Dimethyl Fumarate Sandoz
Brand Information
| Brand name | Dimethyl Fumarate Sandoz |
| Active ingredient | Dimethyl fumarate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Dimethyl Fumarate Sandoz.
Summary CMI
Dimethyl Fumarate Sandoz®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I taking Dimethyl Fumarate Sandoz?
Dimethyl Fumarate Sandoz contains the active ingredient dimethyl fumarate. Dimethyl Fumarate Sandoz is used to treat relapsing multiple sclerosis (MS).
For more information, see Section 1. Why am I taking Dimethyl Fumarate Sandoz? in the full CMI.
2. What should I know before I take Dimethyl Fumarate Sandoz?
Do not use if you have ever had an allergic reaction to dimethyl fumarate or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take Dimethyl Fumarate Sandoz? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Dimethyl Fumarate Sandoz and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take Dimethyl Fumarate Sandoz?
- The recommended starting dose of Dimethyl Fumarate Sandoz is 120 mg taken twice daily. After 7 days the recommended dose is 240 mg twice daily.
More instructions can be found in Section 4. How do I take Dimethyl Fumarate Sandoz? in the full CMI.
5. What should I know while taking Dimethyl Fumarate Sandoz?
| Things you must do |
|
| Things you must not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking Dimethyl Fumarate Sandoz? in the full CMI.
6. Are there any side effects?
Common side effects are reddening of the face or body or body feeling warm, hot, burning or itchy (flushing), loose stools (diarrhoea), feeling sick (nausea), stomach pain or stomach cramps, inflammation of the lining of the intestines (gastroenteritis), being sick (vomiting), indigestion (dyspepsia), inflammation of the lining of the stomach (gastritis), gastrointestinal disorder, burning sensation, hot flush, feeling hot, itchy skin (pruritus), rash, pink or red blotches on the skin (erythema), runny nose (rhinorrhoea), hair thinning (alopecia). Serious side effects are signs of infection (e.g. unexplained fever, severe diarrhoea). For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Dimethyl Fumarate Sandoz®
Active ingredient(s): dimethyl fumarate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Dimethyl Fumarate Sandoz. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Dimethyl Fumarate Sandoz.
Where to find information in this leaflet:
1. Why am I taking Dimethyl Fumarate Sandoz?
2. What should I know before I take Dimethyl Fumarate Sandoz?
3. What if I am taking other medicines?
4. How do I take Dimethyl Fumarate Sandoz?
5. What should I know while taking Dimethyl Fumarate Sandoz?
6. Are there any side effects?
7. Product details
1. Why am I taking Dimethyl Fumarate Sandoz?
Dimethyl Fumarate Sandoz contains the active ingredient dimethyl fumarate.
Dimethyl Fumarate Sandoz is used to treat relapsing multiple sclerosis (MS).
Dimethyl Fumarate Sandoz slows down the progression of physical disability in people with relapsing forms of MS and decreases the number of flare ups (relapses).
Some people feel better when they start to take Dimethyl Fumarate Sandoz. However Dimethyl Fumarate Sandoz cannot repair damage that has already been caused by MS.
When you start Dimethyl Fumarate Sandoz you might not notice an improvement, but Dimethyl Fumarate Sandoz may still be working to help prevent your MS from becoming worse.
The cause of MS is not yet known, MS affects the brain and spinal cord. In MS, the body's immune system reacts against its own myelin (the ‘insulation’ surrounding nerve fibres). In relapsing forms of MS, people have ‘exacerbations’ from time to time (e.g. blurred vision, weakness in the legs or arms, or loss of control of bowel or bladder function). They are followed by periods of recovery. Recovery may be complete or incomplete. If it is incomplete there is ‘progression of disability’.
Dimethyl fumarate decreases the inflammation in your brain that is caused by MS and thereby reduces nerve damage. It works by reducing inflammatory responses in cells and helps to protect the central nervous system cells against attack. Inflammation of the brain is an important part of the MS disease process.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
Dimethyl fumarate has not been studied in patients with chronic progressive MS.
2. What should I know before I take Dimethyl Fumarate Sandoz?
Warnings
Do not use Dimethyl Fumarate Sandoz if:
- you are allergic to any medicine containing dimethyl fumarate or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other body parts
- rash, itching or hives on the skin. - you are being treated with other medicines containing fumaric acid (creams or tablets/capsules).
Check with your doctor if you:
- have any other medical conditions, such as
- liver problems
- kidney problems
- infection - recently received a vaccination
- take any other medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Shingles
Tell your doctor at the earliest opportunity if you suspect you may have shingles (a painful viral infection with a painful rash that develops on one side of the face or body).
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
There is no information on the use of Dimethyl Fumarate Sandoz during pregnancy. Your doctor will discuss the risks and benefits of taking it if you are pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known whether Dimethyl Fumarate Sandoz passes into breast milk. Your doctor will discuss the risks and benefits of taking it if you are breastfeeding or planning to breastfeed.
Children
Safety and effectiveness in children younger than 18 years have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Dimethyl Fumarate Sandoz and affect how it works. These include:
- medicines that contain fumaric acid (creams or tablets/capsules)
- medicines which affect immune function including other medicines to treat MS such as fingolimod, natalizumab or mitoxantrone or some other commonly used cancer medicines
- medicines which affect the kidneys, including some antibiotics (used to treat infections), "water tablets" (diuretics), certain types of painkillers (such as ibuprofen and other similar anti-inflammatory medicines and medicines purchased without a doctor's prescription) and medicines that contain lithium
- live vaccines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Dimethyl Fumarate Sandoz.
4. How do I take Dimethyl Fumarate Sandoz?
How much to take
- The recommended starting dose of Dimethyl Fumarate Sandoz is 120 mg taken twice daily. After 7 days the recommended dose is 240 mg twice daily.
When to take Dimethyl Fumarate Sandoz
- Take one capsule twice a day. Taking it at the same time each day (e.g. at morning during breakfast and at night during dinner) will help you remember when to take it.
- Dimethyl Fumarate Sandoz can be taken with or without food. For those patients who experience gastrointestinal side effects or flushing, taking Dimethyl Fumarate Sandoz with food may help reduce these effects.
- Your doctor may tell you to take Dimethyl Fumarate Sandoz with aspirin or may temporarily reduce your dose.
- Do not reduce your dose unless your doctor tells you to.
How to take Dimethyl Fumarate Sandoz
- Swallow each capsule whole with a glass of water. Do not crush, divide or dissolve the capsule or its contents.
How long to take Dimethyl Fumarate Sandoz
- Continue taking Dimethyl Fumarate Sandoz for as long as your doctor tells you.
- Dimethyl Fumarate Sandoz helps to control your condition but does not cure it. The positive effects of Dimethyl Fumarate Sandoz may not be seen immediately. it is important to keep taking your medicine even if you feel well. It is important not to interrupt treatment with Dimethyl Fumarate Sandoz unless your doctor tells you to.
