DBL Naloxone Hydrochloride Injection
Brand Information
| Brand name | DBL Naloxone Hydrochloride Injection |
| Active ingredient | Naloxone hydrochloride |
| Schedule | S3 | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the DBL Naloxone Hydrochloride Injection.
Summary CMI
DBL™ Naloxone Hydrochloride
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given DBL Naloxone Hydrochloride?
DBL Naloxone Hydrochloride contains the active ingredient naloxone hydrochloride dihydrate. Naloxone is used to reverse the effects of opium-like substances for example, morphine, heroin, methadone or codeine.
For more information, see Section 1. Why am I being given DBL Naloxone Hydrochloride? in the full CMI.
2. What should I know before I am given DBL Naloxone Hydrochloride?
Do not start treatment if you have ever had an allergic reaction to naloxone or any of the ingredients listed at the end of this CMI.
Talk to your doctor if you have an addiction to, or regularly use opioids (including babies of mothers who fall into this category); have heart, lung, kidney or liver disease, or any other medical conditions; take any other medicines; or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given DBL Naloxone Hydrochloride? in the full CMI.
3. What if I am taking other medicines?
Some medicines, including some pain medicines and cold and cough medicines, may interfere with DBL Naloxone Hydrochloride and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is DBL Naloxone Hydrochloride given?
DBL Naloxone Hydrochloride is given as an injection into a muscle, under the skin or as a slow injection into a vein.
Your dose depends on whether you are an adult, child or baby and how you are receiving DBL Naloxone Hydrochloride.
More instructions can be found in Section 4. How is DBL Naloxone Hydrochloride given? in the full CMI.
5. What should I know while I am being given DBL Naloxone Hydrochloride?
| Things you should do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while I am being given DBL Naloxone Hydrochloride? in the full CMI.
6. Are there any side effects?
Symptoms of opioid withdrawal if you or your baby are dependent on opioids.
Side-effects not specific to opioid withdrawal may include: nausea/vomiting; sweating; agitation; headache; light-headedness, dizziness; hyperventilating; tremors; seizures/fits; fast or abnormal heartbeat; coughing up bloody or frothy mucus; coma; heart suddenly stops beating which may lead to death.
Allergic reaction, which may be serious. Symptoms may include: shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, and rash, itching or hives on the skin.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
DBL™ Naloxone Hydrochloride
Active ingredient(s): naloxone hydrochloride dihydrate
Consumer Medicine Information (CMI)
This leaflet provides important information about using DBL Naloxone Hydrochloride.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using DBL Naloxone Hydrochloride.
Where to find information in this leaflet:
1. Why am I being given DBL Naloxone Hydrochloride?
2. What should I know before I am given DBL Naloxone Hydrochloride?
3. What if I am taking other medicines?
4. How is DBL Naloxone Hydrochloride given?
5. What should I know while I am being given DBL Naloxone Hydrochloride?
6. Are there any side effects?
7. Product details
1. Why am I being given DBL Naloxone Hydrochloride?
DBL Naloxone Hydrochloride contains the active ingredient naloxone hydrochloride dihydrate.
It belongs to a group of medicines known as opioid antagonists.
DBL Naloxone Hydrochloride is used to reverse the effects of opium-like substances such as morphine, heroin, methadone or codeine.
It may also be used after surgical operations when powerful pain killers which have been given during the operation are no longer required.
It acts very quickly, within one or two minutes when injected into a vein and can be a life-saving measure in those people who have received an overdose of an opioid-like drug.
DBL Naloxone Hydrochloride is not addictive.
2. What should I know before I am given DBL Naloxone Hydrochloride?
Warnings
You must not be given DBL Naloxone Hydrochloride if:
- you are allergic to naloxone or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Check with your doctor if you:
Have or have had any other medical conditions, especially the following:
- heart disease
- lung disease
- kidney disease
- liver disease
- drug addiction (including babies born to mothers with opioid addiction or regular opioid use)
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Naloxone crosses the placenta and can be absorbed by the unborn baby. This may result in withdrawal symptoms in the baby as well as the mother, if the mother has regularly taken opioids over the course of her pregnancy.
