Demazin Original Cold + Flu Relief Day + Night
Brand Information
| Brand name | Demazin Original Cold + Flu Relief Day + Night |
| Active ingredient | Paracetamol + Pseudoephedrine hydrochloride, Paracetamol + Pseudoephedrine hydrochloride + Chlorphenamine maleate (chlorpheniramine maleate) |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Demazin Original Cold + Flu Relief Day + Night.
Full CMI
Demazin® Original Cold + Flu Relief Day + Night
Active ingredients: Day Tablet: Paracetamol, Pseudoephedrine hydrochloride
Night tablet: Paracetamol, Pseudoephedrine hydrochloride, Chlorphenamine maleate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Demazin Original Cold + Flu Relief Day + Night tablets. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Demazin.
Where to find information in this leaflet:
1. Why am I taking Demazin?
2. What should I know before I take Demazin?
3. What if I am taking other medicines?
4. How do I take Demazin?
5. What should I know while taking Demazin?
6. Are there any side effects?
7. Product details
1. Why am I taking Demazin?
Demazin contains two different tablets. The white DAY tablet contains the active ingredients paracetamol and pseudoephedrine hydrochloride. The pink NIGHT tablet contains paracetamol, pseudoephedrine hydrochloride and chlorphenamine maleate.
Demazin is a cold and flu medicine. The Day tablets contain a decongestant and pain reliever. The Night tablet contains a decongestant, pain reliever and antihistamine. Pseudoephedrine hydrochloride is a decongestant and is used to relieve nasal congestion and runny nose. It works by reducing congestion in the upper respiratory tract, including the nose, nasal passages and sinuses, and making it easier to breathe.
Paracetamol is a pain reliever and works by blocking the pain messages from getting through to the brain. It also acts in the brain to reduce fever.
Chlorphenamine maleate is an antihistamine. Antihistamines help reduce allergic symptoms such as sneezing and itchy watery eyes by preventing the effects of a substance called histamine. Histamine is produced by the body in response to foreign substances that the body is allergic to.
Demazin is used to provide temporary relief from the following symptoms of cold and flu:
- runny nose
- sinus pain
- blocked nose and sinus
- fever
- headache
- body aches and pain.
The NIGHT tablet also aids rest by providing relief from:
- watery, itchy eyes
- sneezing.
2. What should I know before I take Demazin?
Warnings
Do not take Demazin if you:
- are allergic to paracetamol, pseudoephedrine, chlorphenamine, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - have any of the following conditions:
- very high blood pressure
- severe coronary artery disease (heart disease caused by poor blood flow or narrowing of the blood vessels of the heart)
- narrow angle glaucoma (high pressure in the eyes)
- stomach obstruction, including due to a peptic ulcer
- enlarged prostate
- bladder obstruction. - are taking a medicine for depression known as a monoamine oxidase inhibitor (MAOI) or if you have taken one in the past 14 days.
Check with your doctor or pharmacist if you:
- have any other medical conditions including:
- high blood pressure
- overactive thyroid
- diabetes mellitus
- heart disease
- glaucoma
- enlarged prostate
- epilepsy
- impaired kidney or liver function. - take any medicines for any other condition, especially anti-depressant medication.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Your doctor can discuss the risks and benefits of taking Demazin during pregnancy.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Demazin should not be used when breastfeeding. Paracetamol, pseudoephedrine and chlorphenamine pass into breast milk.
Use in children
- Demazin is not recommended for children under 12 years of age.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket, or health food shop.
Some medicines may interfere with Demazin and affect how it works. These include:
- Medicines which may increase blood pressure including:
- Tricyclic antidepressants and monoamine oxidase inhibitors (MAOIs).
- Decongestants used in cold medications
- Beta blockers and methyldopa used to lower blood pressure
- Appetite suppressants
- Phenytoin used for epilepsy - Medicines which may increase the sedating effects of Demazin including:
- Medicines to help you sleep or relax
- Opioid pain killers - Other medicines including:
- Warfarin used to prevent blood clots. Your warfarin dose may require reduction if you use this product for a long period of time.
- Probenecid used for gout
- Medicines for urinary tract infections or bladder problems
- Colestyramine used to lower cholesterol
- Metoclopramide used to control nausea and vomiting.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Demazin.
4. How do I take Demazin?
How much to take
- Follow the instructions provided with the medicine.
