EMLA
Brand Information
| Brand name | EMLA |
| Active ingredient | Lidocaine (lignocaine) + Prilocaine |
| Schedule | S2 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the EMLA.
Summary CMI
EMLA
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using EMLA?
EMLA contains the active ingredient lidocaine and prilocaine. EMLA is used to numb the skin to allow minor surgical procedures to be performed with a significant reduction in pain.
For more information, see Section 1. Why am I using EMLA? in the full CMI.
2. What should I know before I use EMLA?
Do not use if you have ever had an allergic reaction to EMLA or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use EMLA? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with EMLA and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use EMLA?
- EMLA is available as a cream or a patch. Instructions on how to use it are included in the box. More instructions can be found in Section 4. How do I use EMLA? in the full CMI.
5. What should I know while using EMLA?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using EMLA? in the full CMI.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using EMLA. Some of the mild side effects include local reactions such as itching, swelling, paleness, redness or a burning sensation. Some of the serious side effects include a rash at a spot where EMLA is not being used, difficulty breathing, dizziness, shaky hands, blurred vision and areas of skin becoming blue. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
EMLA
Active ingredient(s): Lidocaine and prilocaine
Consumer Medicine Information (CMI)
This leaflet answers some common questions about EMLA. It does not contain all the available information that is known about EMLA. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you or your child using EMLA against the benefits they expect it will have for you.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using EMLA.
Keep this leaflet with the medicine.
You may need to read it again.
Where to find information in this leaflet:
1. Why am I using EMLA?
2. What should I know before I use EMLA?
3. What if I am taking other medicines?
4. How do I use EMLA?
5. What should I know while using EMLA?
6. Are there any side effects?
7. Product details
1. Why am I using EMLA?
EMLA is a mixture of two local anaesthetics, lidocaine and prilocaine. These local anaesthetics are combined with special agents which allow the products to pass through the skin. Once through the skin the numbing effect they produce allows minor surgical procedures to be performed with a significant reduction in pain.
These procedures can include taking blood samples, vaccination, skin grafting, cleaning leg ulcers, inserting an intravenous catheter, minor superficial cosmetic procedures and procedures on genital skin.
EMLA is available as a cream which is applied then covered with a dressing, or as a patch, which has the cream and dressing already to use.
Follow all directions given to you by your doctor carefully.
They may differ from the information contained in this leaflet.
Your doctor may prescribe this medicine for another use. Ask your doctor if you want more information.
EMLA is not addictive.
2. What should I know before I use EMLA?
Warnings
Do not apply EMLA cream to any open wounds except leg ulcers.
EMLA should not be used in premature babies. We do not have enough knowledge yet to be sure it is safe in premature babies.
Do not use EMLA if
You or your child have allergies to:
- any of the ingredients listed at the end of this leaflet
- other local anaesthetics, for example those used at the dentist.
- adhesives or sticking plasters.
If you have an allergic reaction, you may get a skin rash, hayfever, asthma or feel faint.
Do not use after the use by (expiry) date printed on the pack or the packaging is torn or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
Do not use it to treat any other complaints unless your doctor says it is safe.
Do not give this medicine to anyone else.
Check with your doctor if you:
Have any of the following medical conditions:
- dermatitis
- methaemoglobinaemia
- glucose-6-phosphate dehydrogenase deficiency
- mollusca contagiosa.
It may not be safe for you or your child to use EMLA if you have any of these conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use EMLA if you are pregnant or breastfeeding unless your doctor says to do so. Ask your doctor about the risks and benefits involved.
The ingredients in EMLA have been used for many years and no ill effects have been shown if they are used while you are pregnant.
Only very small amounts of EMLA get into the blood so even though your baby can take in EMLA from breast milk if you are breastfeeding, it is unlikely to cause any problems.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and EMLA may interfere with each other. These include:
- a sulphonamide antibiotic eg sulfatrimoxazole
- medicines used to treat irregular heartbeat such as amiodarone
- other local anaesthetics
Your doctor or pharmacist can tell you what to do if you are taking any of these medicines.