If you forget to take Dimethyl Fumarate Sandoz
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much Dimethyl Fumarate Sandoz
If you think that you have taken too much Dimethyl Fumarate Sandoz, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking Dimethyl Fumarate Sandoz?
Things you should do
Take Dimethyl Fumarate Sandoz exactly as your doctor has prescribed.
Keep all of your doctor's appointments so that your progress can be checked.
If you are about to have any blood or urine tests, tell your doctor that you are taking Dimethyl Fumarate Sandoz. Blood and urine test results may be affected by treatment with Dimethyl Fumarate Sandoz.
Tell your doctor if you are going to be vaccinated.
If you are about to start on any new medicine, remind your doctor and pharmacist that you are taking Dimethyl Fumarate Sandoz.
Remind any doctor, dentist or pharmacist you visit that you are taking Dimethyl Fumarate Sandoz.
Tell your partner or caregiver about your treatment.
Call your doctor straight away if you:
- think you have an infection, have fever, or feel like you have the flu.
Dimethyl Fumarate Sandoz may decrease lymphocyte (white blood cell) counts. White blood cells fight infection. You may get infections more easily while you are taking Dimethyl Fumarate Sandoz. Any infection that you already have may get worse. Infections could be serious and sometimes life-threatening. If you have a serious infection, your doctor may recommend that you stop taking Dimethyl Fumarate Sandoz until you recover. - think you are experiencing symptoms similar to an MS relapse, new or worsening weakness on one side of the body, clumsiness, changes in vision, thinking, or memory, or confusion or personality changes lasting for more than several days.
These could be signs of a rare and very serious brain infection called progressive multifocal leukoencephalopathy (PML). The symptoms of PML may be similar to an MS relapse. Having low lymphocyte levels, particularly for a long period of time can increase your risk of PML.
Things you should not do
- Do not take Dimethyl Fumarate Sandoz to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not stop taking Dimethyl Fumarate Sandoz or lower the dosage without checking with your doctor.
Blood and urine tests
- Before you start Dimethyl Fumarate Sandoz, your doctor will do a blood test to check the number of your white blood cells. Your doctor may also test these periodically during treatment.
- Before you start Dimethyl Fumarate Sandoz, your doctor will make sure you have results from a recent urine test to check your kidney function and may repeat the test periodically during treatment. Dimethyl Fumarate Sandoz may cause proteins (such as albumin) to be detected in a urine test.
- Dimethyl Fumarate Sandoz may also cause increases in the level of liver enzymes that will show up in a blood test.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Dimethyl Fumarate Sandoz affects you.
Looking after your medicine
- Keep your capsules in the pack until it is time to take them.
- Store below 25°C.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General disorders:
| Call your doctor straight away if you notice any of these serious side effects |
Very serious side effects
| Very serious side effects | What to do |
Allergic reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these very serious side effect. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Dimethyl Fumarate Sandoz contains
| Active ingredient (main ingredient) | dimethyl fumarate |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Dimethyl Fumarate Sandoz looks like
Dimethyl Fumarate Sandoz 120 mg capsules – white and light-green printed with '120 mg' in black ink on the capsule body (AUST R 346056)
Available in blisters containing 14 or 56 capsules packed in a box.
Dimethyl Fumarate Sandoz 240 mg capsules – light-green printed with '240 mg' in black ink on the capsule body (AUST R 346055)
Available in blisters containing 14 or 56 capsules packed in a box.
Further information
You can obtain more information from your doctor, pharmacist or the MS Society in your State, or by telephoning the MS Alliance on 1800 852 289 in Australia or 0800 852 289.
Who distributes Dimethyl Fumarate Sandoz
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
This leaflet was prepared in April 2025.
® Registered Trade Mark. The trade marks mentioned in this material are the property of their respective owners.
Brand Information
| Brand name | Dimethyl Fumarate Sandoz |
| Active ingredient | Dimethyl fumarate |
| Schedule | S4 |
MIMS Revision Date: 01 February 2026
1 Name of Medicine
Dimethyl fumarate.
2 Qualitative and Quantitative Composition
Dimethyl Fumarate Sandoz (dimethyl fumarate [DMF]) is formulated as enteric coated granules enclosed within hard gelatin capsules, containing the active ingredient dimethyl fumarate.
Each Dimethyl Fumarate Sandoz capsule contains 120 mg or 240 mg DMF. The inactive ingredients of Dimethyl Fumarate Sandoz are: croscarmellose sodium, colloidal anhydrous silica, sodium stearyl fumarate, methacryclic acid copolymer, purified talc, triethyl citrate, methacrylic acid - ethyl acrylate copolymer (1:1) dispersion, glyceryl monostearate 40-55 per cent, polysorbate 80, gelatin, titanium dioxide, iron oxide yellow, brilliant blue FCF, printing ink (shellac glaze, iron oxide black, propylene glycol).
3 Pharmaceutical Form
Modified release capsules.
120 mg capsules. Dimethyl Fumarate Sandoz is supplied as white and light-green capsules printed with "120 mg" on the capsule body. Each capsule contains 120 mg DMF.
240 mg capsules. Dimethyl Fumarate Sandoz is supplied as light-green capsules printed with "240 mg" on the capsule body. Each capsule contains 240 mg DMF.
4 Clinical Particulars
4.1 Therapeutic Indications
Dimethyl Fumarate Sandoz is indicated in patients with relapsing multiple sclerosis to reduce the frequency of relapses and to delay the progression of disability.
4.2 Dose and Method of Administration
The starting dose for Dimethyl Fumarate Sandoz is 120 mg twice a day orally. After 7 days, increase to the recommended dose of 240 mg twice a day orally.
The capsule or its contents should not be crushed, divided or dissolved as the enteric coating of the granules prevents irritant effects on the gut.
Temporary dose reduction to 120 mg twice a day may reduce the occurrence of flushing and gastrointestinal (GI) side effects. Within 1 month, the recommended dose of 240 mg twice a day orally should be resumed.
Dimethyl Fumarate Sandoz can be taken with or without food. For those patients who may experience gastrointestinal or flushing side effects, taking Dimethyl Fumarate Sandoz with food may improve tolerability.
Administration of 325 mg non-enteric coated aspirin prior to DMF dosing reduced the occurrence and severity of flushing in a healthy volunteer study (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
DMF has not been studied in patients with renal or hepatic impairment. Based on clinical pharmacology studies, no dose adjustments are needed.
4.3 Contraindications
Dimethyl Fumarate Sandoz is contraindicated in patients with known hypersensitivity to DMF or any excipients in this product.