- Take 2 white DAY tablets morning and afternoon if necessary.
- Take 2 pink NIGHT tablets at bedtime if necessary.
- Do not exceed the recommended dosage. The maximum dose is 6 DAY tablets and 2 NIGHT tablets in 24 hours.
If you are over 65 years of age, talk to your pharmacist or doctor about how much to take.
Elderly patients are more likely to have side effects from taking this medicine.
If you take too much Demazin
If you think that you have used too much Demazin, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning because of the risk of delayed, serious liver damage.
5. What should I know while taking Demazin?
Things you should do
If symptoms persist, see your doctor or pharmacist.
Unless advised by your doctor, Demazin should not be used for more than a few days at a time in adults and in children aged between 12 and 17 years for more than 48 hours.
Remind any doctor, dentist or pharmacist you visit that you are using Demazin.
Things you should not do
- Do not take Demazin with any other cold and flu medicines unless your doctor or pharmacist has advised you to.
- Do not take Demazin with any other paracetamol containing products unless your doctor or pharmacist has advised you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Demazin affects you.
The NIGHT tablets may cause drowsiness and may increase the effects of alcohol. Drowsiness may continue the following day. If you are affected do not drive or operate machinery.
Drinking alcohol
Tell your doctor or pharmacist if you drink alcohol.
Alcohol may increase the sedative effects of this medicine making you more drowsy.
Looking after your medicine
- Store below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Demazin contains
Day Tablet
| Active ingredients (main ingredients) | Paracetamol Pseudoephedrine hydrochloride |
| Other ingredients (inactive ingredients) | Microcrystalline cellulose Crospovidone Magnesium stearate Povidone Pregelatinised maize starch Stearic acid |
Night Tablet
| Active ingredients (main ingredients) | Paracetamol Pseudoephedrine hydrochloride Chlorphenamine maleate |
| Other ingredients (inactive ingredients) | Microcrystalline cellulose Crospovidone Magnesium stearate Povidone Pregelatinised maize starch Stearic acid Erythrosine |
Do not take this medicine if you are allergic to any of these ingredients.
What Demazin looks like
Demazin Original Cold + Flu Relief Day + Night contains a Day tablet and a Night tablet.
The Day tablet is a white round tablet with a break line.
The Night tablet is a pink round tablet with a break line.
Each pack contains 16 day tablets and 8 night tablets.
Registration number: AUST R 286002.
Who distributes Demazin
iNova Pharmaceuticals (Australia) Pty Limited
Level 10, 12 Help Street
Chatswood NSW 2067
Tel: 1800 630 056
This leaflet was prepared in December 2022.
Brand Information
| Brand name | Demazin Original Cold + Flu Relief Day + Night |
| Active ingredient | Paracetamol + Pseudoephedrine hydrochloride, Paracetamol + Pseudoephedrine hydrochloride + Chlorphenamine maleate (chlorpheniramine maleate) |
| Schedule | S3 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Day tablets: Paracetamol and pseudoephedrine hydrochloride.
Night tablets: Paracetamol, pseudoephedrine hydrochloride and chlorphenamine maleate.
2 Qualitative and Quantitative Composition
Each Day tablet contains: Paracetamol 500 mg; pseudoephedrine hydrochloride 30 mg.
Each Night tablet contains: Paracetamol 500 mg; pseudoephedrine hydrochloride 30 mg; chlorphenamine maleate 2 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Tablet.
Day Tablet: white, round tablet with a break-line.
Night Tablet: pink, round tablet with a break-line.
4 Clinical Particulars
4.1 Therapeutic Indications
Temporary relief from the symptoms of colds and flu.
Day tablet. Temporary relief from the following symptoms: Runny nose, nasal congestion, sinus pain, headache, body aches and pain, and fever.
Night tablet. Temporary relief from the following symptoms: Runny nose, nasal congestion, headache, pain and fever, and sneezing.
4.2 Dose and Method of Administration
This medicine should not be taken with other medicines containing paracetamol unless advised to do so by a doctor or pharmacist.
Adults and children over 12 years. Day tablet. Two tablets morning and afternoon if necessary.
Night tablet. Two tablets at bedtime if necessary. Maximum 6 Day and 2 Night tablets in 24 hours.
Use in adults. Keep to the recommended dose. This medicine should not be taken for more than a few days at a time except on medical advice.
