DBL Cisplatin Injection
Brand Information
| Brand name | DBL Cisplatin Injection |
| Active ingredient | Cisplatin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the DBL Cisplatin Injection.
Summary CMI
DBL™ Cisplatin Injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being treated with DBL Cisplatin Injection?
DBL Cisplatin Injection contains the active ingredient cisplatin. DBL Cisplatin Injection is used to treat testicular cancer, ovarian cancer, bladder cancer and cancers of the head and neck.
For more information, see Section 1. Why am I being treated with DBL Cisplatin Injection? in the full CMI.
2. What should I know before treatment with DBL Cisplatin Injection?
DBL Cisplatin Injection should not be used if you have ever had an allergic reaction to cisplatin or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have kidney disease, hearing difficulties, an infection, low blood cell production or any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before treatment with DBL Cisplatin Injection? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with DBL Cisplatin Injection and affect how it works. Examples include some medicines to treat bacterial or fungal infections, cancer, and gout; medicine to prevent fits or to reduce the amount of water in the body; and blood thinners, including aspirin.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is DBL Cisplatin Injection given?
Your doctor will give you DBL Cisplatin Injection as a slow injection (drip) into a vein and decide how much you will be given and for how long.
More instructions can be found in Section 4. How is DBL Cisplatin Injection given? in the full CMI.
5. What should I know during treatment with DBL Cisplatin Injection?
| Things you should do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know during treatment with DBL Cisplatin Injection? in the full CMI.
6. Are there any side effects?
Side effects include sudden life-threatening allergic reactions (shortness of breath, wheezing or difficulty breathing, flushing, swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin); infection (fever, fatigue, chills, a sore throat, aches and pains, or coughing); severe nausea and vomiting; reduced urination, joint pain, swelling of feet or lower legs, pain in the lower back or side; headache, seizures, slurred speech and loss of memory, confusion; eye pain, blurring or other changes in your vision; problems with movement or reduced reflexes, and leg weakness; paralysis or numbness of the face, arm or leg, tingling or a loss of sensation in the hands or feet; unusual bleeding such as bleeding gums, blood in urine/wee or bowel motions/poo, unusual bruising or pinpoint red spots on the skin; changes in the rhythm or rate of the heartbeat, tightness or pain of the chest, neck, back or arms; shortness of breath, chest pain or coughing, yellowing of the skin, eyes or mouth, dark urine.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
DBL™ Cisplatin Injection
Active ingredient(s): cisplatin
Consumer Medicine Information (CMI)
This leaflet provides important information about using DBL Cisplatin Injection. You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about using DBL Cisplatin Injection.
Where to find information in this leaflet:
1. Why am I being treated with DBL Cisplatin Injection?
2. What should I know before treatment with DBL Cisplatin Injection?
3. What if I am taking other medicines?
4. How is DBL Cisplatin Injection given?
5. What should I know during treatment with DBL Cisplatin Injection?
6. Are there any side effects?
7. Product details
1. Why am I being treated with DBL Cisplatin Injection?
DBL Cisplatin Injection contains the active ingredient cisplatin. DBL Cisplatin Injection is a type of medicine called an anti-neoplastic or cytotoxic agent. You may also hear it referred to as chemotherapy. Cisplatin blocks DNA production in cancer cells which helps to weaken and destroy them. It may be used alone or in combination with other anti-cancer therapies.
DBL Cisplatin Injection is used to treat testicular cancer, ovarian cancer, bladder cancer and cancers of the head and neck.
2. What should I know before treatment with DBL Cisplatin Injection?
Warnings
DBL Cisplatin Injection should not be used if you have:
- an allergy to cisplatin, other platinum containing compounds, or any of the ingredients listed at the end of this leaflet
- kidney problems
- hearing problems
- reduced amounts of red or white blood cells or platelets
- any infection
- if you are pregnant or breast feeding.
Check with your doctor if you:
- have or have had low iron levels
- have heart problems
- will require dental work or an invasive medical procedure
- have low magnesium or calcium levels
- are going to be given a vaccine
- have been exposed to an infection recently, especially chicken pox or shingles
- have muscle numbness or weakness, or seizures
- have liver problems.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you or are pregnant or you or your partner intend to become pregnant.
DBL Cisplatin Injection is potentially harmful to the fetus.
Women should use effective contraception during treatment with DBL Cisplatin Injection and for at least 7 months after treatment.
Men should use effective contraception during treatment with DBL Cisplatin Injection and for at least 4 months after treatment.
DBL Cisplatin Injection may affect male and female fertility. Check with your doctor for advice on fertility preservation before you begin treatment.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
DBL Cisplatin injection has been shown to pass into breast milk.
