Cyclogyl
Brand Information
| Brand name | Cyclogyl |
| Active ingredient | Cyclopentolate hydrochloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Cyclogyl.
Summary CMI
CYCLOGYL™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using CYCLOGYL?
CYCLOGYL contains the active ingredient cyclopentolate hydrochloride. CYCLOGYL is used to enlarge (dilate) the pupil and to prevent the eye from focusing during an eye examination or before a surgical procedure. CYCLOGYL may also be used to relax the muscles of your eye if it is inflamed (swollen and or irritated).
For more information, see Section 1. Why am I using CYCLOGYL? in the full CMI.
2. What should I know before I use CYCLOGYL?
Do not use if you have ever had an allergic reaction to CYCLOGYL or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, allergies, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use CYCLOGYL? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with CYCLOGYL and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use CYCLOGYL?
- Your doctor will put the correct number of drops of CYCLOGYL into your eye(s) before you have an operation or an eye examination.
- If you have been prescribed CYCLOGYL for use at home, the usual adult dose is one drop in the eye(s) followed by a second drop 5 minutes later.
More instructions can be found in Section 4. How do I use CYCLOGYL? in the full CMI.
5. What should I know while using CYCLOGYL?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using CYCLOGYL? in the full CMI.
6. Are there any side effects?
All medicines can have side effects although not everybody gets them. The most common side effects of CYCLOGYL occur in, or around, the eye, including increased pressure in the eye, burning and irritation, sensitivity to light, blurred vision, redness and irritation of the eyelid, dilation of the pupil (which may continue for some time), and eye pain. Occasionally, some people notice unwanted effects in the rest of their body. There is a greater chance of such side effects occurring if CYCLOGYL is used in children.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
CYCLOGYL™
Active ingredient(s): cyclopentolate hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using CYCLOGYL. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using CYCLOGYL.
Where to find information in this leaflet:
1. Why am I using CYCLOGYL?
2. What should I know before I use CYCLOGYL?
3. What if I am taking other medicines?
4. How do I use CYCLOGYL?
5. What should I know while using CYCLOGYL?
6. Are there any side effects?
7. Product details
1. Why am I using CYCLOGYL?
CYCLOGYL contains the active ingredient cyclopentolate hydrochloride. CYCLOGYL (cyclopentolate hydrochloride) belongs to a group of medicines known as cycloplegics / mydriatics.
CYCLOGYL is used to enlarge (dilate) the pupil and to prevent the eye from focusing. Your doctor may use CYCLOGYL prior to a surgical procedure or prior to examining the inside of your eye.
CYCLOGYL may also be used to relax the muscles of your eye. This may be necessary when some parts of the eye are inflamed (iridocyclitis).
The effect of CYCLOGYL is usually no longer than 24 hours.
Ask your doctor if you have any questions or concerns about why this medicine has been used on your eye(s).
Your doctor may have prescribed it for another reason.
2. What should I know before I use CYCLOGYL?
Warnings
Do not use CYCLOGYL if:
- You are allergic to cyclopentolate hydrochloride, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, or other parts of the body
- rash, itching or hives on the skin. - Do not use CYCLOGYL if the expiry date has passed, the packaging is torn or the safety seal around the closure and neck area is broken. If it has expired or is damaged, return it to your pharmacist for disposal.
- CYCLOGYL has been developed for use in the eye only. It is not to be swallowed or injected.
- If you are not sure whether you should be given CYCLOGYL talk to your doctor.
Check with your doctor if you:
- have any other medical conditions, especially angle-closure glaucoma. Prior to using CYCLOGYL your doctor may check your eyes to see if you may be prone to angle-closure glaucoma.
- have any allergies to any other medicines, foods, preservatives or dyes.
- have had a severe reaction to a medicine called atropine.
- take any medicines for any other conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Your doctor will discuss the possible risks and benefits of using CYCLOGYL during pregnancy.
Check with your doctor if you are breastfeeding or intend to breastfeed. Your doctor will discuss the possible risks and benefits involved.
Use in children
CYCLOGYL should not be used in small infants.
Concentrations greater than 0.5% are not recommended due to the risk of serious side effects.
Infants, small children, children with brain damage or spastic paralysis, Down's syndrome, epilepsy (fits) or fair-skinned, blue-eyed children are susceptible to the effects of CYCLOGYL.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket, or health food shop.
Some medicines may interfere with CYCLOGYL and affect how it works. These includes:
- Other medicines used to treatment glaucoma.
- Amantadine (Symmetrel) used to treat Parkinson's disease or type-A influenza.
