Cyproterone Sandoz
Brand Information
| Brand name | Cyproterone Sandoz |
| Active ingredient | Cyproterone acetate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Cyproterone Sandoz.
Summary CMI
Cyproterone Sandoz®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Cyproterone Sandoz?
Cyproterone Sandoz contains the active ingredient cyproterone acetate. In men, Cyproterone Sandoz is used for reduction of sex drive in cases of sexual deviations in men, as well as for antiandrogen treatment in inoperable prostate cancer. In women, Cyproterone Sandoz is used to slow or stop moderately severe to severe signs of androgenisation such as excessive hairiness, loss of scalp hair, acne, oily skin and dandruff.
For more information, see Section 1. Why am I using Cyproterone Sandoz? in the full CMI.
2. What should I know before I take Cyproterone Sandoz?
Do not use if you have ever had an allergic reaction to cyproterone acetate or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take Cyproterone Sandoz? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Cyproterone Sandoz and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take Cyproterone Sandoz?
In men, for prostate cancer, the usual daily dose is 50-300 mg of Cyproterone Sandoz. In women, the dose is 50-100 mg of Cyproterone Sandoz daily from the 1st to the 10th day of the cycle. Your doctor will advise of the most suitable dose for you to take. Follow all directions given to you by your doctor or pharmacist carefully.
More instructions can be found in Section 4. How do I take Cyproterone Sandoz? in the full CMI.
5. What should I know while taking Cyproterone Sandoz?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking Cyproterone Sandoz? in the full CMI.
6. Are there any side effects?
The serious side effects include yellowing of the skin and/or eyes, light coloured bowel motions, dark coloured urine, severe upper abdominal pain, vomiting blood or material that looks like coffee grounds, bleeding from the back passage, black sticky bowel motions (stools) or bloody diarrhoea, sudden severe headache, loss of vision, loss of coordination, slurred speech, shortness of breath, chest pain, numbness, heat or swelling in the arms and legs, hearing loss, memory loss, seizures and weakness in the arms or legs.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Cyproterone Sandoz®
Active ingredient(s): cyproterone acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Cyproterone Sandoz. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Cyproterone Sandoz.
Where to find information in this leaflet:
1. Why am I using Cyproterone Sandoz?
2. What should I know before I take Cyproterone Sandoz?
3. What if I am taking other medicines?
4. How do I take Cyproterone Sandoz?
5. What should I know while taking Cyproterone Sandoz?
6. Are there any side effects?
7. Product details
1. Why am I using Cyproterone Sandoz?
Cyproterone Sandoz contains the active ingredient cyproterone acetate. Cyproterone Sandoz is an antiandrogen medicine. It works by blocking the effect of androgens, which are natural sex hormones produced mainly in males but are also produced, to a slight extent, in females.
In men, androgens may help cancer cells to grow in some types of prostate cancer. By blocking these hormones, Cyproterone Sandoz may slow or stop the growth of cancer. Cyproterone Sandoz may also be used in combination with other medicines or, following surgical removal of the testes, to treat side effects such as “hot flushes” or “sweats” and to prevent any initial worsening of the disease.
Cyproterone Sandoz is also used to reduce abnormal sex drive in men.
In women, androgens may increase hair growth, loss of scalp hair and secretion of oil from the sweat glands. By blocking these hormones, Cyproterone Sandoz may slow or stop excessive hairiness, loss of scalp hair, acne, oily skin and dandruff.
