Dysport
Brand Information
| Brand name | Dysport |
| Active ingredient | Botulinum toxin type A |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Dysport.
Summary CMI
Dysport®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given Dysport?
Dysport contains the active ingredient clostridium botulinum type A toxin-haemagglutinin complex. In adults, Dysport is used to treat: muscle spasms around the eyes, in the face, in the neck; muscle stiffness and tightness (spasticity) in the arms, shoulders, and legs; and, to treat lines between the eyebrows, and lines near the outside corner of the eyes. In children, Dysport is also used to treat muscle stiffness and tightness (spasticity) in the arms and legs.
For more information, see Section 1. Why am I being given Dysport? in the full CMI.
2. What should I know before I am given Dysport?
Do not use if you have ever had an allergic reaction to Dysport or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given Dysport? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Dysport and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How am I given Dysport?
- Dysport should only be given to you by a doctor who has received training and is experienced in giving Dysport injections.
- Your doctor will give you injections of Dysport into your muscle (intramuscular) or under the skin (subcutaneous).
More instructions can be found in Section 4. How am I given Dysport? in the full CMI.
5. What should I know while being given Dysport?
| Things you should do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while being given Dysport? in the full CMI.
6. Are there any side effects?
Like all medicines, Dysport can cause side effects, although not everybody gets them. Some side effects may occur in any patient treated with Dysport, whilst other side effects may depend on the condition being treated. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Tell your doctor straight away if you notice any of the following side effects, you may need urgent medical attention:
- problems swallowing
- problems breathing or with your speech
- worsened muscle weakness
- shortness of breath
- very dry eyes
Full CMI
Dysport®
Active ingredient(s): Clostridium botulinum type A toxin-haemagglutinin complex
Consumer Medicine Information (CMI)
This leaflet provides important information about using Dysport. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Dysport.
Where to find information in this leaflet:
1. Why am I being given Dysport?
2. What should I know before I am given Dysport?
3. What if I am taking other medicines?
4. How am I given Dysport?
5. What should I know while being given Dysport?
6. Are there any side effects?
7. Product details
1. Why am I being given Dysport?
Dysport contains the active ingredient clostridium botulinum type A toxin-haemagglutinin complex. It works by stopping your muscles contracting. It does this by stopping the release of a chemical messenger which acts between the nerves and muscles to make the muscles contract. This helps to reduce abnormal muscle contractions known as spasms.
Dysport is used to treat:
- muscle stiffness or tightness in the arm, shoulders and/or hand muscles (upper limb spasticity) in adults and children 2 years and older.
- muscle stiffness or tightness in the leg and/or foot muscles (lower limb spasticity) in adults and children 2 years and older.
- a condition which causes an involuntary turning movement of the neck leading to an unusual and painful position of the head and shoulder (spasmodic torticollis, also known as cervical dystonia) in adults.
- a condition that causes uncontrollable blinking and closing of the eyelids (blepharospasm) in adults.
- a condition that causes muscles on one side of the face to uncontrollably contract or spasm (hemifacial spasm) in adults.
- vertical frown lines between the eyebrows (glabellar lines) in adults.
- lines that appear near the outside corner of your eyes ('crow's feet' or lateral canthal lines) in adults.
2. What should I know before I am given Dysport?
Warnings
You must not be given Dysport if:
- you have a muscle disorders medical condition called myasthenia gravis or a myasthenic syndrome.
- you have any signs of infection or inflammation in the muscles where the doctor will inject Dysport.
- you are allergic to clostridium botulinum type A toxin-haemagglutinin complex, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- skin redness, rash, itching or hives on the skin
Check with your doctor if you have:
- any difficulties in swallowing.
- a history of bronchitis, pneumonia or problems with your breathing.
- had any reactions such as skin rash or 'flu-like' symptoms to any injections of toxin in the past.
- worsening or changes in your muscle spasms.
- any excessive weakness, shrinkage or wastage of muscles, or previous prolonged periods of muscle weakness.
- bleeding problems, you bleed easily or have a history of prolonged bleeding times.
- ever had facial surgery.
- been given Dysport or another brand of botulinum toxin in the past few weeks.
- received any facial cosmetic procedures recently, particularly if your doctor will inject Dysport into your facial muscles.
- any other medical conditions.
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed. It is not recommended to be given Dysport while you are breastfeeding.
Elderly
- If you are elderly, your doctor may give you a smaller amount of Dysport.
Children
- Dysport can be used for the treatment of upper and lower limb spasticity in children (2 years of age and older).
- It is not recommended to use Dysport in children under 2 years of age.
- Dysport should not be used for treatment of any other conditions in children.
Other
- Dysport contains a small amount of albumin that has been obtained from human blood. The risk of a viral infection cannot be eliminated completely when using human blood or products made from human blood.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Dysport and affect how it works:
- muscle relaxing drugs (muscle relaxants)
- antibiotics (aminoglycoside antibiotics, spectinomycin, polymyxins, tetracyclines and lincomycin)
- other medicines, in particular penicillamine and procainamide
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Dysport.
4. How am I given Dysport?
How much is given
- Your doctor will choose the correct dosage of medicine to use. This will depend on what you are being treated for.
- Your doctor may divide the dose amount between your affected muscles.
- The dose may depend on the size, number, and location of muscles affected, how severe your condition is, and taking into account any local muscle weakness or your previous response to treatment.
Upper limb spasticity:
- In adults, your dose of Dysport can be up to 1000 units in total.
- In children, your dose of Dysport can be up to 640 units or 16 units/kg for one arm or up to 840 units or 21 units/kg for both arms, whichever amount is lower.
Lower limb spasticity:
- In adults, your dose of Dysport can be up to 1500 units in total.
- In children, your dose of Dysport can be up to 1000 units or 30 units/kg, whichever amount is lower.
Upper and lower limb spasticity:
- If both your arms and legs are being treated in the same session, your dose can be up to 1500 units total in adults. For children, it can be up to 1000 units or 30 units/kg, whichever is lower, in total.
Spasmodic torticollis (cervical dystonia):
- Your first dose of Dysport will usually be 250 to 500 units in total.
- Your dose of Dysport can be up to 1000 units.
Blepharospasm or hemifacial spasm:
- Your first dose will be approximately 40 units of Dysport per eye or on the affected side of your face.
- On the next visits, your dose of Dysport can be up to 120 units per eye.
Frown lines:
- Your dose of Dysport can be up to 50 units divided equally among 5 injection sites.
Crow's feet:
- Your dose of Dysport can be up to 30 units per side divided equally among 3 injection sites per eye (equal to 60 units total at 6 injection sites).
When Dysport is given
- Your doctor will decide how often and for how long you need treatment. This will depend on what you are being treated for.
- Upper or lower limb spasticity, spasmodic torticollis/cervical dystonia, blepharospasm/hemifacial spasm: In adults, your injections will usually be around every 12 to 16 weeks, but not less than 12 weeks.
- Upper or lower limb spasticity: In children, your injections will usually be around every 16 to 28 weeks, but not less than 12 weeks (lower limbs) or 16 weeks (upper limbs).
- Frown lines and crow's feet: Your injections will be not less than 12 weeks apart.
How Dysport is given
- Your doctor will prepare the vial and give the injection to you.
- Your doctor will inject the medicine into your muscle (intramuscular) or under your skin (subcutaneous), depending on what you are being treated for.
- Your doctor will decide where to make the injections and how many injections you will receive.
- You doctor will have received training and be experienced in giving Dysport injections.
If you forget to get your Dysport injection
Tell your doctor if you have missed an injection. They will decide when you need your next injection.
Nothing will happen if you miss an injection other than some of the spasm or muscle stiffness may return.
If you stop getting injections, the muscle relaxing effect will eventually wear off. The muscle movements will return to the way they were before your treatment.
If you are given too much Dysport
Dysport is given to you by a doctor or nurse, so it is unlikely that you or your child will be given too much.
If you are given too much Dysport, muscles other than the ones that were injected may begin to feel weak. There is a risk of the medicine getting into the bloodstream and causing complications (botulinum poisoning). You may experience paralysis of your muscles. If it affects your breathing, you may need to be placed on a respirator and other support systems.
If you think that you have been given too much Dysport, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while being given Dysport?
Things you should do
Call your doctor straight away if you:
- have any problems swallowing, breathing, or with your speech.
- have worsened muscle weakness.
- notice your eyes are very dry.
Remind any doctor, dentist, pharmacist, or clinic staff you visit that you are using Dysport.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Dysport affects you.