- Antihistamines used to treat allergies.
- Phenothiazine used to treat severe mental illnesses.
- Tricyclic antidepressants used to treat depression.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect CYCLOGYL.
Your doctor will advise you and decide whether or not to give you CYCLOGYL.
4. How do I use CYCLOGYL?
How much to use
- Your doctor will put the correct number of drops of CYCLOGYL into your eye(s) before you have an operation or an eye examination.
- The usual adult dose of CYCLOGYL is one drop in the eye(s) followed by a second drop 5 minutes later.
- The usual dose for a child is one drop in the eye(s) followed by a second drop 5 minutes later, if necessary.
- Follow the instructions provided and use CYCLOGYL until your doctor tells you to stop.
When to use CYCLOGYL
CYCLOGYL is to be used by your doctor before you have an operation or an examination. Your doctor might prescribe CYCLOGYL for you to use outside the doctor's surgery.
If you forget to use CYCLOGYL
If you forget to use CYCLOGYL Eye Drops, you should put in the drops that you missed as soon as you remember and then go back to using them normally. If it is almost time for your next dose, skip the dose that you have missed and take your next dose when you are due to.
Never use a double dose to make up for the one that you missed.
If you are not sure what to do, contact your doctor or pharmacist.
If you are given too much CYCLOGYL
If too many drops have been put in accidentally in your eye(s) immediately rinse your eye(s) with warm water.
If you think that you or someone else has swallowed or used too much CYCLOGYL, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre in Australia
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of overdose may include:
- flushed or dry skin, rash (may be present in children)
- blurred vision
- rapid and irregular pulse
- fever
- abdominal swelling in children
- convulsions
- hallucinations
- loss of muscle coordination.
Symptoms of a severe overdose include:
- mental and behavioural disturbances (e.g. hallucinations, seizures or fits, disorientation)
- coma
- circulation failures
- breathlessness.
Symptoms of a severe overdose can result in death.
5. What should I know while using CYCLOGYL?
Things you should do
Tell your doctor, dentist, or pharmacist that you are using CYCLOGYL Eye Drops before you start taking any other medicines.
If you become pregnant or decide to breastfeed while using CYCLOGYL, tell your doctor immediately.
Make sure that you take appropriate steps to protect your eye(s) from light.
Your eyes will be very sensitive to light while you are using this medicine.
Things you should not do
Do not let children handle CYCLOGYL Eye Drops. If a child accidentally swallows any of the drops, read the instructions under "If you are given too much CYCLOGYL”.
Do not stop using CYCLOGYL Eye Drops without first asking your doctor. If you stop using CYCLOGYL your condition may worsen.
Do not give this medicine to anyone else, even if they appear to have the same condition as you.
Do not put CYCLOGYL Eye Drops into your eye(s) while you are wearing contact lenses. The preservative in CYCLOGYL, benzalkonium chloride, may be deposited in contact lenses.
Driving or using machines
While your eye(s) are affected by CYCLOGYL, you should not drive, operate machinery or engage in any other hazardous activities.
Looking after your medicine
Keep CYCLOGYL at 2°C to 8°C. Refrigerate. Do not freeze.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place, away from moisture, heat or sunlight, for example. Do not store it
- in the bathroom, near a sink, or
- in the car or a windowsill.
Heat and humidity can destroy some medicines.
Keep CYCLOGYL where children cannot reach it. A locked cupboard at least one and a half metres above the ground is a good place to store medicines.
When to discard your medicine (as relevant)
Discard the container 4 weeks after opening.
Write the date on the bottle label when it was opened to remind you when to discard the bottle.
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicines that is left over.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use any medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
In or around the eye (ocular):
There is a greater chance of such side effects occurring if CYCLOGYL Eye Drops are used in children. | Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic reaction:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor, nurse, or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What CYCLOGYL contains
| Active ingredient (main ingredient) | cyclopentolate hydrochloride 1% (10 mg/mL) |
| Other ingredients (inactive ingredients) | benzalkonium chloride 0.1 mg/mL as preservative boric acid disodium edetate potassium chloride sodium carbonate and/or hydrochloric acid to adjust pH purified water. |
| Potential allergens | CYCLOGYL does not contain: gluten, latex, corn/corn derivatives, mercury, dyes, sulpha, sulphites or sulphate, phthalates (DEHP/PVC), dairy, eggs, BSE/TSE, and alcohol. |
Do not take this medicine if you are allergic to any of these ingredients.
