Diazepam Elixir
Brand Information
| Brand name | Diazepam Elixir |
| Active ingredient | Diazepam |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Diazepam Elixir.
Summary CMI
DIAZEPAM ELIXIR®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Diazepam Elixir®?
Diazepam Elixir® contains the active ingredient diazepam. Diazepam Elixir® is used for anxiety, to relax muscles, to treat trembling, confusional states or anxiety associated with alcohol withdrawal. It is also used to treat panic attacks.
For more information, see Section 1. Why am I using Diazepam Elixir®? in the full CMI.
2. What should I know before I use Diazepam Elixir®?
Do not use if you have ever had an allergic reaction to Diazepam Elixir®, any other benzodiazepine medicine, or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, have drug or alcohol addiction, drink alcohol, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Diazepam Elixir®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Diazepam Elixir® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Diazepam Elixir®?
Follow all directions given to you by your doctor or pharmacist carefully. Your doctor or pharmacist will tell you how much Diazepam Elixir® you will need to take each day. This depends on your condition and whether or not you are taking any other medicines.
More instructions can be found in Section 4. How do I use Diazepam Elixir®? in the full CMI.
5. What should I know while using Diazepam Elixir®?
| Things you should do | Use Diazepam Elixir® exactly as your doctor has prescribed. Remind any doctor, dentist or pharmacist you visit that you are using Diazepam Elixir®. If you become pregnant while you are taking Diazepam Elixir® tell your doctor immediately. Keep enough Diazepam Elixir® to last weekends and holidays. |
| Things you should not do | Do not take Diazepam Elixir® for a longer period of time than your doctor has prescribed. Do not stop taking Diazepam Elixir® or lower the dose, without first checking with your doctor, especially if you have epilepsy. |
| Driving or using machines | Be careful before you drive or use any machines until you know how Diazepam Elixir® affects you. |
| Drinking alcohol | Alcohol may increase the effects of Diazepam Elixir®. Your doctor may suggest that you avoid alcohol or reduce the amount of alcohol you drink while you are taking Diazepam Elixir®. |
| Looking after your medicine | Keep Diazepam Elixir® in a cool dry place, away from moisture, heat or sunlight, where the temperature stays below 25°C. Keep it where children cannot reach it. |
For more information, see Section 5. What should I know while using Diazepam Elixir®? in the full CMI.
6. Are there any side effects?
Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects: sudden anxiety or excitation, restlessness, agitation, irritability, anger, hallucinations or delusions, severe sleep disturbances, difficulties in breathing or choking or coughing, or yellowing of the skin and/or eyes (jaundice).
Speak to your doctor if you have any of these less serious side effects and they worry you: tiredness, drowsiness, dizziness, unsteadiness, loss of memory, inattentiveness, confusion, lack of concentration, headache, hangover feeling in the morning, slurred speech or unpleasant dreams.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
DIAZEPAM ELIXIR®
Active ingredient: Diazepam 10mg/10mL
Consumer Medicine Information (CMI)
This leaflet provides important information about using Diazepam Elixir®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Diazepam Elixir®.
Where to find information in this leaflet:
1. Why am I using Diazepam Elixir®?
2. What should I know before I use Diazepam Elixir®?
3. What if I am taking other medicines?
4. How do I use Diazepam Elixir®?
5. What should I know while using Diazepam Elixir®?
6. Are there any side effects?
7. Product details
1. Why am I using Diazepam Elixir®?
Diazepam Elixir® contains the active ingredient diazepam.
Diazepam is used for anxiety. Anxiety or tension associated with the normal stress of everyday life usually does not require treatment with medicines.
Diazepam is used to relax muscles.
Diazepam can also be used to treat trembling, confusional states or anxiety associated with alcohol withdrawal. It is also used to treat panic attacks.
Your doctor, however, may have prescribed Diazepam Elixir® for another purpose. Ask your doctor if you have any questions about why Diazepam Elixir® has been prescribed for you.
Diazepam Elixir® belongs to a group of medicines called benzodiazepines. They are thought to work by their action on brain chemicals.