2. What should I know before I take Cyproterone Sandoz?
Warnings
Do not use Cyproterone Sandoz if:
- you are allergic to cyproterone acetate, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- Do not use Cyproterone Sandoz if you have any of the following medical conditions:
- liver disease, previous or existing liver tumours unless they are caused by metastases from prostate cancer (your doctor would have told you if you have this)
- Dubin-Johnson or Rotor syndrome (your doctor would have told you if you have either of these conditions)
- history of jaundice (yellow skin or eyes), herpes or persistent itching during a previous pregnancy
- previous or existing benign brain tumour (meningioma)
- wasting disease (a disease-causing muscle loss or loss of strength, with the exception of prostate cancer)
- severe and persistent depression
- previous or existing conditions relating to formation of blood clots
- severe diabetes with blood vessel changes (your doctor would have told you if you have this)
- sickle-cell anaemia (your doctor would have told you if you have this)
Check with your doctor if you:
- have any other medical conditions, especially following:
- diabetes
- history of blood clotting or sickle cell anaemia
- osteoporosis, a family history of osteoporosis or risk factors for developing osteoporosis (such as smoking, a diet low in calcium, poor mobility, a slight build or treatment with steroid medicines) - take any medicines for any other condition
- have an intolerance to some sugars, Cyproterone Sandoz tablets contains lactose monohydrate
- have any allergies to any other medicines, foods, preservative or dyes
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
If taken during pregnancy, Cyproterone Sandoz may lead to signs of feminisation in the male foetus. Therefore, your doctor will check that you are not pregnant before you start taking Cyproterone Sandoz. Women should use a reliable form of contraception while taking Cyproterone Sandoz.
Do not take this medicine if you are pregnant or suspect you may be pregnant.
It may affect your developing baby if you take it during pregnancy.
Do not breastfeed if you are taking this medicine.
The active ingredient in Cyproterone Sandoz passes into breast milk and there is a possibility that your baby may be affected.
Tell your doctor if fertility after treatment is important
- For men it is recommended that a sperm count is taken to establish fertility before commencing Cyproterone Sandoz. It can take 3-20 months for fertile sperm production to be re-established after stopping this medicine.
- The long-term effects of Cyproterone Sandoz on female fertility are not known.
Cyproterone Sandoz should not be taken by children and adolescents below 18 years of age or girls who have not completed puberty.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Cyproterone Sandoz and affect how it works.
- phenytoin, a medicine used to treat epilepsy
- medicines used to treat fungal infections, including ketoconazole, itraconazole, clotrimazole
- ritonavir, a medicine used in the treatment of HIV
- rifampicin, an antibiotic used to treat infections such as tuberculosis and leprosy
- St John's wort, a herbal remedy used to treat mood disorders
- statins (HMGCoA inhibitors), medicines used to lower cholesterol levels in people with or at risk of cardiovascular disease
- medicines used to treat diabetes
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Cyproterone Sandoz.
4. How do I take Cyproterone Sandoz?
How much to take
Follow the instructions provided and take Cyproterone Sandoz until your doctor tells you to stop.
MEN:
- For Prostate cancer, the dose is 50-300 mg of Cyproterone Sandoz. Your doctor may request you take Cyproterone Sandoz with other medicines and/or change your dose during treatment.
- For Reduction of abnormal sex drive, generally treatment is started with 50 mg of Cyproterone Sandoz twice daily and may be increased to 100 mg twice daily or three times daily before reducing gradually to the lowest effective dose. Your doctor may change your dose during treatment.
WOMEN:
- If you are of childbearing age, you should commence your tablet taking on the 1st day of your cycle (= 1st day of bleeding). If you have no menstrual periods (amenorrhoea) your treatment can start immediately. In this case, the first day of treatment is to be regarded as the 1st day of the cycle.
- Starting from day 1 take 50-100 mg (as advised by your doctor) of Cyproterone Sandoz daily from the 1st to the 10th day of the cycle (= for 10 days). Additionally, your doctor will advise the most appropriate contraceptive for you to take to provide the necessary contraceptive protection and to stabilise your cycle.
- Your doctor will re-evaluate your treatment when you reach menopause. Long-term use (years) of Cyproterone Sandoz should be avoided.
- If you are postmenopausal or have had a hysterectomy, Cyproterone Sandoz may be administered alone. The usual dose is 25-50 mg of Cyproterone Sandoz once daily for 21 days, followed by a 7-day tablet-free interval.
- Shortness of breath may occur at high doses.
Cyproterone Sandoz helps to control your condition but does not cure it. It is important to keep taking your medicine even if you feel well.
When to take Cyproterone Sandoz
- Cyproterone Sandoz should be taken whole, with some liquid after meals.
- Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
Missed Cyproterone Sandoz tablets may diminish the effectiveness of treatment and may lead to breakthrough bleeding in women.
If you forget to use Cyproterone Sandoz
- If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
- Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting an unwanted side effect.