If you have not told your doctor about any of these things, tell them before you use any EMLA.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect EMLA.
4. How do I use EMLA?
How to use EMLA
EMLA is available as a cream or a patch.
Instructions on how to use it are included in the box. Please read them carefully. The instructions also give maximum doses for adults and children. Do not exceed the maximum doses as this can cause harmful side effects, particularly in infants under 3 months of age.
On intact skin
The most important thing to do is to be sure that you apply EMLA at least one hour before you are due to have the procedure except for skin grafting where a two hour application time is required or for procedures on male genital skin where only fifteen minutes is required.
Leg ulcers
If you are using EMLA on leg ulcers you only need to apply the cream 30 minutes before your ulcer is going to be cleaned.
EMLA can be left on for several hours and still work.
If you forget to use it
Apply EMLA as soon as you realise it if it is less than an hour until you are supposed to have the procedure. Tell your doctor exactly when you put EMLA on and they will decide when the procedure can take place.
If you use too much EMLA
If you think that you have used too much EMLA, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Do not exceed the maximum recommended dose or length of administration. Children, particularly those aged below 3 months of age, are at an increased risk of serious adverse effects in overdose.
For most procedures, it is very difficult to use too much EMLA. Be careful not to use too much in procedures where larger amounts may be required, such as cosmetic hair removal (see Package Insert for maximum amounts).
Using too much EMLA may cause unwanted side effects. Some of these can be serious, such as methaemoglobinaemia (a condition where the blood cannot take enough oxygen to your body) or cardiovascular effects (effects on your heart and lungs).
If you use too much EMLA, you may notice the following:
- nervousness
- dizziness
- difficulty breathing
- numbness of the mouth
- your skin turning blue (a symptom of methaemoglobinaemia)
- blurred vision
- shaky hands
- a rash where EMLA has not been applied.
5. What should I know while using EMLA?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using EMLA.
Remember to apply EMLA on intact skin at least one hour before your procedure is due. For leg ulcers remember to apply EMLA 30 minutes before your ulcer is due to be cleaned. Apply EMLA two hours before split skin grafting and at least fifteen minutes before a procedure on male genital skin.
If you do not, your or your child's appointment may have to be delayed, or the procedure may hurt more than it would otherwise.
If you or your child are using EMLA on leg ulcers, only use the tube once. Throw out any cream left in the tube after use.
Be sure to follow the instructions in the pack on how to apply EMLA cream or patch carefully.
Make sure the patch or the dressing covering the cream is firmly fixed, especially on young children.
If EMLA is not covered carefully it may not work effectively.
Things you should not do
Do not use EMLA cream on leg ulcers for longer than 2 months without checking with your doctor or pharmacist.
Do not to let EMLA get into the eyes.
It may cause eye irritation. If EMLA does get into your eyes, immediately rinse them with large amounts of water and contact your doctor or pharmacist for advice.
Looking after your medicine
Keep your cream or patches in the pack until it is time to use them.
If you squeeze EMLA out of the tube or open the patches it will not keep well.
Keep EMLA cream in a cool dry place where the temperature stays below 30°C. Do not let it freeze.
Keep EMLA patches in a cool dry place where the temperature stays below 25°C. Do not let them freeze.
Do not store it or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
When to discard your medicine (as relevant)
Ask your pharmacist what to do with any EMLA you have left over if your doctor tells you to stop using it, or you find that the expiry date has passed.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you or your child do not feel well while using EMLA.
EMLA helps most people to undergo minor procedures without feeling pain, but it may have unwanted side-effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not.
You may need medical treatment if you or your child get some of the side effects.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these mild side effects and they worry you |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. You may need urgent medical attention or hospitalisation |
Tell your doctor or pharmacist if you notice anything else that may be making you or your child feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What EMLA contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What EMLA looks like
EMLA Cream is a white, soft, homogenous cream, packed in an aluminium tube.
