Diphereline
Brand Information
| Brand name | Diphereline |
| Active ingredient | Triptorelin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Diphereline.
Summary CMI
Diphereline®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I being given Diphereline?
Diphereline contains the active ingredient triptorelin. In adult men, Diphereline is used to treat prostate cancer that has spread into surrounding tissue and/or to other parts of the body. In children, Diphereline 22.5 mg is used to treat puberty that occurs at a very young age (Central Precocious Puberty).
For more information, see Section 1. Why am I being given Diphereline? in the full CMI.
2. What should I know before I am given Diphereline?
Do not use if you have ever had an allergic reaction to triptorelin or any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I am given Diphereline? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Diphereline and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How am I given Diphereline?
Your doctor or nurse will give you an injection of Diphereline into your muscle (intramuscular). More instructions can be found in Section 4. How am I given Diphereline? in the full CMI.
5. What should I know while being given Diphereline?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given Diphereline? in the full CMI.
6. Are there any side effects?
Like all medicines, Diphereline can cause side effects, although not everybody gets them. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Some side effects may need medical attention, tell your doctor straight away if you notice any of the following:
- In men: severe back pain, difficulty passing urine. These could be signs of temporary worsening of your prostate tumour.
- In men: chest pain. This could be a sign of heart-related problems.
- If your child complains of: vision problems, severe headaches, pain behind the eye, ringing in the ears, dizziness, nausea. These could be signs of increased pressure around the brain.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
Diphereline®
Active ingredient(s): triptorelin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Diphereline. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about being given Diphereline.
Where to find information in this leaflet:
1. Why am I being given Diphereline?
2. What should I know before I am given Diphereline?
3. What if I am taking other medicines?
4. How am I given Diphereline?
5. What should I know while being given Diphereline?
6. Are there any side effects?
7. Product details
1. Why am I being given Diphereline?
Diphereline contains the active ingredient triptorelin. Diphereline belongs to a group of medicines called Gonadotrophin Releasing Hormone agonists (GnRHa).
In males, Diphereline works by lowering the production of testosterone. Testosterone is a natural male sex hormone. In some types of prostate cancer, testosterone may help the cancer cells to grow. By lowering testosterone, Diphereline may slow or stop the growth of cancer.
In females, Diphereline lowers the levels of the hormone oestrogen. Oestrogen is a natural female sex hormone.
In adult men, Diphereline is used to treat prostate cancer that has spread into surrounding tissue and/or to other parts of the body. It is not a cure for prostate cancer.
In children, Diphereline 22.5 mg is used to treat puberty that occurs at a very young age (Central Precocious Puberty).
2. What should I know before I am given Diphereline?
Warnings
You should not be given Diphereline if:
- there are signs that the tumour doesn't rely on hormones to grow (check with your doctor).
- you have had surgical removal of your testes.
- you have severe back pain as a result of your prostate cancer spreading and pressing into the nerves of your backbone.
- you are allergic to triptorelin, polysorbate 80, Gonadotropin Releasing Hormone (GnRH) or any other Gonadotrophin Releasing Hormone agonists (GnRHa), or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include:
- rash, itching or hives on the skin
- swelling of the face, lips, tongue or other parts of the body
- shortness of breath, wheezing or trouble breathing
Check with your doctor if you:
- take any medicines for any other condition.
- have an enlargement (benign tumour) of your pituitary gland. If you are unaware of any enlargement, it may be discovered during treatment with Diphereline.
- are allergic to food, dyes, preservatives or any other medicines.
In men:
- have cancer related pain (metastatic pain).
- experience difficulty or pain when passing urine.
- have osteoporosis, a family history of osteoporosis or risk factors for developing osteoporosis (such as heavy drinking, smoking, a diet low in calcium, poor mobility, a slight build or treatment with steroid medicines or anticonvulsants).
- get sudden headaches, and/or have blurred vision.
- have high blood sugar or diabetes.
- have heart or vascular problems or other cardiovascular risk factors.
- have any heart or blood vessel conditions, including heart rhythm problems (arrhythmia), or are being treated with medicines for these conditions. The risk of heart rhythm problems may be increased when using Diphereline.
- are taking medicines to lower your blood pressure.
In children:
- If your child has progressive brain tumour, tell their doctor. This may affect the way your doctor decides to treat your child.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
You should not be given Diphereline if you are pregnant or breastfeeding.
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Mood changes and depression
There have been reports of mood changes and depression in patients taking GnRH analogues, such as Diphereline, which may be severe. In very rare cases, severe depression can lead to suicidal thoughts or attempts. If you are being given Diphereline and develop depressed mood or have any thoughts of self-harm, inform your doctor or nurse straight away.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Diphereline and affect how it works.
- medicines used to prevent blood clots (anti-coagulants), including warfarin. There is a possible risk of haematoma formation (bruising, bleeding) at the site of the injection.
- medicines that increase levels of another hormone, prolactin.
- medicines affecting secretion of gonadotrophins.
Diphereline might interfere with some medicines used to treat heart rhythm problems, such as:
- quinidine
- disopyramide
- amiodarone
- sotalol
Diphereline might increase the risk of heart rhythm problems when used with some other drugs, such as:
- methadone (used for pain relief and part of drug addiction detoxification)
- moxifloxacin (an antibiotic)
- antipsychotics (used for serious mental illnesses)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Diphereline.
