Dalacin V Cream 2%
Brand Information
| Brand name | Dalacin V Cream 2% |
| Active ingredient | Clindamycin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Dalacin V Cream 2%.
Summary CMI
DALACIN® V Cream 2%
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using DALACIN V Cream 2%?
DALACIN V Cream 2% contains the active ingredient clindamycin phosphate. Dalacin V is used to treat vaginal infection.
For more information, see Section 1. Why am I using DALACIN V Cream 2%? in the full CMI.
2. What should I know before I use DALACIN V Cream 2%?
Do not use if you have ever had an allergic reaction to DALACIN medicines, clindamycin, lincomycin, or any of the ingredients listed at the end of the CMI or have/have previously had inflammatory bowel disease or antibiotic-associated colitis.
Talk to your doctor if you have had any bowel problems, or any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use DALACIN V Cream 2%? in the full CMI.
3. What if I am taking/using other medicines?
Some medicines may interfere with DALACIN V Cream 2% and affect how it works.
A list of these medicines is in Section 3. What if I am taking/using other medicines? in the full CMI.
4. How do I use DALACIN V Cream 2%?
Vaginal use only, insert one applicator-full of cream, once-a-day, at bedtime, for seven days in a row.
More instructions on how to use DALACIN V Cream 2% can be found in Section 4. How do I use DALACIN V Cream 2%? in the full CMI.
5. What should I know while using DALACIN V Cream 2%?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using DALACIN V Cream 2%? in the full CMI.
6. Are there any side effects?
Vaginal pain/discomfort or unusual smell; skin rash, swellings, itching, redness; headache; dizziness, spinning sensation; stomach pain, indigestion, bloating, excessive wind; constipation; diarrhoea; nausea, vomiting; bad breath; change in taste; back pain; pelvic pain; nosebleed; abnormalities during or in-between periods. Uncommon but serious side effects that may need immediate medical attention: severe diarrhoea which may have blood and mucus; severe pelvic pain; signs of an allergic reaction; discomfort when urinating; irregular heart activities; slow or no progress in labour; abnormal infections.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
DALACIN® V Cream 2%
Active ingredient(s): Clindamycin phosphate
Consumer Medicine Information (CMI)
This leaflet provides important information about using DALACIN V Cream 2%. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using DALACIN V Cream 2%.
Where to find information in this leaflet:
1. Why am I using DALACIN V Cream 2%?
2. What should I know before I use DALACIN V Cream 2%?
3. What if I am taking/using other medicines?
4. How do I use DALACIN V Cream 2%?
5. What should I know while using DALACIN V Cream 2%?
6. Are there any side effects?
7. Product details
1. Why am I using DALACIN V Cream 2%?
DALACIN V Cream 2% contains the active ingredient clindamycin phosphate. DALACIN V Cream 2% is an antibiotic cream used in the vagina to treat vaginal infection. It works by stopping the growth of the bacteria causing the vaginal infection.
Ask your doctor if you have any questions about why DALACIN V Cream 2% has been prescribed for you. Your doctor may have prescribed DALACIN V Cream 2% for another reason.
This medicine is available only with a doctor's prescription.
2. What should I know before I use DALACIN V Cream 2%?
Warnings
You should not be having vaginal sex or using other vaginal products (such as tampons or douches) while using this product.
Do not use condoms or vaginal contraceptive devices as methods of contraception while using DALACIN V Cream 2% or within 72 hours after the end of your treatment with DALACIN V Cream 2%.
You must tell your doctor, and should not use DALACIN V Cream 2% if you:
- have ever had an allergic reaction to Dalacin medicines, clindamycin or lincomycin or any of the ingredients listed at the end of this leaflet.
- have, or have previously had inflammatory bowel disease, or antibiotic-associated colitis (severe diarrhoea associated with the use of antibiotics).
Check with your doctor if you:
- have bowel disease or any gastrointestinal (stomach or gut) problems.
- take/use any medicines for any other conditions.
You should not be using this medicine if the expiry date printed on the pack has passed or if the packaging is torn or shows signs of tampering.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Talk to your doctor if you are:
- pregnant or intend to become pregnant.
- breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved.
DALACIN V Cream 2% has not been shown to be effective or safe in children.
3. What if I am taking/using other medicines?
Tell your doctor or pharmacist if you are taking/using any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with DALACIN V Cream 2% and affect how it works, including:
- some medicines used during surgery.
- other medicines used in the vagina.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking/using and if these affect DALACIN V Cream 2%.
4. How do I use DALACIN V Cream 2%?
How much to use and how long to use it
The recommended dose of DALACIN V Cream 2% is one applicator-full (about 5 grams) of cream, once-a-day, at bedtime, for seven days in a row.
How to use it
The plastic applicators provided with DALACIN V Cream 2% are designed to allow the correct placement of the cream in the vagina. To load and use the applicator correctly, follow these steps:
- Wash hands thoroughly with soap and water before and after using the vaginal applicator.
- Remove the cap from the tube of cream.
- Screw the open end of the applicator onto the tube. Gently squeeze from the base of the tube, forcing the cream into the applicator (see diagram 1).
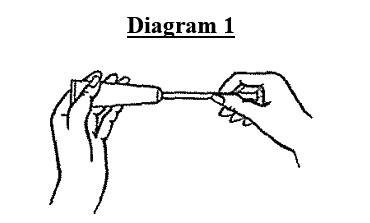
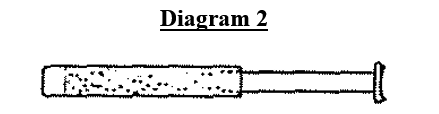
- As the cream enters the applicator, the plunger will be pushed back until it reaches the stopping point. At this stage, unscrew the applicator from the tube (see diagram 2) and replace the cap.
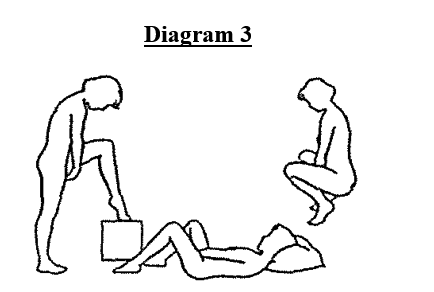
- Choose a position to apply DALACIN V Cream 2% which is comfortable for you, keeping in mind that the applicator must reach as high as is comfortable in the vagina. This may be the position which you usually use for the insertion of a tampon (see diagram 3). For example:
a) squatting;
b) standing with your legs apart or with one leg raised on a chair;
c) lying on your back with your legs raised.
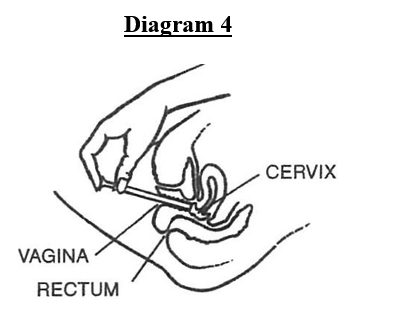
- The lips of the vagina should be parted with the fingers of one hand. Grasp the applicator between the thumb and middle finger of the other hand and insert it (open end first) into the vagina as deeply as it will comfortably reach (see diagram 4).
- Holding the applicator in place, slowly push the plunger until it stops, so depositing the cream in the vagina.
- Carefully withdraw the applicator from the vagina and dispose of the applicator safely where children cannot find it.
If you forget to use DALACIN V Cream 2%
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to. Otherwise, use it as soon as you remember, and then go back to using DALACIN V Cream 2% as you would normally.
Do not use a double dose to make up for the dose that you missed.
If you use too much DALACIN V Cream 2%
It is important that you do not use more DALACIN V Cream 2% than your doctor has prescribed. If you use too much DALACIN V Cream 2%, you may experience unwanted side effects.
If DALACIN V Cream 2% is Swallowed
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If DALACIN V Cream 2% gets into eyes
You should immediately rinse the eye with lots of cool tap water.
5. What should I know while using DALACIN V Cream 2%?
Things you should do
- Wash hands thoroughly with soap and water after using the vaginal applicator.