5 x 5 g tubes (with 10 dressings)
1 x 5 g tube (with 2 dressings)
or 30 g tube.
EMLA Patch is circular cellulose disc on a square, thin, flexible aluminium/plastic laminate with adhesive surface and peel-off protective liner.
Packs of 2 or 20 patches.
Who distributes EMLA
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was prepared in July 2025.
Australian Registration Number (ARTG)
EMLA Cream
1 or 5 x 5 g - 12046
30 g - 12886
EMLA Patch – 60820
Trademarks are owned by or licensed to the Aspen Group of companies.
© 2025 Aspen Group of companies or its licensor. All rights reserved
Brand Information
| Brand name | EMLA |
| Active ingredient | Lidocaine (lignocaine) + Prilocaine |
| Schedule | S2 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
EMLA (Eutectic Mixture of Local Anaesthetics) is a 1:1 oil/water emulsion of an eutectic mixture of lidocaine and prilocaine.
Lidocaine is the new medicine ingredient name for lignocaine and is used in this product information.
2 Qualitative and Quantitative Composition
When lidocaine and prilocaine are mixed in equal amounts, the solid pure bases of lidocaine and prilocaine form an oil at temperatures above 16°C (i.e. a eutectic mixture). By avoiding the need for a non-aqueous solvent, higher concentrations of local anaesthetic in the cream can be achieved and maintained during application.
EMLA cream 5% contains in 1 g lidocaine 25 mg, prilocaine 25 mg as the active ingredients; and carbomer 974P, polyoxyethylene hydrogenated castor oil, sodium hydroxide to a pH of approximately 9.2 and purified water to 1 g.
3 Pharmaceutical Form
EMLA patch 5% is a single dose unit in the form of an occlusive dressing. It is composed of a laminate backing, an absorbent cellulose disc and an adhesive tape ring. The disc provides a contact area of approximately 10 cm2 and contains 1 g of EMLA emulsion. Each 1 g of EMLA emulsion contains lidocaine 25 mg and prilocaine 25 mg as active ingredients, and excipients as for EMLA cream 5%. The emulsion contains a lower concentration of the thickening agent, carbomer 974P.
EMLA is non-sterile and does not contain preservatives.
4 Clinical Particulars
4.1 Therapeutic Indications
EMLA cream and EMLA patch. Topical anaesthesia of the skin prior to insertion of i.v. catheters, blood sampling; vaccination; superficial surgical procedures, including split skin grafting.
EMLA cream. Topical anaesthesia of leg ulcers to facilitate mechanical cleansing or debridement.
Topical anaesthesia of genital skin prior to superficial surgical procedures or infiltration anaesthesia.
Topical anaesthesia of the skin prior to minor superficial cosmetic procedures.
4.2 Dose and Method of Administration
In order to avoid cross-contamination, infection control procedures and principles should be strictly adhered to during application of EMLA.
Pharmacokinetic data for application longer than 4 hours is not available in children. In adults, there is no benefit in application times longer than 5 hours, as the analgesic effectiveness of the cream dissipates over time.
Use in the elderly. No dosage adjustment is required when EMLA is applied to intact skin in the elderly.
Use in premature infants with a gestational age of less than 37 weeks is not recommended (see Section 4.4 Special Warnings and Precautions for Use).
EMLA cream 5%. The protective membrane of the tube is perforated by reversing the cap and piercing the membrane. When used on leg ulcers discard the tube with any remaining EMLA after each occasion that a patient is treated.
See Table 1.

A 1 g dose of EMLA cream can also be achieved by squeezing a length of EMLA of approximately 3.5 cm from the tube.
EMLA patch 5%. Maximum doses. Neonates and infants up to 3 months old should not have more than 1 patch applied at the same time.
Infants aged 3 months up to 12 months old should not have more than 2 patches applied at the same time.
Children aged 1 year up to 12 years old should not have more than 5 patches applied at the same time.