What CYCLOGYL looks like
CYCLOGYL Eye Drops is a sterile solution that comes in a 15 mL dropper bottle (DROP-TAINER®) with screw cap. The safety seal must be removed before use. (AUST R 25273)
Who distributes CYCLOGYL
CYCLOGYL is distributed in Australia by:
Alcon Laboratories (Australia) Pty Ltd
ABN 88 000 740 83054
15 Talavera Road
Macquarie Park NSW 2113
Telephone: 1800 224 153
This leaflet was prepared in September 2025. Internal document code cyc150925c.
You should ensure that you speak to your doctor to obtain the most up to date information on the medicine.
You can also obtain a copy of the most up to date leaflet from your pharmacist. The updates may contain important information about the medicine and its use of which you should be aware.
Brand Information
| Brand name | Cyclogyl |
| Active ingredient | Cyclopentolate hydrochloride |
| Schedule | S4 |
MIMS Revision Date: 01 October 2023
1 Name of Medicine
Cyclopentolate hydrochloride.
2 Qualitative and Quantitative Composition
Cyclogyl eye drops contain cyclopentolate hydrochloride 10 mg/mL (1.0%).
It also contains benzalkonium chloride 0.1 mg/mL as a preservative.
For the full list of excipients see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Eye drops.
Cyclogyl eye drops have been formulated as a sterile, multi-dose product for topical ophthalmic use. The pH of Cyclogyl eye drops is approximately 4.5.
4 Clinical Particulars
4.1 Therapeutic Indications
For mydriasis and cycloplegia in diagnostic procedures. For some pre- and post-operative states when mydriasis is required and when a shorter acting mydriatic and cycloplegic is needed in the therapy of iridocyclitis.
4.2 Dose and Method of Administration
Adults. One drop, followed by a second drop in 5 minutes.
Complete recovery usually occurs in 24 hours.
Children. One drop is instilled in each eye, followed 5 minutes later by a second application if necessary.
Pretreatment with Cyclogyl eye drops on the day prior to examination usually is not necessary.
In order to minimise systemic absorption, apply pressure to the tear duct for two minutes immediately after administration.
4.3 Contraindications
Should not be used:
when narrow-angle glaucoma or anatomically narrow angles are present;
when there is hypersensitivity to any component of this preparation (see Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
For ophthalmic use only - not for injection.
This preparation may cause psychotic reactions, behavioural disturbances and other CNS disturbances in patients with increased susceptibility to anticholinergic drugs. This is especially true in younger age groups, but may occur at any age. Premature and small infants are especially prone to CNS and cardiopulmonary side effects from systemic absorption of cyclopentolate and, therefore, Cyclogyl eye drops should not be used in these patients.
Use with caution in patients, especially children, who have previously had a severe systemic reaction to atropine.
Cyclogyl may cause increased intraocular pressure. The possibility of undiagnosed glaucoma should be considered in some patients, such as elderly patients. Caution should be observed when considering the use of this medication in the presence of Down's syndrome and in those predisposed to angle-closure glaucoma. To avoid inducing angle-closure glaucoma, determine the intraocular pressure and an estimation of the depth of the anterior chamber should be made prior to the initiation of therapy.
Complete recovery of accommodation usually occurs within 24 hours, however, in some individuals complete recovery may require several days.
Because of risk of provoking hyperthermia, use with caution in patients, especially children, who may be exposed to elevated environmental temperatures or who are febrile.
Use in hepatic impairment. No formal studies have been conducted in patients with hepatic impairment.
Use in renal impairment. No formal studies have been conducted in patients with renal impairment.
Use in the elderly. In the elderly and others where increased intraocular pressure may be encountered, mydriatics and cycloplegics should be used with caution.
Elderly patients may be a higher risk for undiagnosed glaucoma as well as cyclopentolate induced psychotic reactions and behavioural disturbances.
Paediatric use. Cyclogyl 1% eye drops should not be used in small infants as concentrations greater than 0.5% are not recommended due to the risk of serious systemic side effects (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects); Section 4.9 Overdose).
Increased susceptibility to cyclopentolate has been reported in infants, young children and in children with Down syndrome, spastic paralysis or brain damage to central nervous system disturbances, and cardiopulmonary and gastrointestinal toxicity from systemic absorption of cyclopentolate. Cyclopentolate should not, therefore, be used in premature and small infants and should be used with great caution in young children and in children with Down syndrome, spastic paralysis or brain damage.
Seizures and acute psychosis induced by cyclopentolate are especially prominent in children. Cyclopentolate should be used with caution in children, with known epilepsy.
Fair-skinned children with blue eyes may exhibit an increased response and/or increased susceptibility to adverse reactions.
