Dalacin C Injection
Brand Information
| Brand name | Dalacin C Injection |
| Active ingredient | Clindamycin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Dalacin C Injection.
Summary CMI
DALACIN® C Injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about receiving this medicine, speak to your doctor or nurse.
1. Why am I being treated with Dalacin C injection?
Dalacin C injection contains the active ingredient clindamycin phosphate. Dalacin C injection is used to treat infections in different parts of the body caused by bacteria.
For more information, see Section 1. Why am I being treated with Dalacin C injection? in the full CMI.
2. What should I know before treatment with Dalacin C injection?
You should not be treated with Dalacin C Injection if you have ever had an allergic reaction to Dalacin C injection, clindamycin, lincomycin, or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have liver, kidney, bowel, stomach or gut problems, or have had severe diarrhoea associated with the use of antibiotics, allergy to latex or history of allergy to different things, or any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before treatment with Dalacin C injection? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Dalacin C injection and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Dalacin C injection given?
Your doctor will decide what dose you will receive. Dalacin C Injection is given as an injection into a muscle or as a slow drip into a vein.
More instructions can be found in Section 4. How is Dalacin C injection given? in the full CMI.
5. What should I know during or after treatment with Dalacin C injection?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
For more information, see Section 5. What should I know during or after treatment with Dalacin C injection? in the full CMI.
6. Are there any side effects?
Side effects may include discomforts at injection site; pain/stiffness/swelling in joints; skin rash; stomach ache; vomiting; nausea; change/loss of taste; vaginal infection; red rash on folds of the buttocks, groin, armpit or behind the knee.
Serious side effects may include low blood pressure; severe diarrhoea which may have blood and mucus; weight loss and fevers, night sweats, fatigue, cough, pain, weakness, confusion; severe chills and sore throat or mouth ulcers; bleeding/bruising more easily than normal; signs of allergy on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or trouble breathing; little or no urine; yellowing of the eyes or skin; severe swelling and redness along a vein; chest and heart discomfort, fast/pounding/irregular heartbeat.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
DALACIN® C Injection
Active ingredient(s): [Clindamycin phosphate]
Consumer Medicine Information (CMI)
This leaflet provides important information about Dalacin C injection. You should also speak to your doctor or nurse if you would like further information or if you have any concerns or questions about using Dalacin C injection.
Where to find information in this leaflet:
1. Why am I being treated with Dalacin C injection?
2. What should I know before treatment with Dalacin C injection?
3. What if I am taking other medicines?
4. How is Dalacin C injection given?
5. What should I know during or after treatment with Dalacin C injection?
6. Are there any side effects?
7. Product details
1. Why am I being treated with Dalacin C injection?
Dalacin C injection contains the active ingredient clindamycin phosphate. Dalacin C injection is an antibiotic. It is used to treat infection in different parts of the body caused by bacteria.
This medicine works by killing or stopping the growth of the bacteria causing your infection.
It will not work against viral infections such as colds or flu.
Dalacin C injection is recommended for patients who are allergic to penicillin or for whom penicillin is not suitable.
Ask your doctor if you have any questions about why Dalacin C injection has been prescribed for you. Your doctor may have prescribed Dalacin C injection for another reason.
2. What should I know before treatment with Dalacin C injection?
Warnings
You should not be given Dalacin C Injection if you have ever had an allergic reaction to clindamycin, lincomycin or any of the ingredients listed at the end of this leaflet.
Tell your doctor if you:
- have had severe diarrhoea associated with the use of antibiotics
- have liver disease
- have kidney disease
- have or have a history of bowel disease or any gastrointestinal (stomach or gut) problems
- have a history of allergic reactions to different things
- take any medicines for any other conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy
Tell your doctor if you are pregnant or intend to become pregnant. Clindamycin and benzyl alcohol contained in Dalacin C injection cross the placenta. Your doctor will discuss the benefits and risks of using Dalacin C injection in pregnancy.
Breastfeeding
Tell your doctor if you are breastfeeding or intend to breastfeed. Dalacin C injection may affect your baby and is not recommended during breastfeeding.