Dysport may cause dizziness in some people. You may experience weakness in the muscles that are being treated, or general weakness.
Looking after your medicine
Your doctor will keep the medicine at the doctor's clinic or surgery, or at the pharmacy.
If you need to store Dysport at home:
- Keep it in a refrigerator between 2°C to 8°C.
- Do not freeze it.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
When to discard your medicine
A vial of Dysport will be used only for you, and only for a single treatment session. Your doctor or clinic staff will discard any unused medicine.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
| General for any condition: Signs of effects of Dysport away from the site of injection:
Signs of allergic reaction (hypersensitivity):
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Less serious side effects
| Less serious side effects | What to do |
General for any condition:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Dysport contains
| Active ingredient (main ingredient) | clostridium botulinum type A toxin-haemagglutinin complex |
| Other ingredients (inactive ingredients) | albumin lactose |
Do not take this medicine if you are allergic to any of these ingredients.
What Dysport looks like
Dysport is available in 125 units, 300 units, and 500 units (AUST R 235282, 170651, 74124).
These IPSEN units apply to Dysport only and are not the same for other medicines containing botulinum toxin.
Each pack contains 1 vial of Dysport. Each vial contains a small pellet of white powder that must be mixed with sterile sodium chloride solution before injection.
Australian Sponsor of Dysport
Ipsen Pty Ltd
Level 5, 627 Chapel Street
South Yarra VIC 3141
DYSPORT® is a registered trademark of Ipsen Biopharm Limited.
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Dysport |
| Active ingredient | Botulinum toxin type A |
| Schedule | S4 |
MIMS Revision Date: 01 February 2023
1 Name of Medicine
Clostridium botulinum type A toxin - haemagglutinin complex.
2 Qualitative and Quantitative Composition
Dysport Powder for Injection contains 125, 300 or 500 Ipsen units per vial of Clostridium botulinum type A toxin-haemagglutinin complex, 125 microgram human serum albumin and 2.5 mg lactose in a sterile, lyophilised form without a preservative.
One Ipsen unit is not equivalent to one unit of any other botulinum toxin preparation.
From now on in this product information the term Ipsen unit will simply be replaced by the term unit.
Clostridium botulinum type A toxin-haemagglutinin complex has a molecular weight of about 900,000 D and is a complex of proteins.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Dysport 125 U, 300 U or 500 U is a white lyophilised powder for injection in a clear glass vial.
4 Clinical Particulars
4.1 Therapeutic Indications
Dysport is indicated for symptomatic treatment of focal spasticity of:
upper limbs in adults;
lower limbs in adults;
upper limbs in children aged 2 years and older;
lower limbs in children aged 2 years and older.
Dysport is indicated in adults for the treatment of:
spasmodic torticollis;
blepharospasm;
hemifacial spasm;
moderate to severe glabellar lines and/or lateral canthal lines (crow's feet).
4.2 Dose and Method of Administration
The units of Dysport are specific to the preparation and are not interchangeable with other preparations of botulinum type A toxin.
Training. Dysport should only be administered by appropriately trained physicians. The product distributor can facilitate training in administration of Dysport injections.
Focal spasticity affecting the upper limbs in adults. Dosing in initial and sequential treatment sessions should be tailored to the individual based on the size, number and location of muscles involved, severity of spasticity, the presence of local muscle weakness, the patient's response to previous treatment, and/or adverse event history with Dysport. In clinical trials, doses of 500 U, 1000 U and 1500 U were divided among selected muscles (Table 1) at a given treatment session. The maximum recommended dose is 1000 U in a single treatment session. Doses greater than 1000 U and up to 1500 U, when the shoulder muscles were also injected, have been used but have not been adequately studied.
No more than 1 mL should generally be administered intramuscularly at any single injection site. Doses exceeding 1500 U of Dysport were not investigated for the treatment of upper limb spasticity in adults.
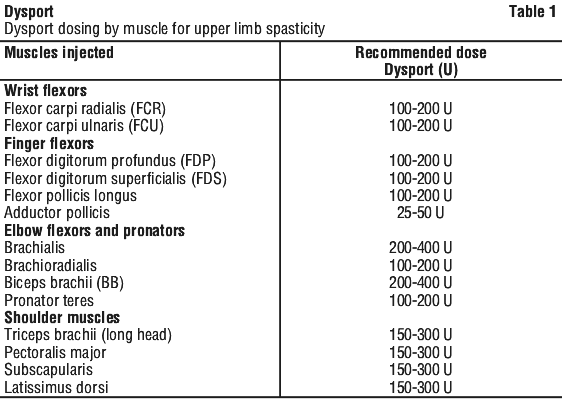
Repeat Dysport treatment should be administered when the effect of a previous injection has diminished, but no sooner than 12 weeks after the previous injection. A majority of patients in clinical studies were retreated between 12-16 weeks; however, some patients had a longer duration of response, i.e. 20 weeks. The degree and pattern of muscle spasticity at the time of re-injection may necessitate alterations in the dose of Dysport and muscles to be injected. Clinical improvement may be expected one week after administration of Dysport.
Focal spasticity affecting the lower limbs in adults. Doses of up to 1500 U may be administered intramuscularly in a single treatment session. The exact dosage in initial and sequential treatment sessions should be tailored to the individual based on the size and number of muscles involved, the severity of the spasticity, also taking into account the presence of local muscle weakness and the patient's response to previous treatment. However, the total dose should not exceed 1500 U. No more than 1 mL should generally be administered at any single injection site. See Table 2.
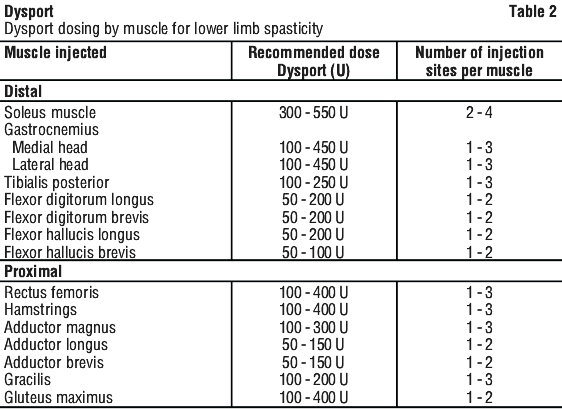
Although actual location of the injection sites can be determined by palpation, the use of injection guiding techniques, e.g. electromyography, electrical stimulation or ultrasound are recommended to help accurately target the injection sites.
Repeat Dysport treatment should be administered every 12 to 16 weeks, or longer as necessary, based on return of clinical symptoms and no sooner than 12 weeks after the previous injection.
Focal spasticity affecting the upper and lower limbs in adults. If treatment is required in the upper and lower limbs during the same treatment session, the dose of Dysport to be injected in each limb should be tailored to the individual needs, without exceeding a total body dose of 1500 U.
Spasmodic torticollis. The doses recommended for the treatment of torticollis are applicable to adults of all ages providing they are of normal weight and have no evidence of reduced neck muscle mass. A lower dose may be appropriate if the patient is markedly underweight, or in the elderly and in women, where a reduced muscle mass may exist.
The recommended initial dose for the treatment of spasmodic torticollis is 250-500 units per patient given as a divided dose and administered into the two or three most active neck muscles.
For rotational torticollis distribute the optimal dose by administering 70% of the dose into the splenius capitis muscle, ipsilateral to the direction of the chin/head rotation and 30% of the dose into the sternomastoid muscle, contralateral to the rotation.
For laterocollis, distribute the optimal dose by administering 70% of the dose into the ipsilateral splenius capitis muscle and 30% of the dose into the ipsilateral sternomastoid muscle. In cases associated with shoulder elevation the ipsilateral trapezoid or levator scapulae muscles may also require treatment, according to visible hypertrophy of the muscle or electromyographic (EMG) findings. Where injections of three muscles are required, distribute 60% of the optimal dose into the splenius capitis, 20% into the sternomastoid and 20% into the third muscle.
For retrocollis distribute the optimal dose by administering 50% of the dose into each of the splenius capitis muscles. Bilateral splenii injections may increase the risk of neck muscle weakness.
All other forms of torticollis are highly dependent on specialist knowledge and EMG to identify and treat the most active muscles. EMG should be used diagnostically for all complex forms of torticollis, for reassessment after unsuccessful injections in non complex cases, and for guiding injections into deep muscles or in overweight patients with poorly palpable neck muscles.
On subsequent administration, the doses may be adjusted according to the clinical response and side effects observed. Doses within the range of 250-1000 units are recommended, although the higher doses may be accompanied by an increase in side effects, particularly dysphagia. The maximum dose administered must not exceed 1000 units.