Women should not breastfeed during treatment with DBL Cisplatin Injection and for at last 1 month after treatment.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may affect or be affected by DBL Cisplatin Injection. These include:
- some medicines to treat bacterial infections, e.g. gentamicin
- some medicine to treat fungal infections e.g. amphotericin B
- some medicine to treat cancer, e.g. ifosfamide, etoposide, paclitaxel, bleomycin
- methotrexate, a medicine used to treat some forms of arthritis, psoriasis and cancer
- medicine to reduce the amount of water in the body e.g. loop diuretics like furosemide
- medicine to treat gout e.g. allopurinol, colchine, probenecid, sulfinpyrazone
- medicine to prevent seizures/fits e.g. phenytoin
- blood thinners e.g. coumarins, warfarin
- lithium, used to treat bipolar disorder or schizophrenia.
Check with your doctor, nurse or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect DBL Cisplatin Injection.
4. How is DBL Cisplatin Injection given?
How is DBL Cisplatin Injection given
- You will be given DBL Cisplatin Injection as a slow injection (drip) into a vein.
How much will be given
- Your doctor will decide how much DBL Cisplatin Injection you will be given and how long you will be given it.
If you are given too much too much DBL Cisplatin Injection
As DBL Cisplatin Injection doses are carefully worked out and given by a doctor experienced in its use and who will monitor you during treatment, it is extremely unlikely that you will be given too much.
5. What should I know during treatment with DBL Cisplatin Injection?
Things you should do
Drink plenty of water prior to and after your treatment with DBL Cisplatin injection to ensure you are well hydrated as this may help to protect the kidneys.
Keep all of your doctor's appointments and attend any tests as directed, as treatment with DBL Cisplatin Injection requires your doctor to monitor your kidneys, liver, naturally occurring chemicals in your blood and blood cells.
During treatment with DBL Cisplatin Injection you may be at increased risk of bleeding, bruising and infection. Care should be taken to minimise cuts, impacts or exposure to infection.
Inform your doctor immediately if you experience:
- problems with your hearing or a buzzing/ringing or other noise in your ears
- abnormal bruising or bleeding, including in your urine/wee, bowel motion/poo, or vomit
- signs of infection such as fever or chills
- signs of anaemia such as shortness of breath and dizziness
- severe vomiting that will not stop
- swelling of the face, wheezing, shortness of breath, skin rash or itchiness
- chest pain
- muscle spasms/cramps, which may also have numbness/tingling
- problems with your vision
- fits/seizures.
Discuss any dental work you feel may be required with your doctor before visiting your dentist.
Remind any doctor, nurse, dentist, surgeon, anaesthetist, or pharmacist you visit that you are being treated with DBL Cisplatin Injection.
Things you should not do
Avoid taking aspirin as it may increase the risk of bleeding in the mouth, throat, stomach and gut.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how DBL Cisplatin Injection affects you.
Drinking alcohol
Alcohol should be avoided as it may increase the risk of bleeding in the mouth, throat, stomach and gut.
Looking after the medicine
DBL Cisplatin Injection will be stored appropriately at the hospital.
Getting rid of any unwanted medicine
The hospital staff will dispose of any leftover DBL Cisplatin Injection.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Side effects
| Side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
| Speak to your doctor as soon as possible if you notice any of these side effects. |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Some of these side effects can only be found when your doctor does tests from time to time to check your progress.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/safety/reporting-problems.
By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What DBL Cisplatin Injection contains
| Active ingredient (main ingredient) | Cisplatin |
| Other ingredients (inactive ingredients) | Mannitol Sodium chloride Water for injections |
This medicine should not be used if you are allergic to any of these ingredients.
DBL Cisplatin Injection does not contain lactose, sucrose, gluten, tartrazine or other azo dyes.
What DBL Cisplatin Injection looks like
DBL Cisplatin Injection is a clear colourless to pale yellow solution (AUST R 47275 and AUST R 47276).
Who distributes DBL Cisplatin Injection
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in September 2025.
Brand Information
| Brand name | DBL Cisplatin Injection |
| Active ingredient | Cisplatin |
| Schedule | S4 |
MIMS Revision Date: 01 June 2024
1 Name of Medicine
Cisplatin.
2 Qualitative and Quantitative Composition
Each mL of DBL Cisplatin Injection contains 1 mg cisplatin.
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
Solution for injection.
DBL Cisplatin Injection is a clear, colourless to pale yellow sterile solution of cisplatin 1 mg/mL, mannitol 1 mg/mL and sodium chloride 9 mg/mL in water for injections. The solution does not contain any preservative.
4 Clinical Particulars
4.1 Therapeutic Indications
DBL Cisplatin Injection is indicated for the palliative treatment of metastatic non-seminomatous germ cell carcinoma, advanced stage refractory ovarian carcinoma, advanced stage refractory bladder carcinoma and refractory squamous cell carcinoma of the head and neck. It may be used as a single agent or in combination with other chemotherapeutic agents. It may be employed, in appropriate circumstances, in addition to other modalities, e.g. radiotherapy or surgery.