Your doctor can discuss with you the risks and benefits involved.
Use in children
Dalacin C injection contains benzyl alcohol. Benzyl alcohol has been associated with serious side effects in newborns and infants. Your doctor will decide if treatment is appropriate.
Use in the elderly
Patients over 60 years of age are more likely to have severe gastrointestinal effects. It is important to tell your doctor immediately if you suffer from severe stomach pain or diarrhoea which may also involve blood in your stool (poo). This may occur more than 2 months after treatment with Dalacin C injection is stopped.
3. What if I am taking other medicines?
Tell your doctor or nurse if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Dalacin C injection and affect how it works including:
- erythromycin, ciprofloxacin, clarithromycin, rifampicin, rifaximin - used to treat bacterial infections
- fluconazole, itraconazole, ketoconazole, voriconazole - used to treat fungal infections
- carbamazepine, phenobarbital, phenytoin, primidone - used to manage seizures
- imatinib, enzalutamide, apalutamide, midostaurin - used to treat certain cancers
- aprepitant - used to treat nausea and vomiting associated with cancer treatment
- lumacaftor - used for the treatment of cystic fibrosis
- St. John's wort - used to assist emotional health
- dexamethasone - used to treat various inflammatory, endocrine or rheumatic conditions
- diltiazem, verapamil - used to treat angina or high blood pressure
- ritonavir - used to treat HIV/AIDS
- some medicines that may be used during surgery in an intensive care setting.
Check with your doctor or nurse if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Dalacin C injection.
4. How is Dalacin C injection given?
How much Dalacin C injection is given
Your doctor will decide the dose, how often, and how long it should be given according to your age, condition and body weight.
How is Dalacin C injection given
Dalacin C injection may be given as an intravenous infusion (as a slow drip into a vein) or as an injection into a muscle. It is given by a doctor or trained nurse.
If you receive too much Dalacin C injection
As Dalacin C injection is usually given to you in hospital under the close supervision of your doctor, it is unlikely that you will receive too much. However, if you experience severe side effects after being given Dalacin C injection, including severe diarrhoea which may include blood, severe stomach pain, feeling dizzy or faint, unusual heartbeat or chest pain tell your doctor or nurse immediately.
Tell your doctor or nurse immediately if you are concerned that you may have been given too much Dalacin C injection.
5. What should I know during or after treatment with Dalacin C injection?
Things you should do
Tell your doctor or nurse immediately while receiving or after Dalacin C injection has been stopped as you may need urgent medical care:
- If you develop severe stomach pain or severe diarrhoea. Do this even if it occurs several weeks after Dalacin C injection has been stopped. Diarrhoea may mean that you have a serious condition affecting your bowel.
- If you get a severe skin rash. A severe skin rash may mean you are having an allergic reaction to Dalacin C injection.
- If you have chest pain, shortness of breath, nausea and vomiting, sweating and fast or irregular heartbeats. This may mean you are having an allergic reaction to Dalacin C injection.
- If you feel lightheaded or dizzy.
Tell your doctor:
- If you get a sore, white mouth or tongue, vaginal itching or discharge. This may mean you have a fungal/yeast infection called thrush. Sometimes the use of Dalacin C injection allows fungi/yeast to grow and the above symptoms to occur. Dalacin C does not work against fungi/yeast.
- If the symptoms of your infection do not improve within a few days, or if they become worse.
- If you are about to have any blood tests. Dalacin C injection may interfere with the results of some tests.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you should not do
- Do not stop receiving Dalacin C injection because you are feeling better, unless advised by your doctor.
- Do not take any medicines for diarrhoea without first checking with your doctor.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Dalacin C affects you.
Drinking alcohol
No information available.
Looking after your medicine
Dalacin C injection will be stored in the hospital pharmacy or on the ward.