Use in children and adolescents. Not recommended for children under 12 years of age.
Keep to the recommended dose. This medicine should not be taken for more than 48 hours except on medical advice.
4.3 Contraindications
Demazin Original Cold + Flu Relief Day + Night Tablets is contraindicated for use in patients with the following conditions.
Known hypersensitivity or idiosyncratic reaction to paracetamol, pseudoephedrine, chlorphenamine (or substances of a similar chemical structure) or any of the other ingredients in this medicine.
Severe or uncontrolled hypertension.
Severe coronary artery disease.
Severe acute or chronic kidney disease/renal failure.
Taking monoamine oxidase inhibitors (MAOIs) or who have taken MAOIs within the previous 14 days.
Narrow-angle glaucoma.
Stenosing peptic ulcer.
Symptomatic prostatic hypertrophy.
Bladder neck obstruction.
Pyloroduodenal obstruction.
Lactating women.
For additional information see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
4.4 Special Warnings and Precautions for Use
Identified precautions. Use with caution in patients with the following conditions: hypertension; hyperthyroidism; diabetes mellitus; coronary heart disease; ischaemic heart disease; glaucoma; prostatic hypertrophy; epilepsy.
This medicine may be dangerous when used in large amounts or for long periods. Hepatotoxicity may occur with paracetamol even at therapeutic doses, after short treatment duration and in patients without pre-existing liver dysfunction. Hepatotoxicity may develop following as little as 10 to 15 g of paracetamol and hepatic failure is known to occur occasionally with long term use of paracetamol. To avoid the risk of overdose patients should be advised not use this medicine with other paracetamol containing products.
Caution is advised in patients with underlying sensitivity to aspirin and/or to non-steroidal anti-inflammatory drugs (NSAIDs).
Patients with known analgesic intolerance or known bronchial asthma must only use this medication after having consulted a physician (hypersensitivity reactions including bronchospasm are possible).
Severe cutaneous adverse reactions (SCARs). Life threatening cutaneous reactions Stevens-Johnson Syndrome (SJS), and Toxic Epidermal Necrolysis (TEN) have been reported with the use of paracetamol. Patients should be advised of the signs and symptoms and monitored closely for skin reactions. If symptoms or signs of SJS and TEN (e.g. progressive skin rash often with blisters or mucosal lesions) occur, patients should stop treatment immediately and seek medical advice.
This medicine should be used with caution in patients with Gilbert's syndrome or Glucose-6-phosphate dehydrogenase deficiency.
Cases of high anion gap metabolic acidosis (HAGMA) due to pyroglutamic acidosis have been reported in patients with severe illness such as severe renal impairment and sepsis, or in patients with malnutrition or other sources of glutathione deficiency (e.g. chronic alcoholism) who were treated with paracetamol at therapeutic dose for a prolonged period or a combination of paracetamol and flucloxacillin. If HAGMA due to pyroglutamic acidosis is suspected, prompt discontinuation of paracetamol and close monitoring, including measurement of urinary 5-oxoproline, is recommended. The measurement of urinary 5-oxoproline may be useful to identify pyroglutamic acidosis as underlying cause of HAGMA in patients with multiple risk factors.
Effects on sleep. Chlorphenamine may cause drowsiness and may increase the effects of alcohol. Drowsiness may continue the following day. Those affected should not drive or operate machinery; alcohol should be avoided.
This medicine contains pseudoephedrine which may cause sleeplessness if taken up to several hours before going to bed.
Ischaemic colitis. Some cases of ischaemic colitis have been reported with pseudoephedrine. Pseudoephedrine should be discontinued, and medical advice sought if abdominal pain, rectal bleeding or other symptoms of ischaemic colitis develop.
Posterior reversible encephalopathy syndrome (PRES) and reversible cerebral vasoconstriction syndrome (RCVS). Cases of PRES and RCVS have been reported with the use of pseudoephedrine-containing product (see Section 4.8 Adverse Effects (Undesirable Effects)). The risk is increased in patients with severe or uncontrolled hypertension, or with severe acute or chronic kidney disease/renal failure (see Section 4.3 Contraindications).
Pseudoephedrine should be discontinued and immediate medical assistance sought if the following symptoms occur: sudden severe headache or thunderclap headache, nausea, vomiting, confusion, seizures and/or visual disturbances. Most reported cases of PRES and RCVS resolved following discontinuation and appropriate treatment.
