Ego DermAid 1% Cream
Brand Information
| Brand name | Ego DermAid 1% Cream |
| Active ingredient | Hydrocortisone |
| Schedule | S2 | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ego DermAid 1% Cream.
What is in this leaflet
This leaflet answers some common questions about DermAid 1% cream.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
If you have any concerns about this medicine, ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What is DermAid 1% cream
The name of your medicine is DermAid 1% cream. It is a soft, glossy cream.
DermAid 1% cream contains hydrocortisone at 1% w/w as the active ingredient. DermAid also contains water, cetomacrogol 1000, cetostearyl alcohol, cetyl alcohol, self-emulsifying glyceryl monostearate, macrogol 400, propylene glycol and benzyl alcohol (as preservative).
DermAid 1% cream is available in 15 g and 30 g tubes. It is packed in tubes with a tamper evident seal for your protection. Do not use if the seal on the tube is broken.
What DermAid 1% cream is used for
DermAid 1% cream is used on the skin for temporary relief of redness, swelling, itching and discomfort of many skin problems such as:
- dermatitis
- eczema
- psoriasis
- rashes due to cosmetics and jewellery
- itching genital and anal areas
- sunburn
- other types of dermatitis
Your doctor or pharmacist, however, may have recommended DermAid 1% cream for another purpose.
How it works
The active ingredient in DermAid 1% cream, hydrocortisone, reduces inflammation and itching. It is used for the relief of symptoms associated with conditions such as dermatitis, eczema and psoriasis.
Before using DermAid 1% cream
Contraindications (when you must not use this product)
Do not use DermAid 1% cream if you have ever had an allergic reaction to:
- hydrocortisone
- any other corticosteroid
Do not use DermAid 1% cream in the eyes.
Do not use DermAid 1% cream if you have:
- a viral skin infection (such as cold sores, shingles or chicken pox)
- a fungal skin infection (such as thrush, tinea or ringworm)
- a bacterial skin infection (such as impetigo or boils) unless your doctor or pharmacist tells you.
Precautions
DermAid 1% cream reduces inflammation and itching. As a result, signs of infection may be masked. The area being treated should be kept clean at all times. If there is a possibility of infection in the area being treated then the infection should be controlled first.
Do not use under occlusion (plastic film) unless specified by your doctor.
Be careful not to get DermAid 1% cream in your eyes. If this happens rinse your eyes with clean water. If irritation persists tell your doctor or pharmacist.
If irritation or sensitivity occurs discontinue use.
Interactions
Although there are no generally recognised drug interactions attributed to hydrocortisone you should tell your doctor or pharmacist if you are using other creams, ointments or lotions or taking any medicine.
Pregnancy and lactation
You must tell your doctor if you are pregnant or breast-feeding. Your doctor will tell you if you can use DermAid 1% cream during pregnancy or while you are breast-feeding. DermAid 1% cream should not be applied to breasts before breast-feeding.
Use in Children
Do not use DermAid 1% cream on children under 2 years of age unless your doctor tells you.
How to use DermAid 1% cream
Directions
DermAid 1% cream is for external use only.
Apply a thin layer of DermAid 1% cream to your skin 1 to 2 times a day as required.
Your doctor or pharmacist may have recommended a different dosage. It is important to use DermAid 1% cream exactly as directed. If you use it less often than you should, it may not work as well and your skin problem may not improve. Using it more often than you should may not improve your skin problem any faster and may cause or increase side effects.
Cleanse with a soap alternative such as 'Pinetarsol Solution' or 'Pinetarsol Gel' as soap may irritate the skin.
If you have dry skin you may find an emollient such as "QV Cream" provides further relief between applications of DermAid 1% cream. Apply "QV Cream" as often as required.
How long to use it
If your condition persists for more than 7 days then discontinue use and see your doctor or pharmacist.
If you forget to use it
If you forget to use DermAid 1% cream, use it as soon as you remember and then go back to your normal times for applying DermAid 1% cream. Do not try to make up for the amount you missed by using more than you would normally use.
Side effects
DermAid 1% cream helps most people with skin problems but it may have unwanted side effects in a few people. After the application of DermAid 1% cream a slight stinging sensation may occasionally be noticed. This transient symptom is most likely to disappear after several applications.
Side effects that have been reported by some people using cortisones include:
- burning
- itching
- dryness
- redness
- allergy
- thinning of the skin
- blurred vision or other vision disturbances
If you have these or any other side effects, check with your doctor or pharmacist.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
In case of overdose
If you have accidentally applied too much DermAid 1% cream, then wipe off any excess. Use DermAid 1% cream exactly as directed. If you use it more often than directed your skin problem may not improve any faster and you may cause or increase side effects. If you experience any side effects contact your doctor or pharmacist.