Disposal of the medicine
The hospital staff will dispose of any leftover Dalacin C injection.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Side effects
| Side effects | What to do |
| Speak to your doctor or nurse if you have any of these side effects and they worry you. |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Rare instances following too rapid intravenous administration
| Serious side effects | What to do |
| Tell your doctor or nurse straight away. |
Tell your doctor or nurse if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/safety/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or nurse or pharmacist before you decide to stop receiving/taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Dalacin C injection contains
| Active ingredient (main ingredient) | clindamycin phosphate |
| Other ingredients (inactive ingredients) | benzyl alcohol disodium edetate hydrochloric acid sodium hydroxide water for injections |
Tell your doctor or nurse if you are allergic to any of these ingredients.
What Dalacin C injection looks like
Dalacin C injection appears as a clear colorless solution and comes in 2 mL or 4 mL ampoules
AUST R 12294 DALACIN C clindamycin 300mg/2mL (as phosphate) injection ampoule
AUST R 47636 DALACIN C clindamycin 600mg/4mL (as phosphate) injection ampoule
Who distributes Dalacin C injection
Pfizer Australia Pty Ltd
Level 17, 151 Clarence Street SYDNEY NSW 2000
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in December 2025.
®Registered Trademark
© Pfizer
Brand Information
| Brand name | Dalacin C Injection |
| Active ingredient | Clindamycin |
| Schedule | S4 |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Clindamycin phosphate.
2 Qualitative and Quantitative Composition
Each 1 mL of Dalacin C contains clindamycin phosphate equivalent to 150 mg clindamycin base; 9.45 mg benzyl alcohol; 0.5 mg disodium edetate; and water for injections q.s. When necessary, pH is adjusted with sodium hydroxide and/or hydrochloric acid.
Clindamycin is a semi synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent compound lincomycin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for Injection.
Dalacin C (clindamycin phosphate injection) is a sterile solution of a water soluble ester of clindamycin and phosphoric acid.
4 Clinical Particulars
4.1 Therapeutic Indications
Dalacin C is indicated in the treatment of serious infections caused by susceptible anaerobic bacteria.
Dalacin C is also indicated in the treatment of serious infections due to susceptible strains of Streptococci, Pneumococci and Staphylococci.
Its use should be reserved for penicillin allergic patients or other patients for whom, in the judgement of the physician, a penicillin is inappropriate.
Anaerobes. Serious respiratory tract infections such as empyema, anaerobic pneumonitis and lung abscess; serious skin and skin structure infections; septicaemia; intra-abdominal infections such as peritonitis and intra-abdominal abscess (typically resulting from anaerobic organisms resident in the normal gastrointestinal tract) and infections of the female pelvis and genital tract such as endometritis, non-gonococcal tubo-ovarian abscess, pelvic cellulitis and post-surgical vaginal cuff infection, all when given in conjunction with an antibiotic of appropriate Gram negative aerobic spectrum.
Streptococci. Serious respiratory tract infections; serious skin and skin structure infections; septicaemia.
Staphylococci. Serious respiratory tract infections; serious skin and skin structure infections; septicaemia; acute haematogenous osteomyelitis.
Pneumococci. Serious respiratory tract infections.
Adjunctive therapy. In the surgical treatment of chronic bone and joint infections due to susceptible organisms.
Indicated surgical procedures should be performed in conjunction with antibiotic therapy. Bacteriological studies should be performed to determine the causative organisms and their susceptibility to clindamycin.
4.2 Dose and Method of Administration
For Intramuscular and Intravenous Use.
Dosage and route of administration should be determined by the severity of the infection, the condition of the patient and the susceptibility of the causative microorganism.
Dalacin C IM administration should be used undiluted.
Dalacin C IV administration should be diluted (see Dilution for IV use and IV infusion rates below).
Adults (IM or IV administration). The usual daily adult dosage of clindamycin phosphate for infections of the intra-abdominal area, female pelvis and other complicated or serious infections is 1200-2700 mg given in 2, 3 or 4 equal doses. Doses of up to 4800 mg daily have been used successfully. Less complicated infections due to more susceptible organisms may respond to lower doses such as 600-1200 mg/day administered in 3 or 4 equal doses.