- If you are also taking an oral contraceptive and more than 12 hours has elapsed from the time Cyproterone Sandoz was due to be taken, note that contraceptive protection in this cycle may be reduced and thus there is an increased risk of becoming pregnant.
If you use too much Cyproterone Sandoz
If you think that you have used too much Cyproterone Sandoz, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking Cyproterone Sandoz?
Things you should do
- Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
- Your doctor will check your liver function during treatment with Cyproterone Sandoz and whenever any symptoms or signs suggesting liver problems are observed.
- If you have diabetes, your doctor will monitor you to ensure that you receive the appropriate dose of oral antidiabetic or insulin whilst taking Cyproterone Sandoz.
- Your doctor will also check your red-blood cell count to ensure you do not become anaemic during treatment with Cyproterone Sandoz.
- If you are a female taking an oral contraceptive during treatment, tell your doctor if your period does not occur during the tablet-free / placebo interval. Your doctor may need to check whether you are pregnant before you can continue treatment.
- If you are a male taking Cyproterone Sandoz to reduce abnormal sex drive, you should consider undertaking additional measures such as therapy or counselling in order to take advantage of the period of reduced drive. These measures may assist in achieving personal and social re-orientation.
Remind any doctor, dentist or pharmacist you visit that you are using Cyproterone Sandoz.
Things you should not do
- Do not stop using this medicine suddenly or lower the dosage without checking with your doctor.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Cyproterone Sandoz affects you.
Cyproterone Sandoz may cause tiredness and can impair the ability to concentrate. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol
Tell your doctor if you drink alcohol.
If you drink alcohol, tiredness and the ability to concentrate may be worse.
Alcohol may prevent Cyproterone Sandoz from working as well as it should in reducing abnormal sex drive.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
If you were fertile before treatment, Cyproterone Sandoz will normally prevent sperm production in men and ovulation in women. In men, fertility is usually regained within a few months of discontinuing therapy. The long term effects on female fertility are not known.
In men Cyproterone Sandoz will also normally result in the inability to get or maintain an erection (impotence). This ability is usually also regained within a few months of discontinuing therapy.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Use of cyproterone acetate has been linked to the development of meningioma (generally benign tumour). The risk increases when used for several years, or with high doses (25 mg per day and above).
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Cyproterone Sandoz contains
| Active ingredient (main ingredient) | cyproterone acetate |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | lactose monohydrate |
Do not take this medicine if you are allergic to any of these ingredients.
What Cyproterone Sandoz looks like
Cyproterone Sandoz 50 mg– white to off white, round tablets engraved with ‘50’ over a break line on one face, plain on the other face. Available in bottles of 20 or 50 tablets. (Aust R 107330).
Cyproterone Sandoz 100 mg – white to off white, capsule shaped tablets with ‘100’ engraved on one face and a break line on the other face. Available in blisters of 50 tablets. (Aust R 107331).
Not all pack sizes may be available in Australia.
Who distributes Cyproterone Sandoz
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
This leaflet was prepared in December 2025.
® Registered Trade Mark. The trade marks mentioned in this material are the property of their respective owners.
Brand Information
| Brand name | Cyproterone Sandoz |
| Active ingredient | Cyproterone acetate |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Cyproterone acetate.
2 Qualitative and Quantitative Composition
Each Cyproterone Sandoz 100 mg tablet contains 100 mg cyproterone acetate.
Excipient with known effect. Sugars as lactose monohydrate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Cyproterone Sandoz 100 mg tablets are white to off white, capsule shaped tablet with '100' engraved on one face, and a break line on the other face.
4 Clinical Particulars
4.1 Therapeutic Indications
Inoperable prostatic carcinoma. To suppress flare with initial luteinising hormone releasing hormone (LHRH) analogue therapy; in long-term palliative treatment where LHRH analogues or surgery are ineffective, not tolerated, contraindicated or where oral therapy is preferred; in the treatment of hot flushes in patients treated with LHRH analogues or who have had orchidectomy.
4.2 Dose and Method of Administration
Dosage. Cyproterone Sandoz 100 mg tablets are to be taken with some liquid after meals.