4. How am I given Diphereline?
How much is given
Your doctor will decide how much to give depending on the condition being treated:
- a 3.75 mg injection given once a month, or
- a 11.25 mg injection given once every 3 months (4 times a year), or
- a 22.5 mg injection given every 6 months (2 times a year).
Only the 22.5 mg injection is used to treat Central Precocious puberty in children.
When am I given Diphereline
- Your doctor will decide how frequently and for how long you will be given the injection.
- Your child's doctor will decide when treatment should be stopped. Normally this will happen when your child is about 12-13 years old if they are a girl, and about 13-14 years old if they are a boy.
How am I given Diphereline
Your doctor or nurse will inject Diphereline into a muscle, usually your bottom.
If you forget to get your Diphereline injection
Tell your doctor as soon as you realise that you have missed an injection. You will then be given your next injection.
Make a doctor's appointment for your next injection date. Keep a diary of when your next dose is due.
If you are given too much Diphereline
Diphereline is given to you by a doctor or nurse, so it is unlikely that you or your child will be given too much. However, if you think that you or your child have been given too much Diphereline, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while being given Diphereline?
Things you should do
Call your doctor straight away if you:
experience any of the following symptoms within the first few weeks of treatment:
- pain in the bones or backbone
- difficulty passing urine
- weakness, tingling or numbness in your arms and legs
At the start of treatment with medicines like Diphereline (GnRHa medicines), you will have an increased amount of testosterone in your body which may cause the symptoms of your cancer to get worse.
Tell your doctor immediately if you continue to experience these symptoms. It may be a sign that the cancer is growing. Your doctor may give you other medicines to manage these symptoms.
Talk to your doctor or pharmacist if:
- your female child experiences some vaginal bleeding in the first month of treatment.
- you feel that your medicine is not helping your condition (or your child's condition).
Remind any doctor, dentist or pharmacist you visit that you (or your child) are using Diphereline.
Keep follow-up appointments with your doctor. It is important to have your follow-up doses at the appropriate times to get the best effects from your treatments.
Your doctor will monitor you during treatment to check that you do not develop high blood sugar or diabetes. You will also be monitored for any symptoms or signs of cardiovascular disease.
Things you should not do
- Do not stop using this medicine suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Diphereline affects you.
Diphereline may cause dizziness, blurred vision, or seizures in some people.
Be careful of your child riding bicycles or climbing (such as trees or playground equipment) until you know how Diphereline affects them.
Drinking alcohol
Tell your doctor if you drink alcohol.
Drinking alcohol may make dizziness or light-headedness worse when you are taking Diphereline.
Looking after your medicine
Your doctor will keep the medicine at the doctor's clinic or surgery, or at the pharmacy.
If you need to store Diphereline at home:
- Keep it in a cool, dry place, where the temperature stays below 25°C.
- Keep it in the original container.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Some side effects (for example, high blood pressure or changes in liver function) can only be found when your doctor does tests to check on your progress.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
Symptoms of an allergic reaction:
Signs of tumour flare:
Signs of thrombocytosis:
Signs of gout:
Signs of enlargement of pituitary gland (benign tumour):
Signs of changes in ECG (record of the electrical activity of the heart) (QT prolongation):
Signs of high blood pressure in the brain:
| Call your doctor or your child's doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Less serious side effects
| Less serious side effects | What to do |
| Prostate cancer: Gastrointestinal related:
Nervous system related:
Heart or circulatory system related:
Mental health related:
Reproductive system and breast related:
Skin related:
Muscle or bone related:
Kidney or bladder related:
General:
Heart or circulatory system related:
Mental health related:
Reproductive system and breast related:
Gastrointestinal related:
Nervous system related:
Skin related:
General:
Skin related:
Muscle or bone related:
Respiratory related:
| Speak to your doctor or your child's doctor if you or your child has any of these less serious side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Diphereline contains
| Active ingredient (main ingredient) | triptorelin embonate |
| Other ingredients (inactive ingredients) | polyglactin mannitol carmellose sodium polysorbate 80 sterile water |
Do not take this medicine if you are allergic to any of these ingredients.
What Diphereline looks like
Diphereline is available in 3.75 mg (1 month formulation), 11.25 mg (3 month formulation), and 22.5 mg (6 month formulation) (AUST R 109854, 109856, 159173).
Each pack contains 1 vial of Diphereline, 1 ampoule of solvent, 1 empty polypropylene syringe and 2 needles.
The vial of Diphereline contains a small pellet of white to slightly yellow powder which must be mixed with the contents of the ampoule (solvent) before injection.
Australian Sponsor of Diphereline
Ipsen Pty Ltd
Level 5, 627 Chapel Street
South Yarra VIC 3141
Diphereline® is a registered trademark of Ipsen Pharma S.A.S.
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Diphereline |
| Active ingredient | Triptorelin |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 September 2025
1 Name of Medicine
Triptorelin embonate.
2 Qualitative and Quantitative Composition
Diphereline 3.75 mg 1 month formulation. Each vial contains a triptorelin content which allows the administration of an effective dose of 3.75 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 1.875 mg of triptorelin.
Diphereline 11.25 mg 3 month formulation. Each vial contains a triptorelin content which allows the administration of an effective dose of 11.25 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 5.625 mg of triptorelin.
Diphereline 22.5 mg 6 month formulation. Each vial contains a triptorelin content which allows the administration of an effective dose of 22.5 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 11.25 mg of triptorelin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Powder and solvent for suspension for injection, prolonged release granules.
White to off-white powder.