In general, benzodiazepines such as Diazepam Elixir® should be taken for short periods only (for example 2 - 4 weeks). Continuous long-term use is not recommended unless advised by your doctor. The use of benzodiazepines may lead to dependence on the medicine.
2. What should I know before I use Diazepam Elixir®?
Warnings
Do not use Diazepam Elixir® if:
- You have an allergy to:
- Diazepam or any of the ingredients listed at the end of this leaflet.
- Any other medicine from the benzodiazepine group of medicines.
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing, or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
- You have severe and chronic lung disease.
- You suffer from severe muscle weakness known as myasthenia gravis.
- You suffer from severe liver disease.
- You have temporary stops breathing during sleep.
- You have drug or alcohol addiction (unless being used to treat symptoms of alcohol withdrawal).
Check with your doctor if you:
- You have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
- You have or have had any other health problems including:
- Liver, kidney or lung disease.
- If you suffer from fits or convulsions.
- If you have low blood pressure.
- If you have glaucoma (high pressure in the eye).
- If you suffer from depression, psychosis or schizophrenia.
If you have not told your doctor about any of the above, tell them before you take any Diazepam Elixir®.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use Diazepam Elixir® if you are pregnant or plan to become pregnant.
It is not known whether Diazepam Elixir® is harmful to an unborn baby when taken by a pregnant woman. If there is a need to take Diazepam Elixir® when you are pregnant your doctor will discuss the risks and benefits to you and the unborn baby.
Do not use Diazepam Elixir® if you are breastfeeding or plan to breastfeed, unless you and your doctor have discussed the risks and benefits involved.
Diazepam may pass into the breastmilk and cause drowsiness and/or feeding difficulties in the infant.
Do not give this medicine to children under 6 months of age, unless advised by the child's doctor.
The safety and effectiveness of Diazepam Elixir® in children under 6 months of age has not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Diazepam Elixir® and affect how it works. These include:
- other sleeping tablets, sedatives or tranquillisers
- medicines for depression
- medicines for allergies or colds e.g. antihistamines or cold tablets
- pain relievers
- muscle relaxants
- medicines to control fits
- cimetidine and omeprazole: a medicine used to treat gastric reflux and ulcers
- disulfiram, a medicine used to treat alcohol abuse
- cisapride, a medicine used to treat gastric reflux
- ketoconazole, a medicine used to treat fungal infections
- antibiotics, medicine used to treat infections e.g. erythromycin
These medicines may be affected by Diazepam Elixir® or may affect how well Diazepam Elixir® works. You may need to take different amounts of your medicine, or you may need to take different medicines. Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking Diazepam Elixir®.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Diazepam Elixir®.
4. How do I use Diazepam Elixir®?
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
How much to take:
Take Diazepam Elixir® exactly as your doctor has prescribed. Your doctor will tell you how much Diazepam Elixir® to take each day.
The dose of Diazepam Elixir® may be different for each person depending on age and the condition being treated.
The usual adult dose is between 5mg (5mL) and 40mg (40mL) daily. Children, elderly and very ill patients may need to take less.
How to take it:
Measure out the dose, decided for you by your doctor using a medicine glass and drink all of it, straight away.
When to take it:
Take Diazepam Elixir® at about the same time each day.
Taking Diazepam Elixir® at the same time each day will have the best effect. It will also help you remember when to take it. Usually it is taken three times daily. It does not matter if you take Diazepam Elixir® before or after food.
How long to take it:
Do not use Diazepam Elixir® for longer than your doctor says.
Usually Diazepam Elixir® should be used for short periods only (for example 2 – 4 weeks). Continuous long-term use is not recommended unless advised by your doctor. The use of benzodiazepines may lead to dependence on the medicine.
Follow the instructions provided and use Diazepam Elixir® until your doctor tells you to stop.
If you forget to take Diazepam Elixir®
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking it as you would normally.
Do not use a double dose to make up for the dose that you missed.
If you are unsure whether to take your next dose, speak to your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you use too much Diazepam Elixir®
If you think that you have used too much Diazepam Elixir®, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Also report any other medicine or alcohol which has been taken.