4.2 Dose and Method of Administration
Dosage. A variety of doses and schedules are used. To obtain optimum therapeutic results with minimum adverse effects, the dosage of cisplatin must be based on the clinical, renal and haematologic status of the patient.
The usual dose in adults and children when used as single agent therapy is 50-100 mg/m2 as a single IV infusion every 3-4 weeks; or 15-20 mg/m2 as a daily IV infusion for 5 days every 3-4 weeks.
Dosage adjustment. Dosage should be reduced in patients with depressed bone marrow function.
Subsequent treatment with cisplatin. A repeat course of cisplatin should not be given until:
the serum creatinine is below 140 micromol/L and/or the plasma urea is below 9 mmol/L; and
circulating blood elements are at an acceptable level (platelets at least 100,000/mm3, WBC at least 4000/mm3).
A base line audiogram should be taken and the patient monitored periodically for auditory deterioration (see Section 4.4 Special Warnings and Precautions for Use).
Hepatic impairment. Human studies show a high uptake of cisplatin in the liver. An elevated aspartate aminotransferase (AST) and alkaline phosphatase (ALP) with clinical signs of liver toxicity has been reported in some cases. The adult dosage should be used with caution in patients with pre-existing hepatic dysfunction.
Renal impairment. Cisplatin displays high tissue uptake in the kidneys, exhibits dose related and cumulative nephrotoxicity and is excreted mainly in the urine. In addition, the plasma elimination half-life of cisplatin is prolonged in renal failure.
Caution should be exercised in patients with pre-existing renal dysfunction. Cisplatin is contraindicated in patients with serum creatinine levels greater than 0.2 mmol/L. Repeat courses are not advised until serum creatinine is below 0.14 mmol/L and/or blood urea below 9 mmol/L.
Administration. (a) Pretreatment hydration. Patients should be adequately hydrated before and for 24 hours after administration of cisplatin to ensure good urinary output and minimise nephrotoxicity. Hydration may be achieved by IV infusion of 2 litres of either sodium chloride IV infusion 0.9% or glucose-saline (e.g. glucose 4% in one-fifth sodium chloride IV infusion 0.9%) over a 2 hour period. During the last 30 minutes of the pretreatment hydration or after the hydration, 375 mL of 10% mannitol injection may be administered via a side-arm drip.
(b) Preparation of cisplatin infusion. DBL Cisplatin Injection should be added to 1 litre of sodium chloride IV infusion 0.9%.
Aluminium containing equipment should not be used for administration of cisplatin (see Section 6.2 Incompatibilities).
(c) Treatment. Following prehydration, administer the cisplatin infusion over 1-2 hours. It has been proposed that a longer infusion time of 6-8 hours may decrease gastrointestinal and renal toxicities.
The IV flask should be covered to preclude light.
(d) Post-treatment hydration. Adequate hydration and urinary output must be maintained during the 24 hours following infusion. It has been suggested that IV hydration continue after treatment with the aim to administer 2 litres of sodium chloride IV infusion 0.9% or glucose-saline over a period of 6-12 hours.
The product and its admixtures contain no antimicrobial agent. In order to reduce microbiological hazards it is recommended that further dilution be effected immediately prior to use and infusion commenced as soon as practicable after preparation of the admixture. Infusion should be completed within 24 hours of preparation and the residue discarded.
Handling precautions. As with all antineoplastic agents, trained personnel should prepare cisplatin injection. This should be performed in a designated area (preferably a cytotoxic laminar flow cabinet). Care should be taken to prevent inhaling particles and exposing the skin to cisplatin. Protective gown, mask, gloves and appropriate eye protection should be worn while handling cisplatin. In the event of contact with the eyes, wash with water or saline; where solution accidentally contacts skin or mucosa, the affected area should be immediately washed thoroughly with soap and water and in both cases seek medical advice. Seek immediate medical attention if the drug is ingested or inhaled. It is recommended that pregnant personnel not handle cytotoxic agents such as cisplatin.
Luer-Lock fitting syringes and giving sets to avoid leakage are recommended. Large bore needles are recommended to minimise pressure and possible formation of aerosols. Aerosols may also be reduced by using a venting needle during preparation.
Items used to prepare cisplatin, or articles associated with body waste should be disposed of by placing in a double sealed polythene bag, and incinerating at 1100°C.
Spills and disposal. If spills occur, restrict access to the affected area. Wear two pairs of gloves (latex rubber), a respirator mask, a protective gown and safety glasses. Limit the spread of the spill by covering with a suitable material such as absorbent towel or adsorbent granules. Spills may also be treated with 5% sodium hypochlorite. Collect up absorbent/adsorbent material and other debris from spill and place in a leak proof plastic container and label accordingly.