4 Clinical Particulars
4.1 Therapeutic Indications
Diphereline is indicated for the treatment of hormone dependent locally advanced or metastatic prostate cancer.
Diphereline 22.5 mg 6 month formulation only. Diphereline 22.5 mg is indicated for the treatment of children 2 years and older with central precocious puberty (CPP).
4.2 Dose and Method of Administration
Diphereline 3.75 mg - 1 month formulation. The recommended dose of Diphereline is 3.75 mg triptorelin (1 vial) administered once a month as a single intramuscular injection.
Diphereline 11.25 mg - 3 month formulation. The recommended dose of Diphereline is 11.25 mg triptorelin (1 vial) administered every three months as a single intramuscular injection.
Diphereline 22.5 mg - 6 month formulation. The recommended dose of Diphereline is 22.5 mg triptorelin (1 vial) administered every six months as a single intramuscular injection.
The lyophilised microgranules are to be reconstituted using 2 mL sterile water for injection (see Section 4.2 Dose and Method of Administration). The injection site should be varied periodically.
Since Diphereline is a suspension of microgranules, inadvertent intravascular injection must be strictly avoided.
Special populations. Patients with renal or hepatic impairment. No dosage adjustment is necessary for patients with renal or hepatic impairment (also see Section 5.2 Pharmacokinetic Properties). Diphereline must be administered under the supervision of a physician.
Paediatric population. Precocious puberty (before 8 years in girls and 9 years in boys). Prior to initiation of treatment a clinical diagnosis of CPP should be confirmed by measurement of blood concentrations of LH (basal or stimulated with a GnRH analogue), sex steroids, and assessment of bone age versus chronological age. Baseline evaluations should include height and weight measurements, diagnostic imaging of the brain (to rule out intracranial tumour), pelvic/testicular/adrenal ultrasound (to rule out steroid secreting tumours), human chorionic gonadotropin levels (to rule out a chorionic gonadotropin secreting tumour), and adrenal steroid measurements to exclude congenital adrenal hyperplasia.
The treatment of children with Diphereline 22.5 mg should be under the overall supervision of a paediatric endocrinologist or of a paediatrician or an endocrinologist with expertise in the treatment of central precocious puberty.
Treatment should be stopped around the physiological age of puberty in boys and girls and should not be continued in girls with a bone maturation of more than 12-13 years. There are limited data available in boys relating to the optimum time to stop treatment based on bone age, however it is advised that treatment is stopped in boys with a bone maturation age of 13-14 years.
Method of administration. The solvent for suspension should be drawn into the injection syringe and transferred to the vial containing the powder. The vial should be gently shaken to thoroughly disperse particles and obtain a uniform suspension. The suspension will appear milky. The suspension obtained should be drawn back into the injection syringe. The injection needle has to be changed and the produced suspension for injection should be administered immediately.
The suspension should be discarded if not used immediately after reconstitution.
Diphereline contains no antimicrobial agent. The product is for treatment of one patient only on one occasion. Discard any remaining contents. Used injection needles should be disposed in a designated sharp container. Any remaining product should be discarded.
Diphereline must be administered under the supervision of a healthcare professional.
4.3 Contraindications
Diphereline is contraindicated in patients with known hypersensitivity to the active substance triptorelin, GnRH, other GnRH agonist analogues or to any of the excipients (see Section 6.1 List of Excipients). Note that polysorbate 80 has been observed to induce hypersensitivity reactions in some patients.
Diphereline must not be administered if there are indications that the tumour is not hormone-dependent or following surgical castration.
Diphereline is contraindicated in patients with spinal cord compression secondary to prostate cancer metastases.
Diphereline is contraindicated in pregnancy and during lactation.
4.4 Special Warnings and Precautions for Use
Prostate cancer. Initially triptorelin causes a transient increase in serum testosterone levels. As a consequence, isolated cases of transient worsening of signs and symptoms of prostate cancer may occasionally develop during the first weeks of treatment. During the initial phase of treatment, consideration should be given to the additional administration of a suitable antiandrogen to counteract the initial rise in serum testosterone levels and the worsening of clinical symptoms.
A small number of patients may experience a temporary worsening of signs and symptoms of prostate cancer (tumour flare) and a temporary increase in cancer related pain (metastatic pain), which can be managed symptomatically.
As with other GnRH agonists, isolated cases of spinal cord compression or urethral obstruction have been observed. If spinal cord compression or renal impairment develops, standard treatment of these complications should be instituted, and in extreme cases an immediate orchiectomy should be considered. Careful monitoring is indicated during the first weeks of treatment, particularly in patients suffering from vertebral metastases, at the risk of spinal cord compression, and in patients with urinary tract obstruction.
After surgical castration, triptorelin does not induce any further decrease in serum testosterone levels. Once the castration levels of testosterone have been achieved by the end of the first month, serum testosterone levels are maintained for as long as the patients receive their injection every one, three or six months. The effectiveness of treatment can be monitored by measuring serum levels of testosterone and prostate specific antigen.
QT prolongation. Androgen deprivation therapy may prolong the QT interval.
In patients with a history of or risk factors for QT prolongation and in patients receiving concomitant medicinal products that might prolong the QT interval (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions) physicians should assess the benefit risk ratio including the potential for torsades de pointes prior to initiating Diphereline.
Precocious puberty. Treatment of children with progressive brain tumours should follow a careful individual appraisal of the risks and benefits.
Pseudo-precocious puberty (gonadal or adrenal tumour or hyperplasia) and gonadotropin-independent precocious puberty (testicular toxicosis, familial Leydig cell hyperplasia) should be precluded.