Your doctor or pharmacist will discuss the risks and benefits of being given naloxone during pregnancy.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
It is not known whether naloxone passes into breast milk.
Your doctor or pharmacist will discuss the possible risks and benefits of being given naloxone when breast-feeding.
If you have not told your doctor about any of the above, tell them before you are given this medicine.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with DBL Naloxone Hydrochloride and affect how it works. These include:
- Pain killers (e.g. codeine, buprenorphine or pentazocine)
- Cough and cold remedies
- Heart and blood pressure medication.
These medicines may be affected by DBL Naloxone Hydrochloride or may affect how well it works. You may need different amounts of your medicine, or you may need to take/use different medicines. Your doctor or pharmacist will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect DBL Naloxone Hydrochloride.
4. How is DBL Naloxone Hydrochloride given?
How much is given
For a known or suspected overdose in an adult, an initial dose of 1 to 5 ampoules of DBL Naloxone Hydrochloride may be used. This may be repeated at 2 to 3 minute intervals, up to a total of 25 DBL Naloxone Hydrochloride ampoules being used.
For a known or suspected overdose in a child, medical advice must be obtained to determine how much is given.
Seek emergency services if delivered outside a hospital setting.
In hospital settings, your doctor will decide what dose you will receive and how often.
How it is given
DBL Naloxone Hydrochloride may be given either:
- as an injection into a muscle (intramuscular)
- just under your skin (subcutaneous) or
- as a slow injection into a vein (intravenous).
Injection into a vein is the most common site in an emergency.
If you are given too much DBL Naloxone Hydrochloride
Symptoms of an overdose include the side effects listed below in Section 6. Are there any side effects? but are usually of a more severe nature.
If you think that too much DBL Naloxone Hydrochloride has been given and/or if you experience severe side effects, you should immediately:
- contact your doctor, or
- phone the Poisons Information Centre
(by calling 13 11 26), or - go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
Severe withdrawal symptoms can be produced in patients who are regular users of opioids (or babies born to women who are regularly take opioids) if too much naloxone is used.
Ask your doctor or pharmacist if you have any concerns.
5. What should I know while I am being given DBL Naloxone Hydrochloride?
Things you should do
Remind any doctor, nurse, pharmacist or dentist you visit that you are receiving DBL Naloxone Hydrochloride.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are receiving this medicine.
If you become pregnant while receiving DBL Naloxone Hydrochloride, tell your doctor or pharmacist immediately.
Tell your doctor if you have heart disease, lung disease, kidney disease, liver disease or drug addiction.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly.
Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Driving or using machines
Be careful driving or using any machines or tools until you know how DBL Naloxone Hydrochloride affects you.
Drinking alcohol
Be careful when drinking alcohol while you are receiving this medicine.
Looking after your medicine
DBL Naloxone Hydrochloride will usually be stored in the pharmacy or on the ward.
However, if you need to store DBL Naloxone Hydrochloride, it should be kept in a cool dry place, protected from light by keeping the ampoules in the box, and where the temperature stays below 25°C.
For example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Abrupt reversal of the effects of opium-like substances may result in withdrawal symptoms, which can be serious.
| Side effect | What to do |
The symptoms of opioid withdrawal that may be experienced if you or your baby are dependent on opioids may include:
Additional symptoms in babies may include:
General side effects not specific to opioid withdrawal may include:
| Tell your doctor or nurse or go immediately to Accident and Emergency if you notice any of the following during treatment with DBL Naloxone Hydrochloride. You may need urgent medical attention. |
Symptoms of an allergic reaction may include:
| Tell your doctor or nurse immediately if you notice any of the following These may be more serious side effects. You may need urgent medical attention. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/safety/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop treatment with any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What DBL Naloxone Hydrochloride contains
| Active ingredient (main ingredient) | Naloxone hydrochloride dihydrate |
| Other ingredients (inactive ingredients) | Hydrochloric acid Sodium Chloride Water for Injections |
Do not use this medicine if you are allergic to any of these ingredients.