- Tell your doctor immediately if you suffer mild diarrhoea or seek immediate emergency medical attention if you experience severe diarrhoea which may contain blood. Do this even if it occurs several weeks after stopping DALACIN V Cream 2%.
- Tell your doctor if you feel that DALACIN V Cream 2% is not helping your condition.
- Tell your doctor if you have persistent symptoms (infections) following treatment with DALACIN C Cream 2%, as you may be infected with other bacteria.
- Tell your doctor or pharmacist if you do not feel well while using DALACIN V Cream 2%.
- If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using DALACIN V Cream 2%.
- Do not use any medicines for diarrhoea or strong pain unless you have spoken to your doctor, nurse or pharmacist first.
- Tell any other doctors, dentists, and pharmacists who treat you that you are using this medicine.
- If you become pregnant or plan to breastfeed while using DALACIN V Cream 2%, tell your doctor immediately.
- Keep all your doctor's appointments so that your progress can be checked.
Things you should not do
- DALACIN V Cream 2% must only be used in the vagina. It must not be used on skin, in the mouth or in eyes.
- You should not be having vaginal sex or using other vaginal products (such as tampons or douches) while using this product.
- Do not use condoms or vaginal diaphragm contraceptive devices as methods of contraception while using DALACIN V Cream 2% or within 72 hours after the end of your treatment with DALACIN V Cream 2%.
- Do not stop using this medicine without talking to your doctor.
- Do not take/use any medicines/products for diarrhoea without first checking with your doctor.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not use DALACIN V Cream 2% to treat any other illnesses unless your doctor tells you to.
Driving or using machines
No information available.
Drinking alcohol
No information available.
Looking after your medicine
Store it in a cool dry place where the temperature stays below 25°C, away from moisture, heat, or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window-sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half meters above the ground is a good place to store medicines.
When to discard your medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Side effects
| Side effects | What to do |
| Speak to your doctor if you have any of these side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight-away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side-effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/safety/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What DALACIN V Cream 2% contains
| Active ingredient (main ingredient) | clindamycin phosphate |
| Other ingredients (inactive ingredients) | benzyl alcohol cetostearyl alcohol cetyl esters wax liquid paraffin polysorbate 60 propylene glycol purified water sorbitan monostearate stearic acid |
Do not use this medicine if you are allergic to any of these ingredients.
What DALACIN V Cream 2% looks like
DALACIN V Cream 2% is a white semi-solid cream. It is available in a 40g tube. Each DALACIN V Cream 2% pack also contains 7 disposable applicators.
AUST R 48431 - DALACIN V clindamycin 2% (20mg/g) (as phosphate) cream tube
Who distributes DALACIN V Cream 2%
Pfizer Australia Pty Ltd
Level 17, 151 Clarence Street SYDNEY NSW 2000
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in July 2025.
® Registered Trademark
© Pfizer
Brand Information
| Brand name | Dalacin V Cream 2% |
| Active ingredient | Clindamycin |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Clindamycin phosphate.
2 Qualitative and Quantitative Composition
Dalacin V Cream 2% contains the equivalent of 2% (20 mg/g) free clindamycin as a water soluble ester of clindamycin and phosphoric acid (clindamycin phosphate). Each unit dose of Dalacin V Cream 2% (approximately 5 grams) represents 100 mg of clindamycin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Intravaginal cream.
4 Clinical Particulars
4.1 Therapeutic Indications
Dalacin V Cream 2% is indicated for the treatment of symptomatic bacterial vaginosis.
Note. For the purposes of this indication, bacterial vaginosis is usually defined by positive results to at least three of the four following criteria.
1. Vaginal discharge with pH > 4.5.
2. Vaginal discharge demonstrating an amine ("fishy") odour with the addition of 10% potassium hydroxide.
3. Vaginal discharge with "clue cells" on microscopy.
4. A Gram stain consistent with a diagnosis of bacterial vaginosis (Lactobacillus morphotype absent or markedly decreased, Gardnerella morphotype predominant flora, white blood cells absent or few, Mobiluncus morphotype may or may not be present).