Minor procedures (e.g. needle insertion). The patch is applied to the skin area selected. Care should be taken that the patch does not become detached during the minimum application time of 60 minutes (see Section 4.4 Special Warings and Precautions for Use).
The EMLA patch should be applied at least 1 hour prior to the start of the procedure.
4.3 Contraindications
Hypersensitivity to prilocaine, lidocaine or any local anaesthetics of the amide type.
Hypersensitivity to any of the excipients of EMLA cream or emulsion, or to the adhesive used in the patch.
Glucose-6-phosphate dehydrogenase deficiency or congenital or idiopathic methaemoglobinaemia.
4.4 Special Warnings and Precautions for Use
Open wounds. EMLA should not be applied to open wounds other than leg ulcers, due to insufficient data on absorption from these sites.
Atopic dermatitis. Care should be taken when applying EMLA to skin areas with atopic dermatitis. A shorter application time (15-30 minutes) may be sufficient.
Eyes. EMLA should not be applied to or near to the eyes since it causes corneal irritation. Damage to the eye may also occur from undetected foreign bodies. Special care should be employed to reduce the risk of rubbing the eyes with EMLA. It is therefore important that the patch or occlusive dressing should be secured against accidental dislocation, especially in young children.
Middle ear. EMLA is not recommended in any clinical situation in which its penetration into the middle ear is possible. In studies in rodents (guinea pigs), EMLA was found to have an ototoxic effect when instilled directly into the middle ear, however no abnormalities were observed when EMLA was applied to the animals' external auditory canal. EMLA cream 5% caused minor structural damage to the tympanic membrane in rats when applied directly to the membrane. The relevance of these findings to the clinical situation is unknown.
Genital mucosa. EMLA is presently not recommended for use on genital mucosa. Available data suggest that the anaesthetic efficacy of EMLA on genital mucosa may be variable.
Application of EMLA patch 5%. Care should be taken that the patch does not become detached (especially in young children) during the 60 minute wait. The possibility that this may occur can be reduced if the patch is applied to the non-dominant hand; or lightly bandaged if applied to the antecubital fossa to protect it from lifting if rubbed by clothing.
Vaccination. Lidocaine and prilocaine have bactericidal and antiviral properties in concentrations above 0.5 - 2%. A clinical trial with MMR vaccine administered subcutaneously demonstrated that EMLA does not adversely affect antibody response. There are no data on effects of EMLA on other live viral vaccines administered subcutaneously. When EMLA is used prior to intradermal BCG vaccination, the results of vaccination should be monitored.
Anti-arrhythmic drugs class III. Patients treated with anti-arrhythmic drugs class III (e.g. amiodarone) should be kept under close surveillance and ECG monitoring considered, since cardiac effects may be additive.
Drugs reducing clearance of lidocaine. Drugs that reduce the clearance of lidocaine (for example, cimetidine or betablockers) may cause potentially toxic plasma concentrations when lidocaine (e.g. EMLA) is given in repeated high doses over a long time period. Such interactions should therefore be of no clinical importance following short term treatment with lignociane (e.g. EMLA) at recommended doses.
Use in the elderly. No data available.
Paediatric use. Until further clinical data are available, EMLA should not be used in infants between 0 and 12 months of age receiving treatment with methaemoglobin-inducing agents such as sulphonamides (also see Section 4.9 Overdose) or in preterm infants with a gestational age less than 37 weeks.
Studies have been unable to demonstrate the efficacy of EMLA for heel lancing in neonates.
EMLA should not be applied to the genital mucosa of children owing to insufficient data on absorption. However, when used in neonates for circumcision (genital skin), a dose of 1.0 g EMLA on the prepuce has proven to be safe.