DBL Naloxone Hydrochloride does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
What DBL Naloxone Hydrochloride looks like
DBL Naloxone Hydrochloride is a clear, colourless solution.
It should not be given if there are any crystals or particles visible in the solution.
DBL Naloxone Hydrochloride is available as follows:
- 400 microgram/mL, 5 x 1 mL ampoules
(AUST R 16282)
Who distributes DBL Naloxone Hydrochloride
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in September 2025.
™ Trademark
Brand Information
| Brand name | DBL Naloxone Hydrochloride Injection |
| Active ingredient | Naloxone hydrochloride |
| Schedule | S3 | S4 |
MIMS Revision Date: 01 May 2023
1 Name of Medicine
Naloxone hydrochloride dihydrate.
2 Qualitative and Quantitative Composition
DBL Naloxone Hydrochloride contains 400 micrograms/1 mL naloxone hydrochloride dihydrate and sodium chloride in water for injections. The preparation has a pH of approximately 3.5.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
DBL Naloxone Hydrochloride Injection is a sterile, clear, colourless solution, free from visible particulates.
Naloxone is a semi-synthetic opioid antagonist which differs structurally from oxymorphone only in that the methyl group on the nitrogen atom of oxymorphone is replaced by an allyl group.
Naloxone hydrochloride dihydrate is 17-allyl-4,5α-epoxy- 3,14-dihydroxymorphinan-6-one hydrochloride.
Naloxone hydrochloride dihydrate occurs as a white to slightly off-white powder and is soluble in water, dilute acids and strong alkalis and is slightly soluble in alcohol. It is practically insoluble in ether or chloroform.
4 Clinical Particulars
4.1 Therapeutic Indications
Naloxone is indicated for the complete or partial reversal of opioid depression, including respiratory depression, induced by natural and synthetic opioids including propoxyphene, methadone, codeine, morphine and heroin, and the mixed opioid agonist-antagonist analgesics such as pentazocine. Naloxone is also indicated for the diagnosis of suspected acute opioid overdosage.
4.2 Dose and Method of Administration
Dosage. Use in adults. Opioid overdosage - known or suspected. An initial dose of 400 microgram (0.4 milligrams) to 2 milligrams of naloxone hydrochloride may be administered intravenously. This dose may be repeated at 2 to 3 minute intervals, if necessary. If no response is seen after a total of 10 milligrams has been administered, the diagnosis of opioid-induced or partial opioid-induced toxicity should be questioned. If the intravenous route is not available, the medicine may be administered by intramuscular or subcutaneous injection.
Post-operative opioid depression. For the partial reversal of opioid-induced depression following the use of opioids during surgery, smaller doses of naloxone are usually sufficient. The dose should be titrated according to the patient's response. The usual initial dosage is 100 to 200 microgram (0.1 to 0.2 milligrams) administered intravenously at 2 to 3 minute intervals until the desired degree of reversal is achieved, i.e. adequate ventilation and alertness without significant pain or discomfort. Doses of naloxone larger than necessary may result in significant reversal of analgesia and increase in blood pressure. Too rapid reversal may induce nausea, vomiting, sweating or circulatory stress.
Repeat doses of naloxone may be required within one or two hours, depending on the type and amount of opioid administered and the time interval since the last dose. Supplemental intramuscular doses have been shown to produce a longer lasting effect.
Use in children. Opioid overdose - known or suspected. The usual initial dose in children is 10 microgram (0.01 milligrams) per kg body weight given intravenously. If the desired degree of clinical improvement is not seen, a subsequent dose of 100 microgram (0.1 milligrams) per kg may be administered. If the intravenous route is not available, naloxone may be administered by intramuscular or subcutaneous injection in divided doses. If necessary, naloxone can be diluted with sterile water for injections.