In girls, initial ovarian stimulation at treatment initiation, followed by the treatment-induced oestrogen withdrawal, may lead, in the first month, to vaginal bleeding of mild or moderate intensity.
After discontinuation of treatment the development of puberty characteristics will occur.
Information with regards to future fertility is still limited but future reproductive function and fertility appears to be unaffected by GnRH treatment. In most girls, regular menses will start on average one year after ending the therapy.
Bone mineral density may decrease during GnRH agonist therapy for central precocious puberty due to the expected effects of oestrogen suppression. However, after cessation of treatment subsequent bone mass accrual is preserved and peak bone mass in late adolescence does not seem to be affected by treatment.
Slipped capital femoral epiphysis can be seen after withdrawal of GnRH agonist treatment. The suggested theory is that the low concentrations of oestrogen during treatment with GnRH agonists weaken the epiphysial plate. The increase in growth velocity after stopping the treatment subsequently results in a reduction of the shearing force needed for displacement of the epiphysis.
Cardiovascular disease. An increased risk of developing myocardial infarction, sudden cardiac death and stroke has been reported in association with use of GnRH agonists in men. The risk appears low based on the reported odds ratio, and should be evaluated carefully along with cardiovascular risk factors when determining a treatment for patients with prostate cancer. Patients receiving a GnRH agonist should be monitored for symptoms and signs suggestive of development of cardiovascular disease and managed according to current clinical practice.
Osteoporosis and bone mineral density. The use of GnRH agonists may cause reduction in bone mineral density. In men, preliminary data suggest that the use of a bisphosphonate in combination with an GnRH agonist may reduce bone mineral loss. Particular caution is necessary in patients with additional risk factors for osteoporosis (e.g. chronic alcohol abuse, smokers, long-term therapy with drugs that reduce bone mineral density, e.g. anticonvulsants or corticoids, family history of osteoporosis, malnutrition).
Long-term androgen deprivation either by bilateral orchiectomy or administration of GnRH analogues is associated with increased risk of bone loss and may lead to osteoporosis and increased risk of bone fracture. This may also lead to an incorrect diagnosis of bone metastases.
Depression. In patients undergoing treatment with GnRH agonists, an increased risk of mood changes and depression (which may be severe and includes very rare cases of suicidal ideation or suicide attempts from post-marketing experience) was reported. Patients should be informed accordingly and treated as appropriate if symptoms occur. Patients with known depression should be monitored closely during therapy.
Hyperglycaemia and diabetes. Hyperglycaemia and an increased risk of developing diabetes have been reported in men receiving GnRH agonists. Patients may experience metabolic changes (e.g. glucose intolerance, fatty liver). Hyperglycaemia may represent development of diabetes mellitus or worsening of glycaemic control in patients with diabetes. Monitor blood glucose and/or glycosylated haemoglobin (HbA1c) periodically in patients receiving a GnRH agonist and manage with current practice for the treatment of hyperglycaemia or diabetes.
Other identified precautions. Treatment with GnRH analogues may reveal the presence of a previously unknown gonadotroph cell pituitary adenoma. These patients may present a pituitary apoplexy which is characterised by sudden headache, vomiting, visual impairment and ophthalmoplegia.
Adjustment of antihypertensive therapy may be required in patients receiving such medication.
Caution is required with intramuscular injection in patients treated with anticoagulants, due to the potential risk of haematomas at the site of injection.
Administration of triptorelin in therapeutic doses results in suppression of the pituitary gonadal system. Normal function is usually restored after treatment is discontinued. Diagnostic tests of pituitary gonadal function conducted during treatment and after discontinuation of therapy with a GnRH agonist may therefore be misleading.
Pseudotumour cerebri (PTC)/ idiopathic intracranial hypertension has been reported in women and children receiving GnRH analogues including triptorelin. Monitor patients for signs and symptoms of PTC/ idiopathic intracranial hypertension, including severe or recurrent headache, papilledema, blurred vision, diplopia, loss of vision, pain behind the eye or pain with eye movement, tinnitus, dizziness, and nausea. Refer the patient to an ophthalmologist to confirm the presence of papilledema. If PTC/ idiopathic intracranial hypertension is confirmed, treat the patient in accordance with established treatment guidelines and permanently discontinue use of triptorelin.
Convulsions have been reported with GnRH analogues, particularly in women and children. Some of these patients had risk factors for seizures (such as a history of epilepsy, intracranial tumours or co-medication with drugs known to present a risk of seizure reactions). Convulsions have also been reported in patients in the absence of such risk factors.
Due to androgen deprivation, treatment with analogues of GnRH can increase the risk of anaemia in men. This risk should be assessed in treated patients and monitored appropriately.
All formulations of Diphereline contain less than 1 mmol (23 mg) sodium per dose.
Use in the elderly. No data available.
Paediatric use. The Diphereline 22.5 mg 6 month formulation is indicated for use in children 2 years and older with central precocious puberty (CPP).
The safety and efficacy of triptorelin embonate 3.75 mg (1 month) and 11.25 mg (3 month) formulations have not been reviewed in neonates, infants, children and adolescents, and are therefore not indicated for use in these populations.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Since androgen deprivation treatment may prolong the QT interval, the concomitant use of Diphereline with medicinal products known to prolong the QT interval or medicinal products able to induce torsades de pointes such as class IA (e.g. quinidine, disopyramide) or class III (e.g. amiodarone, sotalol, dofetilide, ibutilide) antiarrhythmic medicinal products, methadone, moxifloxacin, antipsychotics, etc. should be carefully evaluated (see Section 4.4 Special Warnings and Precautions for Use).