4.4 Special Warnings and Precautions for Use
Infection. Decreases in lymphocyte counts observed in patients treated with DMF in clinical trials were not associated with increased frequencies of infections. However, due to the risk of serious, possibly fatal infection, patients who develop lymphopenia as a result of treatment with Dimethyl Fumarate Sandoz require close monitoring. Patients should be instructed to report symptoms of infection to their physician. For patients with signs and symptoms of serious infections, interrupting treatment with Dimethyl Fumarate Sandoz should be considered until the infection(s) resolves.
Lymphopenia. Dimethyl Fumarate Sandoz may decrease lymphocyte counts (see Section 4.8 Adverse Effects (Undesirable Effects)). In the MS placebo controlled trials, mean lymphocyte counts decreased by approximately 30% during the first year of treatment with DMF and then remained stable. WBC counts < 3.0 x 109/L and lymphocyte counts < 0.5 x 109/L were reported in 6 to 7% of subjects given DMF. Prior to initiating treatment with Dimethyl Fumarate Sandoz, a recent complete blood count (CBC) including lymphocytes (i.e. within 6 months) is recommended. A CBC, including lymphocytes, is also recommended, after 6 months of treatment and every 6 to 12 months thereafter, and as clinically indicated. In clinical studies, 9% of patients had lymphocyte counts ≥ 0.5 x 109/L and < 0.8 x 109/L for at least six months. 2% experienced lymphocyte counts < 0.5 x 109/L, for at least six months and in this group the majority of lymphocyte counts remained < 0.5 x 109/L with continued therapy. In controlled and uncontrolled clinical studies, the mean time for lymphocyte counts to return to normal after discontinuing DMF treatment was 4.7 weeks in patients without prolonged, severe lymphopenia and 29 weeks in patients with prolonged, severe lymphopenia (patients with lymphocyte counts < 0.5 x 109/L for six months or greater).
Consider interruption of Dimethyl Fumarate Sandoz in patients with lymphocyte counts < 0.5 x 109/L persisting for more than six months. Lymphocytes counts should be followed until recovery. Assess the benefit/risk in patients that experience moderate lymphopenia for more than 6 months.
Interrupting treatment should be considered in patients with serious infections until the infection(s) resolved. Dimethyl Fumarate Sandoz has not been studied in patients with pre-existing low lymphocyte counts and caution should be exercised when treating these patients.
Progressive multifocal leukoencephalopathy. Progressive multifocal leukoencephalopathy (PML) has occurred in the setting of lymphopenia (< 0.91 x 109/L) in patients with multiple sclerosis (MS) treated with DMF (see Section 4.8 Adverse Effects (Undesirable Effects)). These PML cases have occurred predominantly in the setting of prolonged moderate to severe lymphopenia. PML is an opportunistic viral infection of the brain that may lead to death or severe disability.
The symptoms of PML may be similar to a MS relapse. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes. At the first sign or symptom suggestive of PML, withhold Dimethyl Fumarate Sandoz and perform an appropriate diagnostic evaluation.
Anaphylactic reactions. Cases of anaphylaxis have been reported following DMF administration. These reactions generally occurred after the first dose, but may occur at any time during treatment, and may be serious and life threatening. Patients should be instructed to discontinue Dimethyl Fumarate Sandoz and seek immediate medical care if they experience signs or symptoms of anaphylaxis. Treatment should not be restarted (see Section 4.8 Adverse Effects (Undesirable Effects), Post-marketing experience).
Herpes zoster infections. Serious cases of herpes zoster have occurred with DMF, including disseminated herpes zoster, herpes zoster ophthalmicus, herpes zoster meningoencephalitis and herpes zoster meningomyelitis. These events may occur at any time during treatment. Monitor patients on Dimethyl Fumarate Sandoz for signs and symptoms of herpes zoster. If herpes zoster occurs, appropriate treatment for herpes zoster should be administered. Consider withholding Dimethyl Fumarate Sandoz treatment in patients with serious infections until the infection has resolved (see Section 4.8 Adverse Effects (Undesirable Effects), Post-marketing experience).
Vaccination. Patients taking Dimethyl Fumarate Sandoz may receive non-live vaccines (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). The safety of administration of live attenuated vaccines during treatment with DMF has not been evaluated in clinical trials. Live vaccines have a potential risk of clinical infection and are not recommended during treatment with Dimethyl Fumarate Sandoz.
Serious gastrointestinal reactions. Serious gastrointestinal reactions, including perforation, ulceration, haemorrhage, and obstruction, some with fatal outcomes, have been reported in the post-marketing setting with the use of fumaric acid esters, including DMF, with or without concomitant aspirin use. The majority of these events have occurred within 6 months of fumaric acid ester treatment initiation. In controlled clinical trials, the incidence of serious gastrointestinal adverse events was 1% in patients treated with DMF; these events, none of which were fatal, included vomiting (0.3%) and abdominal pain (0.3%) (see Section 4.8 Adverse Effects (Undesirable Effects)).
Use in renal impairment. In clinical trials with patients with multiple sclerosis, adverse events of proteinuria (proteinuria, microalbuminuria and urine albumin present) were reported at slightly higher frequencies in patients treated with DMF compared to patients that received placebo. The significance of these clinical observations is not known at this time.
Prior to initiating treatment with Dimethyl Fumarate Sandoz, urinalysis should be available (within 6 months prior to starting therapy). During treatment, urinalysis is recommended annually and as clinically indicated.
The use of DMF in patients who receive chronic treatment with medications that are associated with potential nephrotoxic risk (e.g. aminoglycosides, diuretics, NSAIDs, lithium) has not been evaluated. Therefore, caution should be exercised if Dimethyl Fumarate Sandoz is used in patients receiving chronic treatment with such medications.
Use in the elderly. There are limited data available for the use of DMF in patients aged 65 years and over, therefore it is unknown whether elderly patients respond differently to younger patients.
Paediatric use. The safety and effectiveness of DMF in paediatric patients with multiple sclerosis below the age of 18 have not been established.
Effects on laboratory tests. There are no data available on whether DMF interferes with laboratory tests.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In humans, DMF is extensively metabolised by esterases before it reaches the systemic circulation and further metabolism occurs through the tricarboxylic acid (TCA) cycle, with no involvement of the cytochrome P450 (CYP) system. Potential drug interaction risks were not identified from in vitro CYP-inhibition and induction studies, a p-glycoprotein study, or studies of the protein binding of DMF and MMF.
A pharmacokinetic study with a combined oral contraceptive has been performed with dimethyl fumarate. There were no relevant effects of dimethyl fumarate on the pharmacokinetic profile of norelgestromin and ethinyl estradiol. No interaction studies have been performed with oral contraceptives containing other progestogens; however an effect of Dimethyl Fumarate Sandoz on their exposure is not expected.