Single IM doses of greater than 600 mg are not recommended.
Children over one month of age (IM or IV administration). Clindamycin should be dosed based on total body weight regardless of obesity.
Serious infections. 15-25 mg/kg/day in 3 or 4 equal doses.
More severe infections. 25-40 mg/kg/day in 3 or 4 equal doses.
As an alternative to dosing on a bodyweight basis, children may be dosed on the basis of square metres of body surface.
Serious infections. 350 mg/m2/day.
More severe infections. 450 mg/m2/day.
In severe infections it is recommended that children be given no less than 300 mg per day regardless of bodyweight.
Parenteral therapy may be changed to oral clindamycin (Dalacin C Capsules) when the condition warrants and at the discretion of the physician.
In cases of β-haemolytic streptococcal infections, treatment should be continued for at least 10 days.
Dilution for IV use and IV infusion rates. Dalacin C must be diluted prior to IV administration. The concentration of clindamycin in diluent for infusion should not exceed 12 mg per mL and infused at a rate of not more than 30 mg/minute as indicated in Table 1.

Compatibility. Dalacin C has been known to be physically and chemically compatible for at least 24 hours in glucose 5% water and sodium chloride injection solutions containing the following antibiotics in usually administered concentrations: amikacin sulfate, aztreonam, cefamandole nafate, cefazolin sodium, cefotaxime sodium, cefoxitin sodium, ceftazidime sodium, ceftizoxime sodium, gentamicin sulfate, netilmicin sulfate, piperacillin and tobramycin. The compatibility and duration of stability of drug mixtures will vary depending on concentration and other conditions.
No incompatibility has been demonstrated with the antibiotics cefalotin, kanamycin, gentamicin, penicillin or carbenicillin.
4.3 Contraindications
This drug is contraindicated in individuals with a history of hypersensitivity to preparations containing clindamycin, lincomycin or any of the ingredients listed under Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Serious anaphylactoid reactions require immediate emergency treatment with adrenaline. Oxygen, colloidal infusion, antihistamines and intravenous corticosteroids should also be administered as indicated.
Severe hypersensitivity reactions, including severe skin reactions such as drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), acute generalised exanthematous pustulosis (AGEP), and Kounis syndrome have been reported in patients receiving clindamycin therapy. If a hypersensitivity or severe skin reaction occurs, clindamycin should be discontinued and appropriate therapy should be initiated (see Section 4.3 Contraindications; Section 4.8 Adverse Effects (Undesirable Effects)).
The use of clindamycin can lead to the development of severe colitis. Fatalities have been reported. Therefore, Dalacin C should be reserved for serious infections where less toxic antimicrobial agents are inappropriate as described in Section 4.1 Therapeutic Indications. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections.
A toxin produced by Clostridioides difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with the use of antibiotics, including parenteral clindamycin. Symptoms may occur up to several weeks after cessation of antibiotic therapy.
Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone, however in moderate to severe cases appropriate therapy with suitable oral antibacterial agents effective against C. difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated.
Antibiotic associated colitis and diarrhoea (due to C. difficile), occur more frequently and may be more severe in debilitated and/or elderly patients (> 60 years). When clindamycin is indicated in these patients, they should be carefully monitored for change in bowel frequency.
C. difficile associated diarrhoea (CDAD) has been reported with use of nearly all antibacterial agents, including clindamycin, and may range in severity from mild diarrhoea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhoea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
Dalacin C should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Dalacin C should be used with caution in patients with a history of regional enteritis, ulcerative colitis or antibiotic associated colitis.
Dalacin C should be prescribed with caution in atopic individuals.
During prolonged therapy, periodic liver function tests and blood counts should be performed.
Clindamycin is potentially nephrotoxic. Acute kidney injury including acute renal failure has been reported. Therefore, monitoring of renal function should be considered during therapy of patients with pre-existing renal dysfunction or taking concomitant nephrotoxic drugs and monitoring of renal function should be performed if therapy is prolonged.