If you take too much Diazepam Elixir® you may feel drowsy, confused, tired, and dizzy, have difficulty breathing, feel weak or become unconscious.
5. What should I know while using Diazepam Elixir®?
Things you should do
Use Diazepam Elixir® exactly as your doctor has prescribed.
Remind any doctor, dentist or pharmacist you visit that you are using Diazepam Elixir®.
If you become pregnant while you are taking Diazepam Elixir® tell your doctor immediately.
Tell your doctor or pharmacist that you are taking Diazepam Elixir® if you are about to be started on any new medicines.
Tell your doctor if you feel Diazepam Elixir® is not helping your condition.
Visit you doctor regularly. Your doctor needs to check your progress and see whether you need to keep taking Diazepam Elixir®.
Always discuss with your doctor any problems or difficulties during or after taking Diazepam Elixir®. Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed. Otherwise your doctor may think that it was not effective and change your treatment unnecessarily.
Keep enough Diazepam Elixir® to last weekends and holidays.
If you are being treated for anxiety, be sure to discuss with your doctor any problems you may have and how you feel, especially if your anxiety attacks are getting worse or more frequent. This will help your doctor to determine the best treatment for you.
Things you should not do
Even if you take Diazepam Elixir® at night, you may still be drowsy or dizzy the next day.
Do not take Diazepam Elixir® for a longer period of time than your doctor has prescribed. Diazepam Elixir® should be taken for short periods only (for example 2 to 4 weeks), unless advised otherwise by your doctor.
Do not stop taking Diazepam Elixir® or lower the dose, without first checking with your doctor.
Stopping this medicine suddenly may cause some unwanted effects. Your doctor will explain how you should slowly reduce your dose before you can stop using Diazepam Elixir® completely.
Do not suddenly stop taking Diazepam Elixir® if you suffer from epilepsy. Stopping this medicine suddenly may make your epilepsy worse.
Do not use this medicine to treat any other complaints unless your doctor says to.
Do not give Diazepam Elixir® to anyone else, even if their symptoms seem similar to yours.
Things to be careful of:
Be careful if you are elderly, unwell or taking other medicines.
Some people may experience side effects such as drowsiness, confusion, dizziness and unsteadiness, which may increase the risk of a fall.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Diazepam Elixir® affects you.
Diazepam Elixir® may cause drowsiness or dizziness in some people and therefore may affect alertness.
Drinking alcohol
Tell your doctor if you drink alcohol.
Be careful when drinking alcohol while taking Diazepam Elixir®. Combining Diazepam Elixir® and alcohol can make you more sleepy, dizzy or lightheaded.
Your doctor may suggest that you avoid alcohol or reduce the amount of alcohol you drink while you are taking Diazepam Elixir®.
Looking after your medicine
- Store below 25°C
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Follow the instructions on the label on how to take care of your medicine properly.
Keep your Diazepam Elixir® in its bottle until it is time to take it. If you take the elixir out of the bottle it may not keep well.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not use Diazepam Elixir® if the bottle or cap is broken or shows signs of tampering.
When to discard your medicine
After opening discard after 90 days.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date. It may have no effect at all, or worse, an entirely unexpected effect if you use it after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Nervous system disorders:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Nervous system disorders:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell when you are taking, or soon after you have finished taking, Diazepam Elixir®.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Diazepam Elixir® contains
| Active ingredient (main ingredient) | Diazepam |
| Other ingredients (inactive ingredients) | Ethanol Propylene glycol Glycerol Citric acid Sodium citrate dihydrate Purified water Blackcurrant flavour |
Do not take this medicine if you are allergic to any of these ingredients.
Diazepam Elixir® does not contain gluten, lactose, sucrose, tartrazine or any other azo dyes.
What Diazepam Elixir® looks like
Diazepam Elixir® is a clear, colourless to yellow, slightly viscous liquid with an odour of blackcurrant available in a 100mL amber glass bottle
Australian Registration Number AUST R 42852
Who distributes Diazepam Elixir®
Orion Laboratories Pty Ltd T/A Evaris Pharma
25 – 29 Delawney Street
Balcatta WA 6021
www.evarispharma.com
This leaflet was prepared September 2003
Date of most recent amendment: November 2025
Brand Information
| Brand name | Diazepam Elixir |
| Active ingredient | Diazepam |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Diazepam.