The relief of symptoms of torticollis may be expected within a week after the injection. Injections should be repeated approximately every 16 weeks or as required to maintain a response, but not more frequently than every 12 weeks.
Focal spasticity of lower limbs in children aged 2 years and older. Dosing in initial and subsequent treatment sessions should be tailored to the individual based on the size, number and location of muscles involved, severity of spasticity, the presence of local muscle weakness, the patient's response to previous treatment, and/or adverse event history with botulinum toxins.
The maximum total dose of Dysport administered per treatment session must not exceed 15 units/kg for unilateral lower limb injections or 30 units/kg for bilateral injections. In addition the total Dysport dose per treatment session must not exceed 1000 units or 30 U/kg, whichever is lower. The total dose administered should be divided between the affected spastic muscles of the lower limb(s). When possible, the dose should be distributed across more than 1 injection site in any single muscle. No more than 0.5 mL of Dysport should be administered in any single injection site. See Table 3 for recommended dosing.
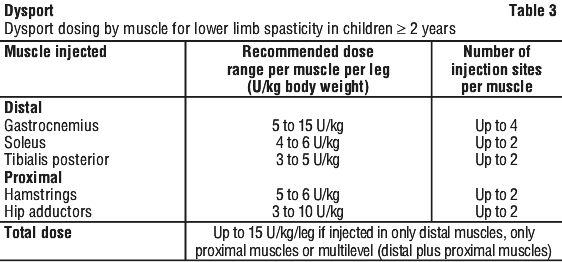
Repeat Dysport treatment should be administered when the effect of a previous injection has diminished, but no sooner than 12 weeks after the previous injection. A majority of patients in clinical studies were retreated between 16-22 weeks; however, some patients had a longer duration of response, i.e. 28 weeks. The degree and pattern of muscle spasticity at the time of re-injection may necessitate alterations in the dose of Dysport and muscles to be injected.
Focal spasticity of upper limbs in children aged 2 years and older. Dosing in initial and sequential treatment sessions should be tailored to the individual based on the size, number and location of muscles involved, severity of spasticity, the presence of local muscle weakness, the patient's response to previous treatment, and/or adverse event history with botulinum toxins.
The maximum dose of Dysport administered per treatment session when injecting unilaterally must not exceed 16 U/kg or 640 U whichever is lower. When injecting bilaterally, the maximum Dysport dose per treatment session must not exceed 21 U/kg or 840 U, whichever is lower.
The total dose administered should be divided between the affected spastic muscles of the upper limb(s). No more than 0.5 mL of Dysport should be administered in any single injection site. See Table 4 for recommended dosing.

Repeat Dysport treatment should be administered when the effect of a previous injection has diminished, but no sooner than 16 weeks after the previous injection. A majority of patients in the clinical study were retreated between 16-28 weeks; however, some patients had a longer duration of response, i.e. 34 weeks or more. The degree and pattern of muscle spasticity at the time of re-injection may necessitate alterations in the dose of Dysport and muscles to be injected.
Focal spasticity of upper and lower limbs in children aged 2 years and older. When treating combined upper and lower limb spasticity in children aged 2 years or older refer to the above sections for the individual indication, i.e. treatment of focal spasticity of the upper limbs or lower limbs in children 2 years of age and older. The dose of Dysport to be injected for concomitant treatment should not exceed a total dose per treatment session of 30 U/kg or 1000 U, whichever is lower.
Retreatment of the upper and lower limbs combined should be considered no sooner than a 12 to 16-week window after the previous treatment session. The optimal time to retreatment should be selected based on individuals progress and response to treatment.
Blepharospasm and hemifacial spasm. In a dose ranging clinical trial of the use of Dysport for the treatment of benign essential blepharospasm a dose of 40 units per eye was significantly effective. A dose of 80 units per eye resulted in a longer duration of effect. Thus, if a dose of 40 units per eye is chosen for the initial treatment, the patient may benefit from a dose of 80 units per eye for subsequent treatments if a longer duration of action is required. However, the incidence of local adverse events, specifically ptosis, was dose related.
For an initial dose of 40 units per eye, injection of 10 units should be made medially and of 10 units should be made laterally into the junction between the preseptal and orbital parts of both the upper (3 and 4) and lower orbicularis oculi muscles (5 and 6) of each eye. Dependant on the muscles involved in the blepharospasm symptoms in the patient treated, additional injections in sites 1 and 2 may be necessary.
In order to reduce the risk of ptosis, injections near the levator palpebrae superioris should be avoided.
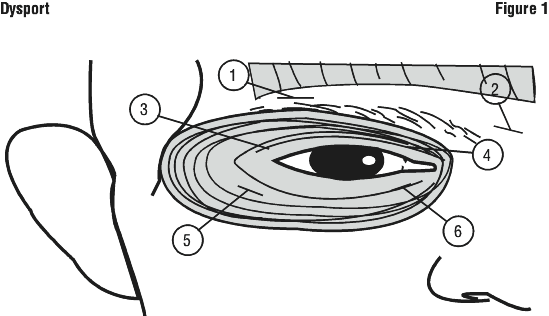
Injections should be repeated approximately every twelve weeks or as required to prevent recurrence of symptoms but not more frequently than every twelve weeks. On such subsequent administrations, if the response from the initial treatment is considered insufficient, the dose per eye may need to be increased to 60 units: 10 units medially and 20 units laterally, 80 units: 20 units medially and 20 units laterally or up to 120 units: 20 units medially and 40 units laterally above and below each eye in the manner previously described.
In cases of unilateral blepharospasm the injections should be confined to the affected eye. Patients with hemifacial spasm should be treated as for unilateral blepharospasm. The doses recommended are applicable to adults of all ages including the elderly.
In the treatment of blepharospasm and hemifacial spasm, the maximum dose must not exceed the total dose of 120 units per eye.
Glabellar lines. Remove any make-up and disinfect the skin with a local antiseptic. Anatomical landmarks can be more readily identified if observed and palpated at maximum frown. Before injection, place the thumb or index finger firmly below the orbital rim in order to prevent extravasation below the orbital rim.
Intramuscular injection should be performed at right angles to the skin using a sterile suitable gauge needle. The needle should be pointed upward and medially during the injection.
The recommended dose is 50 units of Dysport to be divided equally among 5 injection sites. 10 units are to be administered intramuscularly into each of the 5 sites shown on the diagram below as follows:
Corrugator muscles. A total of 4 injections (2 into each of the left and right corrugator muscles) at 5 mm intervals. The more medial of the two corrugator points on each side is localised on a vertical line, parallel to but 8 mm lateral to the medial vertical line and at a point on this vertical line 8 mm above the superior orbital margin. The more lateral corrugator injections should be placed at least 1 cm (i.e. 10 mm) above the bony supraorbital ridge (orbital rim) and 5 mm from the more medial injection site.
Procerus muscle. One injection into the procerus muscle at the intersection of the medial vertical line and the horizontal line at the level of the naso-frontal angles.
In order to reduce the risk of ptosis, injection near the levator palpebrae superioris muscle must be avoided, particularly in patients with larger brow-depressor complexes (depressor supercilii).
The treatment interval depends on the individual patient response following assessment. In clinical studies, some patients were still responding to treatment for up to 4 months after injection. Some patients were still responders at 5 months (see Section 5.1, Clinical trials). There should be a minimum interval of 12 weeks between treatments.
Children. Use of the product is not recommended for the temporary improvement of moderate to severe glabellar lines in patients under 18 years of age.
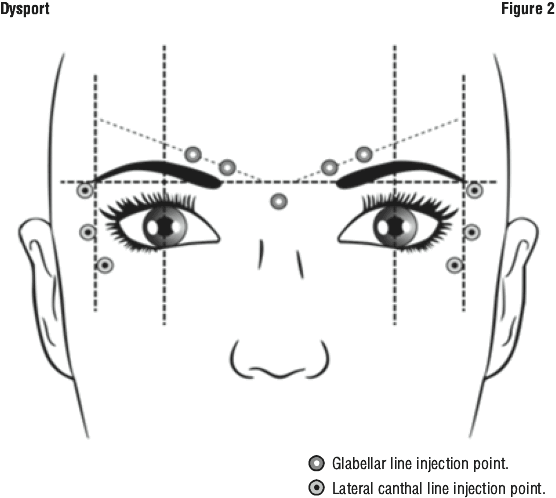
The recommended dose per side is 30 units (60 units for both sides) of Dysport, to be divided into 3 injection sites; 10 units are to be administered intramuscularly into each injection point. Injection should be lateral (20 - 30° angle) to the skin and very superficial. All injection points should be at the external part of the orbicularis oculi muscle and sufficiently far from the orbital rim (approximately 1 - 2 cm) as shown in Figure 2.