Patients with very severe renal disease and/or very severe hepatic disease accompanied by severe metabolic aberrations should be dosed with caution, and serum clindamycin levels monitored during high dose therapy.
Indicated surgical procedures should be performed in conjunction with antibiotic therapy. The use of Dalacin C may result in overgrowth of nonsusceptible organisms, particularly yeasts. Should superinfection occur, appropriate measures should be taken as indicated by the clinical situation.
Dalacin C should not be injected intravenously undiluted as a bolus, but should be infused over at least 10-60 minutes as directed in Section 4.2 Dose and Method of Administration. Drugs which delay peristalsis (e.g. opiates and diphenoxylate with atropine (Lomotil)) may prolong and/or worsen the condition and should not be used.
Rare instances of cardiopulmonary arrest and hypotension have been reported following too rapid intravenous administration (see Section 4.2 Dose and Method of Administration).
Local irritation, pain, induration and sterile abscess have been reported after intramuscular injection and thrombophlebitis after intravenous infusion (see Section 4.8 Adverse Effects (Undesirable Effects)). Reactions can be minimised by giving deep intramuscular injections and avoiding prolonged use of indwelling intravenous catheters.
Usage in meningitis. Since clindamycin does not diffuse adequately into the cerebrospinal fluid, the drug should not be used in the treatment of meningitis.
Use in the elderly. No data available.
Paediatric use. When Dalacin C is administered to newborns and infants, appropriate monitoring of organ system functions is desirable.
This product contains benzyl alcohol which is associated with severe adverse effects, including fatal "gasping syndrome", in paediatric patients. The minimum amount of benzyl alcohol at which toxicity may occur is unknown. The risk of benzyl alcohol toxicity depends on the quantity administered and the liver and kidneys' capacity to detoxify the chemical. Premature and low birthweight infants may be more likely to develop toxicity.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used with caution in patients receiving such agents.
Antagonism has been demonstrated between Dalacin C and erythromycin in vitro. Because of possible clinical significance these two drugs should not be administered concurrently.
In vitro studies of human liver and intestinal microsomes showed that clindamycin is metabolised predominantly by CYP3A4, and to a lesser extent by CYP3A5, to the major metabolite clindamycin sulfoxide and minor metabolite N-desmethylclindamycin. Therefore inhibitors of CYP3A4 and CYP3A5 may reduce clindamycin clearance and inducers of these isoenzymes may increase clindamycin clearance. In the presence of strong CYP3A4 inducers such as rifampicin, monitor for loss of effectiveness.
In vitro studies indicate that clindamycin does not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2E1 or CYP2D6 and only moderately inhibits CYP3A4.
See Section 4.2 Dose and Method of Administration, Compatibility; Section 6.2 Incompatibilities for physicochemical interactions.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Clindamycin crosses the placenta in humans. After multiple doses, amniotic fluid concentrations were approximately 30% of maternal concentrations.
Clindamycin should be used in pregnancy only if clearly needed.
Benzyl alcohol can cross the placenta (see Section 4.4 Special Warnings and Precautions for Use).
Use in lactation. Clindamycin has been reported to appear in human breast milk in ranges from < 0.5 to 3.8 micrograms/mL. Clindamycin has the potential to cause adverse effects on the breastfed infant's gastrointestinal flora such as diarrhoea or blood in the stool, or rash. Therefore, clindamycin is not recommended for nursing mothers.