2 Qualitative and Quantitative Composition
The active ingredient of Diazepam Elixir is diazepam 10 mg/10 mL.
Excipients with known effect. Ethanol and sodium citrate dihydrate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Diazepam Elixir contains diazepam 10 mg per 10 mL. It is a clear, colourless to yellow, slightly viscous liquid with an odour of blackcurrant and is available in a 100 mL amber glass bottle.
4 Clinical Particulars
4.1 Therapeutic Indications
Diazepam is indicated in the management of anxiety disorders and for the short term relief of the symptoms of anxiety. Diazepam is a useful adjunct for the relief of reflex muscle spasm due to local trauma (injury, inflammation) to muscles, bones and joints. It is also indicated in the management of cerebral spasticity due to upper motor neuron lesions such as cerebral palsy and paraplegia, and spasticity in athetosis and stiff man syndrome. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis.
4.2 Dose and Method of Administration
General. The lowest dose which can control the symptoms should be used. The dosage should be carefully individualised for maximum benefit. Dosage may need to be reduced in patients with hepatic or renal disease as the elimination half-life may be prolonged in this group. Treatment with benzodiazepines should always be withdrawn gradually. Elderly patients should be given a reduced dose. These patients should be checked regularly at the start of treatment in order to minimize the dosage and/or frequency of administration to prevent overdose due to accumulation.
In general, benzodiazepines should be prescribed for short periods (for example 2-4 weeks). Long term use of diazepam is not recommended. There is evidence that tolerance develops to the sedative effects of benzodiazepines. Withdrawal symptoms can occur after one week's treatment (e.g. rebound insomnia, following cessation of benzodiazepines).
Usual adult dose. 5 to 40 mg (5 to 40 mL) daily.
Average dosage for ambulatory patients. 2 mg (2 mL) three times daily or 5 mg (5 mL) in the evening and 2 mg (2 mL) once or twice during the day.
Elderly or debilitated patients. 2 mg (2 mL) twice daily or half the usual adult dose.
Children. Benzodiazepines should not be given to children without careful assessment of the indication; duration of treatment must be kept to a minimum.
Children younger than 6 months of age. Use not recommended.
6 months to 3 years. 1 to 6 mg (1 to 6 mL) daily.
4 to 14 years. 4 to 12 mg (4 to 12 mL) daily or calculated from 0.1 to 0.3 mg/kg (0.1 to 0.3 mL/kg) bodyweight.
Hospital treatment of tension, excitation, motor unrest. 10 to 15 mg (10 to 15 mL) three times daily until the acute symptoms subside.
4.3 Contraindications
Diazepam Elixir is contraindicated in patients with:
known hypersensitivity to benzodiazepines or to the excipients of the products (see Section 6.1 List of Excipients);
chronic obstructive airway disease with incipient respiratory failure;
severe respiratory insufficiency;
severe hepatic insufficiency;
sleep apnoea syndrome;
myasthenia gravis;
alcohol dependence unless being treated for acute alcohol withdrawal.
Diazepam should not be used as sole therapy for the treatment of depression or anxiety associated with depression as suicide may occur in such patients, nor should it be used as primary treatment of psychotic illness.
Diazepam Elixir is not recommended to control status epilepticus or other acute management situations.
4.4 Special Warnings and Precautions for Use
Use with caution in the following circumstances. Hypotension. Diazepam should be administered with caution to patients in whom a drop in blood pressure might lead to cardiac or cerebral complications, although hypotension has occurred rarely. This is particularly important in elderly patients.
Amnesia. Transient amnesia or memory impairment has been reported in association with the use of benzodiazepines. Anterograde amnesia can occur using therapeutic doses and the risk increases with higher doses. Amnesic effects may be associated with inappropriate behaviour.
Acute narrow angle glaucoma. Caution should be used in the treatment of patients with acute narrow angle glaucoma (because of atropine-like side effects).