Inoperable prostatic carcinoma. Suppression of flare with initial LHRH analogue therapy. Initially 100 mg twice daily alone for 5-7 days, then 100 mg twice daily for 3-4 weeks together with an LHRH agonist at the dosage recommended by the manufacturer.
Long-term palliative treatment without orchidectomy. 100 mg two or three times daily. Treatment should not be interrupted, nor the dosage reduced, after improvement or remissions have occurred.
Treatment of hot flushes (in patients treated with LHRH analogues or postorchidectomy). Low initial dose of 50 mg once to three times daily, with upward titration to 100 mg three times daily if necessary.
See Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects); Section 4.9 Overdose.
Dosage adjustment. Renal impairment. There are no data suggesting the need for dosage adjustment in patients with renal impairment.
Hepatic impairment. The use of cyproterone acetate is contraindicated in patients with liver diseases.
Paediatric use. Cyproterone acetate is not recommended for use in male children and adolescents below 18 years of age due to a lack of data on safety and efficacy.
Elderly. There are no data suggesting the need for dosage adjustment in elderly patients.
4.3 Contraindications
Liver diseases.
Dubin-Johnson syndrome, Rotor syndrome.
Previous or existing liver tumours (only if these are not due to metastases from carcinoma of the prostate).
Presence or history of meningioma.
Wasting diseases (with the exception of inoperable carcinoma of the prostate).
Severe chronic depression.
Existing thromboembolic processes.
Hypersensitivity to any of the components of Cyproterone Sandoz 100 mg.
4.4 Special Warnings and Precautions for Use
Cyproterone Sandoz 100 mg is for use only in men.
During treatment, liver function, adrenocortical function and red blood cell count should be checked regularly.
As with other antiandrogenic treatments, in male patients long-term androgen deprivation with cyproterone acetate may lead to osteoporosis.
In men of procreative age, for whom fertility could be important after conclusion of the medication, it is advisable to make at least one control spermatogram as a precaution before the start of treatment in order to counter any unjustified claims of later infertility as a result of the antiandrogen therapy. Spermatogenesis has taken 3-20 months to return to normal after discontinuing therapy.
Meningioma. The occurrence of meningiomas (single and multiple) has been reported in association with long-term use (years) of cyproterone acetate at doses of 25 mg/day and above. The risk of meningioma increases with increasing cumulative doses of cyproterone acetate. If a patient treated with cyproterone is diagnosed with meningioma, treatment with cyproterone containing products, including Cyproterone Sandoz must be permanently stopped (see Section 4.3 Contraindications).
Diabetes. Strict medical supervision is necessary if the patient suffers from diabetes, because the requirement for oral antidiabetics or insulin can change during cyproterone acetate treatment (see Section 4.3 Contraindications).
Shortness of breath. A sensation of shortness of breath may occur under high-dosed treatment with cyproterone acetate. The differential diagnosis in such cases must include the stimulating effect on breathing known for progesterone and synthetic progestogens which is accompanied by hypocapnia and compensatory alkalosis and which is not considered to require treatment.
Thromboembolic events. The occurrence of thromboembolic events has been reported in patients using cyproterone acetate although a causal relationship has not been established. Patients with previous arterial or venous thrombotic/thromboembolic events (e.g. deep venous thrombosis, pulmonary embolism, myocardial infarction) or with a history of cerebrovascular accidents or with advanced malignancies are at increased risk of further thromboembolic events.
In patients with a history of thromboembolic processes or suffering from sickle-cell anaemia or from severe diabetes with vascular changes, a careful risk:benefit evaluation must be carried out in each individual case before cyproterone acetate is prescribed.
Adrenocortical function. During treatment, adrenocortical function should be checked regularly, as preclinical data suggest a possible suppression due to the corticoid-like effect of cyproterone acetate with high doses.
Anaemia. Anaemia has been reported during treatment with cyproterone acetate. Therefore, the red-blood cell count should be checked regularly during treatment.
Other conditions. Cyproterone Sandoz tablets contain lactose. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose mal-absorption should not take this medicine.
The sexual drive-reducing effect of cyproterone acetate can be diminished under the influence of alcohol.