Commonly used drugs in patients with multiple sclerosis, intramuscular (IM) interferon beta-1a and GA, were clinically tested for potential drug-interactions with DMF and did not alter the pharmacokinetic profile of DMF. Aspirin (non-enteric coated), 325 mg, when administered approximately 30 minutes before DMF, did not alter the pharmacokinetic profile of DMF.
Patients treated with DMF were able to mount an effective immune response to inactivated neoantigen (first vaccination), recall antigen (re-exposure), or polysaccharide antigen in a clinical study in patients with relapsing forms of MS. This response was comparable to patients treated with non-pegylated interferons. Patients taking Dimethyl Fumarate Sandoz may receive non-live vaccines. No clinical data are available on the efficacy and safety of live attenuated vaccines in patients taking DMF.
During treatment with Dimethyl Fumarate Sandoz, simultaneous use of other fumaric acid derivatives (topical or systemic) should be avoided as such clinical scenarios have not been studied.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Data from nonclinical studies do not suggest that DMF would be associated with an increased risk of reduced fertility.
Administration of DMF to male rats at daily oral doses of up to 7-9 times the maximum recommended human dose (MRHD) based on mg/m2 prior to and during mating had no effects on fertility. Administration of DMF to female rats at daily oral doses of up to 5-6 times the MRHD based on mg/m2 prior to and during mating, and continuing to Day 7 of gestation, delayed oestrus cycling at the highest dose but had no effects on fertility.
Use in pregnancy. (Category B1)
Oral treatment of pregnant rats and rabbits during the period of organogenesis with dimethyl fumarate showed no evidence of teratogenicity. In rats, the high dose of 250 mg/kg/day (9 times the MRHD based on AUC) reduced foetal weight and caused minor impairment of ossification in foetuses, concomitant with maternal toxicity; the no-effect dose for foetal effects was 100 mg/kg/day (4 times the MRHD based on AUC). In rabbits, the high dose of 150 mg/kg/day (14 times the MRHD based on AUC) elicited toxicity and abortions in does, but did not affect embryofoetal development.
The effects of DMF on labour and delivery are unknown. In rats given oral dimethyl fumarate from early gestation to the end of lactation, there were no effects on delivery at doses up to 250 mg/kg/day (9 times the MRHD based on AUC).
Dimethyl Fumarate Sandoz should be used during pregnancy only if clearly needed and if the potential benefit justifies the potential risk to the foetus.
Use in lactation. It is not known whether this drug is excreted in milk. A risk to the newborn/infant cannot be excluded. A decision must be made whether to discontinue breastfeeding or to discontinue Dimethyl Fumarate Sandoz treatment. The benefit of breast-feeding for the child and the benefit of treatment for the woman should be taken into account.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems (Australia) or nzphvc.otago.ac.nz/reporting/ (New Zealand).
The most common adverse reactions (incidence ≥ 10% and > 2% than placebo) for DMF were flushing and gastrointestinal (GI) events (i.e. diarrhoea, nausea, abdominal pain, upper abdominal pain).
The most commonly reported adverse events leading to discontinuation (incidence > 1%) in patients treated with DMF were flushing (3%) and gastrointestinal events (4%).
In placebo-controlled and uncontrolled clinical studies, a total of 2513 patients have received DMF for periods up to 12 years and been followed for periods up to 13 years with an overall exposure equivalent to 11318 person-years. A total of 1169 patients have received at least 5 years of treatment with DMF, and 426 patients have received at least 10 years of treatment with DMF. The experience in uncontrolled clinical trials is consistent with the experience in the placebo-controlled clinical trials.
In the two Phase 3 placebo-controlled trials, 1529 patients received DMF with an overall exposure of 2371 person-years (see Section 5.1 Pharmacodynamic Properties, Clinical trials). The adverse reactions presented in Table 2 are based on safety information from 769 patients treated with DMF 240 mg twice a day and 771 patients treated with placebo.
The adverse reactions are presented as MedDRA preferred terms under the MedDRA system organ class.
The incidence of the adverse reactions in Tables 1 and 2 is expressed according to the following categories: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000).
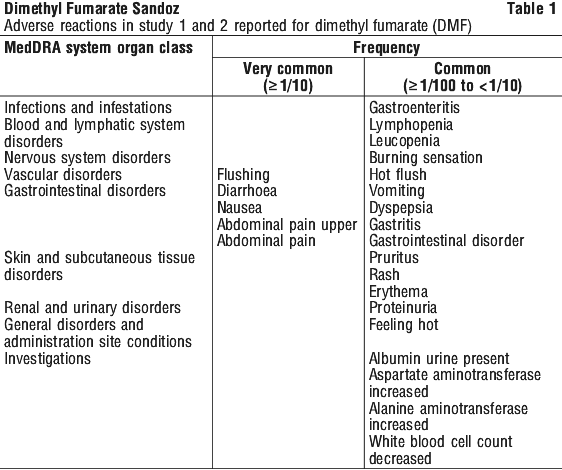
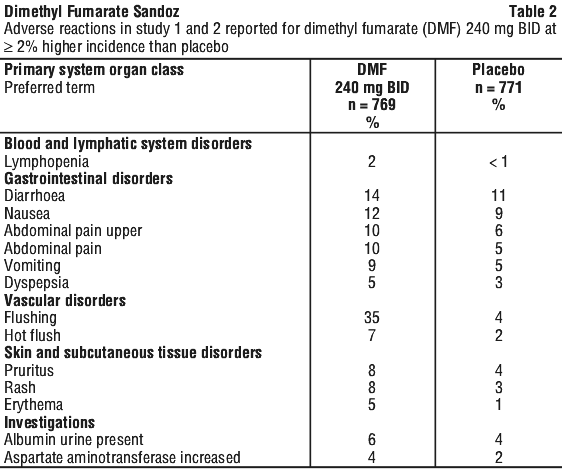
Description of selected adverse events. Flushing. The incidence of patients with flushing events (e.g. warmth, redness, itching, burning sensation) was higher early in the course of treatment (primarily in month 1) and decreased over time, which might indicate that this symptom became less prevalent with continued use. In patients with flushing, the majority had flushing events that were mild or moderate in severity. Overall, 3% of patients treated with DMF discontinued due to flushing. The incidence of serious flushing which may be characterised by generalised erythema, rash and/or pruritus was seen in less than 1% of patients treated with DMF (see Section 4.2 Dose and Method of Administration).
Gastrointestinal. The incidence of patients with GI events (e.g. nausea, vomiting, diarrhoea, abdominal pain, upper abdominal pain and dyspepsia) was higher early in the course of treatment (primarily in month 1) and decreased over time in patients treated with DMF compared with placebo. Four percent (4%) of patients treated with DMF discontinued due to gastrointestinal events. The incidence of serious GI events, including gastroenteritis and gastritis, was seen in less than 1% of patients treated with DMF.