Ischaemic optic neuropathy. Cases of ischaemic optic neuropathy have been reported with pseudoephedrine. The product should be discontinued if sudden loss of vision or decreased visual acuity such as scotoma occurs.
For additional information see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.7 Effects on Ability to Drive and Use Machines.
Use in hepatic impairment. Use with caution in patients with impaired hepatic function. Underlying liver disease increases the risk of paracetamol-related liver damage.
Patients who have been diagnosed with liver impairment must seek medical advice before taking this medication.
Cases of hepatic dysfunction/failure have been reported in patients with depleted glutathione levels, such as those who are severely malnourished, anorexic, have a low body mass index, are chronic heavy users of alcohol or have sepsis.
In patients with glutathione depleted states the use of paracetamol may increase the risk of metabolic acidosis.
Use in renal impairment. Use with caution in patients with impaired renal function. Administration of paracetamol to patients with moderate to severe renal impairment may result in accumulation of paracetamol conjugates.
Patients who have been diagnosed with kidney impairment must seek medical advice before taking this medicine.
Use in the elderly. The elderly may experience paradoxical excitation with chlorphenamine. The elderly are more likely to have central nervous system (CNS) depressive side effects, including confusion.
Paediatric use. Not recommended for children under 12 years of age.
This medicine should not be taken for more than 48 hours except on medical advice see Section 4.2 Dose and Method of Administration.
Children may experience paradoxical excitation with chlorphenamine.
Effects on laboratory tests. Uric acid and blood glucose. Intake of paracetamol may affect the laboratory determination of uric acid by phosphotungstic acid and of blood glucose by glucose oxidase-peroxidase.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following interactions with paracetamol have been noted:
Anticoagulants. Paracetamol may increase the risk of bleeding in patients taking warfarin and other antivitamin K medicines. Anticoagulant dosage may require reduction, and patients should be monitored for appropriate coagulation and bleeding complications.
Drugs which affect motility. Paracetamol absorption is increased by drugs which increase gastric emptying, e.g. metoclopramide and domperidone. Paracetamol absorption is decreased by substances that decrease gastric emptying, e.g. propantheline, antidepressants with anticholinergic properties, and narcotic analgesics.
Cholestyramine. Reduces the absorption of paracetamol if given within 1 hour of paracetamol. Chelating resins can decrease the intestinal absorption of paracetamol and potentially decrease its efficacy if taken simultaneously. In general, there must be an interval of more than 2 hours between taking the resin and taking paracetamol, if possible.
Glutathione depletion states. Co-administration of flucloxacillin with paracetamol may lead to high anion gap metabolic acidosis, particularly in patients presenting risk factors of glutathione depletion, such as sepsis, malnutrition or chronic alcoholism.
Probenecid. Paracetamol excretion may be affected, and plasma concentrations altered when given with probenecid.
Hepatotoxic drugs and microsomal liver enzyme inducers/inhibitors (CYP1A2). The risk of paracetamol toxicity may be increased in patients receiving other potentially hepatotoxic drugs or drugs that induce liver microsomal enzymes, such as antiepileptics (such as phenobarbital, phenytoin, carbamazepine, topiramate), alcohol, barbiturates and rifampicin. The induced metabolism results in an elevated production of the hepatotoxic oxidative metabolite of paracetamol. Hepatotoxicity will occur if this metabolite exceeds the normal glutathione binding capacity.
The following interactions with pseudoephedrine have been noted:
Antidepressant medication e.g. tricyclic antidepressants and monoamine oxidase inhibitors (MAOIs) may cause a serious increase in blood pressure or hypertensive crisis.
Other sympathomimetic agents, such as decongestants, appetite suppressants and amphetamine-like psychostimulants may cause an increase in blood pressure and additive effects.
Antihypertensives e.g. methyldopa and β-blockers may cause an increase in blood pressure.
Urinary acidifiers enhance elimination of pseudoephedrine.
Urinary alkalinisers decrease elimination of pseudoephedrine.
The following interactions with chlorphenamine have been noted:
Central nervous system (CNS) depressants (alcohol, sedatives, opioid analgesics, hypnotics) may cause an increase in sedation effects.
Monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs) may prolong and intensify the anticholinergic and CNS depressive effects.