In children/neonates younger than 3 months of age, a transient increase in methaemoglobin is commonly observed up to 12 hours after an application of EMLA. Caution is required in those at risk of tissue hypoxia, e.g. those with anaemia, respiratory and/or cardiac conditions. Prolonged exposure, e.g. greater than 60 minutes application, significantly increases the risk of methaemoglobinaemia. Repeated applications of EMLA in neonates and infants have not been studied and should be avoided.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Methaemaglobinaemia-inducing agents. EMLA may accentuate the formation of methaemoglobin in patients treated with other drugs known to induce methaemaglobinaemia (e.g. sulphonamides).
Other local anaesthetic agents. With large doses of EMLA, the risk of additional systemic toxicity should be considered in patients receiving other local anaesthetics or agents structurally related to local anaesthetics e.g. mexiletine.
Anti-arrhythmic drugs class III. Specific interaction studies with lidocaine and anti-arrhythmic drugs class III (e.g. amiodarone) have not been performed, but caution is advised.
Drugs reducing clearance of lidocaine. Drugs that reduce the clearance of lidocaine (for example, cimetidine or betablockers) may cause potentially toxic plasma concentrations when lidocaine (e.g. EMLA) is given in repeated high doses over a long time period. Such interactions should therefore be of no clinical importance following short term treatment with lidocaine (e.g. EMLA) at recommended doses.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Although the safety of EMLA during pregnancy has not been established in animal reproductive toxicology studies, lidocaine and prilocaine have been used by a large number of pregnant women and women of child-bearing age, without an increased incidence of malformations or other direct or indirect harmful effects on the foetus having been observed.
Use in lactation. No information is available on the excretion of lidocaine, prilocaine or their metabolites into breast milk following the administration of EMLA.
Following parenteral administration, lidocaine is excreted into breast milk. Because of low maternal systemic absorption following application of recommended doses of EMLA, the amount of lidocaine and prilocaine that may be ingested by the breast-fed infant would be extremely small.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Frequency of adverse events. Intact skin. Common events (≥ 1% and < 10%). Skin: transient local reactions at the application site such as, paleness, erythema (redness) and oedema.
Uncommon events (≥ 0.1% and < 1%). Skin: skin sensations (an initial, usually mild burning sensation, itch or warmth at the application site).
Rare events (< 0.1%). General: in rare cases, local anesthetic preparations have been associated with allergic reactions (in the most severe instances anaphylactic shock).
Rare cases of discrete local lesions at the application site, described as purpuric or petechial, have been reported, especially after longer application times in children with atopic dermatitis or mollusca contagiosa.
Increased methaemoglobin level.
Methaemoglobinaemia and/or cyanosis.
Corneal irritation after accidental eye exposure.
Leg ulcer. Common events (≥ 1% and < 10%). Skin: transient local reactions at the application site such as, paleness, erythema (redness) and oedema.
Skin sensations (an initial, usually mild burning sensation, itch or warmth at the application site).
Uncommon events (≥ 0.1% and < 1%). Skin: skin irritation (at the application site).
Rare events (< 0.1%). General: in rare cases, local anesthetic preparations have been associated with allergic reactions (in the most severe instances anaphylactic shock).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
Rare cases of methaemoglobinaemia have been reported.
Prilocaine in high doses may cause an increase in the methaemoglobin level particularly in conjunction with methaemoglobin-inducing agents (e.g. sulphonamides). Clinically significant methaemoglobinaemia should be treated with a slow intravenous injection of methylene blue.
In the unlikely event of systemic toxicity following epidermal application of EMLA, the signs and symptoms anticipated would be similar in nature to those observed following other routes of administration of local anaesthetics. Owing to slow absorption into the circulation from intact skin, a patient with signs of toxicity should be observed for several hours after treatment.
Systemic toxicity to amide type local anaesthetics is initially manifested as CNS excitation and may result in a slow onset of nervousness, dizziness, blurred vision and tremors followed by drowsiness, convulsions, unconsciousness and possibly respiratory arrest.