Alcohol tolerance. Patients should be further advised that their tolerance for alcohol will be diminished and alcohol should therefore be avoided during treatment.
Prolonged use. Benzodiazepines should be prescribed for short periods only (2-4 weeks). Continuous long-term use of diazepam is not recommended. There is evidence that tolerance develops to the sedative effects of benzodiazepines. After as little as one week of therapy, withdrawal symptoms can appear following cessation of recommended doses (e.g. rebound insomnia following cessation of a hypnotic benzodiazepine).
Withdrawal. Following prolonged use of diazepam withdrawal from medication should be gradual. A withdrawal timetable should be planned for each patient. For patients with known or suspected dependence withdrawal periods from four weeks to four months have been suggested.
Depression, psychosis and schizophrenia. Diazepam is not recommended as primary therapy in patients with depression and/or psychosis. In such conditions psychiatric assessment and supervision are necessary if benzodiazepines are indicated. Benzodiazepines may increase depression in some patients, and may contribute to deterioration in severely disturbed schizophrenics with confusion and withdrawal. Suicidal tendencies may be present or uncovered and protective measures may be required.
Paradoxical reactions. Paradoxical reactions such as restlessness, agitation, irritability, aggressiveness, delusion, nightmares, hallucinations, psychoses, inappropriate behaviour and other reactions such as acute rage, stimulation or excitement may occur. If such reactions occur, diazepam should be discontinued. These reactions are more likely to occur in children and the elderly.
Impaired respiratory function. Caution in the use of diazepam is recommended in patients with respiratory depression. In patients with chronic obstructive pulmonary disease, benzodiazepines can cause increased arterial carbon dioxide tension and decreased oxygen tension. Due to the risk of respiratory depression, lower doses are recommended for patients with chronic respiratory insufficiency.
Epilepsy. When diazepam is administered to patients with convulsive disorders an increase in the frequency and/or the severity of grand mal seizures may occur, necessitating increased anticonvulsant medication. Abrupt withdrawal of benzodiazepines in persons with convulsive disorders may be associated with a temporary increase in the frequency and or severity of seizures.
Abuse. Caution must be exercised in administering diazepam to individuals known to be addiction prone or those with a history which suggests they may increase the dosage on their own initiative. It is desirable to limit repeat prescription without adequate medical supervision.
Dependence. The use of benzodiazepines may lead to dependence as defined by a withdrawal syndrome on discontinuation of the drug. The risk of dependence increases with dosage and the duration of treatment. Patients on long term therapy and/or high dosage can have more marked dependence and it occurs in predisposed patients with a history of drug and/or alcohol abuse. Tolerance as defined by a need to increase the dose in order to achieve the same therapeutic effect seldom occurs in patients receiving recommended doses under medical supervision. Tolerance to sedation may occur with benzodiazepines particularly in those with drug seeking behaviour.
Withdrawal symptoms similar to those noted with barbiturates and alcohol have occurred once physical dependence to benzodiazepines has developed or following abrupt discontinuation of benzodiazepines. These symptoms range from insomnia, anxiety, dysphoria, palpitations, panic attacks, vertigo, myoclonus, akinesia, hypersensitivity to light, sound and touch, abnormal body sensations (e.g. feeling of motion, metallic taste), depersonalisation, derealisation, delusional beliefs, hyperreflexia and loss of short term memory, to a major syndrome which may include convulsions, tremor, abdominal and muscle cramps, confusional state, delusional state, delirium, hallucinations, hyperthermia, psychosis, vomiting and sweating. Such manifestations of withdrawal especially the more serious ones are more common in patients who have received excessive doses over a prolonged period. However, withdrawal symptoms have been reported following abrupt discontinuation of benzodiazepines taken continuously at therapeutic levels. Accordingly, diazepam should be terminated by tapering the dose to minimise the occurrence of withdrawal symptoms. Patients should be advised to consult their physician before increasing the dose or abruptly discontinuing the medication.
Rebound phenomena have been described in the context of benzodiazepine use. Rebound insomnia and anxiety mean an increase in the severity of the symptoms beyond pretreatment levels following the cessation of the benzodiazepines. Rebound phenomena in general possibly reflect re-emergence of pre-existing symptoms combined with withdrawal symptoms described earlier. Withdrawal/ rebound symptoms may follow high doses for relatively short periods.