The anatomical landmarks can be more readily identified if observed and palpated at maximal smile. Care must be taken to avoid injecting the zygomaticus major/minor muscles to avoid lateral mouth drop and asymmetrical smile.
The treatment interval depends on the individual patient's response after assessment. Treatment interval should not be more frequent than every three months.
The efficacy and safety of repeat injections of Dysport has been evaluated in lateral canthal lines for up to 12 months and up to 5 repeat treatment cycles.
Children. Use of the product is not recommended for the temporary improvement of moderate to severe lateral canthal lines in patients under 18 years of age.
Method of administration. The exposed central portion of the rubber stopper should be cleaned with alcohol and allowed to dry immediately prior to piercing the septum. Sterile needles of a suitable gauge should be used for drawing up and administering the product. The product should be reconstituted as described below and injected as described above for each specific indication.
Dysport is administered by:
intramuscular injection for the treatment of focal spasticity affecting the upper and/or lower limb in adults, focal spasticity of the upper and/or lower limb in children, spasmodic torticollis, glabellar lines and lateral canthal lines;
subcutaneous injection for the treatment of blepharospasm and hemifacial spasm.
Reconstitution. Dysport is reconstituted with sodium chloride injection BP (0.9%) to yield a solution containing 100, 200 or 500 units per mL of Dysport as described in Table 5. The vial should be gently swirled to ensure complete dissolution of Dysport powder. Do not shake or invert the vial when reconstituting or drawing up Dysport solution.

Instructions for use/handling. Dysport contains no antimicrobial agent. The product should be administered within one hour of reconstitution to reduce microbiological hazard. If required, it may be held between 2°C and 8°C for 24 hours after reconstitution. The product is for treatment of one patient only on one occasion. Discard any remaining contents.
Immediately after treatment of the patient, any residual Dysport which may be present in either vial or syringe should be inactivated with dilute hypochlorite solution (1% available chlorine). Thereafter, all items should be disposed of in accordance with standard hospital practice. Spillage of Dysport should be wiped up with an absorbent cloth soaked in dilute hypochlorite solution.
4.3 Contraindications
Dysport is contraindicated in individuals with known hypersensitivity to any component of Dysport (see Section 6.1 List of Excipients).
Dysport is contraindicated in patients diagnosed with myasthenia gravis or with Eaton-Lambert (myasthenic) syndrome.
Dysport is contraindicated in the presence of any signs of infection at the proposed injection site.
4.4 Special Warnings and Precautions for Use
The recommended dosages and frequencies of administration for Dysport must not be exceeded. Extensive or inappropriate doses outside the recommended dosage range may lead to an increased risk of adverse effects.
Adverse effects resulting from the distribution of the effects of the toxin to sites remote from the site of administration have been reported. Patients treated with therapeutic doses may present with excessive muscle weakness. The risk of occurrence of such undesirable effects may be reduced by using the lowest effective dose possible and by not exceeding the maximum recommended dose.
Dysport should be administered with caution to patients with pre-existing problems in swallowing or breathing as these problems can worsen following the distribution of the effects of toxin into the relevant muscles. Aspiration has occurred in rare cases and is a risk when treating patients who have a chronic respiratory disorder.
Caution should be exercised when treating adult patients, especially the elderly, with focal spasticity affecting the lower limbs, who may be at increased risk of fall. In placebo controlled clinical studies where patients were treated for lower limb spasticity, 6.3% and 3.7% of patients experienced a fall in the Dysport and placebo groups, respectively.
Very rare cases of death, occasionally in the context of dysphagia, pneumopathy and/or in patients with significant asthenia have been reported after treatment with botulinum toxin type A or B. Patients with disorders resulting in defective neuromuscular transmission, difficulty in swallowing or breathing are more at risk of experiencing these effects. In these patients, treatment must be administered under the control of a specialist and only if the benefit of treatment outweighs the risk. Patients and their caregivers must be warned of the necessity of immediate medical treatment in case of problems with swallowing, speech or respiratory disorders.
Dysport should only be used with extreme caution and under close supervision in patients with subclinical or clinical evidence of any defect in neuromuscular transmission (e.g. drug induced neuromuscular weakness (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions) or undiagnosed myasthenic syndromes). Such patients may have an increased sensitivity to agents such as Dysport, which may result in excessive muscle weakness. Dysport is not recommended for use in any patients suffering from any of the motor neurone disorders e.g. amyotrophic lateral sclerosis.
Antibody formation to botulinum toxin has been noted rarely in patients receiving Dysport. The principal risk factors for the formation of neutralizing antibodies after Dysport treatment are high doses (> 600 units per treatment), short intervals (< 3 months) between injections and booster injections (within the first month of treatment). Clinically, neutralising antibodies might be suspected by substantial deterioration in response to therapy and/or the need for consistent use of increased doses. In three clinical studies investigating the use of Dysport to treat upper limb spasticity in adults in whom neutralizing antibodies were evaluated, the presence of such rare antibodies did not appear to have any significant impact on the efficacy of the drug and was not associated with any unexpected safety concerns.
Dry eye has been reported with the use of Dysport in the treatment of glabellar lines, lateral canthal lines, blepharospasm and hemifacial spasm (see Section 4.8 Adverse Effects (Undesirable Effects)). Reduced tear production, reduced blinking, and corneal disorders, may occur with the use of botulinum toxins, including Dysport.
For the treatment of spasticity in children, Dysport should only be used in children 2 years of age or over.
As with any intramuscular injection, Dysport should be used only where strictly necessary and with due caution in patients with prolonged bleeding times. The same caution applies where there are signs of inflammation at the proposed injection site. In this case, infection must be ruled out (see Section 4.3 Contraindications).
Caution should be taken when Dysport is used where the targeted muscle shows excessive weakness or atrophy.
It is essential to study the patient's facial anatomy prior to administering Dysport for correction of moderate to severe glabellar lines. Facial asymmetry, ptosis, excessive dermatochalasis, scarring and any alterations to this anatomy as a result of previous surgical interventions should be taken into consideration.
This product contains a small amount of human albumin. The risk of transmission of viral infection or prion related infection such as Creutzfeldt-Jakob disease (CJD) cannot be excluded with absolute certainty following the use of human blood or blood products.
Serious and/or immediate hypersensitivity reactions have been rarely reported. As with all biological products, adrenaline and other precautions as necessary should be available for immediate administration should an anaphylactic reaction occur.
As with any injection, procedure related injury could occur. An injection could result in localized infection, pain, inflammation, paraesthesia, hypesthesia, tenderness, swelling, erythema and/or bleeding/ bruising. Needle related pain and/or anxiety may result in vasovagal responses, e.g. syncope, hypotension etc. Care should be taken when injecting near vulnerable anatomic structures.
Dysport should only be used to treat a single patient, during a single session. Any unused product remaining should be disposed of in accordance with Instructions for use/handling (see Section 4.2 Dose and Method of Administration). Specific precautions must be taken during the preparation and administration of the product and the inactivation and disposal of any unused reconstituted solution.
Due to the lack of an international unit, Dysport is not therapeutically equivalent to any other botulinum type A toxin preparation currently available on the Australian market. The potencies of Dysport and any other botulinum type A toxin preparation are based on different assay methods. In view of this lack of harmonisation of unit systems for the botulinum type A toxins on the market, extreme caution is required if it should prove necessary to substitute the botulinum type A toxin of one pharmaceutical company by another. The effect of administering different botulinum neurotoxin serotypes at the same time or within several months of each other is unknown. Excessive neuromuscular weakness may be exacerbated by administration of another botulinum toxin prior to the resolution of the effects of a previously administered botulinum toxin.
Use in the elderly. Clinical experience has not identified differences in response between the elderly and younger adult patients. In general, elderly patients should be observed to evaluate their tolerability of Dysport, due to the greater frequency of concomitant disease and other drug therapy. A reduced dose may be appropriate in elderly patients where reduced muscle mass may exist.
Paediatric use. Dysport is approved for the symptomatic treatment of upper and lower limb focal spasticity in children aged 2 years of age and older. For these indications, Dysport should only be used in children 2 years of age and older.
Post-marketing reports of possible distant spread of toxin have been very rarely reported in paediatric patients with comorbidities, predominantly with cerebral palsy. In general, the dose used in these cases was in excess of that recommended (see Section 4.8 Adverse Effects (Undesirable Effects)).