Other pathogens which may be associated with genital infection such as Trichomonas vaginalis, Candida albicans, Chlamydia trachomatis and Neisseria gonorrhoeae should be ruled out by appropriate laboratory means.
4.2 Dose and Method of Administration
The recommended dose of Dalacin V Cream 2% is one applicator full (approximately 5 grams) of cream intravaginally at bedtime for seven (7) consecutive days.
4.3 Contraindications
Clindamycin phosphate cream is contraindicated in patients with a history of hypersensitivity to clindamycin, lincomycin or other components of the cream (see Section 6.1 List of Excipients). Clindamycin phosphate cream is also contraindicated in individuals with a history of inflammatory bowel disease or history of antibiotic associated colitis.
4.4 Special Warnings and Precautions for Use
For intravaginal use only. Not for ophthalmic, dermal or oral use.
Antibiotic associated pseudomembranous colitis has been reported with many antibiotics, including clindamycin. A toxin produced by Clostridium difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with the use of antibiotics, including vaginally administered clindamycin (approximately 4% of the administered dose is absorbed systemically, see Section 5.2 Pharmacokinetic Properties). Symptoms may occur up to several weeks after cessation of antibiotic therapy.
Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone, however, in moderate to severe cases appropriate therapy with a suitable oral antibacterial agent effective against Clostridium difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated.
Drugs which delay peristalsis (e.g. opiates and diphenoxylate with atropine (e.g. Lomotil)) may prolong and/or worsen the condition and should not be used.
Dalacin V Cream 2% should be used with caution in patients with a history of regional enteritis, ulcerative colitis or antibiotic associated colitis.
The use of intravaginal clindamycin phosphate may result in the localised overgrowth of nonsusceptible organisms, particularly yeasts. Clindamycin has shown in vitro activity against Lactobacilli species, which are the predominant bacteria in normal vaginal flora. In clinical trials approximately 14% of patients treated with Dalacin V Cream 2% developed symptomatic cervicitis/ vaginitis predominantly due to C. albicans (see Section 4.8 Adverse Effects (Undesirable Effects)).
The persistence of symptoms following treatment with Dalacin V Cream 2% should alert the clinician to the possibility of concomitant infection with organisms such as Trichomonas vaginalis, Candida albicans, Chlamydia trachomatis and Neisseria gonorrhoeae. Appropriate microbiological investigations and therapy directed at such organisms should be considered.
The patient should be instructed not to engage in vaginal intercourse or use other vaginal products (such as tampons or douches) during treatment with clindamycin vaginal cream.
This cream contains mineral oil. As mineral oil may weaken latex or rubber products such as condoms or vaginal contraceptive diaphragms, the use of these contraceptive devices is not recommended within 72 hours following treatment with Dalacin V Cream 2%.
Dalacin V Cream 2% contains ingredients that will cause burning and irritation of the eyes. In the event of accidental contact with the eyes, rinse the eye with copious quantities of cool tap water.
Use in the elderly. Clinical studies for clindamycin phosphate vaginal cream 2% did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Paediatric use. The safety and effectiveness of Dalacin V Cream 2% has not been established in children.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Cross resistance has been demonstrated between clindamycin, erythromycin and lincomycin.
No systemic drug interactions are known or anticipated with Dalacin V Cream. Antagonism has been demonstrated between clindamycin and erythromycin in vitro, however, this potential interaction would not appear to be applicable unless erythromycin was also applied intravaginally.
Systemic clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used with caution in patients receiving such agents.
As the studies involving Dalacin V Cream did not allow concurrent intravaginal medication to be administered, there are no data regarding the concomitant use of other intravaginal medications.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility studies in rats treated orally with up to 300 mg/kg/day (31 times the human exposure based on mg/m2) revealed no effects on fertility or mating.
Use in pregnancy. (Category A)
Category A where the fetal membranes are intact.
Studies of Dalacin V Cream 2% have not been conducted in women during the first trimester.