Use in hepatic and renal impairment. Patients with impaired renal or hepatic function should use benzodiazepine medication with caution and dosage reduction may be advisable. In rare instances some patients taking benzodiazepines have developed blood dyscrasias, and some have had elevation in liver enzymes; periodic blood counts and liver function tests are recommended.
Use in the elderly. Such patients may be particularly susceptible to the sedative effects of benzodiazepines and associated giddiness, ataxia and confusion, which may increase the possibility of a fall. Lower doses should be used for elderly and debilitated patients.
Paediatric use. Prolonged central nervous system depression has been observed in neonates due to an inability to biotransform diazepam to inactive metabolites and so they, especially the very young, are usually more sensitive to it. Chronic use of diazepam is not recommended in children younger than 6 months due to a lack of adequate clinical experience.
Effects on laboratory tests. Minor EEG changes, usually low voltage, fast activity, of no known clinical significance have been noted with benzodiazepine administration.
Diazepam can inhibit binding of thyroxine and liothyronine to their binding proteins resulting in erroneously abnormal values from a thyroid function test.
No effects on diagnostic tests have been seen when diazepam has been administered orally or rectally.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Diazepam produces additive CNS depressant effects when co-administered with other medications which themselves produce CNS depression, e.g. alcohol, barbiturates, anxiolytics, sedatives, antidepressants including tricyclic antidepressants, non selective MAO inhibitors, hypnotics, antiepileptic drugs, phenothiazines and other antipsychotics, skeletal muscle relaxants, antihistamines, narcotic analgesics and anaesthetics. The effect of these drugs may potentiate or be potentiated by diazepam.
Concomitant use with alcohol is not recommended due to the enhancement of the sedative effect.
The anticholinergic effects of drugs including atropine and similar drugs, antihistamines and antidepressants may be potentiated.
Cisapride may lead to a temporary increase in the sedative effects of orally administered diazepam due to a faster absorption.
There is a potentially relevant interaction between diazepam and drugs that inhibit certain hepatic enzymes, particularly CYP3A4 and CYP2C19 which are the principal isoenzymes involved in the initial oxidative metabolism of diazepam. Potential hepatic enzyme inhibitors (e.g. cimetidine, omeprazole, disulfiram, fluvoxamine, fluoxetine, probenecid, ketoconazole, macrolide antibiotics such as erythromycin and clarithromycin, HIV-protease inhibitors such as ritonavir and indinavir, oral contraceptives) could decrease the rate of diazepam elimination and may lead to increased and prolonged sedation. Patients receiving diazepam should be monitored for signs of an exaggerated response if any of the above drugs are used concomitantly; some patients may require a reduction in diazepam dosage.
Inducers of hepatic enzymes (e.g. rifampicin, carbamazepine, phenytoin, and phenobarbitone) can increase the rate of elimination of diazepam.
Interactions have been reported between some benzodiazepines and anticonvulsants, with changes in the serum concentration of the benzodiazepines or anticonvulsant. It is recommended that patients be observed for altered responses when benzodiazepines and anticonvulsants are prescribed together and that the serum level monitoring of the anticonvulsant is performed more frequently.
There have been reports that the metabolic elimination of phenytoin is affected by diazepam.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Reproductive studies in rats showed decreases in the number of pregnancies and in the number of surviving offspring following administration of oral doses of 100 mg/kg/day (22-fold the MRHD on a body surface area basis) to both males and females prior to and during mating and throughout gestation and lactation. No adverse effects were observed at 10 mg/kg/day (60 mg/m2/day, twice the MRHD).
Use in pregnancy. (Category C)
The categorisation definition of Category C: Drugs which owing to their pharmacological effects, have caused or may be suspected of causing, harmful effects on the human foetus or neonate without causing malformations. These effects may be reversible.