Observe infants closely for at least 30 minutes following instillation.
Feeding intolerance and necrotising enterocolitis (NEC) in preterm infants may follow ophthalmic use of this product in infants. Cases of NEC have been reported in preterm infants following administration. It is recommended that feeding be withheld for 4 hours after examination in infants.
Parents should be warned not to get this preparation in their children's mouth or cheeks and to wash their hands and the child's hands or cheeks following administration.
Effects on laboratory tests. No data available.
Information for patients. Nasolacrimal occlusion or gently closing the eyelid after administration is recommended. This may reduce the systemic absorption of medicinal products administered via the ocular route and result in a decrease in systemic adverse reactions.
Parents should be warned not to get this preparation in their child's mouth and to wash their own hands and the child's hands following administration.
After cap is removed, if tamper evident snap collar is loose, remove before using product.
Do not touch the dropper tip to any surface as this may contaminate the solution.
A transient burning sensation may occur upon instillation.
Contact lenses. Cyclogyl eye drops contain benzalkonium chloride which may cause eye irritation and is known to discolour soft contact lenses. Avoid contact with soft contact lenses. Patients must be instructed to remove contact lenses prior to application of Cyclogyl eye drops and wait 15 minutes before reinsertion.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The effects of Cyclogyl eye drops may be enhanced by concomitant use of other drugs having antimuscarinic properties, such as amantadine, some antihistamines, phenothiazine antipsychotics, and tricyclic antidepressants. Cyclopentolate may interfere with the antiglaucoma action of carbachol or pilocarpine; also, concurrent use of this medication may antagonise the anti-glaucoma and miotic actions of ophthalmic cholinesterase inhibitors.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies have not been performed to evaluate the effects of topical ocular administration of cyclopentolate on fertility.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with cyclopentolate. It is not known whether cyclopentolate can cause foetal harm when administered to a pregnant woman or can affect reproductive capacity. Cyclopentolate should be administered to a pregnant woman only if clearly needed.
Use in lactation. It is not known whether this drug is excreted in human milk. A risk to the suckling child cannot be excluded. Because many drugs are excreted in human milk, use only when considered essential by the physician.
4.7 Effects on Ability to Drive and Use Machines
Cyclopentolate eye drops may cause drowsiness and blurred vision. Patients should be advised not to drive or engage in other hazardous activities while pupils are dilated, unless vision is clear. Patients may experience sensitivity to light and should protect eyes in bright illumination during dilation.
4.8 Adverse Effects (Undesirable Effects)
Ocular. Increased intraocular pressure, burning, photophobia, blurred vision, irritation, hyperaemia, conjunctivitis, blepharoconjunctivitis, punctate keratitis, synechiae.
Systemic. Use of cyclopentolate has been associated with psychotic reactions and behavioural disturbances in children. These disturbances include ataxia, incoherent speech, restlessness, hallucinations, hyperactivity, seizures, disorientation as to time and place, and failure to recognise people.
This drug produces reactions similar to those of other anticholinergic drugs, however, the central nervous system manifestations as noted above are more common. Other toxic manifestations of anticholinergic drugs are tachycardia, hyperpyrexia, vasodilation, urinary retention, diminished gastrointestinal motility and decreased secretion in salivary and sweat glands, pharynx, bronchi and nasal passages. Severe manifestations of toxicity include coma, medullary paralysis and death.
Postmarketing events. The following adverse reactions have been reported following administration of Cyclogyl eye drops. Frequency cannot be estimated from the available data. Within each system organ class, adverse reactions are presented in order of decreasing seriousness.
Eye disorders. Not known: photophobia, drug effect prolonged (mydriasis), eye irritation, vision blurred, eye pain, ocular hyperaemia.
Nervous system disorders. Not known: incoherent, retrograde amnesia, dizziness, headache, somnolence.
Psychiatric disorders. Not known: hallucination, confusional state, disorientation, agitation, restlessness.
Immune system disorders. Not known: hypersensitivity.
Gastrointestinal disorders. Not known: vomiting, nausea, dry mouth.
Skin and subcutaneous tissue disorders. Not known: erythema.
General disorders and administration site conditions. Not known: gait disturbance, pyrexia, fatigue.
Description of selected adverse reactions. This drug produces reactions similar to those of other anticholinergic drugs. The central nervous system manifestations such as ataxia, incoherent speech, restlessness, hallucinations, hyperactivity, seizures, disorientation as to time and place, and failure to recognize people are possible. Other toxic manifestations of anticholinergic drugs are skin rash, abdominal distention in infants, unusual drowsiness, tachycardia, hyperpyrexia, vasodilation, urinary retention, diminished gastrointestinal motility, and decreased secretion in salivary and sweat glands, pharynx, bronchi and nasal passages. Severe reactions are manifested by hypotension with rapid progressive respiratory depression.