When taken concomitantly with phenytoin may cause a decrease in phenytoin elimination.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
Pseudoephedrine has been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed. Studies in animals are inadequate or may be lacking, but available data shows no evidence of an increased occurrence of foetal damage.
Paracetamol can cross the placenta; however, no teratogenic effects have been observed in rats or mice, after doses of up to 250 mg/kg.
A woman in the third trimester of pregnancy ingested 22.5 g paracetamol. Early treatment with oral acetylcysteine resulted in good outcome for both mother and foetus.
Demazin Original Cold + Flu Relief Day + Night Tablets should not be used in pregnancy unless the potential benefits to the patient are weighed against the possible risk to the foetus.
Use in lactation. Paracetamol is excreted in breast milk. The amount available for ingestion by the infant has been reported variously as less than 0.1% of a single 500 mg dose and as 0.04 to 0.23% of a single 650 mg dose. Maternal ingestion of paracetamol in usual analgesic doses does not appear to present a risk to the breastfed infant. It has been estimated that 0.5% to 0.7% of a single dose of pseudoephedrine ingested by the mother will be excreted in the breast milk over 24 hours.
Chlorphenamine is excreted in breast milk.
Therefore, Demazin Original Cold + Flu Relief Day + Night Tablets is not recommended for breastfeeding mothers (see Section 4.3 Contraindications).
4.7 Effects on Ability to Drive and Use Machines
Chlorphenamine may cause drowsiness and may increase the effects of alcohol. Drowsiness may continue the following day. Those affected should not drive or operate machinery; alcohol should be avoided.
4.8 Adverse Effects (Undesirable Effects)
Reports of adverse reactions to paracetamol are rare. See Table 1.
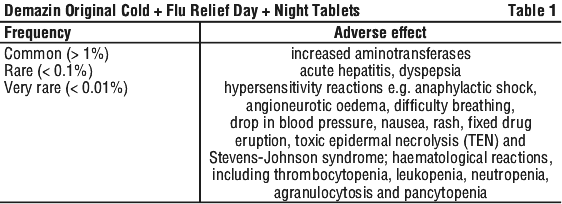
Haemolytic anaemia, particularly in patients with underlying glucose 6-phosphate-dehydrogenase deficiency has been reported. Kounis syndrome has been reported, as have cases of high anion gap metabolic acidosis due to pyroglutamic acidosis in patients with pre-disposing factors for glutathione depletion.
Adverse effects of pseudoephedrine include:
cardiovascular stimulation - elevated blood pressure, tachycardia or arrhythmias;
central nervous system (CNS) stimulation - restlessness, insomnia, anxiety, tremors and (rarely) hallucinations;
skin rashes and urinary retention;
ischaemic colitis (frequency unknown).
Children and the elderly are more likely to experience adverse effects than other age groups.
CNS depressive effects of chlorphenamine include sedation and impaired performance (impaired driving performance, poor work performance, incoordination, reduced motor skills, and impaired information processing). Performance may be impaired in the absence of sedation and may persist the morning after a night-time dose.
CNS stimulatory effects of chlorphenamine may include anxiety, hallucinations, appetite stimulation, muscle dyskinesias and activation of epileptogenic foci.
High doses of chlorphenamine may cause nervousness, tremor, insomnia, agitation, and irritability.
Side effects of chlorphenamine associated with cholinergic blockage include dryness of the eyes, mouth and nose, blurred vision, urinary hesitancy and retention, constipation and tachycardia.
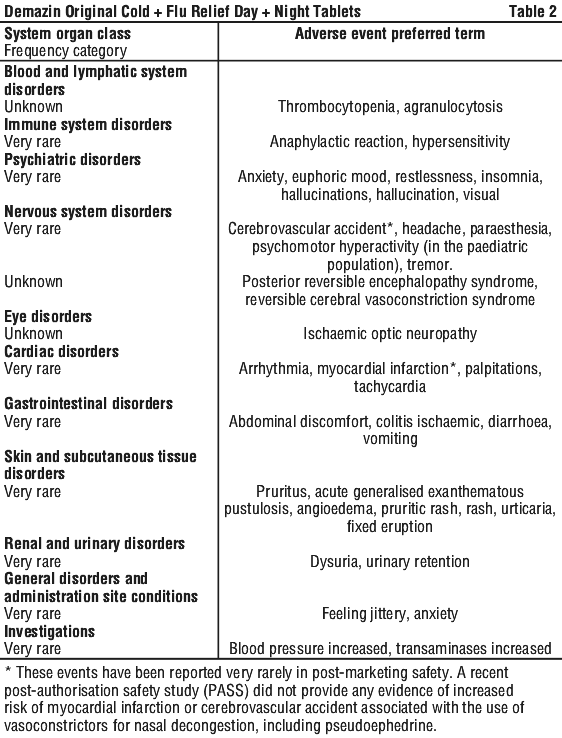
Adverse drug reactions identified during post-marketing experience with the combination appear in Table 2. The frequency category was estimated from spontaneous reporting rates.