When triptorelin is coadministered with drugs affecting pituitary secretion of gonadotrophins, caution should be given and it is recommended that the patient's hormonal status should be supervised.
In the absence of relevant data, there is a theoretical risk that treatment with drugs that increase serum prolactin may decrease the efficacy of GnRH agonists as hyperprolactinaemia reduces the number of pituitary GnRH receptors.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. In chronic toxicity studies at clinically relevant doses, triptorelin induced changes in the reproductive organs of male rats, dogs and monkeys. These were considered to reflect the suppressed gonadal function caused by the pharmacological activity of the compound. These changes would be expected to cause a profound impairment of fertility, but were partly reversed (males) or largely reversed (females) after cessation of treatment. In males, changes included decreases in weight and atrophic histological changes in the testes, epididymis, seminal vesicle and prostate gland, with suppression of spermatogenesis. In females, changes included ovarian atrophy and suppression of ovarian function, with arrest of follicular development and cessation of oestrus cycling; uterine weights were also reduced.
Use in pregnancy. (Category D)
Safe use of Diphereline 22.5 mg in pregnancy has not been established in clinical studies. It should be confirmed that the patient is not pregnant before prescription of Diphereline 22.5 mg.
Diphereline should not be used during pregnancy since concurrent use of GnRH agonists is associated with a theoretical risk of abortion or foetal abnormality.
After subcutaneous administration of 10 microgram/kg/day to rats on days 6 to 15 of gestation, triptorelin did not elicit any embryotoxicity or teratogenicity. At 100 microgram/kg/day, a reduction in maternal bodyweight gain and an increased number of resorptions were observed.
Use in lactation. Diphereline is not recommended for use during lactation.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. However, the ability to drive and use machines may be impaired should the patient experience dizziness, somnolence, epileptic seizures and visual disturbances being possible undesirable effects of treatment or resulting from the underlying disease.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
General tolerance in men. Since patients suffering from locally advanced or metastatic, hormone-dependent prostate cancer are generally old and have other diseases frequently encountered in this aged population, most of the patients included in clinical trials reported adverse events. As seen with other GnRH agonist therapies or after surgical castration, the most commonly observed adverse events related to triptorelin treatment were due to its expected pharmacological effects: initial increase in testosterone levels, followed by almost complete suppression of testosterone. These effects included hot flushes (50%), erectile dysfunction (4%) and decreased libido (3%).
With the exception of immunoallergic reactions (0.2%) and injection site reactions (3%), all adverse reactions are known to be related to testosterone changes.
Tabulated list of adverse reactions in men. The following adverse reactions, considered as at least possibly related to triptorelin treatment, were reported. Most of these are known to be related to biochemical or surgical castration.
In Table 1, the frequency of the adverse reactions is classified as follows: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/1000, < 1/100); rare (≥ 1/10,000, < 1/1000). No very rare (< 1/10,000) adverse reactions were reported.
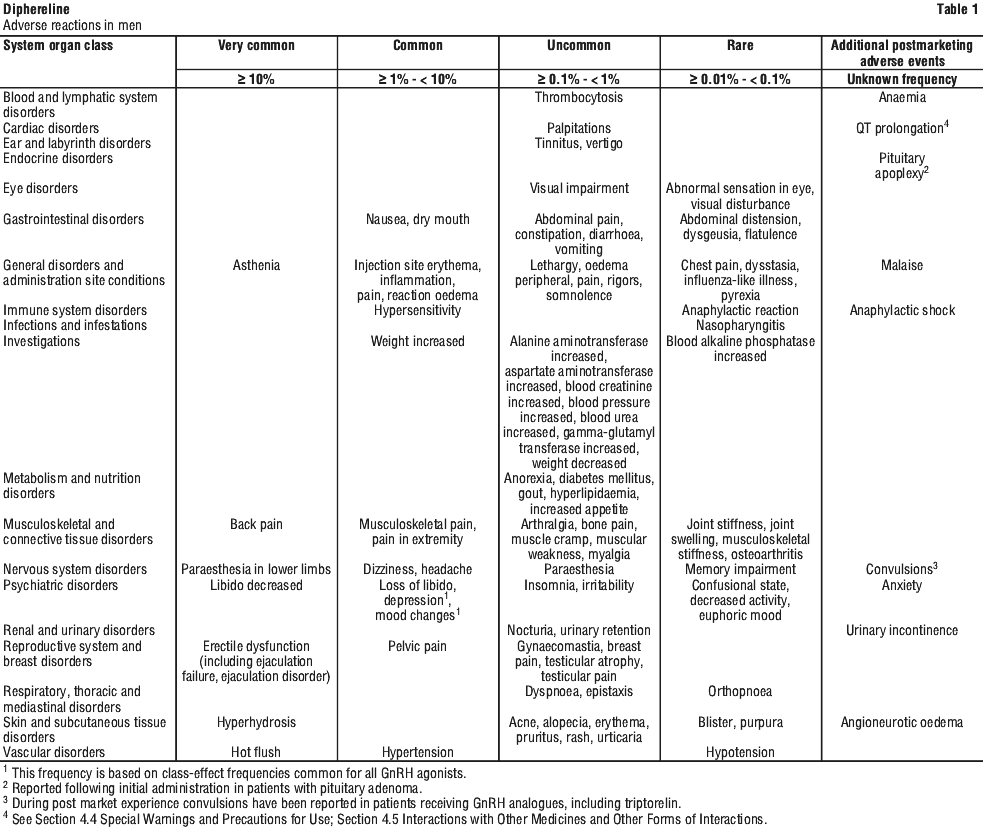
The use of synthetic GnRH agonists, to treat prostate cancer may be associated with increased bone loss and may lead to osteoporosis and increases the risk of bone fracture. This may also lead to an incorrect diagnosis of bone metastases.