Data from observational studies suggest an increased risk of miscarriage with benzodiazepine exposure during pregnancy. The benefits of treatment should be weighed against the risks, and patient should be informed of the increased risk of miscarriage. A woman of childbearing potential should contact her physician regarding the discontinuation of Diazepam Elixir if she intends to become pregnant or suspects that she is pregnant.
The safety of diazepam for use in human pregnancy has not been established. An increased risk of congenital malformation associated with benzodiazepine use in the first trimester has been suggested. Benzodiazepines should be avoided during pregnancy unless there is no safer alternative. Benzodiazepines cross the placenta and may cause hypotension, hypotonia, reduced respiratory function and hypothermia in the newborn infant. Continuous treatment during pregnancy and high doses in connection with delivery should be avoided.
Special care must be taken during labour and delivery; if diazepam is used as single high doses it may produce irregularities in the foetal heart rate, hypotonia, poor sucking, hypothermia, and moderate respiratory depression in the neonate. With newborn infants it must be remembered that the enzyme system involved in the breakdown of the drug is not yet fully developed, especially in premature infants. Withdrawal symptoms in newborn infants have been reported with this class of drugs.
Use in lactation. Breastfeeding is not recommended in patients receiving diazepam. Diazepam is excreted in the breast milk and may cause drowsiness and feeding difficulties in the infant.
4.7 Effects on Ability to Drive and Use Machines
Diazepam Elixir may modify patients' performance at skilled tasks (driving, operating machinery, etc.) to a varying degree depending upon dosage and individual susceptibility. Patients should be warned not to operate machinery or motor vehicles until it is known that they do not become drowsy or dizzy. Abilities may be impaired on the day following use.
4.8 Adverse Effects (Undesirable Effects)
Common (usually dose related). Central nervous system. Fatigue, drowsiness, and ataxia.
Musculoskeletal. Muscle weakness.
Uncommon. Cardiovascular. Hypotension.
Central nervous system. Amnesia (anterograde), confusion, depression, headache, tremor, vertigo, blurred vision, slurred speech and dysarthria.
Dermatological. Skin rash.
Gastrointestinal. Constipation, gastrointestinal disturbances, dry mouth, nausea and hypersalivation.
Genitourinary. Incontinence, urinary retention, increase or decrease in libido.
Rare. Central nervous system. Dizziness with oral diazepam.
Cardiovascular. Cardiac arrest.
Very rare. Haematopoietic. Isolated instances of neutropaenia have been reported.
Hepatic. Jaundice, elevated transaminases and alkaline phosphatase.
Other. Anterograde amnesia may occur using therapeutic dosages, the risk increasing at higher doses. Paradoxical reactions such as acute hyperexcitation, anxiety, hallucinations, increased muscle spasticity, insomnia, rage, sleep disturbances and stimulation have been reported; should these occur, use of diazepam should be discontinued.
Dependence. Chronic use (even at therapeutic doses) of oral diazepam may lead to the development of physical dependence; discontinuation of therapy may result in withdrawal or rebound phenomena.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Overdosage of benzodiazepines is usually manifested by degrees of central nervous system depression ranging from drowsiness to coma. In mild cases, symptoms include drowsiness, mental confusion and lethargy. In more serious cases, symptoms may include ataxia, hypotonia, hypotension, respiratory depression, coma, and very rarely, death.
Treatment. In the management of overdosage with any medication, it should be borne in mind that multiple agents may have been taken. Treatment should be symptomatic and supportive.
Following overdosage, activated charcoal should be administered within one to two hours after ingestion to decrease absorption. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube once the airway is protected. In the management of overdosage special attention should be paid to the respiratory and cardiovascular functions in intensive care. Hypotension and respiratory depression should be managed according to general principles.
Haemoperfusion and haemodialysis are not useful in benzodiazepine intoxication.