Use in lactation. It is not known if clindamycin is excreted in human breast milk following the use of vaginally administered clindamycin phosphate. Clindamycin has been reported to appear in human breast milk in ranges from < 0.5 to 3.8 micrograms/mL following systemic use. Clindamycin has the potential to cause adverse effects on the breastfed infant's gastrointestinal flora such as diarrhoea or blood in the stool, or rash. Therefore, clindamycin is not recommended for nursing mothers.
If clindamycin is required by a nursing mother, it is not a reason to discontinue breastfeeding, but an alternate drug may be preferred. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for clindamycin and any potential adverse effects on the breastfed child from clindamycin or from the underlying maternal condition.
4.7 Effects on Ability to Drive and Use Machines
The effect of clindamycin on the ability to drive or operate machinery has not been systematically evaluated.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials. Non-pregnant women. In clinical trials involving non-pregnant women, medical events judged to be related, probably related, possibly related or of unknown relationship to Dalacin V Cream 2% were reported in 20.7% of the patients receiving treatment for 3 days and 21.3% of the patients receiving treatment for 7 days. Events occurring in ≥ 1% of patients receiving 3 day or 7 day Dalacin V Cream 2% are shown in Table 1.
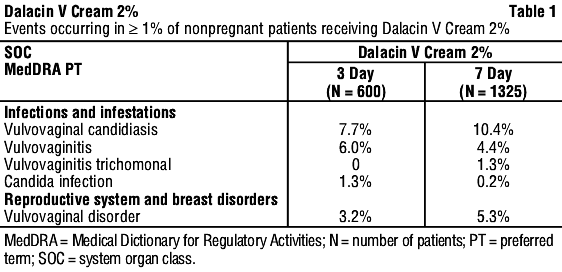
Infections and infestations. Fungal infection, bacterial infection, skin candida, urinary tract infection, vaginal infection.
Immune system disorders. Hypersensitivity.
Endocrine disorders. Hyperthyroidism.
Nervous system disorders. Headache, dizziness.
Ear and labyrinth disorders. Vertigo.
Respiratory, thoracic and mediastinal disorders. Epistaxis.
Gastrointestinal disorders. Abdominal pain, constipation, diarrhoea, nausea, vomiting, breath odour, dyspepsia, flatulence, gastrointestinal disorder.
Skin and subcutaneous tissue disorders. Pruritus (non-application site), rash, maculopapular rash, urticaria, erythema.
Reproductive system and breast disorders. Menstrual disorder, metrorrhagia, vaginal discharge, vulvovaginal pain, endometriosis.
General disorder and administration site conditions. Inflammation.
Investigations. Microbiology test abnormal.
Special senses. Taste perversion.
Pregnant women. In clinical trials involving women during the second trimester of pregnancy, medical events judged to be related, probably related, possibly related or of unknown relationship to Dalacin V Cream 2% were reported in 22.8% of pregnant patients. Events occurring in ≥ 1% of patients receiving Dalacin V Cream 2% or placebo are shown in Table 2.
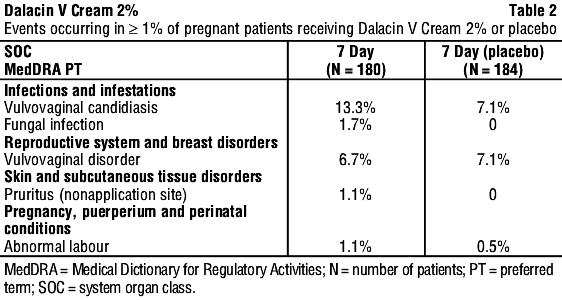
Infections and infestations. Candida infection, upper respiratory tract infection, vulvovaginitis trichomonal.
Gastrointestinal disorders. Diarrhoea.
Skin and subcutaneous tissue disorders. Erythema.
Renal and urinary disorders. Glycosuria, proteinuria, dysuria.
Reproductive system and breast disorders. Metrorrhagia, vulvovaginal pain.
Postmarketing experience. Postmarketing experience of the patients receiving Dalacin V Cream 2% with a frequency category of frequency not known include:
Gastrointestinal disorders. Pseudomembranous colitis, abdominal distension.