Adverse events that have been observed during clinical trials and/or post-marketing use are ranked under the following frequency: Very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 1/10,000 and < 1/1,000), very rare (< 1/10,000).
4.9 Overdose
Elderly persons, small children, patients with liver disorders, chronic alcohol consumption or chronic malnutrition, as well as patients concomitantly treated with enzymes-inducing drugs are at an increased risk of intoxication, including fatal outcome. Overdosage with paracetamol if left untreated can result in delayed severe, sometimes fatal liver damage and rarely, acute renal tubular necrosis.
Symptoms. Immediate medical management is required in the event of an overdose, even if the symptoms of overdose are not present.
Toxic symptoms include vomiting, abdominal pain, hypotension and sweating. Nausea, vomiting, anorexia, pallor and abdominal pain generally appear during the first 24 hours of overdosage with paracetamol. Overdosage with paracetamol may cause hepatic cytolysis which can lead to hepatocellular insufficiency, gastrointestinal bleeding, metabolic acidosis, encephalopathy, disseminated intravascular coagulation, coma and death. Increased levels of hepatic transaminases, lactate dehydrogenase and bilirubin with a reduction in prothrombin level can appear 12 to 48 hours after acute overdosage. Overdosage can also lead to pancreatitis, acute renal failure and pancytopenia. The most serious adverse effect of acute overdosage of paracetamol is a dose-dependent, potentially fatal hepatic necrosis. In adults, hepatotoxicity may occur after ingestion of a single dose of 12 g (24 tablets) of paracetamol; a dose of 25 g (50 tablets) or more is potentially fatal. Symptoms during the first Major manifestations of liver failure such as jaundice, hypoglycaemia and metabolic acidosis may take at least 3 days to develop.
Treatment. If an overdose is taken or suspected, contact the Poisons Information Centre immediately for advice (131 126), or the patient should go to the nearest hospital straight away. This should be done even if they feel well because of the risk of delayed, serious liver damage.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Paracetamol is a p-aminophenol derivative that exhibits analgesic and antipyretic activity. It does not possess anti-inflammatory activity.
Its mechanism of action is believed to include inhibition of prostaglandin synthesis, primarily within the central nervous system. The lack of peripheral prostaglandin inhibition confers important pharmacological properties such as the maintenance of the protective prostaglandins within the gastrointestinal tract. Paracetamol is therefore particularly suitable for patients with a history of acid peptic disease, or on concomitant medication, where peripheral prostaglandin inhibition would be undesirable (such as, for example, those with a history of gastrointestinal bleeding or in the elderly).
Pseudoephedrine has direct and indirect-sympathomimetic activity and is an effective decongestant in the upper respiratory tract. It is a stereoisomer of ephedrine and has a similar action but has been found to have less pressor activity and fewer central nervous system (CNS) effects. Sympathomimetic agents are used as nasal decongestants to provide symptomatic relief. They act by causing vasoconstriction resulting in redistribution of local blood flow to reduce oedema of the nasal mucosa, thus improving ventilation, drainage and nasal stuffiness.
Chlorphenamine competes with histamine at central and peripheral histamine1-receptor sites, preventing the histamine-receptor interaction and subsequent mediator release. It is a highly lipophilic molecule that readily crosses the blood-brain barrier. It is highly selective for histamine1-receptors but has little effect on histamine2 or histamine3 receptors. Chlorphenamine also activates 5-hydroxytryptamine (serotonin) and α-adrenergic receptors and blocks cholinergic receptors.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. After oral administration, paracetamol is absorbed rapidly and completely from the small intestine; peak plasma levels occur 10 to 60 minutes after oral administration. Food intake delays paracetamol absorption.