If you swallow it
Telephone your Poisons Information Centre (13 11 26 in Australia or 03 474 7000 in New Zealand) or contact your doctor or pharmacist.
Storage
Keep DermAid 1% cream where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep DermAid 1% cream in a cool dry place where the temperature stays below 25 degrees C. Do not refrigerate. Do not leave DermAid 1% cream in the car or on windowsills. Heat can reduce the efficacy of some medicines.
Shelf life
Do not use DermAid 1% cream after the expiry date printed on the pack. If you use it after the expiry date has passed, it may not work as well.
For further information
This is not all the information that is available on DermAid 1% cream.
If you have any questions or are not sure about anything, ask your doctor or pharmacist.
Manufacturer
Ego Pharmaceuticals Pty. Ltd.
21-31 Malcolm Road, Braeside 3195
Australia
Distributors
NEW ZEALAND
Douglas Pharmaceuticals Ltd
PO Box 45027, Auckland 8
New Zealand
PAPUA NEW GUINEA, FIJI & SOUTH WEST PACIFIC
Exsel Pacific
PO Box 133, Red Hill
Queensland 4059
Australia
'Ego' and 'DermAid' are trade marks of Ego Pharmaceuticals Pty Ltd
Date of last revision: May 2019
Brand Information
| Brand name | Ego DermAid 1% Cream |
| Active ingredient | Hydrocortisone |
| Schedule | S2 | S3 |
MIMS Revision Date: 01 August 2019
1 Name of Medicine
Hydrocortisone.
2 Qualitative and Quantitative Composition
DermAid 1% cream. Hydrocortisone 1% w/w (10 mg/g).
DermAid 0.5% cream. Hydrocortisone 0.5% w/w (5 mg/g).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
DermAid cream is a white non-greasy cream for topical application.
4 Clinical Particulars
4.1 Therapeutic Indications
DermAid Cream is indicated for topical application for the temporary relief of symptoms associated with acute and chronic corticosteroid responsive conditions including: minor skin irritations, itching and rashes due to eczema, dermatitis, contact dermatitis (such as rashes due to cosmetics and jewellery), psoriasis, anogenital pruritus and sunburn.
4.2 Dose and Method of Administration
DermAid 0.5% cream. A thin layer should be applied to the affected skin one to three times a day as required.
DermAid 1% cream. A thin layer should be applied to the affected skin one to two times a day as required. Once inflammation has subsided the frequency of use may be reduced.
4.3 Contraindications
Like all other topical corticosteroids, DermAid Cream is contraindicated in vaccinia, chicken pox, herpes and other viral infections, bacterial infections, tuberculosis of the skin and syphilitic skin disorders.
Do not use in the eye.
Hypersensitivity to hydrocortisone, other corticosteroids or any other ingredient in the product.
4.4 Special Warnings and Precautions for Use
For external use only.
Long-term continuous topical therapy should be avoided where possible, particularly in children, as adrenal suppression can occur (even without occlusion).
As with other topical corticosteroids, when extensive areas are treated, sufficient systemic absorption may occur to produce the features of hypercorticalism. This effect is more likely to result if occlusive dressings are used or if treatment is prolonged. Rarely, local atrophy or striae may occur after prolonged treatment. This must be borne in mind when treating conditions such as severe eczema and seborrheic dermatitis. If applied to the eyelids, care is needed to ensure that the preparation does not enter the eye as glaucoma may result. Appropriate antimicrobial therapy should be used whenever treating inflammatory lesions that have become infected.
Any spread of the infection requires withdrawal of corticosteroid therapy and systemic administration of antimicrobial agents. Bacterial infection is encouraged by the warm, moist conditions associated by occlusive dressings, so the skin should be cleansed prior to a fresh dressing being applied.
Patients in whom there is a risk of increased systemic absorption should be regularly evaluated for evidence of hypothalamic pituitary adrenal (HPA) axis suppression by using urinary free cortisol (hydrocortisone) tests and monitoring morning plasma cortisol levels.
If there is evidence of suppression, attempts should be made to withdraw the drug or reduce the frequency of application. If hypersensitivity occurs, stop application and institute appropriate therapy. If irritation occurs, discontinue use. Systemic absorption of topical corticosteroids will be increased if extensive body surface areas are treated or if occlusion is used. Suitable precautions should be taken under these conditions or when long-term use is anticipated.
Hydrocortisone may mask signs of infection. If any infection is present, an appropriate anti-infective agent should be used first. DermAid Cream may be used to reduce inflammation but if a favourable response does not occur promptly then use of the product should be discontinued until the infection has been adequately controlled.