Post-operative opioid depression. Follow the recommendations and cautions under "adult post-operative opioid depression". In children the initial dose of naloxone hydrochloride for reversal of respiratory depression should be in increments of 5 to 10 microgram (0.005 to 0.01 milligrams) administered intravenously at 2 to 3 minute intervals until the desired degree of reversal is achieved.
Use in neonates. Opioid-induced depression. The usual initial dose is 10 microgram (0.01 milligrams) per kg body weight administered by intravenous, intramuscular or subcutaneous injection. This dose may be repeated in accordance with the adult administration guidelines for post-operative opioid depression.
Method of administration. DBL Naloxone Hydrochloride Injection may be administered by the intravenous, intramuscular or subcutaneous route. Intravenous administration is recommended in emergency situations, as this route of administration provides the most rapid onset of action.
The duration of action of naloxone may be shorter than that of some opioids. Therefore, the patient should be kept under continued surveillance and repeated doses of naloxone should be administered as necessary.
Continuous intravenous infusion of DBL Naloxone Hydrochloride Injection may be the most appropriate method of administration for patients who require higher doses, or who continue to experience respiratory or central nervous system (CNS) depression after effective therapy with repeated doses, and/or in whom the effects of long acting opioids are being antagonised. For continuous intravenous infusion, 2 milligrams of naloxone hydrochloride may be diluted in 500 mL of sodium chloride 0.9% or glucose 5% injection to produce a solution containing 4 microgram/mL.
DBL Naloxone Hydrochloride Injection should not be mixed with preparations containing bisulfite, metabisulfite, long-chain or high molecular weight anions, or any solution having an alkaline pH. No medicine or chemical agent should be added to naloxone unless its effect on the chemical and physical stability has first been established. Prior to administration, intravenous solutions of DBL Naloxone Hydrochloride Injection should be visually inspected for the presence of particles or discolouration. Diluted solutions of the medicine should be used within 24 hours of preparation and any unused portion discarded after this time.
4.3 Contraindications
Naloxone hydrochloride dihydrate is contraindicated in patients with known hypersensitivity to naloxone or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Naloxone should be administered with caution to patients who have received large doses of opioids, or who are known or suspected to be physically dependent on opioids (including neonates born to women who are opioid dependent), because the drug may precipitate severe withdrawal symptoms.
The signs and symptoms of opioid withdrawal in a patient physically dependent on opioids may include, but are not limited to, the following: body aches, diarrhoea, tachycardia, fever, runny nose, sneezing, pilo-erection, sweating, yawning, nausea or vomiting, nervousness, restlessness or irritability, shivering or trembling, abdominal cramps, weakness, and increased blood pressure.
In the neonate, opioid withdrawal may also include: convulsions, excessive crying, and hyperactive reflexes.
Patients who have satisfactorily responded to naloxone should be carefully monitored since the duration of action of some opioids may exceed that of naloxone. Repeated doses of naloxone should be administered when necessary.
Naloxone is not effective against respiratory depression caused by non-opioid drugs. Reversal of respiratory depression by partial agonists or mixed agonist/antagonists, such as buprenorphine and pentazocine, may be incomplete or require higher doses of naloxone (also see Section 4.2 Dose and Method of Administration). If an incomplete response occurs, respirations should be mechanically assisted as clinically indicated.
When naloxone is used in the management of acute opioid overdosage, other resuscitative measures such as maintenance of a free airway, artificial ventilation, cardiac massage and vasopressor agents should be readily available and used when necessary.
Several instances of hypotension, hypertension, ventricular tachycardia and fibrillation, pulmonary oedema, and cardiac arrest have been reported in post-operative patients following naloxone administration. Death, coma, and encephalopathy have been reported as sequelae of these events. Although a direct cause and effect relationship has not been established, naloxone should be used with caution in patients with pre-existing cardiac disease or patients who have received potentially cardiotoxic drugs, since serious cardiovascular effects, such as hypotension, hypertension, ventricular tachycardia or fibrillation, pulmonary oedema and cardiac arrest have occurred. It has been suggested that the pathogenesis of pulmonary oedema associated with the use of naloxone is similar to neurogenic pulmonary oedema i.e. a centrally mediated massive catecholamine response leading to a dramatic shift of blood volume into the pulmonary vascular bed resulting in increased hydrostatic pressures.