Hepatic transaminases. In placebo-controlled studies, elevations of hepatic transaminases were observed. The majority of patients with elevations had hepatic transaminases that were < 3 times the upper limit of normal (ULN). The increased incidence of elevations of hepatic transaminases in patients treated with DMF relative to placebo was primarily seen during the first 6 months of treatment. Elevations of alanine aminotransferase and aspartate aminotransferase ≥ 3 times ULN, respectively, were seen in 5% and 2% of patients treated with placebo and 6% and 2% of patients treated with DMF. Discontinuations due to elevated hepatic transaminases were < 1% and similar in patients treated with DMF or placebo. Elevations in transaminases ≥ 3 times ULN with concomitant elevations in total bilirubin > 2 times ULN were not observed during placebo-controlled studies but have been observed in the post-marketing experience (see Post-marketing experience below).
Haematological. In the placebo-controlled studies, most patients (> 98%) had normal lymphocyte values prior to initiating treatment. Upon treatment with DMF, lymphocytes counts decreased over the first year with a subsequent plateau. On average, lymphocyte counts decreased by approximately 30% of baseline value. Mean and median lymphocyte counts remained within normal limits. Patients with lymphocyte counts < 0.5 x 109/L were observed in < 1% of patients treated with placebo and 6% of patients treated with DMF. In controlled and uncontrolled clinical studies, 2% of patients experienced lymphocyte counts < 0.5 x 109/L for at least six months. In these patients, the majority of lymphocyte counts remained < 0.5 x 109/L with continued therapy.
The incidence of infections (58% vs 60%) and serious infections (2% vs 2%) was similar in patients treated with placebo or DMF, respectively. An increased incidence of infections and serious infections was not observed in patients with lymphocyte counts < 0.8 x 109/L or 0.5 x 109/L. A transient increase in mean eosinophil counts was seen during the first 2 months of therapy.
Post-marketing experience. In post marketing experience, hypersensitivity reactions including urticaria, angioedema, and difficulty breathing have been reported following DMF administration. Cases of anaphylaxis have also been reported (see Section 4.4 Special Warnings and Precautions for Use, Anaphylactic reactions).
Progressive multifocal leukoencephalopathy has occurred in the setting of lymphopenia (< 0.91 x 109/L) following DMF administration. These PML cases have occurred predominantly in the setting of prolonged moderate to severe lymphopenia (see Section 4.4 Special Warnings and Precautions for Use, Progressive multifocal leukoencephalopathy).
Liver function abnormalities (elevations in transaminases ≥ 3 times ULN with concomitant elevations in total bilirubin > 2 times ULN) have been reported following DMF administration in post marketing experience. These abnormalities resolved upon treatment discontinuation over a varying period of time. Therefore, ongoing monitoring of LFTs is recommended in patients being treated with Dimethyl Fumarate Sandoz, as clinically indicated.
Herpes zoster infection has been reported with DMF administration in post marketing experience. The majority of cases were non-serious (see Section 4.4 Special Warnings and Precautions for Use, Herpes zoster infections).
Other serious opportunistic infections have occurred with DMF, including cases of serious viral (herpes simplex virus, West Nile virus, cytomegalovirus), fungal (Candida and Aspergillus), and bacterial (Nocardia, Listeria monocytogenes, Mycobacterium tuberculosis) infections.
Rhinorrhoea and alopecia has been reported with DMF administration in post marketing experience.
4.9 Overdose
Cases of overdose with DMF have been reported. There are no known therapeutic interventions to enhance elimination of Dimethyl Fumarate Sandoz nor is there a known antidote. In the event of overdose, it is recommended that symptomatic supportive treatment be initiated as clinically indicated.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia) and 0800 POISON or 0800 764766 (New Zealand).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The mechanism by which DMF exerts therapeutic effects in multiple sclerosis is not fully understood. Nonclinical studies indicate that pharmacodynamic responses to DMF appear to be mediated, at least in part, through activation of the nuclear factor (erythroid-derived 2)-like 2 (Nrf2) transcriptional pathway, which is a critical cellular defense system for responding to a variety of potentially toxic stimuli through up-regulation of antioxidant response genes.
Biological response markers of Nrf2 activation (e.g. NAD(P)H dehydrogenase, quinone 1 [NQO1]) are detected at elevated levels in blood from patients with multiple sclerosis following 12 or 48 weeks of oral dosing with DMF. These clinical data appear to be consistent with nonclinical studies demonstrating DMF-dependent up-regulation of Nrf2 antioxidant response genes in multiple tissue types, although the magnitude of up-regulation observed in tissues of the central nervous system was small. The relationships between blood NQO1 levels and the mechanism(s) by which DMF exerts its effects in multiple sclerosis are unknown.
In nonclinical and clinical studies, DMF demonstrates anti-inflammatory and immunomodulatory properties. DMF and monomethyl fumarate (MMF), the primary metabolite of DMF, significantly reduce immune cell activation and subsequent release of pro-inflammatory cytokines in response to inflammatory stimuli, and moreover affects lymphocyte phenotypes through a down-regulation of pro-inflammatory cytokine profiles (TH1, TH17), and biases towards anti-inflammatory production (TH2). DMF demonstrates therapeutic activity in models of inflammatory and neuroinflammatory injury, and also appears to promote improvement in blood brain barrier integrity. All of these anti-inflammatory effects appear consistent with the significant clinical activity of DMF in reducing brain lesions and relapses in multiple sclerosis patients.
In nonclinical studies MMF was shown to penetrate into the central nervous system where it promotes cyto- and neuro-protective responses. DMF and/or MMF significantly improve cell viability after oxidative challenge in primary cultures of astrocytes and neurons, suggesting that DMF and MMF prevent neurodegeneration in response to toxic stress. DMF showed therapeutic benefit in acute neurotoxic injury models and models of neurodegenerative disease. These nonclinical data combined with imaging and functional endpoints from clinical studies suggest DMF may promote a neuroprotective benefit in the central nervous system.
Potential to prolong the QTc interval. Single doses of 240 mg or 360 mg DMF did not have any effect on the QTc interval when compared to placebo in a thorough QTc study.
Clinical trials. The efficacy and safety of DMF was demonstrated in three studies that evaluated DMF taken either twice or three times a day in patients with relapsing-remitting multiple sclerosis (RRMS).
The starting dose for DMF was 120 mg twice or three times a day for the first 7 days, followed by an increase to either 240 mg twice or three times a day. Two of the three studies (Study 1 and Study 2) included patients with Expanded Disability Status Scale (EDSS) scores ranging from 0 to 5, who had experienced at least 1 relapse during the year prior to randomisation, or, within 6 weeks of randomisation had a brain magnetic resonance imaging (MRI) demonstrating at least one gadolinium-enhancing (Gd+) lesion.