If oral or intravenous clindamycin is required by a nursing mother, it is not a reason to discontinue breastfeeding, but an alternate drug may be preferred. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for clindamycin and any potential adverse effects on the breastfed child from clindamycin or from the underlying maternal condition.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
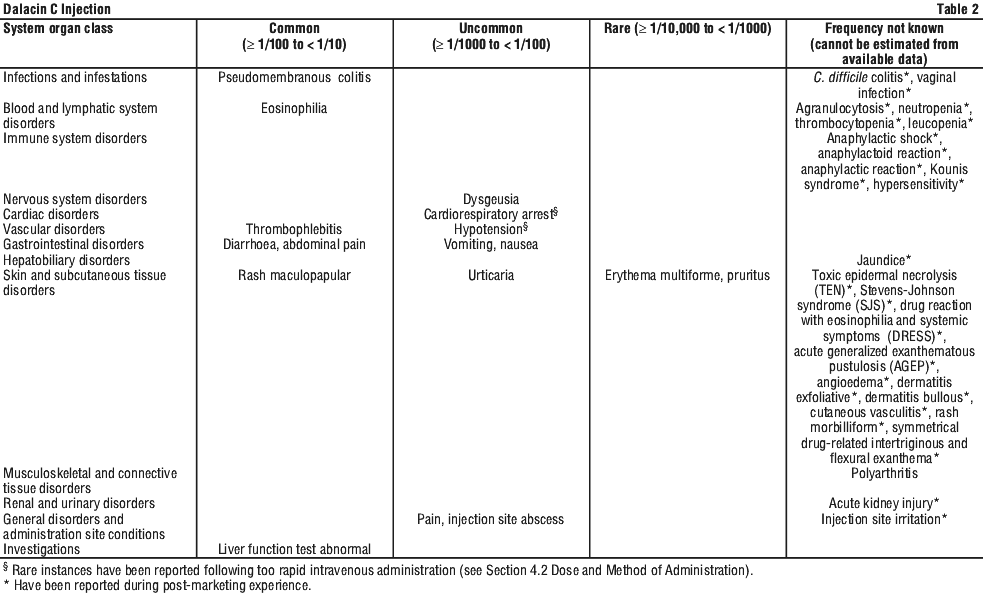
The adverse effects listed in Table 2 are presented by system organ class. Within each frequency category, the adverse effects are presented in the order of frequency and then by decreasing medical seriousness.
4.9 Overdose
Signs and symptoms. The minimal toxic or lethal dose is not well established. At therapeutic doses, the primary toxic effects may involve the gastrointestinal tract and may include severe diarrhoea and pseudomembranous colitis that may result in death. Rapid administration of large doses has resulted in ventricular dysrhythmias, hypotension and cardiac arrest. Dermatitis, nephrotoxicity, hepatotoxicity and various haematological abnormalities are toxic effects that occur less frequently.
Recommended treatment. No specific antidote is known. Support respiratory and cardiac function. In cases of overdose, drug levels of clindamycin are not clinically useful. However, monitoring serum concentrations in patients with markedly reduced renal and hepatic function may be indicated during high dose therapy. Monitor full blood count in patients with significant exposure as clindamycin may produce abnormalities of the haematopoietic system. Because clindamycin may cause hepatotoxicity, monitor liver function tests in patients with significant exposure.
Neither haemodialysis nor peritoneal dialysis appear to be effective in reducing clindamycin levels significantly.
Serious anaphylactoid reactions require immediate emergency treatment with adrenaline. Oxygen and intravenous corticosteroids should also be administered as indicated.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Clindamycin is a lincosamide antibiotic that inhibits bacterial protein synthesis. It binds to the 50S ribosomal subunit and affects both ribosome assembly and the translation process. Although clindamycin phosphate is inactive in vitro, rapid in vivo hydrolysis converts this compound to the antibacterially active clindamycin. At usual doses, clindamycin exhibits bacteriostatic activity in vitro.
Pharmacodynamic effects. Efficacy is related to the time period over which the agent level is above the minimum inhibitory concentration (MIC) of the pathogen (%T/MIC).
Resistance. Resistance to clindamycin is most often due to mutations at the rRNA antibiotic binding site or methylation of specific nucleotides in the 23S RNA of the 50S ribosomal subunit. These alterations can determine in vitro cross resistance to macrolides and streptogramins B (MLSB phenotype). Resistance is occasionally due to alterations in ribosomal proteins. Resistance to clindamycin may be inducible by macrolides in macrolide-resistant bacterial isolates. Inducible resistance can be demonstrated with a disk test (D-zone test) or in broth. Less frequently encountered resistance mechanisms involve modification of the antibiotic and active efflux. There is complete cross resistance between clindamycin and lincomycin. As with many antibiotics, the incidence of resistance varies with the bacterial species and the geographical area. The incidence of resistance to clindamycin is higher among methicillin-resistant staphylococcal isolates and penicillin-resistant pneumococcal isolates than among organisms susceptible to these agents.