Pseudoephedrine is readily absorbed from the gastrointestinal tract.
Chlorphenamine maleate is absorbed relatively slowly from the gastrointestinal tract, with peak plasma concentrations occurring about 2.5 to 6 hours after oral administration. Bioavailability is low, values of 25 to 50% having been reported. A duration of action of 4 to 6 hours has been reported; this is shorter than may be predicted from pharmacokinetic parameters. More rapid and extensive absorption has been reported in children compared to adults.
Distribution. Paracetamol is uniformly distributed throughout most body fluids; the apparent volume of distribution is 1 to 1.2 L/kg. Paracetamol can cross the placenta and is excreted in breast milk. Plasma protein binding is negligible at usual therapeutic concentrations but increases with increasing concentrations.
Small amounts of pseudoephedrine are distributed into breast milk.
Chlorphenamine is widely distributed in the body and enters the CNS. About 70% of chlorphenamine in circulation is bound to plasma proteins.
Metabolism. Paracetamol is metabolised by the hepatic microsomal enzyme system. In adults at therapeutic doses, paracetamol is mainly conjugated with glucuronide (45-55%) or sulfate (20-30%). A minor proportion (less than 20%) is metabolised to catechol derivatives, and mercapturic acid compounds via oxidation.
Paracetamol is metabolised differently by premature infants, newborns, infants and young children compared to adults, the sulfate conjugate being predominant.
Pseudoephedrine is incompletely metabolised (less than 1%) in the liver to an inactive metabolite by N-demethylation.
Chlorphenamine maleate is metabolised extensively. Metabolites include desmethyl- and didesmethylchlorphenamine. Chlorphenamine appears to undergo considerable first-pass metabolism.
Excretion. Paracetamol is excreted in the urine mainly as the glucuronide and sulfate conjugates. Less than 5% is excreted as unchanged paracetamol. 85-90% of the administered dose is eliminated in the urine within 24 hours of ingestion. The elimination half-life is about 1 to 4 hours.
Pseudoephedrine is largely excreted unchanged in the urine, together with small amounts of its hepatic metabolite. It has a half-life of about 5-8 hours; elimination is enhanced and half-life reduced accordingly in acid urine.
Unchanged chlorphenamine and metabolites are excreted primarily in the urine; excretion is dependent on urinary pH and flow rate. Only trace amounts have been found in the faeces. There is wide inter-individual variation in the pharmacokinetics of chlorphenamine; half-life values ranging from 2 to 43 hours have been reported. Faster clearance and a shorter half-life have been reported in children compared to adults.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Contains crospovidone, erythrosine aluminium lake (Night tablets only), magnesium stearate, microcrystalline cellulose, povidone, pregelatinised maize starch, stearic acid.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Blister pack - PVC/PVDC/Aluminium foil.
Pack size: 24 tablets containing 16 white day tablets and 8 pink night tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
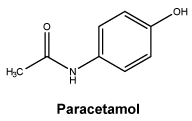
Chemical structure. Paracetamol (day and night tablets).
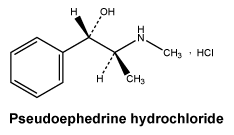
Pseudoephedrine hydrochloride (day and night tablets).
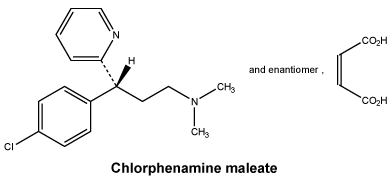
Chlorphenamine maleate (night tablets only).
Paracetamol is a white or almost white crystalline powder. It is sparingly soluble in water, freely soluble in alcohol and very slightly soluble in methylene chloride.
Pseudoephedrine hydrochloride is a white or almost white crystalline powder or colourless crystals. It is freely soluble in water and in ethanol (96 per cent), sparingly soluble in methylene chloride. Its melting point is at about 184⁰C.
Chlorphenamine maleate is a white or almost white, crystalline powder. It is freely soluble in water and soluble in ethanol (96 per cent).
CAS number. Paracetamol. 103-90-2.
Pseudoephedrine hydrochloride. 345-78-8.
Chlorphenamine maleate. 113-92-8.
7 Medicine Schedule (Poisons Standard)
Pharmacist Only Medicine (S3).
Date of First Approval
22 February 2017
Date of Revision
15 December 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.