Colofac
Brand Information
| Brand name | Colofac |
| Active ingredient | Mebeverine hydrochloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Colofac.
Summary CMI
COLOFAC®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about taking this medicine, speak to your doctor or pharmacist.
1. Why am I taking COLOFAC?
COLOFAC contains the active ingredient mebeverine hydrochloride. COLOFAC is taken to relieve the symptoms of irritable bowel syndrome, which can include, abdominal pain and cramps, bowel spasm, diarrhoea and flatulence.
For more information, see Section 1. Why am I taking COLOFAC? in the full CMI.
2. What should I know before I take COLOFAC?
Do not take if you have ever had an allergic reaction to COLOFAC or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take COLOFAC? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with COLOFAC and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take COLOFAC?
- Take COLOFAC at about the same time each day. The usual dose is one tablet three times a day.
- Take COLOFAC immediately before or during a meal. Swallow the tablets with a glass of water.
More instructions can be found in Section 4. How do I take COLOFAC? in the full CMI.
5. What should I know while taking COLOFAC?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking COLOFAC? in the full CMI.
6. Are there any side effects?
Speak to your doctor if you have any of these less serious side effects and they worry you: indigestion or heartburn, constipation, dizziness, headache, tiredness, difficulty sleeping, loss of appetite. Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects: swelling of the face, lips or tongue, difficulty swallowing or breathing.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
COLOFAC®
Active ingredient: mebeverine hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about taking COLOFAC. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about taking COLOFAC.
Where to find information in this leaflet:
1. Why am I taking COLOFAC?
2. What should I know before I take COLOFAC?
3. What if I am taking other medicines?
4. How do I take COLOFAC?
5. What should I know while taking COLOFAC?
6. Are there any side effects?
7. Product details
1. Why am I taking COLOFAC?
COLOFAC contains the active ingredient mebeverine hydrochloride. COLOFAC belongs to a group of medicines called known as antispasmodic agents.
COLOFAC works by relaxing the muscles in the gut, thereby relieving the symptoms of irritable bowel syndrome.
COLOFAC is taken to relieve the symptoms of irritable bowel syndrome, which can include, abdominal pain and cramps, bowel spasm, diarrhoea and flatulence.
2. What should I know before I take COLOFAC?
Warnings
Do not take COLOFAC if:
- you are allergic to mebeverine hydrochloride, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can take this medicine.
Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing.
Check with your doctor if you:
- have any of the following medical conditions:
- angina or other heart conditions
- liver problems
- kidney problems
- lactose intolerance - take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect COLOFAC.
4. How do I take COLOFAC?
How much to take
- The usual dose is one tablet three times a day.
- Follow the instructions provided and continue taking COLOFAC until your doctor tells you to stop.
- It is important to keep taking your medicine even if you feel well.
When to take COLOFAC
- Take COLOFAC immediately before or during a meal.
- Take COLOFAC at about the same time each day.
- Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How to take COLOFAC
- Swallow the tablets with a glass of water.
If you forget to take COLOFAC
COLOFAC should be taken at the same time each day. If you miss your dose at the usual time, take it as soon as you remember, and then go back to taking your medicine as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
- This may increase the chance of getting an unwanted side effect.
- If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for advice.
If you take too much COLOFAC
If you think that you have taken too much COLOFAC, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking COLOFAC?
Things you should do
Call your doctor straight away if you:
- become pregnant while you are taking COLOFAC
If you are going to have surgery, tell the surgeon that you are taking COLOFAC.
Remind any doctor, dentist or pharmacist you visit that you are taking COLOFAC.
Things you should not do
- Do not take COLOFAC to treat any other complaints unless your doctor or pharmacist tells you to.
- Do not give COLOFAC to anyone else, even if they have the same condition as you.
- Do not stop taking COLOFAC, or lower the dosage, without checking with your doctor
Driving or using machines
Be careful before you drive or use any machines or tools until you know how COLOFAC affects you.
Looking after your medicine
- Keep your tablets in the pack until it is time to take them.
- If you take the tablets out of the box or the blister pack they may not keep well.
- Keep the medicine in a cool, dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to take this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not take this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
The below list includes the more common side effects of your medicine. They are usually mild and short-lived.