Musculoskeletal and connective tissue disorders. Back pain.
Reproductive system and breast disorders. Pelvic pain.
General disorder and administration site conditions. Pain.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/safety/reporting-problems.
4.9 Overdose
Clindamycin from Dalacin V Cream 2% may be absorbed in sufficient amounts to produce systemic effects. Acute ingestion of clindamycin has not been associated with significant toxicity. Gastrointestinal decontamination is probably not necessary in most cases.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Clindamycin is a lincosamide antibiotic that inhibits bacterial protein synthesis at the level of the bacterial ribosome. The antibiotic binds preferentially to the 50S ribosomal subunit and affects the translation process. Although clindamycin phosphate is inactive in vitro, rapid in vivo hydrolysis converts this compound to the antibacterially active clindamycin.
Clindamycin, like most protein synthesis inhibitors, is predominantly bacteriostatic and efficacy is associated with the length of time the concentration of active ingredient remains above the MIC of the infecting organism.
Resistance to clindamycin is most often due to modification of the target site on the ribosome, usually by chemical modification of RNA bases or by point mutations in RNA or occasionally in proteins. Cross resistance has been demonstrated in vitro between lincosamides, macrolides and streptogramins B in some organisms. Cross resistance has been demonstrated between clindamycin and lincomycin.
Clindamycin is active in vitro against most strains of the following organisms that have been reported to be associated with bacterial vaginosis.
Bacteroides spp.
Gardnerella vaginalis.
Mobiluncus spp.
Mycoplasma hominis.
Peptostreptococcus spp.
Culture and sensitivity testing of bacteria are not routinely performed to establish the diagnosis of bacterial vaginosis and to guide treatment. Standard methodology for the susceptibility testing of the potential bacterial vaginosis pathogens, Gardnerella vaginalis and Mobiluncus spp. has not been defined. Methods for determining the susceptibility of Bacteroides spp. and Gram-positive anaerobic cocci, as well as Mycoplasma spp. have been described by the Clinical and Laboratory Standards Institute (CLSI) and clindamycin susceptibility breakpoints for Gram-negative and Gram-positive anaerobes have been published by both EUCAST and CLSI. Clinical isolates that test susceptible to clindamycin and resistant to erythromycin should also be tested for inducible clindamycin resistance using the D-test. However, the breakpoints are intended to guide systemic, rather than localised, antibiotic treatment.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Following a once a day (for seven days) dosing of approximately 5 grams of Dalacin V Cream containing the equivalent of 100 mg clindamycin, peak serum clindamycin levels averaged 20 nanogram/mL (range 3 to 93 nanogram/mL) in normal volunteers. Approximately 3% (range 0.1 to 11.3%) of the administered dose was absorbed systemically.
In women with bacterial vaginosis being treated with Dalacin V Cream once daily (5 grams) for seven days, the amount of clindamycin absorbed was 4% (range 0.8 to 8.2%), which approximates results seen in normal volunteers.
The levels of clindamycin absorbed following the intravaginal administration of Dalacin V Cream reached steady state within four days of the seven day regimen.
5.3 Preclinical Safety Data
Carcinogenicity. Long-term studies in animals have not been performed with clindamycin to evaluate carcinogenic potential.
Genotoxicity. Genotoxicity tests performed included a rat micronucleus test and an Ames test. Both tests were negative.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sorbitan monostearate, polysorbate 60, propylene glycol, stearic acid, cetostearyl alcohol, cetyl ester wax, liquid paraffin, purified water, benzyl alcohol (each 5 g contains 50 mg benzyl alcohol as preservative).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Dalacin V Cream 2% is supplied in a 40 g collapsible laminate tube. Each 40 gram pack also contains seven (7) single-use disposable applicators, intended to be used once nightly for seven (7) days.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
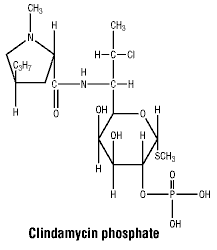
CAS number. CAS Registry Number: 24729-96-2.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
08 April 1994
Date of Revision
09 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.