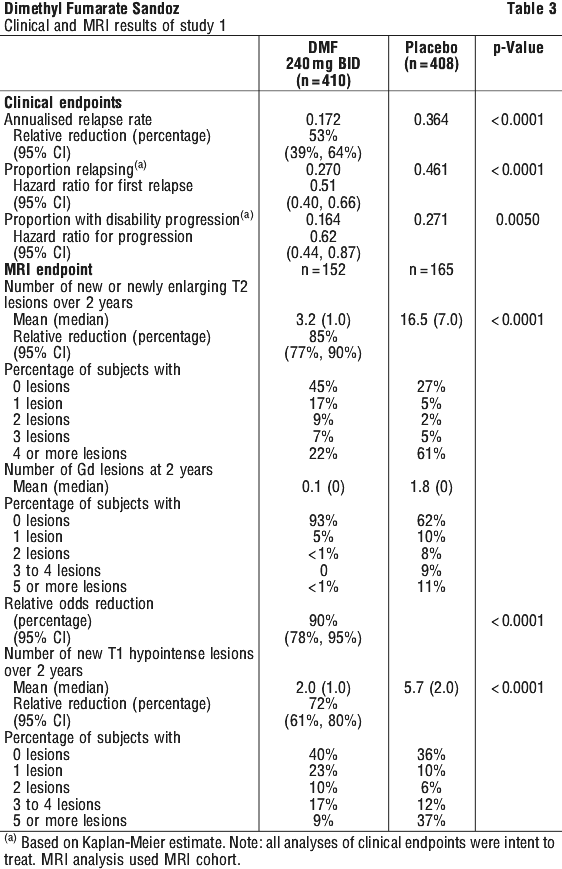
Study 1 (DEFINE) was a 2-year randomised, double-blind, placebo-controlled study in 1234 patients with RRMS who had not received interferon-beta or glatiramer acetate (GA) for at least the previous 3 months or natalizumab for at least the previous 6 months. Neurological evaluations were performed at baseline, every 3 months and at time of suspected relapse. MRI evaluations were performed at baseline, month 6, and year 1 and 2. The primary endpoint in Study 1 was the reduction in the proportion of patients relapsed at 2 years. Patients were randomised to receive DMF 240 mg twice a day (n=410), DMF 240 mg three times a day (n=416), or placebo (n=408) for up to 2 years. Median age: 39 years, median years since diagnosis: 4.0 years and median EDSS score at baseline: 2.0. Median time on study was 96 weeks for all three treatment groups.
The proportion of patients relapsed was significantly lower in the group treated with DMF than in the group treated with placebo at 2 years. Secondary endpoints at 2 years included the number of new or newly enlarging T2 hyperintense lesions, number of Gd-enhancing lesions, annualised relapse rate (ARR), and time to confirmed disability progression. Confirmed disability progression was defined as at least a 1 point increase from baseline EDSS (1.5 point increase for patients with baseline EDSS of 0) sustained for 12 weeks. DMF had a clinically meaningful and statistically significant effect on all primary and secondary study endpoints. The 240 mg three times daily dose resulted in no additional benefit over the DMF 240 mg twice daily dose. The results for this study are shown in Table 3.
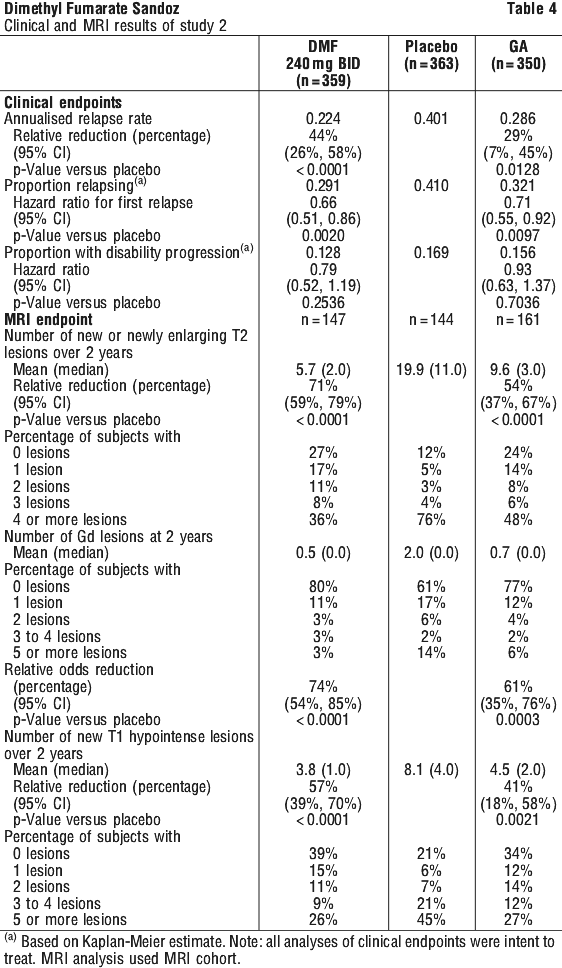
Patients had not received interferon-beta for at least the previous 3 months, natalizumab for at least the previous 6 months and had not previously received GA. The efficacy and safety evaluations were similar to Study 1 and the endpoints were broadly consistent, but the primary endpoint of Study 2 was the annualized relapse rate at 2 years, whereas the primary endpoint of Study 1 was the proportion of subjects relapsed at 2 years. Median age: 37 years, median years since diagnosis: 3.0 years and median EDSS score at baseline: 2.5. Patients were randomised to receive DMF 240 mg twice a day (n=359), DMF 240 mg three times a day (n=344), placebo (n=363) or glatiramer acetate (n=351) for up to 2 years. Median time on study was 96 weeks for all treatment groups.
The annualised relapse rate was significantly lower in patients treated with DMF than in patients treated with placebo at 2 years. Secondary endpoints at 2 years included the number of new or newly enlarging T2 hyperintense lesions, number of T1 hypointense lesions, proportion of patients relapsed and time to confirmed disability progression defined as in Study 1.
DMF had a clinically meaningful and statistically significant effect on the primary endpoint and secondary relapse and MRI endpoints. In Study 2, the annualised relapse rate for glatiramer acetate versus placebo was 0.286 and 0.401, corresponding to a reduction of 29% (p=0.013) which is consistent with approved product labelling. The results for this study are shown in Table 4.
Study 3 (ENDORSE) enrolled eligible patients from Study 1 and 2 into an 8-year two phase extension study of 1736 patients with RRMS. The first phase was a multicentre, parallel group, randomised, dose blind, dose comparison study in which patients received DMF at a dose of 240 mg twice a day or 240 mg three times a day. The second phase was an open label study during which all patients received DMF at a dose of 240 mg twice a day. Eligible patients were enrolled at Week 96 (Visit 24) of their previous Study 1 or Study 2 visit, which served as the Baseline Visit for this extension study.