Use in hepatic impairment. Direct hepatic toxicity, including jaundice, hepatitis and hepatic failure, has been observed in patients treated with cyproterone acetate. At dosages of 100 mg and above, cases with fatal outcome have been reported. Most reported fatal cases were in men with advanced carcinoma of the prostate. Toxicity is dose-related and develops, usually, several months after treatment has begun. Liver function tests should be performed pre-treatment at regular intervals during treatment and whenever any symptoms or signs suggestive of hepatotoxicity occur. If hepatotoxicity is confirmed, cyproterone acetate should be withdrawn, unless hepatotoxicity can be explained by another cause, e.g. metastatic disease, in which case cyproterone acetate should be continued only if the perceived benefit outweighs the risk.
Cases of benign and malignant liver tumours, which may lead to life-threatening intra-abdominal haemorrhage, have been observed after the cyproterone acetate use. If severe upper abdominal complaints, liver enlargement or signs of intra-abdominal haemorrhage occur, a liver tumour should be included in the differential-diagnostic considerations and, if necessary, discontinuation of the preparation considered.
Use in the elderly. See Section 4.2 Dose and Method of Administration.
Paediatric use. See Section 4.2 Dose and Method of Administration, Paediatric use.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The requirement for oral antidiabetics or insulin can change.
Although clinical interaction studies have not been performed, since this drug is metabolized by CYP3A4, it is expected that ketoconazole, itraconazole, clotrimazole, ritonavir and other strong inhibitors of CYP3A4 inhibit the metabolism of cyproterone acetate. On the other hand, inducers of CYP3A4 such as e.g. rifampicin, phenytoin and products containing St. John's wort (Hypericum perforatum) may reduce levels of cyproterone acetate.
The risk of statin associated myopathy or rhabdomyolysis may be increased when those HMG-CoA inhibitors (statins), which are primarily metabolised by CYP3A4, are coadministered with high therapeutic cyproterone acetate doses since they share the same metabolic pathway.
Based on in vitro CYP450 studies, the recommended clinical doses are likely to inhibit CYP2C8, and an inhibition of the CYP2C9, 2C19, 3A4, and 2D6 is also possible at high therapeutic cyproterone acetate doses of 3 times 100 mg per day.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. See Section 4.4 Special Warnings and Precautions for Use; Section 5.1 Pharmacodynamic Properties.
Use in pregnancy. (Category D)
Cyproterone Sandoz 100 mg is for use only in men.
Use in lactation. No data available.
4.7 Effects on Ability to Drive and Use Machines
It should be pointed out to patients whose occupation demands great concentration (e.g. road users, machine operators) that Cyproterone Sandoz 100 mg can lead to tiredness and diminished vitality and can impair the ability to concentrate (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.8 Adverse Effects (Undesirable Effects)
Adverse effects reported in clinical trials. The following adverse effects have been reported at the approximate frequencies (not necessarily implicating a causal relationship) indicated below.
Very common ≥ 1/10; common ≥ 1/100 and < 1/10; uncommon ≥ 1/1,000 and < 1/100; rare ≥ 1/10,000 and < 1/1,000; very rare < 1/10,000.
General. Very common: tiredness, weight increase. Common: headache, depressive moods.
Cardiovascular. Common: thrombotic phenomena.
Gastrointestinal. Common: nausea and other gastrointestinal complaints.
Reproductive. Very common: diminished libido. Common: mastodynia.
Skin. Rare: rash.
The most frequently observed ADRs in patients receiving cyproterone acetate are decreased libido, erectile dysfunction and reversible inhibition of spermatogenesis.
The most serious ADRs in patients receiving cyproterone acetate are hepatic toxicity, benign and malignant liver tumours which may lead to intra-abdominal haemorrhage, and thromboembolic events.
Over the course of several weeks, cyproterone acetate 100 mg gradually impairs spermatogenesis as a result of the antiandrogenic and antigonadotropic actions. Spermatogenesis recovers gradually within several months of discontinuing therapy.
Cyproterone acetate may lead to gynaecomastia (sometimes combined with tenderness to touch of the breast) which usually regresses after withdrawal of the preparation or reduction of the dose.
As with other antiandrogenic treatments, in male patients long-term androgen deprivation with cyproterone acetate may lead to osteoporosis.
In individual cases, disturbances of liver function, some of them severe, have been reported with high dosed cyproterone acetate treatment.