There have been rare spontaneous reports of death sometimes associated with aspiration pneumonia in children with severe cerebral palsy after treatment with botulinum toxin, including following off label use (e.g. neck area). Extreme caution should be exercised when treating paediatric patients who have significant neurologic debility, dysphagia, or have a recent history of aspiration pneumonia or lung disease. Treatment in patients with poor underlying health status should be administered only if the potential benefit to the individual patient is considered to outweigh the risks.
The safety and effectiveness of Dysport for the approved adult indications have not been demonstrated in children.
There were no adverse effects on postnatal growth (including skeletal evaluation), reproductive, neurological and neurobehavioral development when juvenile, rats received 11 administrations over 10 weeks (up to a total cumulative dose of approximately 70 U/kg) from the age of weaning on Postnatal Day 21 up to 13 weeks of age (comparable to children of 2 years old, to young adulthood).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Effect of other medicinal products on Dysport. Any drugs which interfere with neuromuscular transmission, such as muscle relaxants, or drugs which interfere with the intraneuronal concentrations of Ca2+, have the potential to interact with botulinum type A toxin. Aminoglycoside antibiotics cause flaccid paralysis by a similar mechanism to that of botulinum neurotoxin. Therefore, in patients undergoing treatment with Dysport, the additive action of aminoglycoside antibiotics may raise the total neuromuscular blockade to the minimum required for an overt effect. Other drugs that may react pharmacologically with botulinum type A toxin include penicillamine, procainamide, spectinomycin, polymyxins, tetracyclines and lincomycin. Such drugs should be used with caution in patients treated with botulinum toxin.
Effect of Dysport on other medicinal products. No data available.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility in rats was decreased at intramuscular doses of Clostridium botulinum type A toxin-haemagglutinin complex of 33 units per kg per week in males and 80 units per kg per week in females, due to reduced mating secondary to muscle paralysis.
Use in pregnancy. (Category B3)
There are limited data from the use of Dysport in pregnant women. There was no evidence of teratogenicity in rats and rabbits given Clostridium botulinum type A toxin-haemagglutinin complex during the period of organogenesis at respective doses up to 50 and 12 units per kg by daily or weekly intramuscular injection. Maternal toxicity and implantation losses were observed at high doses in both species. Intramuscular administration of Clostridium botulinum type A toxin-haemagglutinin complex to rats during gestation and lactation was associated with slightly reduced pup birth weight and weight gain at severe maternotoxic doses (50 units per kg). Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryofetal development, parturition or postnatal development other than at doses causing maternal toxicity.
Dysport should be used during pregnancy only if the benefit justifies any potential risk to the fetus. Caution should be exercised when prescribing to pregnant women.
Use in lactation. It is not known whether Clostridium botulinum type A toxin-haemagglutinin complex is excreted in human or animal milk. The use of Dysport during lactation cannot be recommended.
4.7 Effects on Ability to Drive and Use Machines
There is a potential risk of muscle weakness or visual disturbances which, if experienced, may temporarily impair the ability to drive or operate machinery.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
The following section is presented in terms of, firstly, those adverse effects which have been reported in association with the use of Dysport in all approved indications and, secondly, any additional adverse effects associated with each specific indication for which Dysport may be used. Only those adverse effects which are considered possibly or probably related to treatment with Dysport are included in this section and the frequency of reporting is indicated as follows:
Tabulated list of adverse reactions. Very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000 to < 1/100); rare (≥ 1/10,000 to < 1/1000); very rare (< 1/10,000); not known (cannot be estimated from the available data).
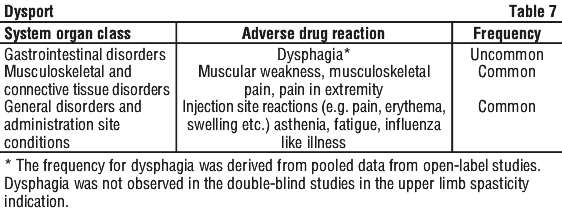
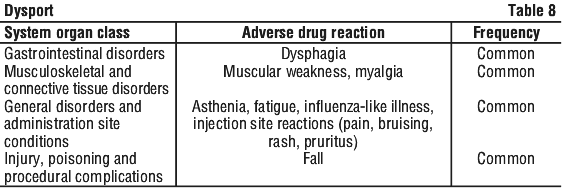
General. In clinical trials in patients suffering from blepharospasm, hemifacial spasm, torticollis or spasticity associated with cerebral palsy or stroke, approximately 30% of patients treated with Dysport experienced an adverse event. See Table 6.

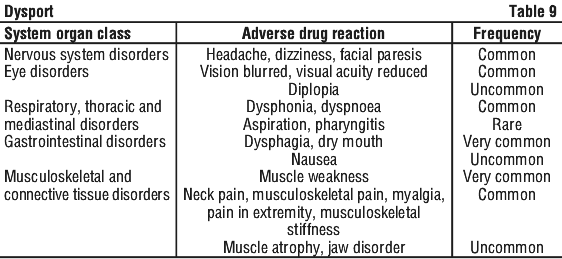
Spasmodic torticollis. In 21 clinical trials involving approximately 4100 patients the following adverse effects were reported. See Table 9.
In addition, during clinical trials, there have been reports of increased salivation in two patients, the causality of which was not assessed by the reporting investigator.
Focal spasticity of lower limbs in children aged 2 years and older. The following adverse events were observed in paediatric patients treated with Dysport for lower limb spasticity. See Table 10.


Blepharospasm and hemifacial spasm. In 13 clinical trials involving approximately 1400 patients treated with Dysport the following adverse effects were reported. See Table 12.

Glabellar lines. In clinical studies, over 1500 patients with moderate to severe glabellar lines have been treated at the recommended dose of 50 units in double blind placebo controlled (Y-97-52120-718 and Y-97-52120-719) and long-term open label (Y-97-52120-720) studies.
In double blind placebo controlled single dose studies, 22.5% of patients treated at the recommended Dysport dose (50 U) and 16.6% of patients treated with placebo, experienced an adverse effect that was related to treatment. In the long-term open label dose phase III study in which patients received multiple injection cycles, 26% of patients experienced at least one treatment related adverse effect after the first injection. The incidence of treatment related adverse effects decreased over repeat cycles.
The most frequently occurring related adverse effects are headache and injection site reactions. Most of the adverse effects reported were of mild to moderate severity.
Patients receiving the recommended dose of 50 units experienced the following adverse effects. See Table 13.
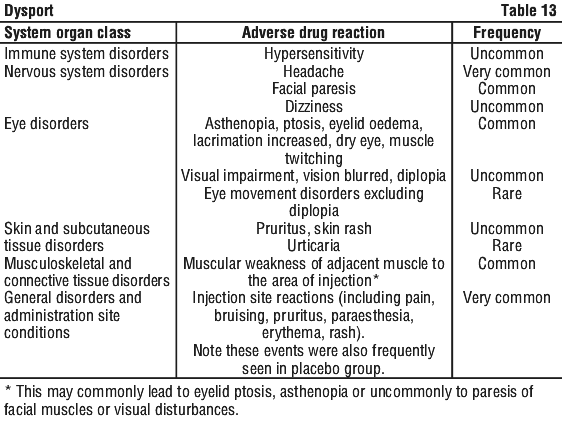
Side effects may occur due to deep or misplaced injections of Dysport temporarily paralysing other nearby muscle groups.
Lateral canthal lines. Based on placebo-controlled clinical trials, patients could experience an adverse effect after the first injection of Dysport at a rate of 6.2% for the treatment of lateral canthal lines (2.9% for placebo). Most of these adverse effects were of mild to moderate severity and reversible. The most frequent adverse effects were injection site reactions, headache and eyelid oedema for lateral canthal lines. See Table 14.

The safety profile of Dysport for concomitant treatment of glabellar lines and lateral canthal lines was evaluated in the open label part of a phase III study; the nature and frequency of adverse events were comparable to what was observed when patients were treated for the individual indications.
Post-marketing reports. The profile of adverse effects reported to the company during post-marketing use reflects the pharmacology of the product and those seen during clinical trials. See Table 15.

During a post-marketing surveillance study a number of reports of neck/ shoulder pain (22), unspecified pain (10), heavy head/ neck/ shoulder (5), local pain (2), rigid neck (1), muscle soreness (1), ear pain (1), back pain (1), neck tension (1), arm pain (1), heavy arm (1), neuralgia (1) and muscle pain (1) were received which were assessed by the reporters as being related to treatment.