Toxic cardiovascular reactions to local anaesthetics are usually depressant in nature, may occur rapidly and with little warning and can lead to peripheral vasodilation, hypotension, myocardial depression, bradycardia and possible cardiac arrest. Severe neurological symptoms (convulsions, CNS depression) must be treated symptomatically by respiratory support and the administration of anticonvulsive drugs.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Lidocaine and prilocaine are both amide-type local anaesthetic agents. Both agents stabilise the neuronal membrane preventing the initiation and conduction of nerve impulses thereby effecting local anaesthetic action.
EMLA provides dermal anaesthesia. The depth and quality of anaesthesia depends upon the application time and the applied dose.
Local anaesthesia with EMLA is achieved after 60 minutes application. EMLA should be applied as EMLA patch; or EMLA cream should be applied under an occlusive, impermeable dressing. Following the application of EMLA cream for 1 - 2 hours, the duration of anaesthesia is at least 2 hours after removal of the occlusive dressing.
Reliable anaesthesia for the cleansing of leg ulcers is achieved after an application time of 30 minutes in most patients. An application time of 60 minutes may improve the anaesthesia. The cleansing procedure should start within 10 minutes of removal of the cream. There is no clinical data regarding cleaning started after 10 minutes of cream removal.
A reduced number of cleansing sessions are required to achieve a clean ulcer when EMLA is used compared to a placebo.
No negative effects on ulcer healing or bacterial flora have been observed when EMLA has been used.
EMLA may cause transient local peripheral vasoconstriction or vasodilation, observed as transient paleness or redness, at the treated area.
Clinical trials. In clinical trials, venepuncture or venous catheterisation was pain-free in 50 - 59% patients, slightly painful in 35 - 40% and painful in 3 - 6%. Anaesthesia may be less for skin structures below the deep fascia.
In clinical trials in adults assessing pain associated with intramuscular influenza vaccination and intramuscular and subcutaneous injections of saline solution, EMLA significantly reduced injection pain relative to placebo.
In clinical trials in infants and children assessing pain associated with subcutaneous and intramuscular vaccination, EMLA significantly reduced injection pain behaviours and pain scores relative to placebo.
In clinical trials assessing the effects of EMLA on intramuscular and subcutaneous, live and non-live vaccines, it was demonstrated that EMLA does not adversely affect antibody response. A clinical trial assessing the effect of EMLA application prior to intracutaneous BCG injection demonstrated that EMLA did not affect the immunisation response.
5.2 Pharmacokinetic Properties
Systemic absorption and anaesthetic efficacy of lidocaine and prilocaine from EMLA is dependent upon the characteristics of the leg ulcer, the applied dose, total application area, application time, thickness of the skin (which varies between different areas of the body), other conditions such as skin diseases, and shaving.
Intact skin. The extent of systemic absorption was approximately 10% following application to the face (10 g/100 cm2 for 2 hours). Maximum plasma levels (mean 0.16 and 0.06 microgram/mL of lidocaine and prilocaine respectively) were reached after approximately 2.5 hours.
After application to the thigh in adults (60 g cream/400 cm2 for 3 hours) the extent of absorption was approximately 5% of lidocaine and prilocaine. Maximum plasma concentrations (mean 0.12 and 0.07 microgram/mL) were reached approximately 2 - 6 hours after the application.
In adults, a thick layer of EMLA Cream 5% (corresponding to approximately 150 g) has been applied to intact skin areas of up to 1,300 cm2 for application times of up to 7 hours. The highest individual plasma levels observed to date were 1.1 microgram/mL lidocaine and 0.2 microgram/mL prilocaine. These levels were below those at which symptoms of toxicity would be expected to occur (5 - 10 microgram/mL either agent; also see Section 4.8 Adverse Effects (Undesirable Effects).
Leg ulcers. Following a single application for 30 minutes of 5 to 10 g of EMLA cream to leg ulcers, the maximum plasma levels of lidocaine (range 0.05 - 0.25 microgram/mL, one individual value of 0.84 microgram/mL) and of prilocaine (0.02 - 0.08 microgram/mL) were reached within 1 - 2.5 hours.