Changes in body weight are possible.
Other adverse events reported at a low incidence are skin discolouration and striae.
Post-marketing information. The following adverse effects have been reported in users of cyproterone acetate (post-marketing data) and are based on post-marketing data and cumulative experience with cyproterone acetate. The most appropriate MedDRA term to describe a certain adverse reaction is listed. Synonyms or related conditions are not listed, but should be taken into account as well.
Very common ≥ 1/10; common ≥ 1/100 and < 1/10; uncommon ≥ 1/1,000 and < 1/100; rare ≥ 1/10,000 and < 1/1,000; very rare < 1/10,000.
General disorders and administration site conditions. Common: fatigue, hot flushes, sweating. Very rare: tiredness, sleep disturbances.
Cardiovascular. Very rare: thrombotic phenomena, tachycardia.
Gastrointestinal. Very rare: nausea, gastrointestinal complaints.
Hepatobiliary. Common: hepatic toxicity, including jaundice, hepatitis, hepatic failure. Rare: increased liver enzymes. Very rare: liver function disturbance.
Reproductive system and breast disorders. Very common: reversible inhibition of spermatogenesis. Common: gynaecomastia. Very rare: breast tenderness, breast pain.
Skin and subcutaneous tissue disorders. Uncommon: rash.
Musculoskeletal and connective tissue disorders. Very rare: osteoporosis.
Immune system disorders. Rare: hypersensitivity reaction.
Metabolism and nutrition disorders. Common: weight increased or weight decreased.
Psychiatric disorders. Very common: libido decreased, erectile dysfunction. Common: depressed mood, restlessness (temporary).
Neoplasms benign and malignant. Very rare: benign and malignant liver tumours*.
Respiratory, thoracic and mediastinal disorders. Common: shortness of breath*.
The ADRs identified only during postmarketing surveillance and for which a frequency could not be estimated are anaemia*, meningioma, intra-abdominal haemorrhage*, thromboembolic events*+.
Under treatment with Cyproterone Sandoz, sexual drive and potency are reduced and gonadal function is inhibited. These changes are reversible after discontinuation of therapy.
Meningiomas have been reported in association with long-term use (several years) of Cyproterone Sandoz doses of 25 mg and above (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
* For further information see Section 4.4 Special Warnings and Precautions for Use.
+ A causal relationship with Androcur has not been established.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is no clinical experience in overdose. Assessment and symptomatic treatment should be initiated as required.
Use of Cyproterone Sandoz 100 mg at high doses has been associated with hepatic toxicity, particularly in the elderly (see Section 4.8 Adverse Effects (Undesirable Effects)).
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Cyproterone acetate is an antiandrogenic hormone preparation.
Cyproterone acetate inhibits competitively the effect of androgens at androgen-dependent target organs, e.g. it shields the prostate from the effect of androgens originating from the gonads and/or the adrenal cortex. Prostatic carcinoma and its metastases are in general androgen dependent, cyproterone acetate therefore exerts a direct antiandrogenic action on the tumour and its metastases.
Cyproterone acetate in addition has a progestogenic action exerting a negative feedback effect centrally on the hypothalamic receptors, so leading to a reduction in gonadotropin release, and hence to diminished production of testicular androgens. Treatment with cyproterone acetate in men results in a reduction of sexual drive and potency and inhibition of gonadal function. These changes are reversible following discontinuation of the therapy.
The antigonadotropic effect of cyproterone acetate is also exerted when the substance is combined with LHRH agonists. The initial increase of testosterone provoked by this substance group is decreased by cyproterone acetate.
Prolactin levels can increase slightly under higher doses of CPA. Studies showed increased prolactin levels increased up to 20 nanogram/mL (normal range 5-15 nanogram/mL). There are no data for periods longer than 6 months.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Following oral administration, cyproterone acetate is completely absorbed over a wide dose range.