Naloxone should also be used with caution in patients with pre-existing pulmonary disease, since sudden exacerbation of underlying pulmonary disease may occur.
Abrupt post-operative reversal of opioid depression may result in nausea, vomiting, sweating, tremulousness, hyperventilation, tachycardia, increased blood pressure, seizures, ventricular tachycardia and fibrillation, pulmonary oedema, and cardiac arrest which may result in death.
Use in hepatic impairment. The safety and effectiveness of naloxone in patients with hepatic disease have not been established in well controlled clinical trials. In one small study in patients with hepatic cirrhosis, plasma naloxone concentrations were approximately six times higher than in patients without hepatic disease. Caution should be exercised when naloxone is administered to patients with hepatic disease.
Use in renal impairment. The safety and effectiveness of naloxone in patients with renal insufficiency/failure have not been established in well controlled clinical trials. Caution should be exercised when naloxone is administered to this patient population.
Use in the elderly. No data available.
Paediatric use. Naloxone should be given with caution to patients who are known or suspected to be physically dependent on opioids (including neonates born to women who are opioid dependent), because the drug may precipitate the onset of severe withdrawal symptoms.
Effects on laboratory tests. None known.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Naloxone reverses the analgesic and other effects of opioid agonist-antagonists such as pentazocine, so may precipitate withdrawal symptoms if used concurrently with these medicines in physically dependent patients.
High doses of naloxone may be required to antagonize buprenorphine since the latter has a long duration of action due to its slow rate of binding and subsequent slow dissociation from the opioid receptor. Buprenorphine antagonism is characterized by a gradual onset of the reversal effects and a decreased duration of action of the normally prolonged respiratory depression.
Naloxone reverses the analgesic and other effects of opioid agonist analgesics, and may precipitate withdrawal symptoms if used concurrently with these medicines in physically dependent patients, including patients receiving methadone to treat opioid dependence.
When naloxone is used post-operatively to reverse the central depressive effects of opioid agonists used as anaesthesia adjuncts, the dose of naloxone must be carefully titrated to achieve the desired effect without interfering with control of post-operative pain, or causing other adverse effects.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No evidence for impaired fertility was observed in a reproductive study in male or female rats given naloxone at doses 8 times the highest recommended human dose (based on body surface area).
Use in pregnancy. (Category B1)
Category B1: Drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus being observed. Studies in animals have not shown evidence of an increased occurrence of foetal damage.
There are no adequate and controlled studies of naloxone in pregnant women. Naloxone should be administered to a pregnant woman only when, in the judgment of the physician, the potential benefits outweigh the possible hazards. Caution should be used when administering naloxone to a pregnant woman who is known to be opioid dependent, since maternal dependence may often be accompanied by foetal dependence. Naloxone crosses the placenta and may precipitate withdrawal in the foetus as well as in the mother.
Reproduction studies in mice and rats at doses of 4 to 8 times the highest recommended human dose (based on body surface area) revealed no evidence of harm to the foetus.
Use in lactation. It is not known whether naloxone is excreted in human milk. Therefore, naloxone should be used with caution in breastfeeding women.
4.7 Effects on Ability to Drive and Use Machines
The effect of naloxone on the ability to drive or use machines has not been systematically evaluated.
4.8 Adverse Effects (Undesirable Effects)
The adverse effects are listed within each system organ class (SOC). See Table 1.
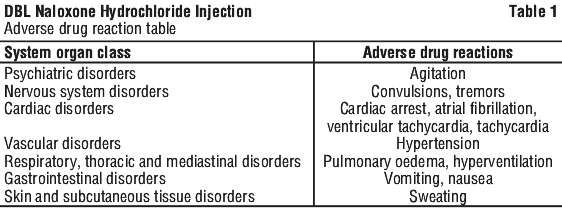
Opioid depression. Abrupt reversal of opioid depression may result in nausea, vomiting, sweating, tachycardia, increased blood pressure, hyperventilation, tremulousness, seizures, ventricular tachycardia and fibrillation, pulmonary oedema, and cardiac arrest which may result in death (see Section 4.4 Special Warnings and Precautions for Use).