General tolerance in children. Tabulated list of adverse reactions in children. The frequency of the adverse reactions is classified as follows: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000, < 1/100). See Table 2.
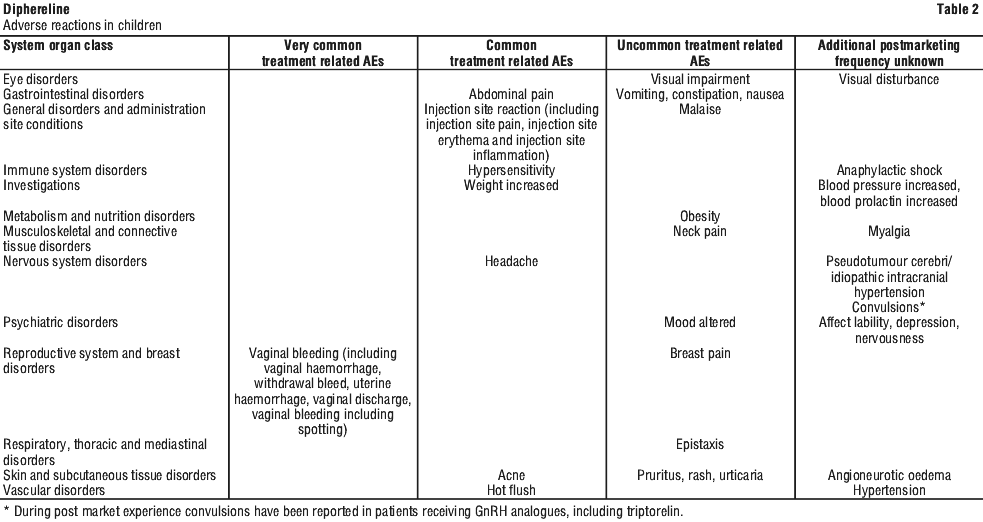
Uncommonly pressure sensitive infiltrations at the injection site have been reported in other triptorelin products after subcutaneous injection.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
The pharmaceutical properties of Diphereline and its route of administration make accidental or intentional overdose unlikely. There is no experience of overdose from clinical trials. Animal tests suggest that no effect other than the intended therapeutic effects on sex hormone concentration and on the reproductive tract will be evident with higher doses of Diphereline.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Hormones and related agents, gonadotropin releasing hormone analogues.
ATC code: L02AE04.
Mechanism of action. Triptorelin, a gonadotrophin releasing hormone (GnRH) agonist, inhibits gonadotrophin secretion when given continuously and in therapeutic doses. Male animal and human studies show that after the administration of triptorelin there is an initial and transient increase in circulating levels of luteinizing hormone (LH), follicle stimulating hormone (FSH), and testosterone. However, chronic and continuous administration of triptorelin results in decreased LH and FSH secretion and suppression of testicular and ovarian steroidogenesis. A reduction of serum testosterone levels into the range normally seen in surgically castrated men occurs approximately 2 to 4 weeks after initiation of therapy. This results in accessory sexual organ atrophy. These effects are generally reversible upon discontinuation of the medicinal product.
In animals, administration of triptorelin resulted in the inhibition of growth of some hormone sensitive prostate tumours in experimental models.
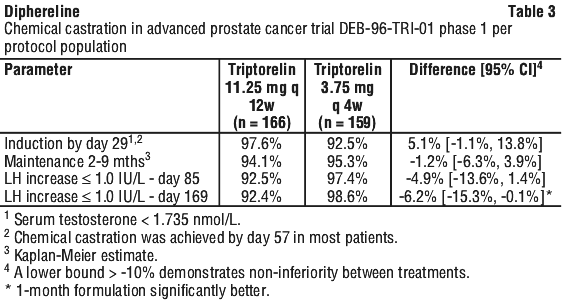
Clinical trials. Prostate cancer. One pivotal, long-term (9 months), controlled, phase III, multicentre study (DEB-96-TRI-01, first phase) compared the 3.75 mg (1 month) and 11.25 mg (3 month) embonate formulations in 348 patients with advanced prostatic cancer. Patients in this study had histologically confirmed stage C (52.9% of patients) or D (46.8% of patients) prostate cancer with testosterone levels greater than 5 nanomol/L at baseline. The mean age of the 348 patients in the safety population was 70.5 years (range 45 to 96 years); mean age at onset of prostate cancer was 69.8 years (range 44 to 96 years) and the mean disease duration was 6.9 months (range 0 to 155 months).
Per protocol and intent to treat analyses produced similar results. The 3 month formulation was noninferior (no worse than 10 percentage point difference in incidence) to the 1 month formulation in inducing and maintaining chemical castration (see Table 3).