The benzodiazepine antagonist, flumazenil may be useful in hospitalised patients for the reversal of acute benzodiazepine overdose effects. The flumazenil product information should be consulted prior to use. The use of flumazenil is not recommended in epileptic patients who have been treated with diazepam. The reversal of the benzodiazepine effect in these patients may induce convulsions.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Diazepam is a long acting benzodiazepine. Benzodiazepines, in general act as depressants of the central nervous system (CNS) and exhibit anxiolytic, anticonvulsant, sedative and muscle relaxant properties. Its actions are presumed to be mediated by enhancement of the activity of gamma-aminobutyric acid (GABA), the major natural inhibitory neurotransmitter in the brain. GABA acts at this receptor to open the chloride channel, allowing the flow of chloride ions into the neuron. Entry of chloride ions results in hyperpolarisation, which inhibits firing of the neuron and translates into decreased neuronal excitability, thus attenuating the effects of subsequent depolarising excitatory transmitters. Diazepam enhances the actions of GABA by causing GABA to bind more tightly to GABA type A receptors.
5.2 Pharmacokinetic Properties
Absorption. Following oral administration diazepam is well absorbed from the gastrointestinal tract with peak plasma levels occurring within about 30 to 90 minutes.
Distribution. Diazepam is highly lipid soluble and widely distributed into body tissues and crosses the blood brain barrier. Diazepam and its metabolites also cross the placental barrier and are also distributed into breast milk. Diazepam is 98% protein bound in the plasma. The plasma concentration time curve is biphasic, with an initial rapid and extensive distribution phase with a half life of up to 3 hours, followed by a prolonged terminal elimination phase (half life 20-48 hours).
Metabolism. Diazepam is extensively metabolised to one major active metabolite (desmethyldiazepam), and two minor active metabolites, 3-hydroxydiazepam (temazepam) and 3-hydroxy-N-diazepam (oxazepam) in plasma. At therapeutic doses, desmethyldiazepam is found in plasma at concentrations equivalent to those of diazepam while oxazepam and temazepam are not usually detectable. Diazepam undergoes hepatic demethylation and hydroxylation, involving cytochrome P450 (CYP) 2C19 and CYP3A4 isoenzymes, followed by glucuronidation.
Excretion. It is largely excreted in the urine (70%), predominately as its conjugated metabolites or in its free form.
5.3 Preclinical Safety Data
Genotoxicity. Limited data from a number of studies have provided weak evidence of a genotoxic potential. Diazepam has been shown to induce aneuploidy in sperm obtained from both mice and humans treated with approximately 10 mg/m2/day (less than the MRHD).
Carcinogenicity. The carcinogenic potential of oral diazepam has been studied in several rodent species. An increase in the incidence of malignant hepatocellular tumours occurred in male rats and mice following lifetime dietary administration of diazepam at 75 mg/kg/day (17- and 8-fold the MRHD on a body surface area basis, respectively). This was not observed in female rats and mice treated with 75 mg/kg/day or hamsters treated with 120 mg/kg/day (18-fold the MRHD).
Teratogenicity. Diazepam was found to be teratogenic in mice at intravenous doses of 45 mg/kg or greater and oral doses of 100 mg/kg or greater (both 10-fold the MRHD on a body surface area basis), as well as in hamsters at 280 mg/kg (41-fold the MRHD). The respective no-effect doses were 50 mg/kg (5-fold the MRHD) in mice and 200 mg/kg (30-fold the MRHD) in hamsters. Malformations included exencephaly, cranioschisis, kinking of the spinal cord, and cleft palate with and without cleft lip. Malformations were not observed in rats or rabbits at respective doses of up to 300 and 50 mg/kg/day (greater than 20-fold the MRHD). Delayed development has been reported in offspring from several animal species treated with diazepam during pregnancy or during pregnancy and lactation.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ethanol, propylene glycol, glycerol, citric acid, sodium citrate dihydrate, purified water and blackcurrant flavour.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
After opening store below 25°C and discard after 90 days.
6.5 Nature and Contents of Container
Diazepam Elixir is available in a 100 mL amber glass bottle.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
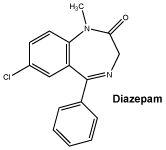
Molecular Formula: C16H13ClN2O.
Molecular weight: 284.74.
Diazepam is a benzodiazepine derivative. It is a colourless, crystalline compound, insoluble in water and soluble in alcohol.
CAS number. 439-14-5.
7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Only Medicine).
Date of First Approval
30 September 2003
Date of Revision
11 February 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.