After an application time of 24 hours the maximum plasma levels of lidocaine (0.19 - 0.71 microgram/mL) and of prilocaine (0.06 - 0.28 microgram/mL) were usually reached within 2 - 4 hours.
Following repeated applications for 30 - 60 minutes of 2 - 10 g EMLA cream 3 - 7 times a week, for up to 15 doses, during a period of one month, there was no apparent accumulation in plasma of lidocaine and its metabolites monoglycinexylidide and 2,6-xylidine or of prilocaine and its metabolite orthotoluidine. The maximum observed plasma levels for lidocaine, monoglycinexylidide and 2,6-xylidine were 0.41, 0.03 and 0.01 microgram/mL respectively. The maximum observed plasma levels for prilocaine and ortho-toluidine were 0.08 microgram/mL and 0.01 microgram/mL respectively.
Children. Following the application of 1.0 g of EMLA cream in neonates below 3 months of age, to approximately 10 cm2 for one hour, the maximum plasma concentrations of lidocaine and prilocaine were 0.135 microgram/mL and 0.107 microgram/mL respectively.
Following the application of 2.0 g of EMLA cream in infants between 3 and 12 months of age, to approximately 16 cm2 for four hours, the maximum plasma concentrations of lidocaine and prilocaine were 0.155 microgram/mL and 0.131 microgram/mL respectively.
Following the application of 10.0 g of EMLA cream in children between 2 and 3 years of age, to approximately 100 cm2 for two hours, the maximum plasma concentrations of lidocaine and prilocaine were 0.315 microgram/mL and 0.215 microgram/mL respectively.
Following the application of 10.0 - 16.0 g of EMLA cream in children between 6 and 8 years of age, to approximately 100 - 160 cm2 for two hours, the maximum plasma concentrations of lidocaine and prilocaine were 0.299 microgram/mL and 0.110 microgram/mL respectively.
5.3 Preclinical Safety Data
Genotoxicity. Genotoxicity tests with lidocaine are inconclusive. In genotoxicity studies, a metabolite of lidocaine, 2,6-xylidine, showed evidence of activity in some tests but not in other tests. A metabolite of prilocaine, o-toluidine, has also shown evidence of mutagenic activity in some genotoxicity tests but not others.
Carcinogenicity. A metabolite of lidocaine, 2,6-xylidine, has been shown to have carcinogenic potential (nasal and subcutaneous tumours) in preclinical toxicological studies evaluating chronic exposure. A metabolite of prilocaine, o-toluidine, has also been shown to have carcinogenic potential (e.g. renal, bladder, spleen, subcutaneous tumours) in preclinical toxicological studies.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this product.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
EMLA cream 5%. Store below 30°C.
EMLA patch 5%. Store below 25°C.
6.5 Nature and Contents of Container
EMLA cream 5%. Containing lidocaine 25 mg and prilocaine 25 mg per gram.
5 g tubes in packs of 5 with 10 occlusive dressings or packs of 1 with 2 occlusive dressings.
5g and 30 g tube.
EMLA patch 5%. Patches containing 1 g of EMLA emulsion in packs of 2 or 20.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Lidocaine.
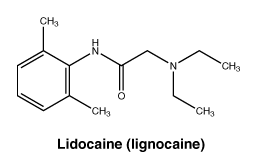
Molecular weight 234.3.
Prilocaine.
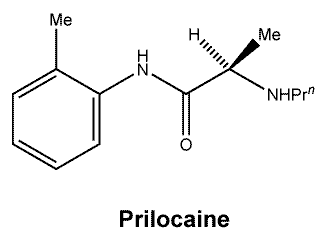
Molecular weight 220.3.
CAS number. Lidocaine. CAS no.137-58-6.
Prilocaine. CAS no. 721-50-6.
7 Medicine Schedule (Poisons Standard)
Pharmacy Medicine: S2.
Date of First Approval
13 August 1991
Date of Revision
10 September 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.