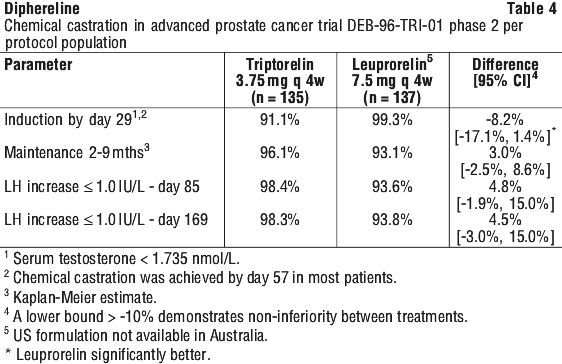
Per protocol and intent to treat analyses produced similar results. Whilst the 1 month formulation was slower in inducing chemical castration than the US 1 month leuprorelin acetate formulation, it was noninferior in maintaining chemical castration (no worse than 10 percentage point difference in incidence). See Table 4.
In trial DEB-TRI6M-301, 120 patients with advanced prostate cancer received Diphereline 22.5 mg 6-month formulation IM on days 1 and 169 and were followed until day 337 (48 weeks). The median age of patients was 70 years (range 50-92). The primary efficacy endpoints were the percentage of patients achieving the castrate level of testosterone (≤ 1.735 nanomol/L) by day 29 and percentage of patients maintaining this level from day 57 to Week 48. 115 patients (96%) completed the study. Three patients died, one was lost to follow-up and one withdrew consent. Of the patients completing the trial, 93% maintained castrate serum testosterone levels from day 57 to week 48 (Table 5 per protocol population). Intent to treat results were similar.
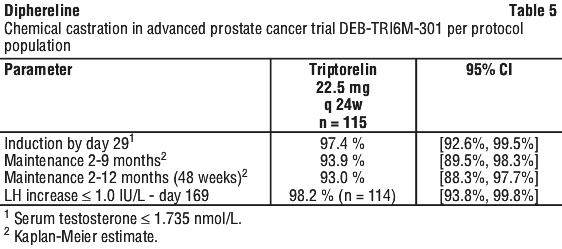
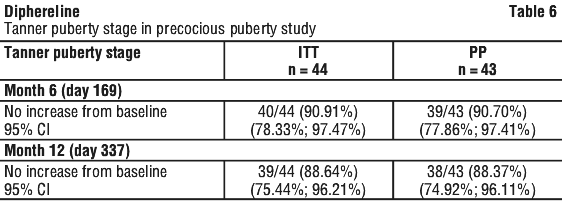
In a non-comparative clinical study, 44 children with central precocious puberty (39 girls and 5 boys) were treated with a total of two intramuscular injections of Diphereline 22.5 mg over 12 months (48 weeks). Suppression of stimulated LH concentrations to prepubertal levels (serum LH ≤ 5 IU/L) was achieved in 95.5% of subjects by month 3, and in 93.2% and 97.7% of subjects at months 6 and 12, respectively.
The consequence is a regression or stabilisation of secondary sex characteristics and slowing down of accelerated bone maturation and growth.
In girls, initial ovarian stimulation at treatment initiation, followed by the treatment-induced oestrogen increase, may lead, in the first month, to uterine 'withdrawal' bleeding of mild or moderate intensity.
The mean chronological age was 94.6 months (range: 31.0 to 118.0 months). The Tanner stage was stable or reduced in 90.9% of patients between baseline and Month 6 and in 88.6% patients between baseline and Month 12. See Table 6.
5.2 Pharmacokinetic Properties
Absorption. In a substudy of the pivotal efficacy trial DEB-96-TRI-01 of the 1 and 3 month doses in patients with prostate cancer, triptorelin exposure based on plasma AUC was comparable after intramuscular doses of the 1 month (x 3 doses at 28-day intervals) and 3-month formulations of triptorelin embonate (Table 7). Triptorelin did not accumulate over 9 months of treatment.
In a substudy in 15 patients with prostate cancer from the efficacy trial of the 6 month formulation (DEB-TRI6M-301), the plasma Cmax after the first injection was comparable to that obtained previously with the 3-month formulation and higher than the 1-month formulation; however, the AUC over 6 months (resulting from an overall dose of 22.5 mg for all 3 formulations) was about half that after the 3-month and 1-month formulations.
The three sustained release formulations of Diphereline cannot be considered strictly bioequivalent and this is due to differences in polymeric matrix which confers different release rate for each of the three strengths. Nonetheless, the three formulations were demonstrated to be pharmacodynamically equivalent.
The relationship between serum triptorelin and serum testosterone is not linear but on/off, so the level of serum triptorelin rather than AUC is important in maintaining castrate serum testosterone levels. After an initial surge, mean ± SD serum testosterone remained below the castrate level (≤ 1.735 nanomol/L) for the 336 days of the trial, except at day 336 when the upper limit of the standard deviation was above the castrate level.

In children with precocious puberty median tmax was 4 (2-8) hours and Cmax following the first injection was 39.9 (19.1-107.0) nanogram/mL.
Distribution. Results of pharmacokinetic investigations conducted in healthy men indicate that after intravenous bolus administration, triptorelin is distributed and eliminated according to a 3 compartment model and corresponding half-lives are approximately 6 minutes, 45 minutes, and 3 hours.
The volume of distribution at steady state of triptorelin following intravenous administration of 0.5 mg triptorelin is approximately 30 L in healthy male volunteers. Since there is no evidence that triptorelin at clinically relevant concentrations binds to plasma proteins, medicinal product interactions involving binding site displacement are unlikely.