Adverse effects resulting from distribution of the effects of the toxin. Adverse effects resulting from distribution of the effects of the toxin to sites remote from the site of injection have been very rarely reported (excessive muscle weakness, dysphagia leading to aspiration pneumonia that may be fatal).
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Excessive doses may produce distant and profound neuromuscular paralysis. Overdose could lead to an increased risk of the neurotoxin entering the bloodstream and may cause complications associated with the effects of oral botulinum poisoning (e.g. dysphagia and dysphonia). Respiratory support may be required where excessive doses cause paralysis of respiratory muscles. General supportive care is advised. In the event of overdose the patient should be medically monitored for any signs and/or symptoms of excessive muscle weakness or muscle paralysis. Symptomatic treatment should be instigated if necessary.
Symptoms of overdose may not present immediately following injection. Should accidental injection or oral ingestion occur the patient should be medically supervised for several weeks for any signs and/or symptoms of excessive muscle weakness or muscle paralysis.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: other muscle relaxants, peripherally acting agents.
ATC Code: M03AX01.
Mechanism of action. Pharmacodynamic effects. Clostridium botulinum type A toxin-haemagglutinin complex blocks peripheral cholinergic transmission at the neuromuscular junction by a presynaptic action at a site proximal to the release of acetylcholine. The toxin acts within the nerve ending to antagonise those events that are triggered by Ca2+ which culminate in transmitter release. It does not affect postganglionic cholinergic transmission or postganglionic sympathetic transmission.
The action of toxin involves an initial binding step whereby the toxin attaches rapidly and avidly to the presynaptic nerve membrane. Secondly, there is an internalisation step in which toxin crosses the presynaptic membrane, without causing onset of paralysis. Finally the toxin inhibits the release of acetylcholine by disrupting the Ca2+ mediated acetylcholine release mechanism, thereby diminishing the endplate potential and causing paralysis.
Recovery of impulse transmission occurs gradually as new nerve terminals sprout and contact is made with the postsynaptic motor endplate, a process which takes 6 - 8 weeks in the experimental animal.
Clinical trials. Focal spasticity affecting the upper limb in adults. The efficacy and safety of Dysport for the treatment of upper limb spasticity was evaluated in a randomized, multi-centre, double-blind, placebo-controlled study that included 238 patients (159 Dysport and 79 placebo) with upper limb spasticity (Modified Ashworth Scale (MAS) score ≥ 2 in the primary targeted muscle group (PTMG: finger, or wrist, or elbow flexors) for toxin naive subjects or MAS score ≥ 3 in the PTMG for toxin non-naive subjects where at least 4 months elapsed since their last botulinum toxin injection, of any serotype) who were at least 6 months post-stroke or post-traumatic brain injury.
The Modified Ashworth Scale (MAS) is the most commonly used measure of efficacy in the reduction of upper limb spasticity and is a direct measure of the degree of spasticity. The MAS assessment of spasticity involves separate assessment of the muscle tone of the elbow, wrist and fingers. The investigator or an appropriate delegate (e.g. physiotherapist) assesses the resistance encountered to passive movement at each joint on a six-point scale as follows:
0 = No increase in muscle tone.
1 = Slight increase in muscle tone, manifested by a catch and release, or by minimal resistance at the end of the range of motion when the affected part is moved in flexion or extension.
1+ = Slight increase in muscle tone, manifested by a catch, or by minimal resistance throughout the remainder (< 1/2) of the range of movement (ROM).
2 = More marked increase in muscle tone through most of ROM, but affected part easily moved.
3 = Considerable increase in muscle tone, passive movement difficult.
4 = Affected part rigid in flexion or extension.
The total volume (i.e. 5.0 mL) of either Dysport 500 U (N = 80), Dysport 1000 U (N = 79), or placebo (N = 79) was injected intramuscularly into the affected upper limb muscles. The volume of either Dysport or placebo injected in the PTMG is presented in Table 16. After injection of the PTMG the remainder of the dose (2.0 or 3.0 mL) was injected into at least two additional upper limb muscles. Muscles suggested to the investigator are listed in Table 16. No more than 1.0 mL was allowed to be administered per injection site. However more than one injection site per muscle was permitted.
An EMG/nerve stimulator was used to assist in proper muscle localization for injection. Patients were followed for 24 weeks.
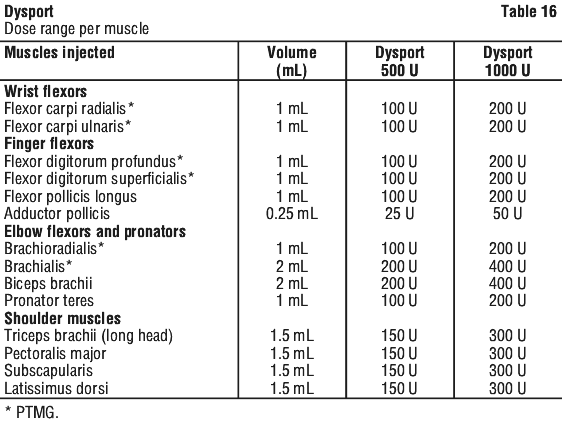
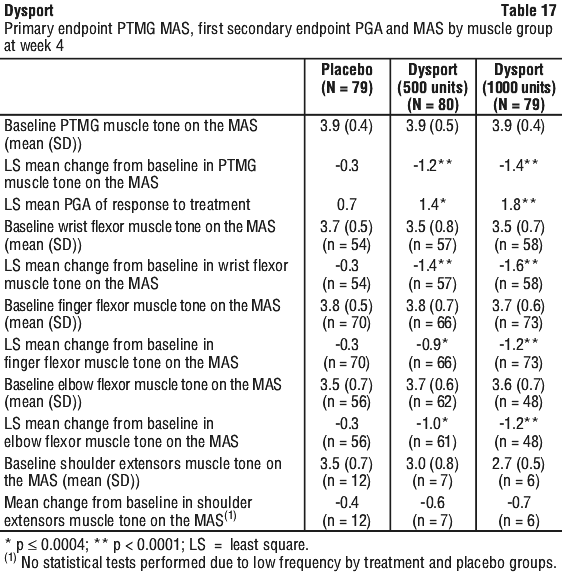

Dysport 1000 U statistically improved the active range of motion (AROM) by clinically meaningful margins in the elbow (+18.3 degrees, baseline 138.7 degrees), wrist (+35.2 degrees, baseline 63.2 degrees) and finger muscles (+11.8 degrees, baseline 47.5 degrees) at week 4 while there was no improvement in placebo group. Dysport 500 U showed similar benefit on finger muscles AROM.
Improvements in ease of applying a splint by the subject were statistically significantly greater in the Dysport 1000 U and 500 U treatment groups than in the placebo group at weeks 4 and 12.
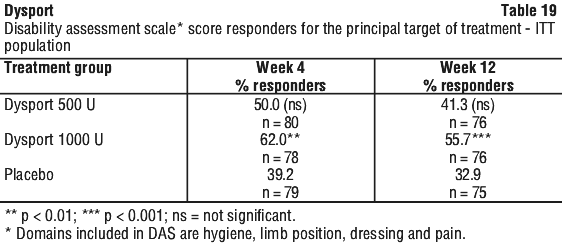
In a subsequent open-label extension study, re-treatment was determined by clinical need after a minimum of 12 weeks. Doses greater than 1000 U and up to 1500 U were permitted when the shoulder muscles were injected. Subjects with co-existing lower limb spasticity were able to receive injections of Dysport 500 U into the affected lower limb in addition to 1000 U in the upper limb, with a maximum total dose of 1500 U. After repeated administration, the efficacy of Dysport is maintained for up to 1 year as assessed by MAS (as evidenced by the responder rates ranging from 75% to 80% in the open label study compared to 75% in the placebo-controlled study) and PGA when injected in the upper limb muscles. Dysport effect was also maintained or improved on passive function (Disability Assessment Scale), spasticity (Tardieu scale), AROM and ease of applying splints.
Focal spasticity affecting the lower limbs in adults. The efficacy and safety of Dysport for the treatment of lower limb spasticity was evaluated in a pivotal randomized, multi-centre, double-blind, placebo-controlled study that included 385 post-stroke and brain injury patients (255 Dysport and 130 placebo treated subjects) with lower limb spasticity. The primary end point was Modified Ashworth Scale (MAS) score assessed at the ankle joint.