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What COLOFAC contains
| Active ingredient (main ingredient) | mebeverine hydrochloride 135 mg per tablet |
| Other ingredients (inactive ingredients) | lactose monohydrate |
| Potential allergens | lactose, sugars and traces of sulfites |
Do not take this medicine if you are allergic to any of these ingredients.
What COLOFAC looks like
COLOFAC tablets are white, round sugar-coated tablets. (AUST R 79335).
COLOFAC tablets are available in boxes of 30 and 90 tablets.
Who distributes COLOFAC
Viatris Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
COLOFAC is made in France.
This leaflet was prepared in February 2022.
COLOFAC® is a Viatris company trade mark
COLOFAC_cmi\Feb22/00
Brand Information
| Brand name | Colofac |
| Active ingredient | Mebeverine hydrochloride |
| Schedule | S4 |
MIMS Revision Date: 01 March 2022
1 Name of Medicine
Mebeverine hydrochloride.
2 Qualitative and Quantitative Composition
Each Colofac tablet contains 135 mg of mebeverine hydrochloride as the active ingredient.
Excipients with known effect. Lactose, sugars and traces of sulfites.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Colofac tablets are sugar-coated, white, round, biconvex, and 11 mm in diameter.
4 Clinical Particulars
4.1 Therapeutic Indications
Colofac tablets are indicated in the management of the irritable bowel syndrome ('irritable colon', 'spastic colon', 'functional bowel disorders', 'spastic constipation', 'nervous diarrhoea'). Colofac is used to treat the symptoms of this condition - i.e. abdominal pain and cramps, persistent non-specific diarrhoea (with or without alternating constipation) and flatulence.
4.2 Dose and Method of Administration
The recommended adult dose is one Colofac mebeverine hydrochloride 135 mg (1 tablet) three times daily, preferably before or with food. In case one or more doses are missed, the patient should continue with the next dose as prescribed, the missed doses are not to be taken in addition to the regular dose.
After a period of several weeks when the desired effect has been obtained, the dosage may be gradually reduced.
4.3 Contraindications
Hypersensitivity to any component of the product.
4.4 Special Warnings and Precautions for Use
Although not reported, Colofac tablets should be used with caution in patients with the following conditions on the basis of potential clinical significance:
Pharmaceutical precaution. Colofac tablets contain lactose monohydrate (80 mg per tablet) and consideration should be given to patients with a potential diagnosis of lactose intolerance simulating irritable bowel syndrome. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine.
The tablets also contain sucrose and should not be used by patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption or sucrase-isomaltase insufficiency.
Cardiac dysrhythmia. In particular patients with partial or complete atrioventricular heart block, and/or angina or severe ischaemic heart disease.
Use in hepatic impairment. Hepatic dysfunction i.e. patients with advanced liver disease e.g. cirrhosis (because of metabolic pathway). Liver function tests may be indicated if patient develops gastrointestinal symptoms or jaundice suggesting hepatic sensitivity.
Use in renal impairment. Advanced renal disease (because of excretory pathway).
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
See Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
Safe use in pregnancy has not been established relative to adverse effects on foetal development. Therefore, Colofac tablets are not recommended during the first trimester of pregnancy, and otherwise risk-benefit must be considered in its use in pregnant women.
Teratogenicity has not been demonstrated in teratology studies in rats and rabbits.
Use in lactation. Mebeverine is secreted in breast milk (< 10 microgram/mL following an oral dose of 100 mg mebeverine hydrochloride). Although problems have not been documented, as a general rule, Colofac tablets should not be given to a woman who is breast feeding unless the anticipated benefits outweigh possible risks.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Because of the low incidence of adverse drug effects reported a meaningful estimate of adverse reactions is difficult to obtain.
The following side effects have been reported in clinical studies: indigestion, heartburn, dizziness, insomnia, anorexia, headache, decrease in pulse rate, constipation, general malaise.
In very rare cases, allergic reactions have been reported, in particular, hypersensitivity, urticaria, angioedema, face oedema and exanthem.
Adverse effects reported during post-marketing use have been consistent with those reported in clinical studies, with the following additional side effect reported:
Immune system disorders. Hypersensitivity (anaphylactic reactions).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
On theoretical grounds, it may be predicted that CNS excitability might occur in cases of overdosage. Observed symptoms of overdose have included those of neurological and cardiovascular nature.
