethal test, mouse micronucleus test and chromosome aberration of bone marrow in Chinese hamsters) were negative.
Carcinogenicity. Two-year oral carcinogenicity studies in rats and mice have revealed no carcinogenic potential at dietary doses up to 6 mg/kg/day for ipratropium bromide.
6 Pharmaceutical Particulars
6.1 List of Excipients
In addition to ipratropium bromide monohydrate, Cipla Ipratropium contains citric acid, purified water, ethanol absolute and the norflurane.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
Avoid storage in direct sunlight or heat. Do not puncture or incinerate, as canister may explode.
Protect from frost. As with most inhaled medications in pressurised canisters, the therapeutic effect of this medication may decrease when the canister is cold. If the Inhaler has been exposed to low temperatures, the patient should take the metal canister out of the plastic case and warm it in their hands for a minimum of two minutes.
6.5 Nature and Contents of Container
Cipla Ipratropium is contained in a aluminium container fitted with a suitable metering valve and a plastic actuator, containing 200 metered doses.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Ipratropium bromide is a synthetic quaternary ammonium compound, chemically related to atropine. The addition of an N-isopropyl group distinguishes the molecule from atropine and is responsible for a lower lipid solubility.
Ipratropium bromide is a white or off-white crystalline substance. It is freely soluble in methanol, soluble in water and slightly soluble in ethanol 96% (v/v).
The chemical name for ipratropium bromide (as monohydrate) is (1R,3r,5S,8r)-3-[(RS)-(3-hydroxy-2-phenyl-propanoyl) -oxy]-8-methyl-8-(1-methylethyl)-8-azoniabicyclo[3.2.1]octane bromide monohydrate.
The molecular formula is C20H30NO3Br.H2O and the molecular weight is 430.4.
Ipratropium bromide has the following structural formula:
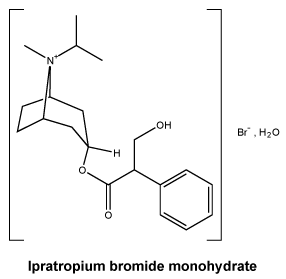
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Date of First Approval
27 May 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.