Cleanse the remaining spill area with copious amounts of water.
4.3 Contraindications
Use of cisplatin is contraindicated in the following conditions:
Renal impairment (see Section 4.2 Dose and Method of Administration);
Hearing disorders (see Section 4.4 Special Warnings and Precautions for Use, Ototoxicity);
Bone marrow depression;
Generalised infections;
During pregnancy or lactation;
In patients with a history of hypersensitivity to cisplatin or platinum-containing compounds.
4.4 Special Warnings and Precautions for Use
Cisplatin is a highly toxic drug with a relatively narrow therapeutic index, and a therapeutic effect is unlikely to occur without some evidence of toxicity. Cisplatin should be administered only under constant supervision by physicians experienced in therapy with cytotoxic agents and only when potential benefits of cisplatin therapy outweigh the possible risks. Appropriate facilities should be available for adequate management of complications should they arise.
To minimise the risk of nephrotoxicity, hydrate before, during and after therapy (see Section 4.2 Dose and Method of Administration). Prior to initial therapy, then before subsequent doses, the following parameters should be monitored: renal function including glomerular filtration rate (GFR), blood urea nitrogen (BUN), serum creatinine and creatinine clearance; electrolytes (magnesium, sodium, potassium and calcium) to detect hypomagnesaemia or hypocalcaemia; auditory function; red blood cells, white blood cells and platelets; liver function and neurological status.
Ototoxicity. Ototoxicity is cumulative and occurs mainly with high dose regimes. Tinnitus or occasional decreased ability to hear normal conversation are indications of ototoxicity which have been frequently observed. Abnormalities of audiometric testing are more common and hearing loss can be unilateral or bilateral; frequency and severity increase with repeated doses and may not be reversible, but mostly occur in the 4,000 - 8,000 Hz range.
Audiometric testing should be performed, if possible, prior to initiation of therapy, and at regular intervals thereafter, particularly if the clinical symptoms of tinnitus or hearing impairment occur. Radiotherapy may enhance ototoxicity. Clinically important deterioration of auditive function may require dosage modifications or discontinuation of therapy.
Myelosuppression. This may occur in patients treated with cisplatin. Haematological toxicity is dose-related and cumulative. The nadirs in circulating platelets and leucocytes generally occur between days 18 - 23 (range 7.3 - 45) with most patients recovering by day 39 (range 13 - 62). Leucopenia and thrombocytopenia are more pronounced at doses greater than 50 mg/m2.
Peripheral blood counts should be monitored frequently in patients receiving cisplatin. Although the haematologic toxicity is usually moderate and reversible, severe thrombocytopenia and leucopenia may occur. In patients who develop thrombocytopenia special precautions are recommended: care in performing invasive procedures; search for signs of bleeding or bruising; test of urine, stools and emesis for occult blood; avoiding aspirin and other NSAIDs. Patients who develop leucopenia should be observed carefully for signs of infection and might require antibiotic support and blood product transfusions.
Subsequent courses of cisplatin should not be instituted until platelets are present at levels greater than 100,000/mm3 and white cells greater than 4,000/mm3.
Anaemia. Anaemia (decrease of greater than 2 g/dL haemoglobin) occurs in a significant number of patients, usually after several courses of treatment. Anaemia occurs at approximately the same frequency but generally with a later onset than leucopenia and thrombocytopenia. Transfusions of packed red cells may be necessary in severe cases.
Rarely, the drug has caused haemolytic anaemia; Coombs-positive results have been reported in a few of these cases. Further courses with cisplatin in sensitised individuals may cause increased haemolysis.
A high incidence of severe anaemia requiring transfusion of packed red cells has been observed in patients receiving combination chemotherapy including cisplatin.
Nausea and vomiting. Marked nausea and vomiting occur in almost all patients treated with cisplatin and are occasionally so severe that dosage reduction or discontinuance of treatment is necessary.
Anaphylaxis. Reactions, secondary to cisplatin therapy, have been occasionally reported in patients who were previously exposed to cisplatin. Patients who are at particular risk are those with a prior history or family history of atopy. Facial oedema, wheezing, tachycardia, hypotension and skin rashes of urticarial non-specific maculopapular type can occur within a few minutes of administration. Serious reactions seem to be controlled by IV adrenaline, corticosteroids or antihistamines.
Patients receiving cisplatin should be observed carefully for possible anaphylactic-like reactions, and the necessary supportive equipment and medication should be readily available to treat such reactions.