Antimicrobial activity. Although clindamycin phosphate is inactive in vitro, rapid in vivo hydrolysis converts this compound to the antibacterially active clindamycin.
Clindamycin has been shown to have in vitro activity against most isolates of the following organisms:
Aerobic bacteria. Gram-positive bacteria. Staphylococcus aureus (methicillin-susceptible isolates), Coagulase-negative staphylococci (methicillin-susceptible isolates), Streptococcus pneumoniae (penicillin-susceptible isolates), Beta-haemolytic streptococci groups A, B, C and G, Viridans group streptococci, Corynebacterium spp.
Gram-negative bacteria. Chlamydia trachomatis.
Anaerobic bacteria. Gram-negative bacteria. Bacteroides spp., Fusobacterium spp., Gardnerella vaginalis, Prevotella spp.
Gram-positive bacteria. Propionibacterium acnes, Actinomyces spp., Eggerthella (Eubacterium) spp., Peptococcus spp., Peptostreptococcus spp. (Finegoldia magna, Micromonas micros), Clostridioides spp. (except C. difficile).
Fungi. Pneumocystis jirovecii.
Protozoans. Toxoplasma gondii, Plasmodium falciparum.
Breakpoints. Dilution or diffusion techniques - either quantitative (MIC) or breakpoint, should be used following a regularly updated, recognised and standardised method (e.g. NCCLS). Standardised susceptibility testing procedures require the use of laboratory control microorganisms to control the technical aspects of laboratory procedures.
The prevalence of acquired resistance may vary geographically and with time for selected species and local information on resistance is desirable, particularly when treating severe infections. As necessary, expert advice should be sought when the local prevalence of resistance is such that the utility of the agent in at least some types of infections is questionable. Particularly in severe infections or therapy failure microbiological diagnosis with verification of the pathogen and its susceptibility to clindamycin is recommended.
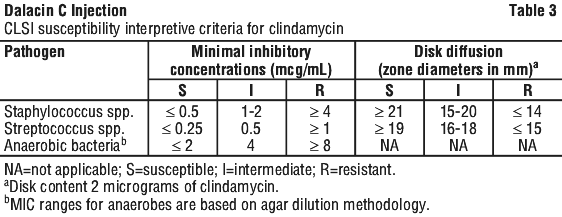
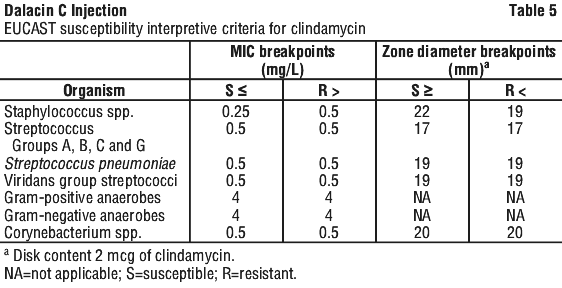
Resistance is usually defined by susceptibility interpretive criteria (breakpoints) established by Clinical and Laboratory Standards Institute (CLSI) or European Committee on Antimicrobial Susceptibility Testing (EUCAST) for systemically administered antibiotics.
Clinical and Laboratory Standards Institute (CLSI) breakpoints for relevant organisms are listed in Table 3.