The primary objective of Study 3 was to evaluate the long-term safety of DMF. The secondary objectives were to evaluate the long-term efficacy of DMF using clinical endpoints (including relapse and ARR) and disability progression (EDSS) and on MS brain lesions on MRI scans.
The median age of patients was 40.0 years. Most patients (945 participants, 54%) were in the study for 7 years or longer and the median time spent in the study (min, max) was 6.759 (0.04, 10.98) years.
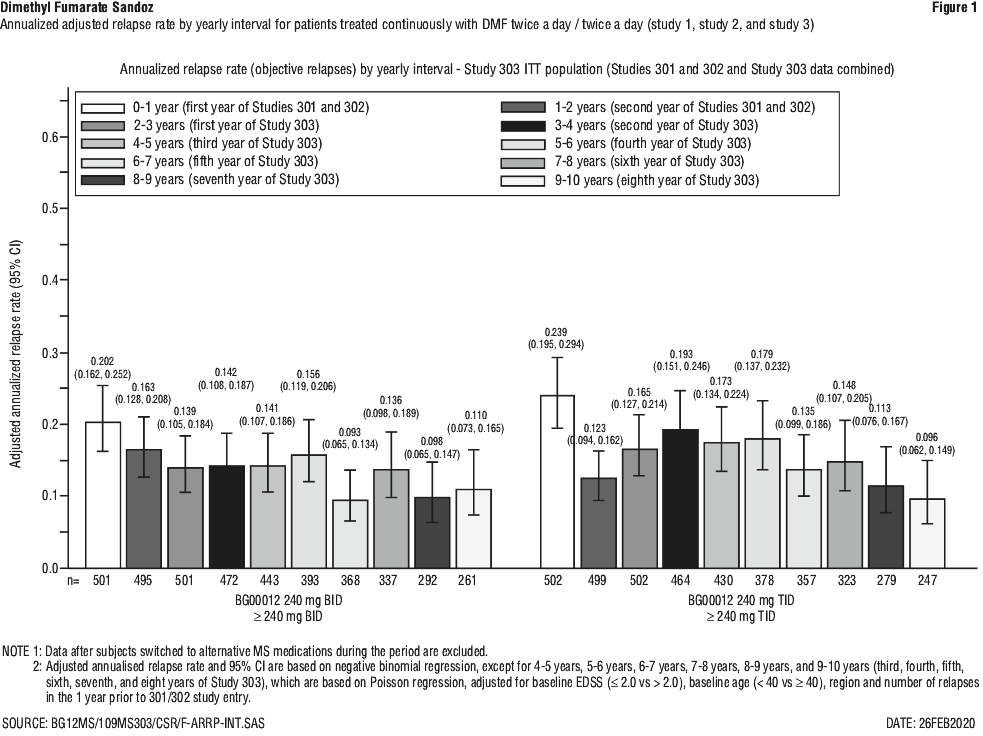
In the first year of treatment with DMF in Study 3, the adjusted ARR (95% CI) ranged from 0.125 (0.084, 0.188) to 0.183 (0.108, 0.308), and remained low in the eighth year, ranging from 0.077 (0.039, 0.153) to 0.129 (0.063, 0.265), in all treatment arms. During the overall study period, the adjusted ARR (95% CI) ranged from 0.126 (0.098, 0.162) to 0.185 (0.129, 0.265) and the majority of patients treated with DMF (between 59% and 69%) had no relapses.
The estimated proportion of relapse (95% CI), at 8 years (384 weeks), in Study 3 ranged from 0.414 (0.314, 0.531) to 0.502 (0.426, 0.584).
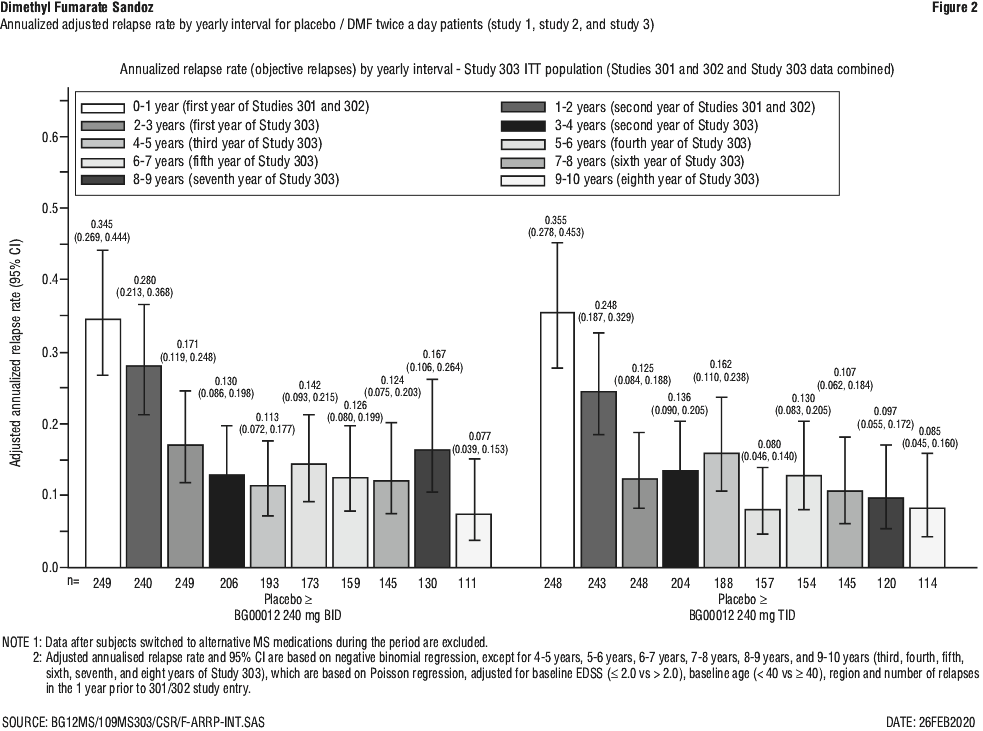
In an integrated analysis of Study 1 and Study 2 with Study 3, for patients continuously treated with DMF twice a day/twice a day (n = 501; patients treated with DMF 240 mg twice a day in Study 1 or 2 and then DMF 240 mg twice a day in Study 3), adjusted ARR was 0.187 (95% CI, 0.156, 0.224) in Study 1 and 2, and was 0.141 (95% CI, 0.119, 0.167) in Study 3. The data in Figure 1 demonstrates that the adjusted ARR in patients treated with DMF was steady throughout the treatment time in Study 1 or 2 and Study 3. For placebo/DMF patients (n = 249; patients treated with placebo in Study 1 or Study 2 and then switched to DMF in Study 3), adjusted ARR was 0.330 (95% CI, 0.266, 0.408) and decreased after initiating DMF, in Study 3, to 0.149 (95% CI, 0.116, 0.190), shown in Figure 2.