Use of the product near the eyes should be avoided. If any skin irritation develops discontinue use and treat appropriately. If extensive areas are treated, or if occlusive dressings are used, the possibility also exists of increased systemic absorption and this in turn could lead to the depression of the hypothalamo-pituitary adrenal axis. In all such patients it is essential to monitor adrenal function at regular intervals.
Use in the elderly. No data available.
Paediatric use. The risk of systemic absorption, and hence systemic toxicity, is greater in children due to a larger skin surface to bodyweight ratio than adults. The preparation is not recommended for use in children under 2 years of age except on the advice of a doctor.
Effects on laboratory tests. No data available.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No interactions known.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Category A: Drugs which have been taken by a large number of pregnant women and women of child bearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed.
Use in lactation. It is not known whether sufficient absorption of topical corticosteroids takes place to be excreted in breast milk. The potential benefits should be weighed against possible hazards to the breastfeeding infant.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
After the application of DermAid Cream a slight stinging sensation may occasionally be noticed. This transient symptom is most likely to disappear after several applications.
The following adverse effects have been reported with topical steroids: burning, itching, irritation, skin atrophy, secondary infection, dryness, acneform eruptions and hypopigmentation. Treatment should be chiefly symptomatic and administration of the steroid should be discontinued.
Intolerance to the occlusive dressing (miliary eruptions, folliculitis) may be expected to be observed, as with other corticosteroids. In such cases the use of an occlusive dressing should be discontinued. Use of the steroid may also need to be reduced or discontinued as local atrophy and striae of the skin may be observed.
In long-term treatment of extensive skin areas with occlusive dressings, one should bear in mind the possibility of inhibition of adrenal function. Therefore, adrenal function should be monitored under these circumstances.
Systemic adverse reactions, such as blurred vision, have also been reported with the use of topical corticosteroids (also see Section 4.4 Special Warnings and Precautions for Use, Visual disturbance).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Percutaneous absorption of corticosteroids may occur, especially under occlusive conditions. The following adverse effects have been reported with topical steroids: burning, itching, irritation, skin atrophy, secondary infection, dryness, acneform eruptions and hypopigmentation. Treatment should be chiefly symptomatic and administration of the steroid should be discontinued.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. DermAid Cream contains dissolved hydrocortisone. Creams with dissolved hydrocortisone have been shown to be pharmacologically more active than creams with suspended hydrocortisone in causing vasoconstriction, as shown in a study by R. Woodford and B.W. Barry, UK, 1984-5. The active component, hydrocortisone, has anti-inflammatory, anti-eczematous, anti-allergic and antipruritic properties.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Metabolism. Hydrocortisone is metabolised in the liver most likely by reduction of the 5,6 double bond and the C3 and C20 keto groups. The resultant hydroxy derivatives are then conjugated with glucuronic acid. Cortisone, an 11-ketosteroid is formed from hydrocortisone; the 11- ketosteroid is formed from hydrocortisone; the 11-ketosteroids are then reduced and conjugated to yield glucuronide metabolites. A small percentage of hydrocortisone is converted to the 17-ketosteroid. The C21 hydroxyl group is conjugated with sulphate.
Excretion. When radioactive carbon ring labelled steroids are injected intravenously in man, most of the radioisotope is recovered in the urine within 72 hours. Neither biliary nor faecal excretion is of any quantitative importance in man. It has been estimated that the liver metabolises at least 70% of the hydrocortisone secreted.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each gram of DermAid cream contains hydrocortisone (10 mg/g or 5 mg/g, as appropriate) in a cream base of purified water, macrogol 400, propylene glycol, cetyl alcohol, self- emulsifying glyceryl monostearate, benzyl alcohol, cetostearyl alcohol and cetomacrogol 1000.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
DermAid 0.5% cream. Store below 25°C. Do not refrigerate.
DermAid 1% cream. Store below 25°C. Do not refrigerate.
6.5 Nature and Contents of Container
DermAid 0.5% cream. 30 g laminate tube with a tamper evident seal packed into a carton.
DermAid 1% cream. 15 g*, 30 g laminate tube with a tamper evident seal packed into a carton.
*Not currently available.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
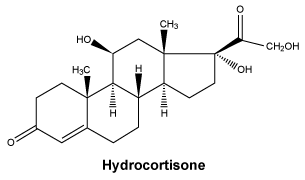
Chemical name: 11β, 17α, 21-trihydroxypregn-4-ene-3,20-dione. Molecular formula: C21H30O5.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S2 (0.5%); S3 (1%).
Date of Revision
24 May 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.