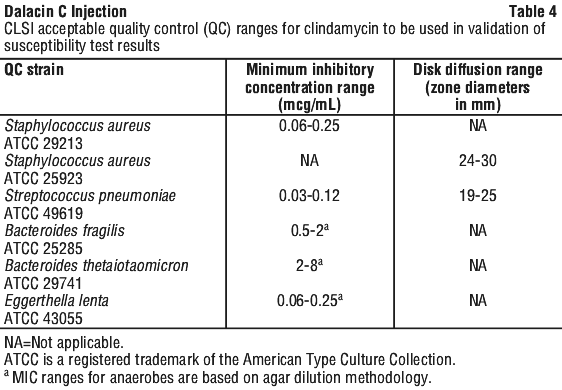
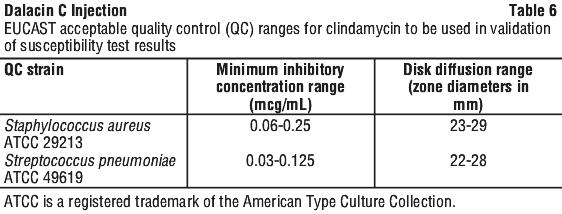
Standardised susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of the supplies and reagents used in the assay, and the techniques of the individuals performing the test. Standard clindamycin powder should provide the MIC ranges in Table 4. For the disk diffusion technique using the 2 mcg clindamycin disk the criteria provided in Table 4 should be achieved.
5.2 Pharmacokinetic Properties
Biologically inactive clindamycin phosphate is rapidly converted to active clindamycin.
By the end of short-term intravenous infusion, peak serum levels of active clindamycin are reached.
In vitro studies in human liver and intestinal microsomes indicated that clindamycin is predominantly oxidised by CYP3A4, with minor contribution from CYP3A5, to form clindamycin sulfoxide and a minor metabolite, N-desmethylclindamycin.
Biologically-inactive clindamycin phosphate disappears rapidly from the serum, the average disappearance half-life is 6 minutes; however, the serum disappearance half-life of active clindamycin is about 3 hours in adults and 2.5 hours in children.
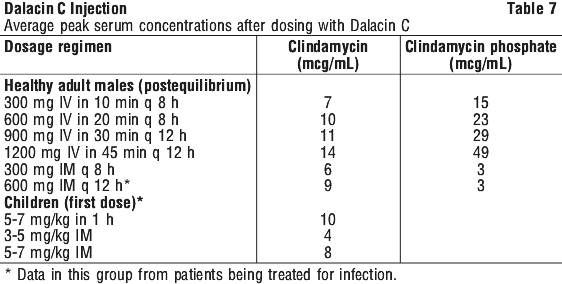
After intramuscular injection of Dalacin C, peak levels of active clindamycin are reached within 3 hours in adults and 1 hour in children. Serum level curves may be constructed from IV peak serum levels as given in Table 7 by application of the disappearance half-lives listed above.
Serum levels of clindamycin can be maintained above the in vitro minimum inhibitory concentrations for most indicated organisms by administration of Dalacin C every 8-12 hours in adults and every 6-8 hours in children, or by continuous intravenous infusion. An equilibrium state is reached by the third dose.
The disappearance half-life of clindamycin is increased slightly in patients with markedly reduced renal or hepatic function; dosage schedules need not be modified in the presence of mild to moderate renal or hepatic disease. No significant levels of clindamycin are attained in the cerebrospinal fluid even in the presence of inflamed meninges.
Obese paediatric patients aged 2 to less than 18 years and obese adults aged 18 to 20 years. An analysis of pharmacokinetic data in obese paediatric patients aged 2 to less than 18 years and obese adults aged 18 to 20 years demonstrated that clindamycin clearance and volume of distribution normalised by total body weight are comparable regardless of obesity.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Benzyl alcohol, disodium edetate, hydrochloric acid, sodium hydroxide, water for injections.
6.2 Incompatibilities
The following drugs are physically incompatible with Dalacin C: ampicillin, phenytoin sodium, barbiturates, aminophylline, calcium gluconate, magnesium sulfate, ceftriaxone sodium and ciprofloxacin.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze.)
6.5 Nature and Contents of Container
The following sizes are available:
2 mL ampoule (300 mg): 10's.
4 mL ampoule (600 mg): 10's.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
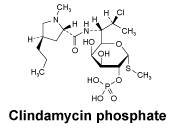
CAS number. CAS Number: 24729-96-2.
7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Only Medicine).
Date of First Approval
02 August 1991
Date of Revision
19 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.