Metabolism. Metabolites of triptorelin have not been determined in humans. However, human pharmacokinetic data suggest that C-terminal fragments produced by tissue degradation are either completely degraded within tissues or are rapidly further degraded in plasma, or cleared by the kidneys.
Excretion. Triptorelin is eliminated by both the liver and the kidneys. Following intravenous administration of intermediate release triptorelin acetate 0.5 mg to 6 young healthy adult males (mean Clcreat 150 mL/min), 42% of the dose was excreted in the urine as intact triptorelin. The mean triptorelin clearance was 212 mL/min.
Pharmacokinetics in special patient populations. Renal and hepatic impairment. In the intravenous study referred to under Excretion, patients with renal and liver impairment were also studied. There were 6 subjects in each group. Compared to young healthy adult males, mean triptorelin clearance was reduced by 43% in subjects with moderate renal impairment (mean Clcreat 40 mL/min), 58% in subjects with severe renal impairment (mean Clcreat 8.9 mL/min) and 73% in subjects with hepatic impairment (Child Pugh score 5-9) and a lower mean Clcreat (90 mL/min) than young healthy adult males. Triptorelin exposure was increased 2- to 4-fold in patients with renal or hepatic impairment.
The effects of age and race on triptorelin pharmacokinetics have not been systematically studied. However, pharmacokinetic data obtained in young healthy male volunteers aged 20 to 22 years with an elevated creatinine clearance (approximately 150 mL/min) indicated that triptorelin was eliminated twice as fast in the young population. This is related to the fact that triptorelin clearance is correlated to total creatinine clearance, which is well known to decrease with age.
Because of the large safety margin of triptorelin and since Diphereline is a sustained release formulation, no dose adjustment is recommended in patients with renal or hepatic impairment.
Pharmacokinetic/pharmacodynamic relationship(s). The pharmacokinetics/pharmacodynamics relationship of triptorelin is nonlinear and time dependent. Thus, after acute administration in naive subjects, triptorelin induces a dose dependent increase of LH and FSH responses.
When administered as a sustained release formulation, triptorelin stimulates LH and FSH secretion during the first days postdosing and, in consequence, testosterone secretion. As shown by the results of the different bioequivalence studies, the maximal increase in testosterone is reached after around 4 days with an equivalent Cmax which is independent from the release rate of triptorelin. This initial response is not maintained despite continuous exposure to triptorelin and is followed by a progressive decrease of testosterone levels. In this case too, the extent of triptorelin exposure can vary markedly without affecting the overall effect on testosterone serum levels.
5.3 Preclinical Safety Data
Genotoxicity. In vitro genotoxicity tests for gene mutations and chromosomal damage, and a mouse micronucleus test have provided no evidence for genotoxic effects.
Carcinogenicity. Carcinogenicity studies were conducted in mice (18 months) and rats (23 months) with triptorelin embonate microgranules administered once monthly IM. In mice, no oncogenic effect was observed at triptorelin doses of up to 6000 microgram/kg/month. In rats, an almost 100% incidence of pituitary tumours was observed at each dose level (120, 600 and 3000 microgram/kg/month), leading to premature death. There were increased incidences of both pituitary adenomas and carcinomas at all dose levels. The increased incidence of pituitary tumours in rats is a common effect associated with GnRH agonist treatment. The clinical relevance of this is not known.
6 Pharmaceutical Particulars
6.1 List of Excipients
Composition of the powder. Polyglactin, mannitol, carmellose sodium, polysorbate 80.
Composition of the solvent. Water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
After opening. Chemical and physical in-use stability has been demonstrated for 24 hours at 25°C.
From a microbiological point of view, the prepared suspension should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2 to 8°C.
Contains no antimicrobial preservative. Use each ampoule in one patient on one occasion only. Discard any residue.
6.4 Special Precautions for Storage
Store below 25°C.
For storage conditions after first opening of the medicinal product, see Section 6.3 Shelf Life.
6.5 Nature and Contents of Container
Diphereline 3.75 mg 1 month formulation. 6 mL type I brown tint glass vial with grey bromobutyl stopper and purple aluminium flip-off capsule.
Diphereline 11.25 mg 3 month formulation. 6 mL type I brown tint glass vial with grey bromobutyl stopper and yellow green aluminium flip-off capsule.
Diphereline 22.5 mg 6 month formulation. 6 mL type I brown tint glass vial with grey bromobutyl stopper and dark green aluminium flip-off capsule.
Solvent: Type I glass ampoule containing 2 mL of sterile solvent for suspension.
Each box contains 1 vial, 1 ampoule and 1 blister containing 1 empty polypropylene injection syringe and 2 injection needles.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. [(L-Pyr)-(L-His)-(L-Trp)-(L-Ser)-(L-Tyr)-(D-Trp)-(L-Leu)-(L-Arg)-(L-Pro)-(Gly-NH2)].
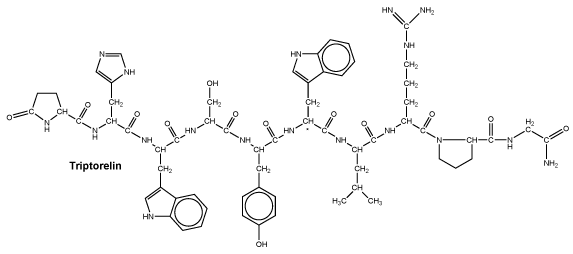
Molecular weight: 1311.5 (triptorelin) + 388.4 (embonate).
CAS number. 57773-63-4.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
28 August 2006
Date of Revision
06 August 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.