The total volume of 7.5 mL of either Dysport 1000 U (N = 127), Dysport 1500 U (N = 128) or Placebo (N = 128) was divided between the gastrocnemius and soleus muscles and at least one other lower limb muscle according to clinical presentation.
When assessing MAS at the ankle with the knee extended (involving all plantar flexors), statistically significant improvement was observed for 1500 U. When assessing MAS at the ankle with the knee flexed (involving all plantar flexors except the gastrocnemius), statistically significant improvement was observed for both 1000 U and 1500 U.
Improvements in the spasticity at the ankle joint were also demonstrated using the Tardieu Scale (TS) with statistically significant improvements in the spasticity severity grade observed at both the 1000 U and 1500 U doses. Dysport treatment was also associated with statistically significant clinical improvement at both doses as measured by the Physician Global Assessment (PGA) Score.
Numerical improvement in ankle dorsiflexion for the higher Dysport dose was seen with the change peaking at 4 weeks post administration. Additional endpoints such as reduction in pain, using walking aids and quality of life measures did not show statistically significant improvement.
On completion of this study, 345 patients entered an open-label extension study in which retreatment with Dysport 1000 U or 1500 U was determined by clinical need. Subjects with co-existing upper limb spasticity were able to receive injections of Dysport 500 U into the affected upper limb in addition to 1000 U in the lower limb, with a maximum total dose of 1500 U. Improvements in efficacy parameters (MAS, PGA and TS) seen after 4 weeks of double blind treatment with Dysport in the lower limb continued to improve over repeated treatment. Improvement in walking speed was not observed after a single treatment in the double blind study but was observed after repeated treatment.
Improvements in 10-m walking speed (comfortable and maximal, with or without shoes) were observed, which increased with successive treatment cycles. No significant improvements in lower limb pain using the SPIN scale, use of walking aids or quality of life measures were observed.
Several small studies in adult lower limb spasticity did not show a significant benefit.
Spasmodic torticollis in adults. In a dose-finding study (n = 74) conducted in 5 neurology clinics in Germany, doses of 250 units (n = 19), 500 units (n = 17) and 1000 units (n = 18) of Dysport were compared with placebo (n = 20) in a randomised, parallel group study in male and female patients aged 18 years or over with rotational torticollis. Improvements in symptoms were statistically significantly better than placebo for the 500 unit and 1000 unit dose groups at 4 weeks using Tsui score. A dose relationship was also demonstrated by patient and investigator assessments of improvement since injection. Compared with placebo, statistically significant differences were observed at 8 weeks for the 500 unit and the 1000 unit treatment groups but at 4 weeks only the comparisons of the 250 unit and the 1000 unit groups were statistically significant. Associated with an increase in dose is an increased risk, particularly of dysphagia and therefore the optimal initial dose appears to be 250-500 units. A dose range of 250-1000 units is appropriate for simple rotational torticollis.
In a double blind study (n = 73) conducted in 7 centres in Sweden and Finland in male or female patients over the legal age of consent where Dysport (n = 38) was compared with the other botulinum toxin preparation available in Australia, a ratio of approximately 3 units of Dysport was found to achieve similar effects to one unit of Botox for the treatment of spasmodic torticollis within the therapeutic dose range (250 to 1000 units).
Focal spasticity of lower limbs in children aged 2 years and older. A double-blind, placebo-controlled multicentre study (study Y-55-52120-141) was conducted in children with dynamic equinus foot deformity due to spasticity in children with Cerebral Palsy. A total of 235 botulinum toxin naive or non-naive patients with a Modified Ashworth Score (MAS) of grade 2 or greater were enrolled to receive Dysport 10 Units/kg/leg, Dysport 15 Units/kg/leg or placebo. Forty one percent of patients were treated bilaterally resulting in a total Dysport dose of either 20 Units/kg or 30 Units/kg. The primary efficacy variable was the mean change from baseline in MAS in ankle plantar flexors at week 4 (Table 20). Secondary efficacy variables were the mean Physicians Global Assessment (PGA) score and Mean Goal Attainment Scaling (GAS) score at week 4 (Table 20). Patients were followed up for at least 12 weeks post-treatment and up to a maximum of 28 weeks.

Both Dysport treatment groups, 10 U/kg/leg and 15 U/kg/leg, demonstrated a significant improvement from baseline in the Observational Gait Scale (OGS) overall score at week 4 when compared to placebo and a statistically significantly higher proportion of patients were treatment responders for initial foot contact on the OGS at week 4 and week 12.
Parents completed the condition-specific Module for CP for the Paediatric Quality of Life Inventory. There was a statistically significant improvement from baseline in fatigue at week 12 in the Dysport 10 U/kg/leg and 15 U/kg/leg Dysport treatment groups compared to placebo. No other statistically significant improvements were observed in the other subscales.
On completion of this study, 216 patients entered an open-label extension study where they could receive re-treatment based on clinical need. Both distal (gastrocnemius, soleus and tibialis posterior) and proximal (hamstrings and hip adductors) muscles were permitted to be injected, including multilevel injections. Efficacy was observed over repeated treatment sessions for up to 1 year as assessed by MAS, PGA and GAS.
Another double-blind, placebo-controlled multicentre study (A-94-52120-094) was conducted for the treatment of hip adductor spasticity in 61 children with cerebral palsy 2 to 10 years of age. Dysport 30 U/kg (15 U/kg/leg) or placebo was injected into the adductor and medial hamstring muscles of both legs.
Significant improvement compared to placebo was observed at week 4 in the primary variables of change in passive range of motion at the hip (mean change from baseline of 4.8 degrees versus 0.5 degrees; p = 0.04) and inter-medial condyle distance at fast stretch (mean change from baseline of 6.4 degrees versus 1.9 degrees, p < 0.001). Significant improvements in muscle tone, measured by the MAS, were observed for adductor muscles and medial hamstrings post-treatment.
There is limited placebo-controlled clinical study data for the treatment of paediatric lower limb spasticity in proximal muscles.
Focal spasticity of upper limbs in children aged two years and older. The efficacy and safety of Dysport for the treatment of upper limb spasticity in children was evaluated in a randomised, multi-centre, double-blind, controlled, study in which doses of 8 U/kg and 16 U/kg in the selected study upper limb were compared with a low dose control group of 2 U/kg. A total of 210 botulinum toxin naive or non-naive patients with upper limb spasticity due to cerebral palsy (Modified Ashworth Scale (MAS) score ≥ 2 in the primary targeted muscle group (PTMG)) were randomised and treated in the study.
After the initial treatment, up to 3 further treatments of Dysport could be administered at planned doses of either 8 U/kg or 16 U/kg, although the investigator could elect to increase or decrease the dose (but not exceeding 16 U/kg).
The total dose of Dysport was injected intramuscularly into the affected upper limb muscles which included the PTMG of either elbow flexors or wrist flexors as well as other upper limb muscles according to the disease presentation. No more than 0.5 mL was allowed to be administered per injection site. However, more than one injection site per muscle was permitted.
An electrical stimulation (ES) and/or ultrasound was used to assist muscle localisation for injection.
The primary efficacy variable was the mean change from baseline in MAS in PTMG at week 6. Secondary efficacy variables were the mean Physicians Global Assessment (PGA) score and mean Goal Attainment Scale (GAS) score at week 6. As shown in Table 21, statistically significant improvements were demonstrated in MAS; improvements in PGA and GAS were not statistically significant.
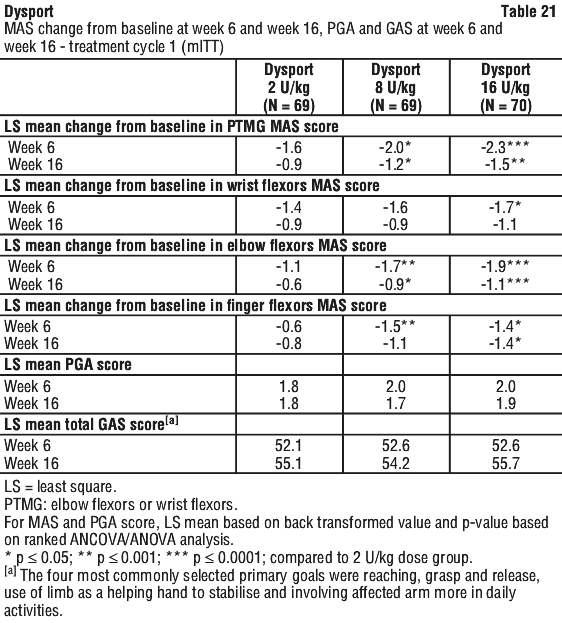
Parents completed the condition-specific module for Cerebral Palsy for the Paediatric Quality of Life Inventory. At week 16, there was a statistically significant improvement from baseline in fatigue (p = 0.0443) in the Dysport 8 U/kg group and, in movement and balance (p = 0.0068) in the 16 U/kg group compared with the Dysport 2 U/kg group. No other statistically significant improvements were observed in the other subscales.