Cardiovascular toxicity. Cisplatin has been found to be associated with cardiovascular toxicity (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients may experience clinically heterogeneous venous thromboembolic events, myocardial infarction, cerebrovascular accidents, thrombotic microangiopathy and cerebral arteritis. Cases of pulmonary embolism (including fatalities) have been reported (see Section 4.8 Adverse Effects (Undesirable Effects)).
Hypomagnesaemia and hypocalcaemia. Hypomagnesaemia occurs quite frequently with cisplatin administration, while hypocalcaemia occurs less frequently. The loss of magnesium seems to be associated with renal tubular damage which prevents resorption of this cation. Where both electrolytes are deficient, tetany may result. It does not appear to be dose related. Monitoring of electrolytes is necessary.
Neurotoxicity and seizures. Cisplatin is known to induce neurotoxicity; therefore, neurologic examination is warranted in patients receiving a cisplatin-containing treatment. Peripheral neuropathy, postural hypotension, myasthenic syndromes, seizures and visual loss may occur with cisplatin treatment. This appears to be more common after prolonged treatment. Since neurotoxicity may result in irreversible damage, the development of clinically significant symptoms should generally contraindicate further cisplatin usage.
Immunosuppressant effects/increased susceptibility to infections. Administration of live or live-attenuated vaccines in patients immunocompromised by chemotherapeutic agents including cisplatin, may result in serious or fatal infections. Extreme caution should be used where patients have recently been exposed to infections, particularly chicken pox and herpes zoster. Vaccination with a live vaccine should be avoided in patients receiving cisplatin. Killed or inactivated vaccines may be administered; however, the response to such vaccines may be diminished.
Dental. The bone marrow depressant effects of cisplatin may result in an increased incidence of microbial infection, delayed healing and gingival bleeding. Dental work should be avoided during cisplatin therapy.
Others. As patients undergoing treatment with cisplatin are at an increased risk of bleeding, bruising and infection, it is recommended that extreme care be used when performing necessary invasive procedures.
Alcohol and aspirin should be avoided because of the risk of gastrointestinal bleeding.
Use in hepatic impairment. Liver function should be monitored periodically.
Use in renal impairment. Cisplatin is contraindicated in patients with renal impairment (see Section 4.3 Contraindications).
Cumulative and dose-related renal insufficiency is the major dose-limiting toxicity of cisplatin. The most commonly observed change in renal function has been a fall in glomerular filtration rate reflected by a rise in serum creatinine and a reduction in effective renal plasma flow.
Pre and post treatment hydration may reduce nephrotoxicity (see Section 4.2 Dose and Method of Administration).
Renal function must return to normal before further doses are given (see Section 4.2 Dose and Method of Administration).
Special care has to be taken when cisplatin-treated patients are given concomitant therapies with other potentially nephrotoxic drugs (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in the elderly. No data available.
Paediatric use. Cisplatin can also be used in children. Cases of delayed-onset hearing loss have been reported in the paediatric population. Long-term follow-up in this population is recommended.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Cisplatin is mostly used in combination with antineoplastic drugs having similar cytotoxic effects. In these circumstances additive toxicity is likely to occur.
Other known drug interactions are reported below.
Nephrotoxic drugs. Potentially nephrotoxic medicines, e.g. aminoglycoside antibiotics and loop diuretics when given concurrently or within 1-2 weeks after cisplatin administration, may potentiate the nephrotoxic effects of cisplatin. Concomitant use of other potentially nephrotoxic drugs (e.g. amphotericin B) is not recommended during cisplatin therapy.
The renal toxicity of ifosfamide may be greater when used with cisplatin or in patients who have previously been given cisplatin.
Ototoxic drugs. Concurrent and/or sequential administration of potentially ototoxic medicines, e.g. aminoglycoside antibiotics and loop diuretics, may potentiate the ototoxic effects of cisplatin, especially in the presence of renal impairment.
Ifosfamide may increase hearing loss due to cisplatin.
Renally excreted drugs. Literature data suggest that cisplatin may alter the renal elimination of bleomycin and methotrexate (possibly as a result of cisplatin-induced nephrotoxicity) and enhance their toxicity. Reduction of the lithium blood levels was noticed in a few cases after treatment with cisplatin combined with bleomycin and etoposide. It is therefore recommended to monitor the lithium values.
Antigout agents. Cisplatin may raise the concentration of blood uric acid. Thus, in patients concurrently receiving antigout agents such as allopurinol, colchicine, probenecid or sulfinpyrazone, dosage adjustment of these drugs may be necessary to control hyperuricemia and gout.
Anticonvulsant agents. Plasma levels of anticonvulsant agents may become subtherapeutic during cisplatin therapy. In patients receiving cisplatin and phenytoin, serum concentrations of the latter may be decreased, possibly as a result of decreased absorption and/or increased metabolism. In these patients, serum levels of antiepileptics should be monitored and dosage adjustments made as necessary.