Opioid dependence (see Section 4.4 Special Warnings and Precautions for Use). Naloxone may precipitate severe withdrawal symptoms in patients known or suspected to be physically dependent on opioids (including neonates born to women who are opioid dependent) (see Section 4.4 Special Warnings and Precautions for Use). Agitation and paraesthesias have been infrequently reported with the use of naloxone. Seizures have occurred rarely following administration of naloxone, however, a causal relationship has not been established. Violent behaviour, nervousness, restlessness, excitement and irritability may also occur.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Symptoms of overdosage would be expected to be similar to the effects seen with therapeutic use (see Section 4.8 Adverse Effects (Undesirable Effects)).
Treatment. Treatment of overdosage is symptomatic and supportive.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Naloxone is essentially a pure opioid antagonist, it has little or no agonistic activity. Naloxone is thought to act as a competitive antagonist at mu, kappa, and sigma opioid receptors in the central nervous system (CNS), although the precise mechanism of action has not been fully determined. Naloxone prevents or reverses the effects of opioids, including respiratory depression, sedation and hypotension. Naloxone can also reverse the psychotomimetic and dysphoric effects of agonist-antagonists such as pentazocine, but higher doses are required. One milligram of naloxone intravenously completely blocks the effects of 25 milligrams of diacetylmorphine (heroin).
When administered in usual doses to patients who have not recently received opioids, naloxone exerts little or no pharmacological effect. Even extremely high doses (10 times the usual therapeutic dose) produce insignificant analgesia, only slight drowsiness, and no respiratory depression, psychotomimetic effects, circulatory changes or miosis.
Naloxone does not produce tolerance or physical or psychological dependence.
Parenteral administration of naloxone will produce withdrawal symptoms in patients physically dependent on opioids or pentazocine.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Naloxone has an onset of action within 1 to 2 minutes following intravenous administration and within 2 to 5 minutes following subcutaneous or intramuscular administration. The duration of action depends on the dose and route of administration and is more prolonged following intramuscular administration than after intravenous administration. The duration of action is reported to be up to several hours but the practical duration is probably 1 hour or less. Repeat doses of naloxone may be required depending on the amount, type and route of administration of the opioid being antagonised, as well as the duration of action of naloxone.
Distribution. Following parenteral administration, naloxone is rapidly distributed into body tissues and fluids.
Metabolism. Naloxone is rapidly metabolised in the liver, principally by conjugation with glucuronic acid.
Excretion. Naloxone is 50% protein-bound and is excreted in the urine. The mean plasma half-life of naloxone has been reported to be about 60 minutes in adults with a range of from about 30 to 80 minutes, and about 3 hours in neonates.
5.3 Preclinical Safety Data
Genotoxicity. In assays for gene mutations, naloxone was positive in the Ames test but negative in the mouse lymphoma assay. Naloxone was also positive in an assay for chromosomal damage (human lymphocytes in vitro).
Carcinogenicity. Studies in animals to assess the carcinogenic potential of naloxone have not been conducted.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hydrochloric acid, sodium chloride, water for injections.
6.2 Incompatibilities
Naloxone hydrochloride solution for injection should not be mixed with preparations containing bisulfites, metabisulfites, long chain or high molecular weight anions, or those with an alkaline pH.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Protect from light. Store below 25°C.
6.5 Nature and Contents of Container
DBL Naloxone Hydrochloride Injection is available in ampoules: 400 micrograms naloxone hydrochloride/1 mL.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
Its chemical structure is shown below:
CAS number.CAS registry number is 357-08-4.
7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Only Medicine).
Date of First Approval
08 October 1991
Date of Revision
27 November 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.