The majority of subjects treated with Dysport were retreated by week 28 (62.3% in the Dysport 8 U/kg group and 61.4% in the Dysport 16 U/kg group), though more than 24% of subjects in both treatment groups had not yet required retreatment by week 34.
Blepharospasm and hemifacial spasm in adults. A phase II, multi-centre, randomized, double-blind, parallel group, placebo-controlled study (study Y-47-52125-706) has been conducted to assess the efficacy and safety of a single administration, in 6 injection sites by subcutaneous injection, of three doses of Dysport (40 U/eye, 80 U/eye, 120 U/eye) for the treatment of benign essential blepharospasm. Results of this study support a recommended starting dose of 40 units per eye, increasing to 80 units per eye where a sustained effect is required.
In open label, uncontrolled studies from the published literature, the treatment of hemifacial spasm was generally the same as for the treatment of unilateral blepharospasm.
The studies showed that visual function improved in the majority of cases, returning to normal or near to normal. Injection of Dysport abolished or reduced muscle spasm in patients with blepharospasm or hemifacial spasm, for whom a benefit was reported in 70-100% of the cases according to the investigator. Discomfort was also reduced, and the patients' facial appearance improved.
Criteria for assessment of results varied from one study to another. However, the assessment techniques were mainly qualitative and subjective, relying on a nominal scale which takes into account criteria such as visual function, frequency of spasm or severity of spasm. Neither the severity of the illness, the length of time it existed before commencement of Dysport injections, nor the gender or age of the patient influenced response to treatment.
Despite the variety of doses and administration techniques reported in the published studies, the overall response profile was favourable across the studies. Following the initial treatment, substantial improvements were reported for both blepharospasm (success rate range: 77-100%) and hemifacial spasm (success rate range: 75-100%).
The onset of improvement post the initial injection varied from 1 day to 3 weeks for blepharospasm and from 2 to 7 days for hemifacial spasm. The duration of effect lasted between 5 and 24 weeks for blepharospasm and between 6 and 24 weeks for hemifacial spasm. The issue of time to peak effect post initial injection was assessed somewhat loosely in only about four of the submitted publications and the latter appeared to range from 3 days to 6 weeks and from 1 week to 6 weeks respectively for blepharospasm and hemifacial spasm. There was a tendency for repeat injection to produce a comparable level of efficacy to the initial injection for both conditions.
There are no satisfactory efficacy and safety data on the use of Dysport for the treatment of blepharospasm and hemifacial spasm in children and adolescents younger than 18 years of age.
Glabellar lines. During the clinical development of Dysport for the treatment of glabellar lines, more than 2600 patients were included in the different clinical trials.
In glabellar lines clinical studies, 1907 patients with moderate to severe glabellar lines have been treated at the recommended dose of 50 Units. Of these, 305 were treated with 50 U in two phase III double-blind placebo-controlled studies (Y-97-52120-718 and Y-97-52120-719) and 1200 treated with 50 U in a long-term open-label repeated dose phase III study (Y-97-52120-720).
The median time to onset of response was 2 to 3 days following treatment.
The maximum effect on the number of responders was observed at day thirty following injection, when the assessment of the investigators showed that 90% (273/305) of patients had responded to treatment (exhibited no or mild glabellar lines at maximum frown), compared to 3% (4/153) of placebo-treated patients. The patient's own assessment at maximum frown after thirty days gave a response rate of 82% (251/305) for those treated with Dysport and 6% (9/153) for those treated with placebo.
In both placebo-controlled phase III studies, Dysport injections significantly reduced the severity of glabellar lines for up to 4 months. In one of these two studies, the effect was still statistically significant (p < 0.001) at 5 months with 17% (32/190) of patients treated with Dysport still responding to treatment compared to 1% (1/92) of placebo treated patients. In the other study the corresponding effect after 4 months (p = 0.002) was 24% (24/99) vs. 4% (2/49) with no statistically significant difference beyond 4 months.
The long-term repeat dose open label study showed that the median time to onset of response of 3 days was maintained across repeated dose cycles. The responder rate at maximum frown as determined by the investigator at day 30 was maintained over repeated cycles (ranging between 80% and 91% over the 5 cycles). In patients who were rated 'moderate' or 'severe' as baseline, the responder rate at rest over repeated dose cycles was also consistent with the single dose studies, with ~70% of Dysport-treated patients considered by investigators to be responders thirty days after treatment.
Lateral canthal lines. In clinical studies, 306 patients with moderate to severe lateral canthal lines (LCL) at maximum smile have been treated at the recommended Dysport dose of 30 units per side in double blind studies and had follow-up data. Of these, 252 were treated in a phase III double-blind placebo controlled study and 54 patients were treated in a double-blind phase II dose-ranging study.
In the phase III study, Dysport injections significantly reduced the severity of LCL compared with placebo (p ≤ 0.001) at 4, 8 and 12 weeks (assessed at maximum smile by the investigators). For the subjects' assessment of satisfaction with the appearance of their LCL, there was a statistically significant difference between Dysport and placebo (p ≤ 0.010) in favour of Dysport at 4, 8, 12 and 16 weeks.
A subset of 241 patients had moderate to severe canthal lines at rest prior to treatment. In this subset, the proportion of subjects exhibiting a one-grade improvement at week 4, according to the investigator assessment at rest, was statistically significantly higher in Dysport-treated patients than in placebo-treated subjects.
The primary efficacy endpoint was at 4 weeks following injection: the assessment of the investigators showed that 47.2% (119/252) of patients had responded to treatment (no or mild LCL at maximum smile), compared to 7.2% (6/83) placebo-treated patients. In a post-hoc analysis, at the same time point, 4 weeks following injection, 75% (189/252) of Dysport treated patients had at least 1 grade improvement at maximum smile compared with only 19% (16/83) of placebo-treated subjects.
A total of 315 subjects entered the open label extension phase of the phase III study in which they could be treated concomitantly for both lateral canthal lines and glabellar lines.
Patients treated with Dysport in the double-blind and open label phases of the phase III study received a median of 3 treatments for LCL. The median interval between injections for LCL, which was largely determined by the protocol design, ranged from 85 to 108 days. The results showed that efficacy is maintained with repeated treatments over the period of one year.
The patient satisfaction levels at weeks 4, 16 and 52 show after the first treatment with Dysport that 165/252 subjects (65.5%) were either very satisfied or satisfied with the appearance of their LCLs.
At week 16, 4 weeks after either a second Dysport treatment for those randomised to Dysport in Part A or the first treatment for those randomised to placebo, the proportion who were very satisfied or satisfied was 233/262 (89.0%). At week 52 when subjects could have had up to five cycles of Dysport treatment with the last one being at week 48, the proportion of very satisfied/satisfied subjects was 255/288 (84.7%).
No patients developed the presence of neutralising antibodies after receiving repeated treatments with Dysport over one year.
5.2 Pharmacokinetic Properties
Pharmacokinetics have not been formally studied in humans or animals. Following intramuscular injection to man, there is usually a delay of 2-3 days with a peak effect between 10 and 21 days after injection. The duration of response varies but on average is 8-12 weeks.
5.3 Preclinical Safety Data
Genotoxicity. No studies to assess the mutagenic potential of Dysport have been conducted.
Carcinogenicity. Long term animal studies to evaluate the carcinogenic potential of Dysport have not been performed.
6 Pharmaceutical Particulars
6.1 List of Excipients
125 microgram human serum albumin and 2.5 mg lactose in a sterile, lyophilised form without a preservative.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Unopened vials must be maintained at temperatures between 2°C and 8°C.
Dysport must be stored in a refrigerator at the hospital or clinic where the injections are to be carried out and should not be given to the patient to store. Dysport should not be frozen.
6.5 Nature and Contents of Container
Dysport is provided in type 1 glass vials with bromobutyl rubber stoppers, containing 125, 300 or 500 units of botulinum toxin type A powder for injection.
Boxes of 1 vial are available.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Clostridium botulinum type A toxin-haemagglutinin complex has a molecular weight of about 900,000 D and is a complex of proteins.
7 Medicine Schedule (Poisons Standard)
S4.
Date of Revision
20 December 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.