No specific information is available on the treatment of overdosage of mebeverine hydrochloride and no specific antidote is available. Therapy with Colofac tablets should be discontinued, and the patients' vital functions monitored closely. Treatment is symptomatic and supportive.
For more information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Category: antispasmodic; smooth muscle relaxant.
Mechanism of action. Mebeverine has a direct non-specific relaxant effect on vascular, cardiac, and other smooth muscle. Studies indicate that the spasmolytic activity of mebeverine is not restricted to one particular system, but the compound possesses a polyvalent spasmolytic action in which at least three types of mechanisms are involved:
a direct musculotropic action involving calcium ion exchange and stabilization of excitable membranes;
a competitive antimuscarinic activity of about 0.05-0.1 times that of atropine;
a local anaesthetic activity together with potentiation of sympathetic inhibitory influences due to blockade of noradrenaline uptake into sympathetic nerve endings.
In in vitro studies mebeverine hydrochloride has been shown to have a papaverine-like spasmolytic effect on the smooth muscle of the ileum, uterus and the gall bladder. It possesses a strong local anaesthetic activity.
When tested in vivo in various species, mebeverine hydrochloride was found to be three to five times more powerful than papaverine in blocking spasm of smooth muscle and in relieving the carbachol-induced spasm of the sphincter of Oddi in rabbits, mebeverine hydrochloride proved to be twenty times more active than papaverine. In vivo studies also demonstrate that mebeverine has only minor effects on normal intestinal peristalsis but possesses spasmolytic activity when hypermotility is induced. The spasmolytic activity is found in all parts of the gastrointestinal tract and, in some experiments, has been found to be more active on colonic smooth muscle.
Studies with mebeverine hydrochloride 100 mg tablets indicate that mebeverine is free of central anticholinergic effects, and practically free of peripheral effects with an activity of less than 0.001 times that of atropine. Mebeverine does not show central depressant or analgesic effects, and only in high doses are some central stimulating effects observed. No ganglion blocking or interference with neuromuscular transmission occurs.
Mebeverine injected intravenously in animals produces transient cardiac arrhythmias, bradycardia and ECG changes.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Following oral administration of 3H and 14C labelled mebeverine hydrochloride in man, absorption was followed by the appearance in the plasma of veratric acid and an oxidised metabolite of the mebeverine alcohol moiety of the drug, mebeverinic acid.
Distribution. Maximum plasma radioactivity levels were found 1-3 hours after dosing. Binding of mebeverine to human serum albumin was 75%.
Metabolism. The primary metabolic step in mebeverine degradation is hydrolysis of the ester function.
Excretion. The major route of excretion of the metabolites is via the urine (95%) and the peak rate of excretion usually occurs within two hours. Virtually 98% urinary recovery of the conjugated and unconjugated metabolites was observed after a period of 24 hours. No unchanged mebeverine was excreted with the urine.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Colofac tablets also contain acacia, carnauba wax, gelatin, lactose monohydrate, magnesium stearate, povidone, potato starch, sucrose and purified talc.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Container type: blister pack [PVC/Al].
Pack sizes: 10, 30 and 90 tablets.
Some strengths, pack sizes and/or pack types may not be marketed.
Australian register of therapeutic goods (ARTG). AUST R 79335 - Colofac mebeverine hydrochloride 135 mg tablet blister pack.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Mebeverine hydrochloride is 4-[ethyl-[2-(4-methoxyphenyl)-1-methylethyl] aminobutyl veratrate hydrochloride, a derivative of phenylethylamine. It is a white to almost white, crystalline powder having a very bitter taste, very soluble in water, freely soluble in ethanol and practically insoluble in ether. The empirical formula is C25H35NO5.HCl. MW: 466.0.
Chemical structure.
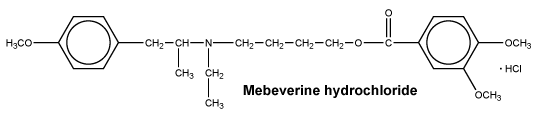
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
16 May 1987
Date of Revision
04 February 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.