The ingestion of 100 mg of cyproterone acetate gives maximum serum levels of 239.2 ± 114.2 nanogram/mL at 2.8 ± 1.1 hours. Thereafter, drug serum levels declined during a time interval of typically 24 to 120 hours, with a terminal half-life of 42.8 ± 9.7 h. The total clearance of cyproterone acetate from serum was determined to be 3.8 ± 2.2 mL/min/kg. The absolute bioavailability of cyproterone acetate is unknown. Relative bioavailability was calculated, in a study of eight young women, from a dose-corrected comparison of area under the curves of serum levels after 100 mg oral and 300 mg intramuscular depot administration and was found to be 80 ± 30% when averaged over all volunteers (range 23%-119%).
Distribution. The major part of circulating cyproterone acetate is bound to serum albumin. In a study in 15 women receiving 2 mg cyproterone acetate in combination with 35 microgram ethinyloestradiol, the free fraction of cyproterone acetate was about 3.5-4%. Because protein binding is non-specific, changes in SHBG (sex hormone binding globulin) levels do not affect the pharmacokinetics of cyproterone acetate.
Metabolism. Cyproterone acetate is metabolised by various pathways, including hydroxylations and glucuronide conjugations. The main metabolite in human plasma is the 15β-hydroxy derivative. Some dose parts are excreted unchanged with bile fluid. Phase I metabolism of cyproterone acetate is mainly catalysed by the CYP450 enzyme CYP3A4.
Excretion. In a study in 6 women administered a 14C labelled dose of 2 mg cyproterone acetate in combination with a 50 microgram oestrogen, approximately 30% of the label was found in the urine and 58% in the faeces. The renal and biliary excretion was determined to proceed with a half-life of 1.9 days. Metabolites from plasma were eliminated at a similar rate (half-life of 1.7 days).
Steady state conditions. According to the long half-life of the terminal disposition phase from plasma (serum) and the daily intake, an accumulation of cyproterone acetate in the serum by a factor of about 3 can be expected during repeated daily administration.
5.3 Preclinical Safety Data
Genotoxicity. Cyproterone acetate (CPA) was negative in a standard battery of genotoxicity studies. However, further tests showed that CPA was capable of producing hepatocyte DNA adducts in rats, dogs and monkeys (and an increase in DNA repair activity in rats) in vivo, and also in freshly isolated rat and human liver cells in vitro. This DNA adduct formation occurred at exposures that might be expected to occur in the recommended dose regimens for Cyproterone Sandoz 100 mg. In vivo consequences of CPA treatment were the increased incidence of focal, possibly preneoplastic, liver lesions in which cellular enzymes were altered in female rats, and an increase of mutation frequency in transgenic rats carrying a bacterial gene as target for mutation. The clinical relevance of these findings presently remains uncertain.
Carcinogenicity. Long-term animal carcinogenicity studies were performed in rats and mice. In one rat study, an increased incidence of hepatomas was reported at oral dose levels of 50 mg/kg CPA and above. In mouse (and a second rat) carcinogenicity studies, increases in benign proliferative changes (nodular hyperplasia) in liver cells of female mice and male and female rats were reported at oral doses of 2 mg/kg. Because of shortcomings in these studies (inadequate pharmacokinetic data and the need to reassess the liver pathology), the carcinogenic potential of CPA in animals could not be determined.
Clinical experience and limited epidemiological data available to date do not appear to have supported an increased incidence of hepatic tumours in humans. However it must be borne in mind that steroidal sex hormones can promote the growth of certain hormone dependent tissues and tumours.
6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, povidone and magnesium stearate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information on interactions with other medicines and other forms of interactions, see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
Protect from light and moisture.
6.5 Nature and Contents of Container
Cyproterone Sandoz 100 mg tablets is available in (PVC/PVDC/Al) blister packs of 50 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Cyproterone acetate is a white to pale yellow crystalline powder. Melting point: 206 to 213°C. It is very soluble in chloroform and dioxane, freely soluble in acetone and benzene, soluble in ethanol, methanol and ethyl acetate, sparingly soluble in solvent hexane and almost insoluble in water.
Chemical structure.
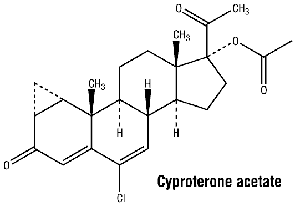
Empirical formula: C24H29ClO4.
Molecular weight: 416.96.
CAS number. 427-51-0.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
15 November 2004
Date of Revision
23 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.