Cyclogyl eye drops may increase intraocular pressure and provoke glaucoma attacks in patients predisposed to acute angle closure in particular geriatric patients (see Section 4.4 Special Warnings and Precautions for Use).
The onset of cyclopentolate toxicity occurs within 20 to 30 minutes of drug instillation, and although usually transient (subsiding in 4 to 6 hours), the symptoms can last 12 to 24 hours.
Paediatric population. Increased risk for systemic toxicity has been observed in premature and small infants, young children, or children with Down syndrome, spastic paralysis or brain damage with this class of drug (see Section 4.4 Special Warnings and Precautions for Use).
Use of Cyclogyl eye drops has been associated with psychotic reactions and behaviour changes in paediatric patients. Central nervous system reactions manifest similar to those listed above. Seizures and acute psychosis induced by cyclopentolate are especially prominent in children.
Feeding intolerance may follow ophthalmic use of the product in infants (see Section 4.4 Special Warnings and Precautions for Use).
A local or generalized allergic-type response to cyclopentolate consisting of an urticarial rash has been described in children.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
An ocular overdose of Cyclogyl eye drops can be flushed from the eye(s) with lukewarm water.
Excessive dosage may produce exaggerated symptoms as noted in Section 4.8 Adverse Effects (Undesirable Effects).
Systemic toxicity may occur following topical use, particularly in children. It is manifested by flushing and dryness of the skin (a rash may be present in children), blurred vision, a rapid and irregular pulse, fever, abdominal distension in infants, convulsions and hallucinations and the loss of neuromuscular coordination. Severe intoxication is characterized by central nervous system depression, coma, circulatory and respiratory failure, and death. Treatment is symptomatic and supportive. In infants and small children the body surface must be kept moist.
In cases of suspected overdose the first action should be to discontinue administration of the drug. In case of severe manifestations of toxicity the antidote of choice is physostigmine salicylate.
Paediatric dose. Slowly inject 0.5 mg physostigmine salicylate intravenously. If toxic symptoms persist and no cholinergic symptoms are produced, repeat at five minutes intervals to a maximum of 2.0 mg.
Adolescent and adult. Slowly inject 2.0 mg physostigmine salicylate intravenously. A second dose of 1-2 mg may be given after 20 minutes if no reversal of toxic manifestations has occurred. Physostigmine salicylate can be administered subcutaneously.1,2,3
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the accommodative and paralysis of muscle of the ciliary body to cholinergic stimulation, producing pupillary dilation (mydriasis) accommodation (cycloplegia). It acts rapidly, but has a shorter duration than atropine.
Clinical trials. No data are available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. Studies in animal or humans have not been conducted to evaluate the potential of these effects.
Carcinogenicity. Studies in animal or humans have not been conducted to evaluate the potential of these effects.
6 Pharmaceutical Particulars
6.1 List of Excipients
Benzalkonium chloride, boric acid, disodium edetate, potassium chloride, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
18 months. Discard 4 weeks after opening.
6.4 Special Precautions for Storage
Store at 2 to 8°C. (Refrigerate. Do not freeze.)
6.5 Nature and Contents of Container
Cyclogyl Eye Drops are supplied in multi-dose, red LDPE bottles with natural LDPE Drop-Tainer dispensers, 15 mL.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
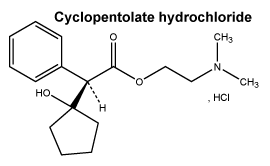
Molecular weight. 327.85.
Chemical name. 2-dimethylaminoethyl-(RS)- 2-(1-hydroxycyclopentyl)- 2-phenylacetate hydrochloride.
CAS number. 5870-29-1.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Date of First Approval
15 October 1991
Date of Revision
15 August 2023
Summary Table of Changes

References
1. Rumack, B.H.: Anticholinergic Poisoning: Treatment with Physostigmine. Pediatrics 52(6): 499-51; 1973.
2. Duvoisin, R.C. and Katz R.: Reversal of Central Anticholinergic Syndrome in Man by Physostigmine. J.Am.Med.Assn. 206(9): 1963-65; 1968.
3. Grant W.M: Toxicology of the Eye. Second Edition, Volume 1. Springfield, Illinois, Charles C. Thomas: 1974.
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.