Anticoagulants. It is advisable to check the international normalised ratio (INR) when oral anticoagulants such as coumarins/warfarin are used simultaneously with cisplatin.
Paclitaxel. Administration of cisplatin prior to an infusion with paclitaxel may reduce the clearance of paclitaxel by 33% and can therefore intensify neurotoxicity.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Female. Based on non-clinical and clinical findings, female fertility may be compromised by treatment with cisplatin. Use of cisplatin has been associated with cumulative dose-dependent ovarian failure, premature menopause and reduced fertility. Non-clinical findings in mice treated with cisplatin (5 mg/kg intraperitoneally) showed that cisplatin caused direct damage to primordial follicle oocytes, leading to apoptosis, and ovarian depletion.
Male. Cisplatin can affect male fertility. Impairment of spermatogenesis and azoospermia have been reported (see Section 4.8 Adverse Effects (Undesirable Effects)). Cisplatin caused testis damage and decreased sperm counts in mice, primarily through effects on differentiated spermatogonia. Although the impairment of spermatogenesis can be reversible, males undergoing cisplatin treatment should be warned about the possible adverse effects on male fertility.
Both men and women should seek advice on fertility preservation before treatment.
Use in pregnancy. (Category D)1
The safety of cisplatin in pregnancy has not been established. Cisplatin can cross the placental barrier. In mice and rats, cisplatin is teratogenic and embryotoxic (at clinically relevant doses) and in both species, deformities have been reported. Studies in rodents have shown that exposure during pregnancy can cause tumours in adult offspring. Cisplatin may be toxic to the fetal urogenital tract. Therefore cisplatin is considered to be potentially harmful to the fetus when administered to a pregnant woman and its use in pregnant women is not recommended. Patients should be advised to avoid becoming pregnant.
If the patient becomes pregnant whilst receiving the drug she should be advised of the hazard to the fetus. Cisplatin should only be used if the potential benefits outweigh the risk of therapy.
Women of childbearing potential/contraception in males and females. Women of childbearing potential should use effective contraception during treatment with cisplatin and for at least 7 months following the last dose. Men with female partners of childbearing potential should be advised to use effective contraception during treatment with cisplatin and for at least 4 months after the last dose.
1 Category D: Drugs which have caused, are suspected to caused or be expected to cause, an increased incidence of human fetal malformations or irreversible damage. These drugs may also have adverse pharmacological effects.
Use in lactation. Cisplatin and its active metabolites have been identified in human milk of treated mothers. Advise pregnant women not to breastfeed during treatment with cisplatin and for 1 month following last dose of treatment or to discontinue treatment, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Ear and labyrinth disorders. Unilateral or bilateral tinnitus and/or high frequency hearing loss (> 4000 Hz) has been observed in up to 31% of patients treated with cisplatin and is usually reversible. The damage to the hearing system appears to be dose related and cumulative, and it is reported more frequently in very young or very old patients. Auditory function should be monitored more closely during treatment.
Eye disorders. Retinal toxicity manifests as blurred vision and altered colour perception. Optic neuritis, papilloedema and cortical blindness have been reported rarely following the administration of cisplatin. These events are usually reversible after drug withdrawal. Retinal pigmentation has also been reported.
Infections and infestations. Infection (infectious complications have led to death), sepsis.
Blood and lymphatic system disorders. Thrombotic microangiopathy (haemolytic uraemic syndrome), bone marrow failure, neutropenia, Coombs positive haemolytic anaemia.
Myelosuppression often occurs during cisplatin therapy. Mild bone marrow toxicity may occur with both leucopenia and thrombocytopenia. These effects are usually reversible after ceasing treatment. Cisplatin may also induce anaemia: this is not clearly dose related and is occasionally caused by haemolysis. Leucopenia and thrombocytopenia are dose-related and more pronounced at doses greater than 50 mg/m2. Leucocyte and platelet nadirs generally occur between days 18 and 23 of treatment, with recovery in most patients by day 39. Anaemia occurs at approximately the same frequency.
There have been rare reports of acute myelogenous leukemias and myelodysplastic syndromes arising in patients who have been treated with cisplatin, mostly when given in combination with other potentially leukemogenic agents.
Immune system disorders. Anaphylactic and anaphylactic-like reactions, consisting principally of flushing, facial oedema, wheezing, tachycardia and hypotension have been reported in patients previously exposed to cisplatin. The reactions usually occur within a few minutes of cisplatin administration and may be controlled by IV adrenaline, corticosteroids and/or antihistamines.