In Study 3, the mean (median) EDSS score at baseline ranged from 2.37 (2.0) to 2.64 (2.0). The estimated proportion of patients with confirmed progression (95% CI) in the eighth year of Study 3 after treatment with DMF ranged from 0.314 (0.268, 0.365) to 0.387 (0.311, 0.475).
In an integrated analysis of Study 1 and Study 2 with Study 3, at Week 480, the estimated proportion (95%) of patients with confirmed disability progression (Study 1, 2, and 3 data combined) was 0.349 (0.302, 0.401) in the DMF twice a day/twice a day group and 0.362 (0.292, 0.443) in the placebo/DMF group.
In Study 3, 752 patients were included in an MRI cohort, which included patients who had previously been included in the MRI cohort of Study 1 or Study 2. Due to sample size restrictions (by year 8 all groups has < 30 patients), MRI results are presented only through Year 6 of Study 3. Across all treatment arms, the adjusted mean number of new or newly enlarging T2 lesions relative to Study 3 baseline over 6 years ranged from 3.911 to 8.650 (the adjusted mean was based on negative binomial regression, adjusted for region and baseline volume of T2 lesions). The median number of new or newly enlarging T2 lesions over 6 years ranged from 1.0 to 3.0. Across all treatment arms, the mean (median) number of Gd+ lesions at 6 years ranged from 0.0 (0.0) to 0.7 (0.0). The percentage of patients with no Gd+ lesions ranged from 84% to 100%. The mean number of new T1 hypointense lesions over 6 years, adjusted for region and baseline volume of T1 lesions (based on negative binomial regression), ranged from 1.060 to 4.326. The median ranged from 1.0 to 2.5.
5.2 Pharmacokinetic Properties
Orally administered DMF undergoes rapid presystemic hydrolysis by esterases and is converted to its primary metabolite, MMF, which is also active. DMF is not quantifiable in plasma following oral administration of DMF. Therefore, all pharmacokinetic analyses related to DMF were performed with plasma MMF concentrations. Pharmacokinetic data were obtained in subjects with multiple sclerosis and healthy volunteers.
Absorption. The Tmax of DMF is 2-2.5 hours. As DMF is protected by an enteric coating, absorption does not commence until the drug leave the stomach (generally less than 1 hour). Following 240 mg administered twice a day with food, the median peak (Cmax) was 1.72 mg/L and overall (AUC) exposure was 8.02 h.mg/L in subjects with MS. Cmax and AUC increased approximately dose proportionally in the dose range studied (120 mg to 360 mg).
Food does not have a clinically significant effect on exposure of DMF. Therefore, Dimethyl Fumarate Sandoz may be taken with or without food.
Distribution. The apparent volume of distribution following oral administration of 240 mg DMF varies between 60 and 90 L. Human plasma protein binding of MMF generally ranges between 27%-40%.
Metabolism. In humans, DMF is extensively metabolised by esterases, which are ubiquitous in the gastrointestinal tract, blood and tissues, before it reaches the systemic circulation. Further metabolism occurs through the tricarboxylic acid (TCA) cycle, with no involvement of the cytochrome P450 (CYP) system. A single 240 mg 14C-DMF dose study identified monomethyl fumarate, fumaric and citric acid, and glucose as the major metabolites in plasma. The downstream metabolism of fumaric and citric acid occurs through the TCA cycle, with exhalation of CO2 serving as a primary route of elimination.
Excretion. Exhalation of CO2 is the primary route of DMF elimination accounting for approximately 60% of the dose. Renal and faecal elimination are secondary routes of elimination, accounting for 15.5% and 0.9% of the dose respectively.
The terminal half-life of MMF is short (approximately 1 hour) and no circulating MMF is present at 24 hours in the majority of individuals. Accumulation of parent drug or MMF does not occur with multiple doses of DMF at the therapeutic regimen.
DMF exposure increases in an approximately dose proportional manner with single and multiple doses in the 120 to 360 mg dose range studied.
Based on the results of ANOVA, body weight is the main covariate of exposure (by Cmax and AUC) in relapsing remitting multiple sclerosis (RRMS) subjects, but did not affect safety and efficacy measures evaluated in the clinical studies. Gender and age did not have a statistically significant impact on Cmax and AUC.
Race and ethnicity have no effect on the pharmacokinetics of Dimethyl Fumarate Sandoz.
Since the renal pathway is a secondary route of elimination for DMF, accounting for less than 16% of the dose administered, evaluation of pharmacokinetics in individuals with renal impairment was not conducted.
As DMF and MMF are metabolised by esterases, without the involvement of the CYP450 system, evaluation of pharmacokinetics in individuals with hepatic impairment was not conducted.
5.3 Preclinical Safety Data
Genotoxicity. DMF and MMF were negative in the following in vitro assays (bacterial reverse mutation test, chromosomal aberration assay in human lymphocytes, and [DMF only] a forward mutation assay in Chinese hamster ovary cells) and in vivo assays (rat micronucleus assay with DMF, bone marrow cytogenetic test with MMF). Results did not suggest a risk of genotoxicity in patients.
Carcinogenicity. Carcinogenicity studies were conducted in mice and rats with oral dosing with DMF for up to 2 years. Doses in mice were 25, 75, 200 and 400 mg/kg/day and in rats were 25, 50, 100 and 150 mg/kg/day.
Incidences of tumours in the nonglandular stomach were increased in mice and rats (squamous cell papillomas and carcinomas in mice and rats; leiomyosarcomas and fibrosarcomas in mice). As the nonglandular stomach of mice and rats does not have a human counterpart, these tumours are not considered to be a risk in patients.
Incidences of renal tubular adenomas (benign) and carcinomas were increased in both mice and rats. Higher incidences of at least one of these tumours were observed at doses of 75 mg/kg/day in mice (1.3 times the MRHD based on AUC) and 100 mg/kg/day in rats (2 times the MRHD based on AUC), with significantly higher incidences at 200 mg/kg/day in mice and 150 mg/kg/day in rats (4 times the MRHD in both species). The clinical relevance of these findings is unclear but they might pose a human risk.
In male rats, an increase in the incidence of benign interstitial cell (Leydig cell) adenoma of the testes was observed at ≥ 100 mg/kg/day (2 times the MRHD based on AUC). The rat is particularly sensitive to developing this tumour type and the relevance of these findings to human risk is considered low.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
120 mg capsules. Capsules are supplied in Al/PVC/PVDC foil blister packs, in pack sizes of 14 and 56.
240 mg capsules. Capsules are supplied in Al/PVC/PVDC foil blister packs, in pack sizes of 14 and 56.
*Not all presentations are currently marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. The structural formula of DMF is shown below.
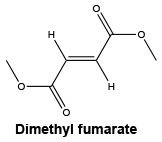
CAS number. The CAS registry number is 624-49-7.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
17 November 2021
Date of Revision
22 December 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.