Metabolism and nutritional disorders. Cisplatin may cause dehydration in patients. Cisplatin may also cause serious electrolyte disturbances, mainly represented by hypomagnesemia, hypocalcemia, and hypokalemia, and associated with renal tubular dysfunction. Hypomagnesemia and/or hypocalcemia may become symptomatic, with muscle irritability or cramps, clonus, tremor, carpopedal spasm, and/or tetany. Hypomagnesaemia and hypocalcaemia may develop during cisplatin therapy or following discontinuance of the drug. Other reported toxicities are hyperuricemia, hyponatremia, hypophosphataemia and syndrome of inappropriate antidiuretic hormone (SIADH). Hyperuricaemia may occur in patients receiving cisplatin, principally as a result of drug-induced nephrotoxicity. Hyperuricaemia is more pronounced with doses greater than 50 mg/m2, with peak levels occurring between 3-5 days after administration of the drug. Allopurinol may be used to reduce serum uric acid levels. Regular monitoring of serum electrolyte levels and replacement where necessary are advisable.
Nervous system disorders. Convulsion, leukoencephalopathy, reversible posterior leukoencephalopathy syndrome, haemorrhagic stroke, ischaemic stroke, ageusia, cerebral arteritis, myelopathy.
Peripheral neuropathies occur infrequently with usual doses of the drug. They are generally sensory in nature (e.g. paraesthesia of the upper and lower extremities), but can also include motor difficulties, reduced or absent reflexes and leg weakness. Autonomic neuropathy, seizures, slurred speech, loss of taste and memory loss have also been reported. These neuropathies usually appear after prolonged therapy, but have also developed after a single drug dose. Areflexia and loss of proprioception and vibratory sensation may be seen, especially if cisplatin is given at higher doses or more frequently than recommended. In some patients they may be irreversible however, they have been partially or completely reversible in others following discontinuance of cisplatin therapy. Cerebrovascular accident has been reported in patients treated with cisplatin. Lhermitte's sign has been reported.
Cardiac disorders. Cardiovascular abnormalities (coronary disease, congestive heart failure, postural hypotension, thrombotic microangiopathy, arrhythmia, bradycardia, tachycardia, cardiac arrest, cardiac disorder etc.)
Vascular disorders. Raynaud's phenomenon.
Venous thromboembolism: a significant increase in the risk of venous thromboembolic events has been reported in patients with advanced solid tumours and treated with cisplatin compared with non-cisplatin-based chemotherapy.
Vascular toxicity coincident with the use of cisplatin in combination with other antineoplastic agents have been reported rarely. The events are clinically heterogeneous and may include myocardial infarction, cerebrovascular accident (haemorrhagic and ischaemic stroke), thrombotic microangiopathy (haemolytic uremic syndrome) or cerebral arteritis. Various mechanisms have been proposed for these vascular complications.
Respiratory, thoracic and mediastinal disorders. Pulmonary embolism.
Pulmonary toxicity has been reported in patients treated with cisplatin in combination with bleomycin or 5-fluorouracil.
Gastrointestinal disorders. Stomatitis, vomiting, nausea, anorexia, hiccups, diarrhoea.
Cisplatin induces severe nausea and vomiting in almost all patients. Severe nausea and vomiting usually begin within 1-4 hours after treatment and may persist for up to a week after treatment. These side effects are only partially relieved by standard antiemetics. The severity of these symptoms may be reduced by dividing the total dose per cycle into smaller doses given once daily for five days. Reported toxicity includes gingival platinum line.
Hepatobiliary disorders. Mild and transient elevations of serum AST and ALT levels may occur infrequently. Liver damage has also been infrequently reported.
Skin and subcutaneous tissue disorders. Mild alopecia. Rarely, urticarial or maculopapular skin rashes have also been observed.
Musculoskeletal and connective tissue disorders. Myalgia, muscle spasms.
Renal and urinary disorders. Acute renal toxicity, which was highly frequent in the past and represented the major dose-limiting toxicity of cisplatin, has been greatly reduced by the use of 6 to 8-hour infusions as well as by concomitant intravenous hydration and forced diuresis. Cumulative toxicity, however, remains a problem and may be severe. Renal impairment, which is associated with tubular damage, may be first noted during the second week after a dose and is manifested by an increase in serum creatinine, BUN, serum uric acid and/or a decrease in creatinine clearance. Renal insufficiency is generally mild to moderate and reversible at the usual doses of the drug (recovery occurring as a rule within 2-4 weeks); however, high or repeated cisplatin doses can increase the severity and duration of renal impairment and may produce irreversible renal insufficiency (sometimes fatal). Renal failure has been reported also following intraperitoneal instillation of the drug.
Reproductive system and breast disorders. Impairment of spermatogenesis and azoospermia have been reported (see Section 4.6 Fertility, Pregnancy and Lactation).
General disorders and administration site conditions. Pyrexia, asthenia, malaise. Local effects such as pain, oedema, erythema, phlebitis, tissue cellulitis, fibrosis, and skin necrosis (following extravasation of the drug) may also occur. Extravasation may result from infusion of solutions greater than 0.5 mg/mL cisplatin.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Acute overdosage with cisplatin may result in an enhancement of its expected toxic effects (e.g. kidney failure, severe myelosuppression, intractable nausea and vomiting, severe neurosensorial toxicities, liver failure etc.). Death may also occur. No proven antidotes are known for cisplatin overdosage. Haemodialysis is only effective, even then partially, up to 3 hours after administration because of the rapid and extensive binding of platinum to plasma proteins. Signs and symptoms of overdosage should be managed with supportive measures. Patients should be monitored for 3 to 4 weeks in case of delayed toxicity. See Section 4.8 Adverse Effects (Undesirable Effects) for possible complications.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Class. Antineoplastic agent.
Mechanism of action. Cisplatin is a platinum compound of which only the cis-isomer is active and has biochemical properties similar to those of bifunctional alkylating agents. The drug inhibits DNA synthesis by producing intrastrand and interstrand crosslinks in DNA. Protein and RNA synthesis are also inhibited to a lesser extent. Cisplatin does not appear to be cell cycle specific.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Distribution. There is good uptake of cisplatin by the kidneys, liver and intestine.
More than 90% of platinum containing species remaining in the blood are bound (possibly irreversibly) to plasma proteins.
The clearance of total platinum from plasma is rapid during the first four hours after intravenous administration, but then proceeds more slowly because of covalent binding to serum proteins. Levels of unbound platinum fall with a half-life of 20 minutes to 1 hour depending on the rate of drug infusion.
Excretion. The elimination of intact drug and various platinum containing biotransformation products is via the urine. About 15 - 25% of administered platinum is rapidly excreted in the first 2 - 4 hours after administration of cisplatin. This early excretion is mostly of intact cisplatin. In the first 24 hours after administration, 20 - 80% is excreted, the remainder representing drug bound to tissues or plasma protein.
Studies aiming at determining plasma elimination half-life of total platinum have shown a very large interindividual and interstudy variation. Most studies reported a half-life of total plasma platinum post cisplatin treatment of approximately 5 days or longer.
5.3 Preclinical Safety Data
Genotoxicity. Cisplatin has been shown to be genotoxic in vitro, in bacterial gene mutation assays, gene mutation assays in yeast (Saccharomyces cerevisiae D7) and mammalian cells (mouse lymphoma cells and Chinese hamster cells), in vitro chromosome aberration assays (in Chinese hamster cells and in human lymphocytes), in vitro DNA repair assays (Saccharomyces cerevisiae D7 and v79 Chinese hamster cells) and in vivo in a chromosome aberration assay in mouse bone marrow cells. Based on these studies, cisplatin is considered to present a genotoxic risk to humans.
Carcinogenicity. No formal carcinogenicity studies were performed. In a transplacental carcinogenicity study, a single IP injection of cisplatin (7.5 mg/kg) to pregnant mice on day 17 of gestation initiated and/or induced thymic lymphomas, lung tumours and proliferative kidney lesions in offsprings at week 25. In another transplacental carcinogenicity study, pregnant rats were given a single IP injection of cisplatin (5 mg/kg) on day 18 of gestation resulted in significantly higher incidences (20/82 in treatment vs 3/75 in control) of hepatocellular adenoma in offspring rats at 79 weeks. Therefore, cisplatin has a high carcinogenic potential in mice and rats.
6 Pharmaceutical Particulars
6.1 List of Excipients
Mannitol, sodium chloride, water for injections.
6.2 Incompatibilities
Cisplatin may interact with aluminium to form a black precipitate. Needles, syringes, catheters or IV administration sets that contain aluminium parts which may come in contact with cisplatin should not be used for preparation or administration of the drug. The stability of cisplatin is adversely affected by the presence of bisulphite, metabisulphite, sodium bicarbonate and fluorouracil.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store between 15°C-25°C. Do not refrigerate. Do not freeze. Protect from light. Single use only. Discard unused portion.
Stability. Cisplatin 0.15 mg/mL in sodium chloride IV infusion 0.9% is chemically stable for 24 hours when stored at room temperature and protected from light.
6.5 Nature and Contents of Container
DBL Cisplatin Injection is available as followed.
Strength and pack size. 50 mg/50 mL: 1 x 50 mL vial.
100 mg/100 mL: 1 x 100 mL vial.
6.6 Special Precautions for Disposal
Cytotoxic waste should be regarded as hazardous or toxic and clearly labelled 'Cytotoxic waste for incineration at 1100°C'. Waste material should be incinerated at 1100°C for at least 1 second.
6.7 Physicochemical Properties
Chemical structure.
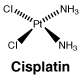
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
31 January 1994
Date of Revision
26 April 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.